Abstract
The COVID-19 pandemic has caused much suffering through disease and death, disruption of daily life, and economic havoc. Global health infrastructure has been challenged, in some cases failing. In the United States, the inability of laboratories to provide adequate testing for the causative pathogen, severe acute respiratory syndrome coronavirus 2, has been the subject of negative press and national debate. Even so, these challenges have prompted pathology practices and clinical labs to change their organizations and operations for the better. The natural positive evolution of the University of Oklahoma Department of Pathology and OU Health Laboratories has been greatly accelerated by the global pandemic. While developing a substantial COVID testing response, our department of pathology and laboratories have evolved a much nimbler organizational structure, established an important research partnership, built a translational research resource, created a significant reference lab capability, and completed many key hires against a national background of hiring freezes and pay cuts. Also, the high visibility of the clinical lab and pathologists during the outbreak has reinforced the value of lab medicine to patient care across our health system. In the midst of significant ongoing changes to the structure and financing of our underlying organizations, high trust among departmental, hospital, health system, and medical school leadership during the pandemic has promoted these positive changes, allowing us to emerge much stronger from this crisis.
Keywords
Introduction
The COVID-19 pandemic has been a unique challenge outside of our direct professional experience. This tragedy has affected all aspects of everyday life. The health care system has been heavily stressed, in some instances breaking down through lack of physical capacity, shortage of providers, disruptions in supply chains, and delayed care of non-COVID maladies. Clinical laboratories have been in the news for failure to provide widespread and timely testing. The vaunted US laboratory infrastructure was at first overwhelmed, lacking necessary reagents, supplies, and appropriately trained personnel. In addition, the initial requirement for emergency use authorization of tests delayed laboratory responses to the crisis. Our early experience was that virtually all COVID-related testing supplies were difficult or impossible to obtain. Vendors often explained that the federal government was directing allocations, and in many cases, no information was available regarding future availability. Despite this ongoing disaster, the OU Health Laboratories and the University of Oklahoma Department of Pathology are headed toward the post-pandemic world with improved capabilities and resources. The natural positive evolution of the department and laboratory has been greatly accelerated by the global pandemic.
Background
For many years, our teaching hospital system was subtotally owned and entirely managed by a for-profit hospital chain. The separate College of Medicine has been a not-for-profit unit of the University of Oklahoma, the state’s flagship institution of higher education. An affiliated not-for-profit entity, OU Medicine, recently rebranded as OU Health, bought out the for-profit company on February 1, 2018. This realignment of the university and its academic health system set the stage for long-term, far-reaching improvements. Essentially, this shift away from shareholders as stakeholders brought local decision-making for any hospital margin. Because expenditures had been curtailed in anticipation of the transition, demand for capital, operating funds, and hires was pent up. Some of the earliest major decisions included building a patient tower (which began a staggered opening in the fall of 2020) that increased the square footage of the adult acute care hospital by approximately 80% and added operating room capacity, designing a multiyear move to the Epic electronic health record from Meditech, upgrading radiology’s picture archiving and communication system, and expanding our children’s intensive care unit. For pathology and laboratory, the changeover first led to a major equipment refresh that was designed and contracted for in 2019 for deployment in 2020. This push did not aid the ramp-up of COVID testing capacity but included an array of analyzers and automation in chemistry, immunoassay, hematology, HLA, and flow cytometry. Upgraded immunohistochemistry capacity and on-site in-situ hybridization were also budgeted for anatomic pathology. Although COVID has slowed down several of these platform implementations, other initiatives are fundamentally changing pathology services at OU.
Emergence of the Global Pandemic Driver
The emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and planned responses were discussed in departmental and institutional meetings in February and early March 2020. On March 6, 2020, the first case of COVID-19 in Oklahoma was reported in Tulsa County. On March 11, the local National Basketball Association (NBA) game was postponed after a visiting team player tested positive for the novel coronavirus. This news galvanized the state. On March 21, 2020, Oklahoma Governor Kevin Stitt issued an executive order directing the OU Medicine Laboratories, Oklahoma State University, and the Oklahoma State Department of Public Health to work together to provide COVID testing. 1 Although not specifying structure or numbers, the executive order did launch a series of regular meetings between these entities to share ideas and expertise and established our laboratory’s role in supporting community testing. Critically, it also aided working with a major vendor of COVID testing kits and equipment. Thus began a highly visible struggle to overcome a myriad of supply chain, personnel, safety, and reporting issues to serve the citizens of Oklahoma. These pressures, superimposed on ongoing organizational transformation, led to a series of positive changes in our department and clinical laboratories.
Positive Evolution of the Department and Laboratory Reporting Structure
OU Health includes hospitals for adults and children and a community-based hospital. The OU Health Labs serve the combined system and are medically directed by consulting College of Medicine faculty members. These pathologists and PhD laboratorians report up through the medical school dean to the OU Health Sciences Center (OUHSC) provost. On the hospital side, just before the pandemic, laboratory personnel reported through 2 levels of nursing leadership (Figure 1). Laboratory staffing, equipment, and reagent decisions were under final control of the hospital. Although this reporting structure sufficed for normal operations, discussions had been underway for several months to create a more formal service line led by an associate vice president for laboratory services partnered with the pathology faculty chief of service. Still in concept, this proposal did not have sufficient buy-in by leadership or a clear timeline.
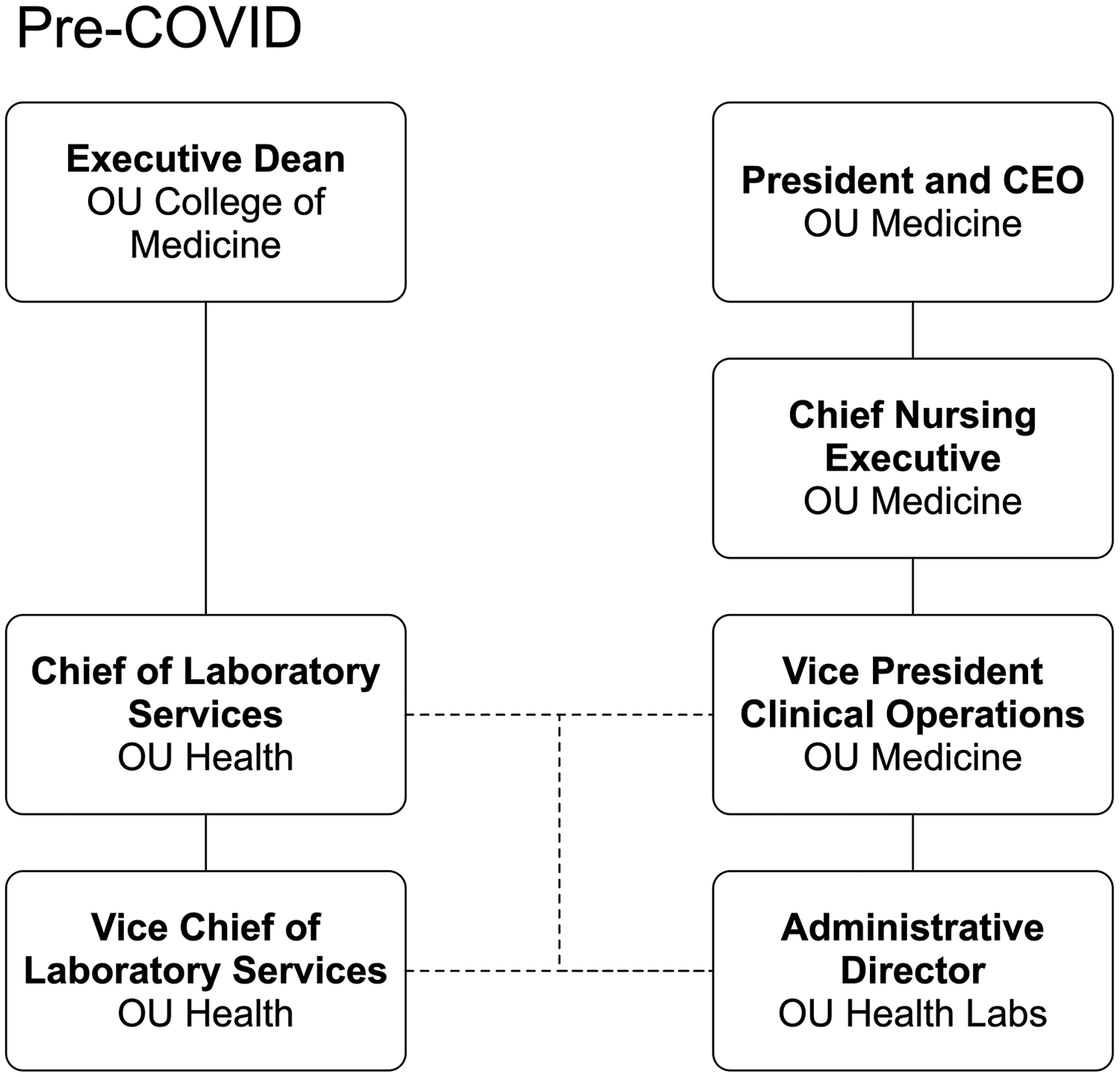
Pre-COVID organizational chart.
As spread of SARS-CoV-2 intensified, the pressure on both nursing and the laboratory made it clear that the existing reporting structure was untenable. Nursing services were consumed by our system ramp-up for COVID, and our leadership could not adequately represent our urgent needs at the highest level of the organization. Oversight of the lab was therefore transferred to a dyad, the chief of the laboratory service and the president of Oklahoma Children’s Hospital, supported by the vice chief of service and the children’s hospital chief operating officer (Figure 2). The chief of laboratory service and the system CEO made this selection, reasoning that the lab needed a strong central voice in administration and that a pediatric hospital would be less likely to be affected by a disease that was particularly devastating for elderly patients. The pairs immediately clicked, yielding an explosion of purchasing, hiring, and problem-solving. Frequent calls from the system CEO, aided by a deep level of trust, expedited moving resource allocation to the level of the dyad. Decision-making was collaborative and immediate: if the top dyad agreed, it simply got done. Some $2 million in testing equipment was purchased, space was repurposed and remodeled, and 15 full-time equivalents (FTEs) were hired, more than doubling the size of our virology staff. Testing in support of the health system and community was top priority, and no resources were held back. Having the only NCI-designated cancer center, level 1 trauma center, and freestanding children’s hospital in the state, we were determined to serve our patients as a surge in COVID cases loomed.
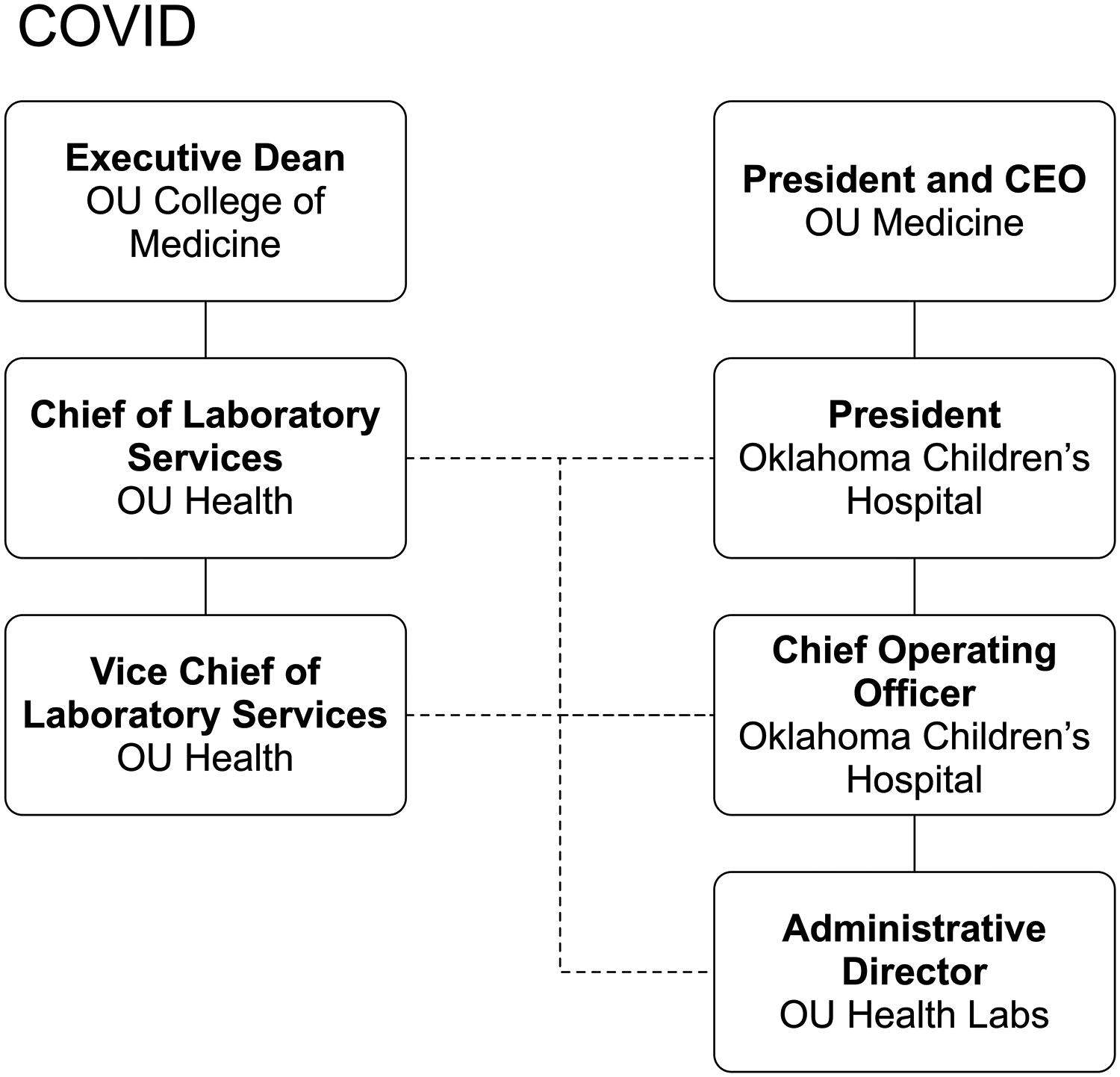
COVID organizational chart.
The leadership dyad noted that neither had ever worked in an environment where decision-making and spending were so free and, at times, it was difficult to absorb, train, and deploy personnel and equipment. Given our rapid expansion, additional effort at the manager level was shifted from anatomic pathology to COVID testing to strengthen staff onboarding, workflow coordination, and occurrence management. Although equipment and personnel spending has slowed down now and is subject to more routine justification and review processes, the successful new reporting model continues, a huge win for the laboratory. An associate vice president for laboratory services position has been posted, and discussions are in progress about pathology and laboratory forming the first true service line in the health system, as has been accomplished at other centers. 2,3 In addition, our productive working relationship and recognition of the effort of the pathology faculty members have also fueled discussions toward an improved hospital contract for the pathology department.
Translational Research Partnership
Across the street from our main clinical lab, the Oklahoma Medical Research Foundation (OMRF) is an independent research institute internationally known for work in immunology and coagulation. In late March, OMRF’s director of translational informatics and clinical research resources (J.M.G.) reached out to propose using the Fluidigm Biomark HD microfluidics platform from his lab to detect SARS-CoV-2. This option offered the possibility of greatly increased testing capacity using reagents that were in less demand. OU Health Sciences Center core laboratory scientists also offered to help develop a COVID assay. To this point, given the history of being managed by a for-profit hospital chain, the OU Health Laboratories typically deployed Food and Drug Administration (FDA)-approved assays, although several of the faculty medical directors had substantial experience with laboratory-developed tests (LDTs), either from training or in the department’s separate molecular pathology laboratory. With the encouragement of the OUHSC vice president for research, OU Health, OMRF, and OUHSC almost immediately decided to develop a high-throughput robotic-assisted polymerase chain reaction (PCR) test using the Biomark HD. After completing space improvements in record time, the needed equipment was moved to an unused area of the OU Health Labs that had been awaiting renovation. Research scientists and technicians initiated the test design and validation process but were gradually replaced by lab technologists, clinical faculty, and a key pathology resident (R.L.) who had previously worked with this research platform. Finally, the director of the infectious diseases lab (C.B.M.) oversaw the completion and deployment of the LDT, the Oklahoma COVID-19 PCR. We submitted for emergency use authorization on April 20, 2020, and again with a revised version of the assay on June 26, 2020. However, as we awaited review, FDA determined that such authorization was not required, so we declined further consideration. This effort has created a much stronger tie between OMRF and the department of pathology, with additional collaborations expected.
Ultimately, the partnership with OMRF yielded a new R&D section of the OU Health Labs. This area is contiguous with our main laboratory and is now part of the infectious diseases section, with OU Health solely responsible for oversight. As COVID testing moderates, this lab will become its own section, with broadened scope and instrumentation. The further development of this capacity to support bench-to-bedside work has been formalized as part of a memorandum of understanding finalizing the department of pathology’s graduate program 7-year review. Thus, campus researchers will have a resource for the translation of novel diagnostics that is fully integrated with our clinical operations.
Creation of Reference Lab Capabilities
Although the OU Health Laboratories have a broad test menu, before COVID, only very limited testing was performed for nonsystem patients or clients, most notably for HLA. Reference testing was discouraged when we were managed by a for-profit, and our HIS/LIS was poor at supporting such. Early in the COVID testing build-out, we recognized that the OU Health Labs would perform large volumes of both medical system and community testing. Testing clients eventually included the state and local health departments, OU student health, a local private school, and a limited number of local businesses. To handle the increased numbers of specimens, a reference lab-like frontend was built to process community swab pod specimens and staffed separately from our core lab’s central receiving and processing area. This created 2 flows of specimens, our preexisting Meditech-based one and a physically separate path using an in-house developed laboratory information management system. The separate high-volume accessioning area was developed in repurposed space to handle high-throughput community testing, which ramped up to over 1000 COVID PCR tests per weekday. We also deployed a commercial software solution for reference work, CareEvolve, allowing for point of collection preaccessioning (OU Health nursing and operations personnel) and flexible reporting, including a patient portal. Besides these features, this system was chosen for its rapid implementation and scalability. As the pandemic subsides, we will use this software package, and, in 2022, the Epic electronic health record, to bolster reference lab testing for the region. To start, we plan to offer new tests in infectious diseases and specialized coagulation, areas of significant existing expertise, further expanding our menu and hopefully supporting additional faculty members.
Visibility and Execution Reinforced Our Value Across the System
Early on, pathology and the lab worked closely with infection control to review biosafety across our clinical, educational, and research missions. Every relevant laboratory process and procedure was also reviewed and modified as needed. Several informative “lab alerts” were distributed to the medical staff, educating them on test characteristics, ordering, and turnaround time expectations. We also set up a multidisciplinary test utilization group composed of our lead infectious disease physician, an inpatient CMO (a pediatric surgeon), an outpatient CMO (a family practitioner), and 3 pathologists to address all types of COVID testing. This workgroup met as needed, designing clinical protocols when tests were initially scarce and eventually prioritizing testing by clinical scenario, as turnaround times ranged from about 1.5 hours for rapid platforms to almost 24 hours for batched assays. In addition, this group regularly reported to our overall incident command for COVID, on which the 3 nonpathologists and the chief pathologist served. Operationally, pathologists also had significant roles in the state testing effort and ongoing infection control. We were also very active in the media, with several interviews with multiple television stations, formal social media events, and newspaper appearances. Internal and external activities such as these have been suggested as ways for academic pathologists to add value. 4
Our efforts to support OU Health continue, as we deploy new SARS-CoV-2 platforms to flexibly support a broad range of testing needs and help guide our colleagues in test selection as influenza and other respiratory disease season progresses.
Growing for the Future
The department of pathology completed 3 recruitments for academic year 2021 before the pandemic. Although none of our more recent faculty hires have been specific for COVID, 6 more positions, including director of anatomic pathology, have been filled under pandemic conditions, positioning the department well for an anticipated increase in case volumes from just completed OR construction. In consultation with the dean, we have continued to push forward with recruiting for future growth. Even though COVID has produced a significant financial drag on budgets across the medical school and the health system, 3 faculty searches are open and strong candidates are being interviewed. We hope that this windfall of talent, at a time while the job market has been in disarray, will strengthen our academic and clinical missions for years to come.
Academics
Academic productivity declined during the first few months of the pandemic, particularly as labs in our basic science division suspended operations. However, our teaching programs adapted rapidly, and the forced exploration and adoption of various low-contact and virtual approaches to education continues to pay dividends. Paradoxically, these new formats increased attendance at some academic activities and important meetings, as our large campus (up to a 12-minute walk between certain venues) was virtually reduced to one’s desktop or laptop. In addition, based on the experience of the virtual Association of Pathology Chairs summer meeting, grand rounds exchanges were trialed with the department of pathology of West Virginia University. Faculty members present by Zoom to both institutions, a particularly cost-effective way for junior faculty to gain experience and exposure. Based on our initial experience, surveyed faculty unanimously agreed to continue the exchange and add additional institutional partners.
COVID-19 has also driven the creation of new academic partnerships. For instance, a member of our faculty submitted a national grant as coinvestigator with the OUHSC College of Public Health, a first for us. Efforts on the main university campus (20 miles to the south) to use 3D printing to make nasopharyngeal swabs led to new collaborations with our department, which previously had used 3D printing to make tools and aids for grossing and impossible-to-source replacement parts for lab equipment.
Five of our 45 faculty members became involved in the pandemic response nearly full time. However, a decrease of approximately 40% in anatomic specimens for 2.5 months, with a subsequent shortfall of approximately 10%, and initial restrictions on bench research (which were gradually relaxed as social distancing could be achieved) created additional time for academic pursuits for some. Grant and manuscript submissions increased markedly as continued academic activities and collaborations were encouraged and promoted. Overall, grants submitted by departmental faculty as PI or Co-PI rose 33% from March to December for 2020 versus 2019. Manuscripts submitted rose by 45% between the 2 periods.
Conclusion
The COVID-19 global pandemic has driven change across the health care landscape, 5 including a marked expansion in telehealth 6 and innovations in vaccine development and production. 7 Clinical laboratories have experienced similar transformations. Like other US laboratories, 8 -11 our anatomic pathology volumes decreased during the pandemic. We also ramped up COVID molecular testing using multiple platforms, a strategy employed by other institutions. 12 -15 Beyond these effects, COVID-19 rapidly accelerated the structural and operational transformation of our pathology department and clinical lab all while our medical school and health system have continued to integrate, with changing ownership, organization, and funds flow.
The responses of our department and clinical lab to COVID-19 have made us more efficient and effective. Perhaps most importantly, we report at least one level higher up. With this change, the current administrative partners more clearly see the capabilities and needs of pathology and the laboratory. Decision-making is much faster, and our reach through the organization is enhanced. Furthermore, the combination of our transition to a true freestanding academic medical center and the threat of COVID-19 has created productive relationships with city, county, and state public health departments, OMRF, the College of Public Health, and main campus research labs. Finally, we have gained translational research capabilities and developed significant reference lab possibilities. Through this chaotic time in which we often acted on instinct, years of organizational evolution occurred in less than one. Moving past the pandemic, clinical laboratories can use the lessons learned and relationships forged from difficult circumstances to continue to advance the science and practice of laboratory medicine.
Footnotes
Acknowledgments
The authors thank our leaders, John Zubialde, MD, Executive Dean, and Chuck Spicer, System CEO, for their trust and unfailing support. The authors also thank Stephen Prescott, MD, OMRF President, for providing generous assistance in the response to COVID-19. The authors gratefully acknowledge the capable secretarial and technical assistance of Rebecca Brum.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
