Abstract
In February of 2020, New York City was unprepared for the COVID-19 pandemic. Cases of SARS-CoV-2 infection appeared and spread rapidly. Hospitals had to repurpose staff and establish diagnostic testing for this new viral infection. In the background of the usual respiratory pathogen testing performed in the clinical laboratory, SARS-CoV-2 testing at the Montefiore Medical System grew exponentially, from none to hundreds per day within the first week of testing. The job of appropriately routing SARS-CoV-2 viral specimens became overwhelming. Additional staff was required to triage these specimens to multiple in-house testing platforms as well as external reference laboratories. Since medical school classes and many research laboratories shut down at the Albert Einstein College of Medicine and students were eager to help fight the pandemic, we seized the opportunity to engage and train senior MD-PhD students to assist in triaging specimens. This volunteer force enabled us to establish the “Pathology Command Center,” staffed by these students as well as residents and furloughed dental associates. The Pathology Command Center staff were tasked with the accessioning and routing of specimens, answering questions from clinical teams, and updating ever evolving protocols developed in collaboration with a team of Infectious Disease clinicians. Many lessons were learned during this process, including how best to restructure an accessioning department and how to properly onboard students and repurpose staff while establishing safeguards for their well-being during these unprecedented times. In this article, we share some of our challenges, successes, and what we ultimately learned as an organization.
Introduction
On December 31, 2019, a cluster of cases of pneumonia was reported in the Hubei province of Wuhan, China. 1 On January 20, the first documented case of COVID-19 in the United States was diagnosed in Seattle, Washington. 2 The first case was diagnosed in New York on March 1, 2020. 3 Our virology laboratory director had been contacted by the Head of the Wadsworth Laboratories of the New York State Department of Health (NYSDOH) on March 1 to inquire about Montefiore willingness to perform on site testing. By March 3, the first patient who met the NYSDOH criteria for persons under investigation (PUI) for COVID-19 presented to Montefiore Medical Center (MMC), and immediately after this sentinel event, our laboratory began to receive requests for COVID-19 testing.
The COVID-19 pandemic continued to surge in New York City through the spring, requiring all New York area hospitals to determine how best to handle this unprecedented crisis, and MMC shifted to care almost entirely for COVID-19 patients. Montefiore Medical Center occupancy swelled from 700 beds in January to 1085 beds during the first peak in April, a 55% increase. During this peak, there were over 1000 concurrent COVID-positive patients being treated by MMC. This increase necessitated changes in laboratory testing protocols for all testing areas, most notably in diagnostic testing for SARS-CoV-2, the causative agent of COVID-19.
As the hospital system began implementing new policies and procedures in order to deal with the emerging pandemic, so too our pathology department needed to pivot many aspects of the laboratory work to meet these new challenges. The most radically different and essential of those changes was the development of a Pathology Command Center (PCC) to handle COVID testing specimens.
Establishing the Command Center
Between March 4 and March 14, our medical center laboratories received 269 upper respiratory swab specimens from COVID-19 PUIs. This sudden influx of specimens increased the demands on our laboratory staff that were already fully occupied performing routine duties. During this time interval, MMC had not yet submitted their request for emergency authorization for SARS-CoV-2 testing to be performed in house. Real-time Reverse Transcriptase Polymerase Chain Reaction (Real-time RT-PCR) testing was only available for Montefiore patients at the NYC Department of Health and Mental Hygiene (DHMH). The amount of testing available was limited due to reagent and personnel constraints. The NYC DHMH required that providers call for preapproval, so requests could be prioritized.
By mid-March, the virology laboratory at MMC finished validation and submission to the FDA of an RT-PCR-based assay for SARS-CoV-2 on the Luminex Aires platform and began offering it clinically. At that time we were also able to shift all send out testing from the NYC DHMH laboratories to in-house testing and external reference laboratories, removing the requirement of preauthorizing SARS-CoV-2 testing. However, while these changes streamlined ordering for clinicians, the volume of specimens overwhelmed accessioning staff. During the week of March 15, nearly 4500 specimens were received for viral testing; approximately half of those required testing for SARS-CoV-2. This was nearly double the volume from 2 weeks prior (Figure 1). In addition to the sheer volume, the laboratory information system (LIS) required separate orders for specimens to reference versus in-house laboratories. Reordering based on specimen routing was overwhelming accessioning staff and leading to delays. Concomitantly, the volume of clinician questions to the laboratory regarding specimen routing, expectations, and delays also increased, putting greater stress on staff. It became clear that additional, dedicated personnel were required to meet the rapidly increasing demands.
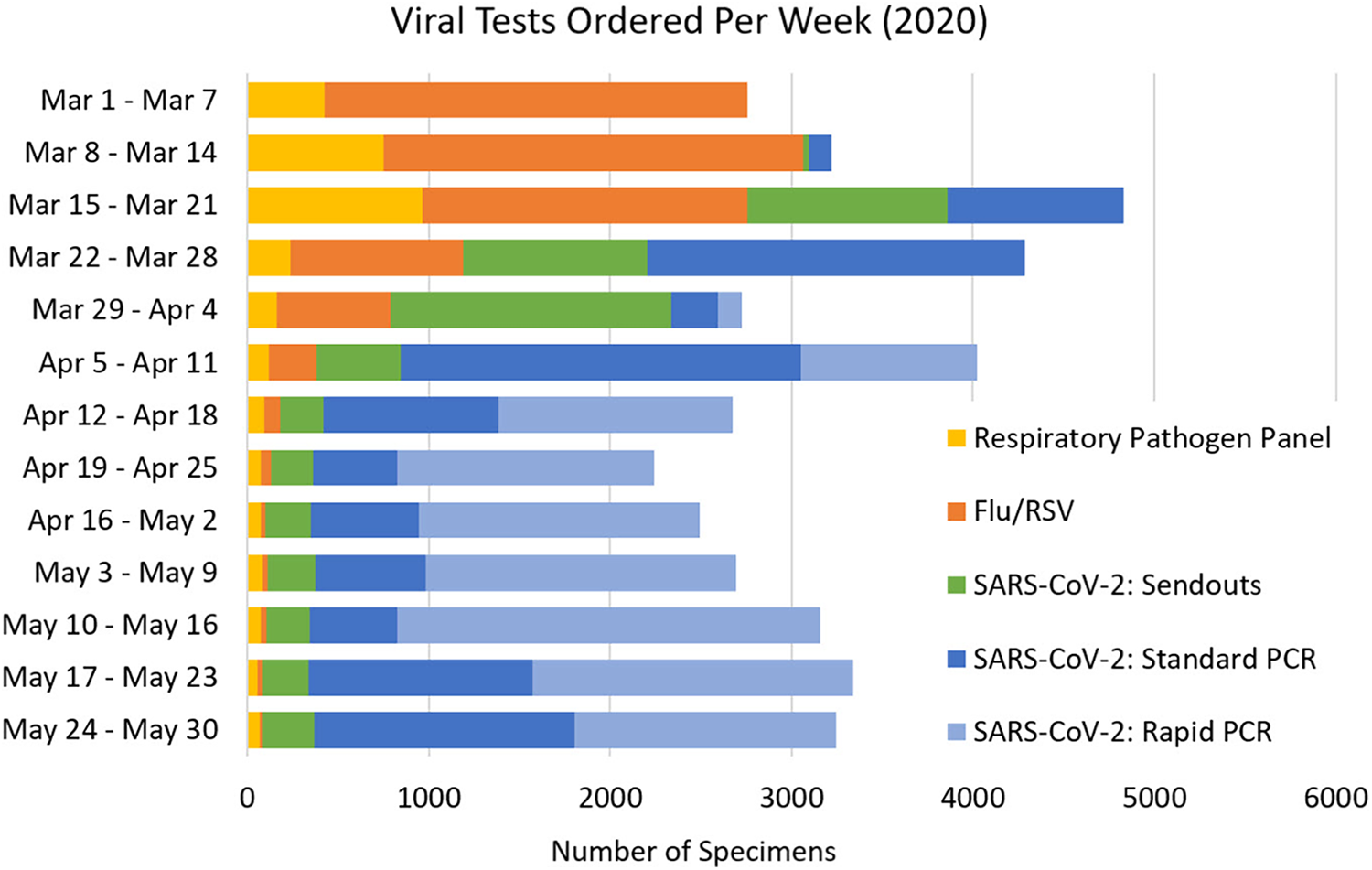
Total number of viral specimens ordered per week from March 1, 2020, to May 30, 2020. Blue represents all SARS-CoV-2 tests performed in-house (microbiology/Rapid Response Laboratory [Rapid PCR: Cepheid] in light blue, virology [Standard PCR: m2000; Panther Fusion, Luminex Aries] in dark blue). Green represents all SARS-CoV-2 tests sent out to NYC DHMH, Viracor, or Quest. Orange represents all Influenza A, Influenza B, and/or Respiratory Syncytial Virus tests. Yellow represents all respiratory pathogen panel tests.
At this time, several departments established “Command Centers” across the hospital system in order to coordinate personnel and standards of care. As a result, a leadership team within the Department of Pathology began to explore a model whereby a centralized area could be established in which all specimen accessioning, tracking, result follow-up, and clinician communication could occur.
The PCC was thus designed and established over the course of a single week, starting on March 16 (Figure 2). On that day (day 1), the goals of the PCC were outlined, the number of staff required for operations were established, and 2 laboratory directors were identified to serve as leads. It was determined that a maximum of 3 concurrent staff would be required for the command center, and that it should be staffed 24/7. This allowed the department to determine the proper location, given social distancing guidelines. A room which had previously been set aside for the construction of a new sequencing laboratory was identified. The room had already been emptied as a result of the expected construction and was capable of housing 3 workstations 6 feet apart as well as a sink and eyewash station. On day 2, the MMC facilities department was engaged and mobilized to order and place computer workstations, label printers, barcode scanners, and telephones. Minor construction of data and phone lines was also required and arranged. Lastly, a specimen refrigerator and specimen racks were acquired and placed in the room. On day 3, one member of the leadership team was diagnosed with COVID-19 and a new director was immediately identified. This ensured continued leadership coverage over weekends and in the event of a further spread of the disease, which was accelerating rapidly in New York and surrounding regions. Also on day 3, 6 senior MD-PhD students with clinical and electronic medical record experience were interviewed over Zoom, hired, and arrived for training. This extremely accelerated time frame was made possible through very close collaboration of the department of pathology, school of medicine, hospital administration, legal department, and human resources department.
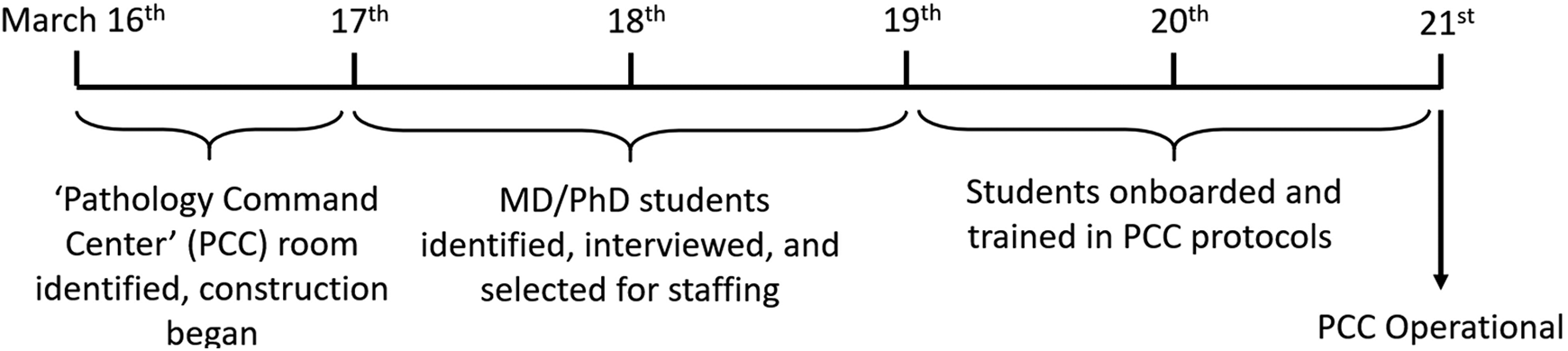
Timeline of the preparation and staffing for the Pathology Command Center.
Over the rest of day 3 and through day 4, the students were trained in infection control and safety, the LIS, and laboratory regulations. Infection control and safety protocols were strictly in line with hospital and Infectious Disease department guidelines. They included the following: use of disposable gowns, gloves, and mouth, nose, and eye coverings at all times, hand washing when changing gloves or exiting the PCC, and a per-shift minimum of cleaning all surfaces and used specimen racks with bleach or other suitable antiviral agent. Rejection of leaking specimens was also strictly enforced to reduce transmission risk for our staff. Laboratory information system consisted of training the MD-PhD students to receive and track orders, entering orders from requisition forms received from outside facilities, using tracking dashboards and pending lists to find delayed specimens, and how to troubleshoot the most common provider questions. Lastly, students were trained in key regulatory compliance issues such as keeping decontamination logs, maintaining a refrigerator temperature log, and requirements for specimen and requisition patient identifiers.
On days 4 and 5, the students drafted a set of protocols based on the training they had received. These protocols consisted of specimen-routing protocols, defining where tests should be sent based on ordering department and clinical need, troubleshooting protocols for answering provider questions, and shift hand-off protocols. These protocols were finalized and approved by the PCC directors, though they would be revised many times over the following weeks in response to the evolving situation. By the end of day 5, phone lines and specimen delivery for all SARS-CoV-2 testing were directed to the PCC. The command center was fully operational.
Command Center Lessons Learned
The formation and subsequent function of the PCC provided many important lessons for the department and hospital system. Decisions needed to be made immediately to accommodate the need for rapid expansion of testing capabilities, the ever-changing clinical protocols, and concomitant LIS build changes to support these. Equally important was the flexibility to recognize that, with experience, some decisions required modification to improve impact. The novel solutions that were devised to overcome these challenges have shown our departments’ new methods that we will carry forward.
Benefits of specimen routing consolidation
The initial creation of the PCC and the centralization of the various accessioning, customer support, and test routing functions had a myriad of benefits for the laboratory. Firstly, it allowed for rapid receipt and routing of specimens at a volume and pace unseen in our laboratory’s history. By April, SARS-CoV-2 testing alone was higher than the virology laboratory’s total volume for any month in 2019 and was steadily rising (Figure 3). Without the extra resources and specialization afforded to us through the PCC, this would have quickly overwhelmed our accessioning staff who also had to contend with rapidly increasing specimen volumes caused by the rise in hospitalized patients with COVID-19. By diverting all respiratory specimens to the PCC, this pressure was somewhat relieved, and accessioning resources could be used to process specimens efficiently from hospitalized patients.
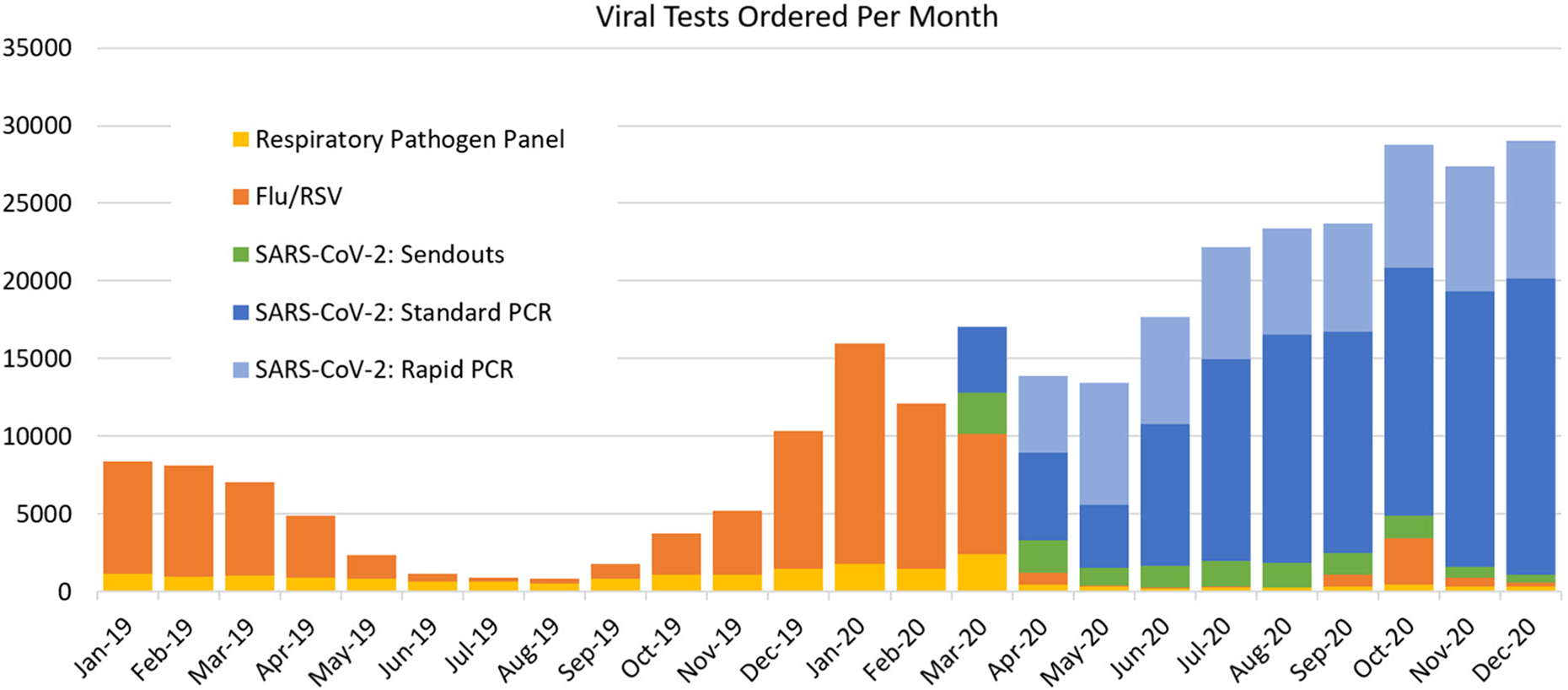
Total number of viral specimens ordered per month in 2019 and 2020. Blue represents all SARS-CoV-2 tests performed in-house (microbiology/Rapid Response Laboratory [Rapid PCR: Cepheid] in light blue, virology [Standard PCR: m2000; Panther Fusion, Luminex Aries] in dark blue). Green represents all SARS-CoV-2 tests sent out to NYC DHMH, Viracor, or Quest. Orange represents all Influenza A, Influenza B, and/or Respiratory Syncytial Virus tests. Yellow represents all respiratory pathogen panel tests.
Evolution of testing platforms
The centralized PCC also allowed the laboratory to make rapid changes to specimen routing that would otherwise have overwhelmed the accessioning staff. The laboratory went live with the Luminex assay on March 11. This was quickly joined by assays on the m2000 (Abbott) and Panther Fusion (Hologic) on March 20 and 25, respectively. The introduction of these 2 instruments allowed the laboratory to shift all reference laboratory testing to in-house testing. This drastically reduced turnaround times (TATs; Table 1) and allowed for expanded employee testing. A further improvement was achieved on April 3 when the GeneXpert SARS-CoV-2 assay (Cepheid) was brought online. The reduced TAT of this assay compared to the other in-house assays made it ideal for patients being considered for admission in order to determine isolation status. However, supplies of these tests were even more restricted than our other assays. The PCC allowed rapid changes in specimen routing to maximize the utilization of the new in-house instruments. The tight shift handoff procedures by the staff ensured continued adherence to any and all changes.
SARS-CoV-2 Test Triaging for the Months of March, April, and May 2020.
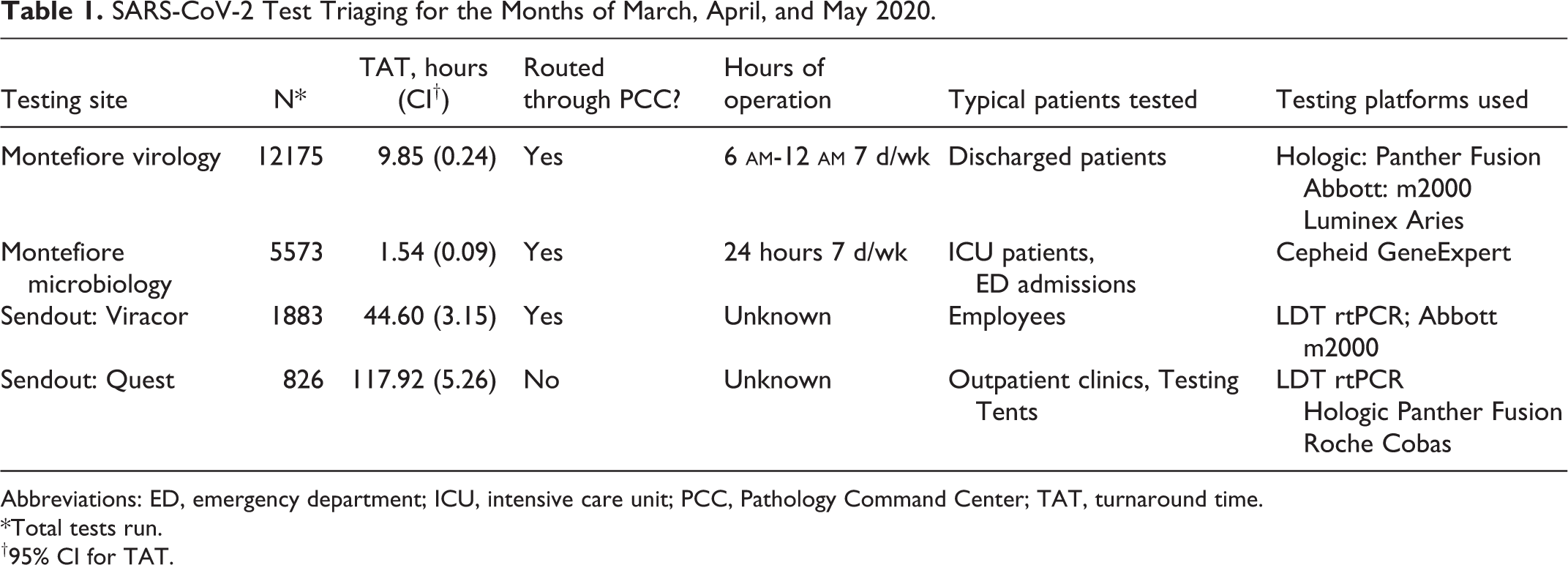
Abbreviations: ED, emergency department; ICU, intensive care unit; PCC, Pathology Command Center; TAT, turnaround time.
*Total tests run.
†95% CI for TAT.
Increased flexibility of dedicated staff
Patient chart review is not ordinarily performed by our accessioning department; however, rapidly changing patient status (ie, order was placed while the patient was stable in the emergency department (ED), but the patient rapidly decompensated and was subsequently admitted to the ICU) made this important. It was feasible to implement because of the availability of the MD-PhD students who had both clinical and laboratory experience. It was essential for proper routing of these high-priority specimens. Laboratory information system changes were implemented to allow PCC accessioning staff to determine the patient’s current status and to easily reroute specimens to reflect the patient’s current status when the specimen was processed. In addition, occasional auditing of patient condition was required to confirm that provider answers to order questions were appropriate. This activity would not have been possible through the ordinary accessioning staff because of the demands of other required duties and lack of medical training required for chart review, but the specialized PCC staff were able to ensure this level of quality control for specimen routing.
It is of note that in New York State, there are additional regulations placed on medical technologists, who have to be licensed by New York State to perform high-complexity testing such as that for SARS-CoV-2. While these requirements were relaxed by NYS Executive Order 202, 4 as the Command Center staff were only accessioning and routing specimens they were not required to be licensed by the state.
In addition to the MD-PhD students originally recruited to this effort, staffing was eventually supplemented by pathology residents who had to be reassigned from other areas in the department due to clinic and autopsy service changes. Residents were able to assist with their considerable clinical and laboratory experience and supplemented our main MD-PhD staff. This allowed for the PCC to have at least 2 staff on hand 24/7 and to account for any last-minute staffing changes.
Importance of prompt LIS support
As previously mentioned, crucial LIS changes were implemented to support specimen routing. In partnership with our LIS team, we designed a testing system that would allow for the PCC staff to make changes to specimen routing without creating ordering complications for our providers. We did this by creating a “shell” order for SARS-CoV-2 RT-PCR testing that allowed consistent ordering by the providers and prompted clinical questions. The answers were provided to the PCC staff through the LIS. This allowed them to route the specimens quickly depending on the current situation and clinical need (Figure 4). This had several advantages. First and foremost, it dramatically simplified ordering for providers, as they only had to choose a single test and answer clear questions about their patient’s status. Secondly, it allowed tracking of the PCC staff’s decision-making. When unexpected routing decisions were made, the PCC staff could answer provider questions through reference to the information available to them in the LIS. In addition, it enabled good laboratory stewardship by conserving precious test resources.
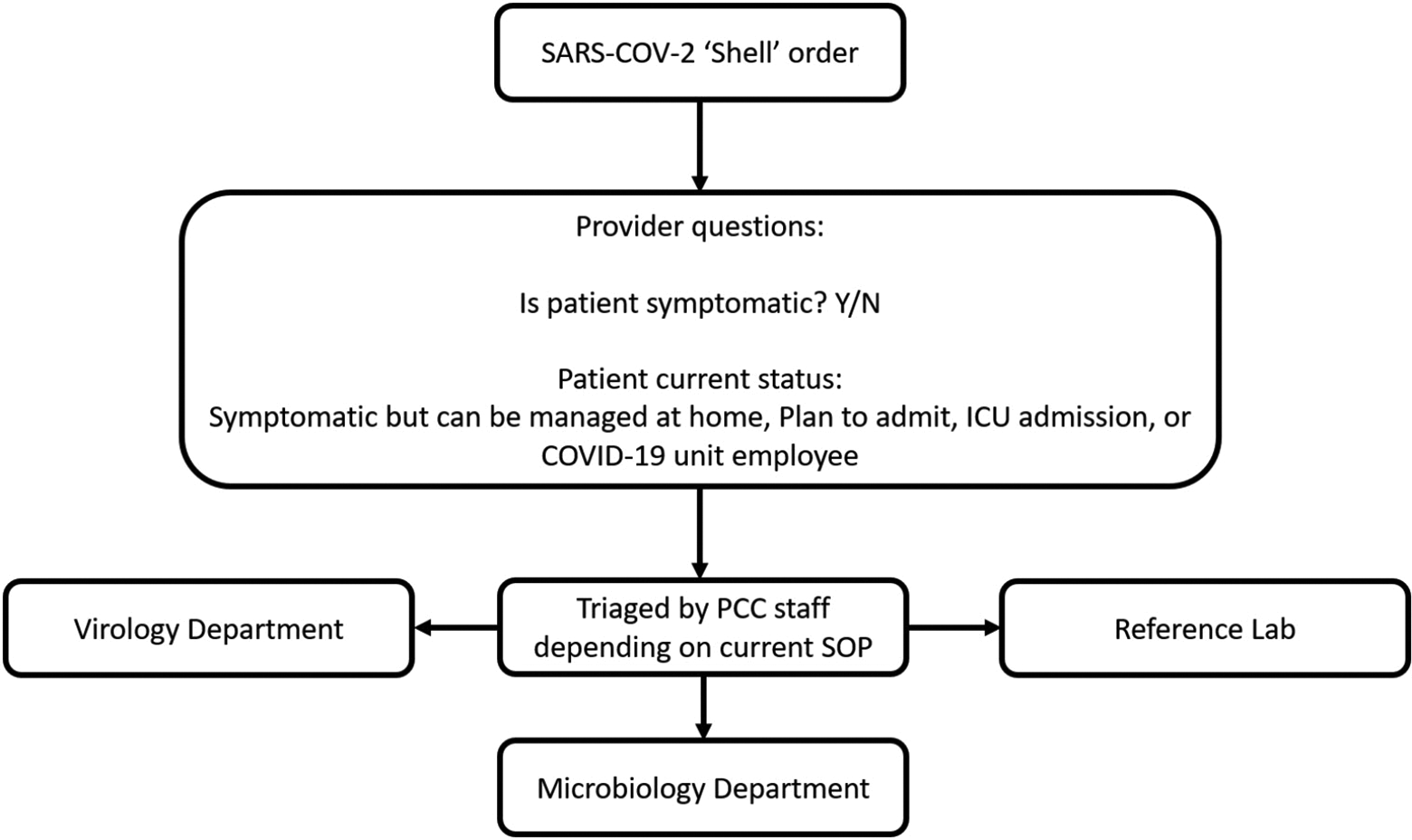
Flowchart of processing of the SARS-COV-2 “shell” order. Providers had access to only one order, which was then triaged by PCC staff depending on the answers to order questions and current laboratory capacity and requirements. PCC indicates Pathology Command Center.
Significance of clear and constant communication
Where the formation of the PCC allowed for centralization of provider questions, it also allowed for centralization of leadership. During the entire lifetime of the PCC, the virology, microbiology, and infectious disease leadership met (virtually) daily and had open lines of communication 24/7. This allowed for the rapid changes to protocols and specimen routing that were required during the chaos of the first surge. Infectious disease leaders were able to inform the laboratory of current and expected changes in patient admission trends and advise on the best use of our limited resources. In addition, the virology and microbiology laboratories were able to share the current and expected reagent allocations so that modifications to specimen routing could be rapidly implemented. This allowed the laboratories to conserve limited resources while meeting provider needs and accounting for unexpected changes.
While the open communication between these teams was a success, retrospective analysis of the PCC operations suggested that communication with other teams could have been handled more effectively. The laboratories communicated mainly with the infectious disease team, who would then communicate to other providers as to the laboratory situation. This communication bottleneck did lead to confusion on the part of various providers as to the testing pathways and expected result TAT. While the PCC was able to handle many provider questions as they occurred, future efforts will involve a heavier investment in provider education in order to inform them of changes in the laboratory expeditiously, especially in such a dynamic situation as the first COVID-19 surge. A weekly written communication from the PCC to the clinical teams outlining the testing platforms utilized, reagent inventory, and explanations for the rationale behind the changes in specimen triaging may have reduced the need for clinical staff to use their limited time to make calls to the PCC.
Impact of MD-PhD Student Participation
Participation of the MD-PhD students as staff afforded them the opportunity to see the functioning of a clinical laboratory in real time; an opportunity not typically provided for these students. None of the students recruited for this effort had matched in pathology, and so this was an experience many of them may never have received. The students were also able to interact with pathology residents and anatomic pathology faculty members who were later brought on to the PCC staff to help in routine activities and supervision, respectively. All of our MD-PhD students described this as an overwhelmingly positive experience and expressed a new appreciation of the work of laboratory staff and the needs of laboratory management.
The MD-PhD students working in the PCC were highly motivated to help in the pandemic response and carried with them lessons that they had learned during their times spent in clinical rotations and in research laboratories. Although they were not utilized for performing the SARS-CoV-2 assays, all were trained to perform RT-PCR testing on the GeneXpert instrument in order to provide supplementary staffing in case of a personnel shortage. Several MD-PhD students were additionally trained and participated in the preparation of plasma specimens for patients enrolled in a clinical trial evaluating convalescent plasma as a treatment against SARS-CoV-2 infection.
The students were able to utilize their clinical training as they performed chart review. After suitable training, students were able to use their clinical judgment to escalate specimens to in-house or more rapid testing platforms based on current patient location, chart review, or a well-justified clinical request. The students also were able to actively practice administrative skills learned from their clinical rotations. The students created a shift hand-off protocol prior to any request from the directors overseeing the PCC. The students would communicate any changes to protocols, any errors they observed, and any ongoing situations requiring follow-up in a communication sheet that was reviewed with those working in the PCC the following shift. This was a form of communication that the students were already familiar with from their clinical duties and worked well in the PCC.
Once the workflow of the PCC became more stable and routine, students began aiding in remnant specimen collection for research. Ultimately, the demand for research specimens exceeded the ability of the clinical laboratory faculty, so additional MD-PhD students were recruited to aid faculty in collecting research specimens to build a biorepository of discarded, deidentified blood, and respiratory specimens. Their clinical training was again utilized, this time in identifying COVID-19 patients and categorizing their disease severity through chart review. Remnant specimens from these patients were collected, aliquoted, deidentified, and added to the COVID-19 biorepository once they were to be discarded from the clinical laboratory. The categorization of these specimen by disease severity ensured broad diversity within the repository. Eventually over 6000 blood and respiratory specimens were collected, organized, and stored. These specimens have been used to establish a lab-developed SARS-CoV-2 antibody detection assay, 5 examined differences in immune responses to SARS-CoV-2 between adult and pediatric patients, 6 discover evidence of secondary thrombotic microangiopathy in COVID-19, 7 establish that extended storage of respiratory specimen doesn’t significantly impact molecular testing results, 8 inspired multicenter collaborations in areas such as antibody studies, 9 and for countless current and future studies that will continue to add to the growing understanding of SARS-CoV-2 and COVID-19 disease.
These training and research opportunities were highly productive and fulfilling for the faculty and students and likely would not have occurred had the medical school not been engaged to provide the PCC with staff. What began as a crisis response grew into an extremely fruitful collaboration both in terms of laboratory operation and science, resulting in new articles authored or assisted by these students. 5 -10 There have been many documented issues with the integration of pathology more fully into medical school curricula. 11 -13 This experience provides ample justification for pathology laboratories to explore avenues to deeper collaborations with medical students.
Pathology Command Center Drawdown
In mid-April, as the census of hospitalized patients at MMC began to decrease, we planned for the discontinuation of the command center. Staffing of the PCC could only be maintained for one additional month since many of the medical students would soon be graduating, while the others would be moving back into their clinical and research responsibilities. In addition, we knew that the PCC dental associates would return to their routine positions when clinics reopened. Because specimen routing protocols had stabilized as a result of some improvement in the reagent supply chain, the decision was made by pathology and infectious disease leadership to transfer the PCC duties back to accessioning staff and pathology customer service. With the continued help of our information technology group, orders for expedited versus standard turn-around-time testing were created, and expedited orders were restricted based on testing location. Emergency department and labor and delivery (L&D) orders could be placed as expedited and were performed using the rapid Cepheid platform, while all other orders would be performed with the standard PCR platforms. Although ED providers had access to both expedited and standard orders, we recommended that standard testing be utilized for patients likely to be discharged. Infectious disease providers could be contacted to place expedited orders for patients outside of the ED and L&D on a case-by-case basis, such as for inpatients requiring emergency procedures. This allowed the volume of rapid tests used to still be a minority, as supplies were still lower. This plan was agreed to by ED and MMC medical directors and implemented on May 18, 2020.
Conclusions
The formation of the PCC in early March at the MMC was an immense benefit to the laboratory and the medical system as a whole. The increased communication between teams, specialization of test routing and customer service abilities, and conservation of laboratory resources allowed the medical system to weather an unprecedented challenge to care delivery. And, in addition, it fostered further education and collaboration between faculty and residents.
The MD-PhD students chosen to staff the PCC gained valuable experience. The students expressed that they never had understood many aspects of diagnostic testing before being immersed in the laboratory. The students also learned important management and collaboration skills, which are translatable outside of the laboratory setting. Finally, the students learned of COVID-19-related research projects in the department, with some working with other faculty in the department on these projects.
Without the formation of the PCC and the engagement of MD-PhD students, specimen routing would have been impossible. Delays in TAT, reagent shortages, and staff burnout would have been measurably worse during an already challenging time. Leadership of many clinical teams have expressed their admiration and appreciation for the work of the clinical laboratory, especially the PCC team. During times of crises, bold collaborative steps can have truly amazing and unanticipated effects. While there is hope that COVID-19 may be relegated to history over the course of this coming year, it is a question of when, not if, the next pandemic will occur. The lessons learned of swift action, communication, and collaboration will be even more important, and the hope is that we might be better prepared for whatever comes next.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
