Abstract
Testing during the COVID-19 pandemic has been crucial to public health surveillance and clinical care. Supply chain constraints—spanning limitations in testing kits, reagents, pipet tips, and swabs availability—have challenged the ability to scale COVID-19 testing. During the early months, sample collection kits shortages constrained planned testing expansions. In response, the University of Vermont Medical Center, University of Vermont College of Medicine, Vermont Department of Health Laboratory, Aspenti Health, and providers across Vermont including 16 area hospitals partnered to surmount these barriers. The primary objectives were to increase supply availability and manage utilization. Within the first month of Vermont’s stay-at-home order, the University of Vermont Medical Center laboratory partnered with College of Medicine to create in-house collection kits, producing 5000 per week. University of Vermont Medical Center reassigned 4 phlebotomists, laboratory educators, and other laboratory staff, who had reduced workloads, to participate (requiring a total of 5.3-7.6 full-time equivalent (FTE) during the period of study). By August, automation at a local commercial laboratory produced 22,000 vials of media in one week (reducing the required personnel by 1.2 FTE). A multisite, cross-institutional approach was used to manage specimen collection kit utilization across Vermont. Hospital laboratory directors, managers, and providers agreed to order only as needed to avoid supply stockpiles and supported operational constraints through ongoing validations and kit assembly. Throughout this pandemic, Vermont has ranked highly in number of tests per million people, demonstrating the value of local collaboration to surmount obstacles during disease outbreaks and the importance of creative allocation of resources to address statewide needs.
Introduction
The novel coronavirus severe acute respiratory syndrome coronavirus 2 has infected over 115 million individuals worldwide with over 2.5 million deaths. 1 The United States demonstrates the highest number of COVID-19 cases, accounting for almost a quarter of the world’s confirmed infections as well as the highest number of total fatalities attributable to COVID-19. 2 Vermont, by contrast, has demonstrated a comparably lower prevalence of COVID-19 3 (Table 1). Coincidingly, the state of Vermont is one of only 9 states that is meeting the COVID-19 testing target, as established by the Harvard Global Health Institutes based on data presented November 1, 2020 4 (Table 1).
Comparison of the United States and Vermont on Select COVID-19 Pandemic Metrics.
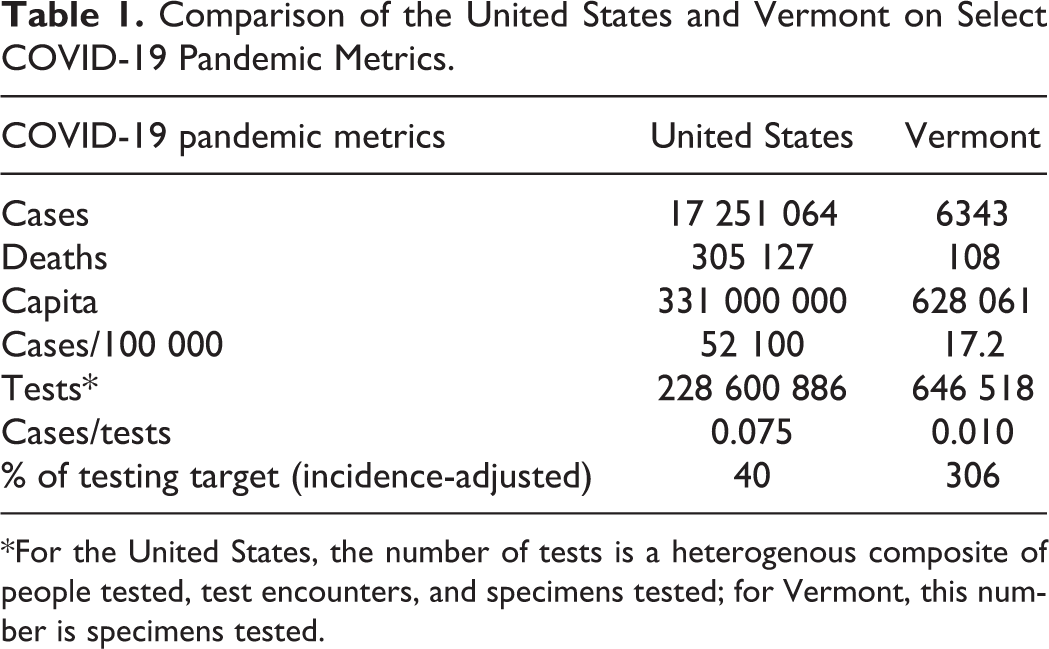
*For the United States, the number of tests is a heterogenous composite of people tested, test encounters, and specimens tested; for Vermont, this number is specimens tested.
Prior to the availability of vaccinations and during the months since they have become available, diagnostic testing has been a primary mechanism to support disease management. 5 Laboratory testing has been instrumental in allowing for detection of virus to identify clinical cases, manage at-risk populations, and identify and manage outbreaks to limit spread. 6
However, while laboratory testing has played a critical role during the COVID-19 pandemic, there have been substantial barriers to scale testing nationwide. One of the most critical barriers to scaling COVID-19 testing has been supply chain resources and management. 7 Although scarcities in health care supplies are well reported for emergencies, natural disasters, and outbreaks, 8 the magnitude, geographic distribution, and time course of this pandemic pose unique challenges. 9
In particular, the availability of preanalytical specimen collection kits has served as a material limitation to increasing COVID-19 testing within the United States. 7 In Vermont, collection kits were quickly identified as a barrier to COVID-19 testing.
In response, we developed a 2-pronged strategy to meet the needs of our state—producing specimen collection kits and managing utilization of kits. In this retrospective, observational study, we identify and assess the impact of these strategies to surmount this barrier to COVID-19 testing in our region. Visibility to the logistical limitations and strategies to overcome these types of barriers may provide insights into planning for future disasters, including pandemics, and enable laboratories to be better prepared for these events.
Methods
We performed a retrospective, descriptive study of operational efforts conducted at the University of Vermont Medical Center (UVMMC) in response to the COVID-19 pandemic. The study period spanned from March 13, 2020, through August 31, 2020. Coincident events in Vermont are depicted in Figure 1 and include school closures beginning as early as March 16, 2020, and the Vermont stay-at-home order issued on March 24, 2020. 10
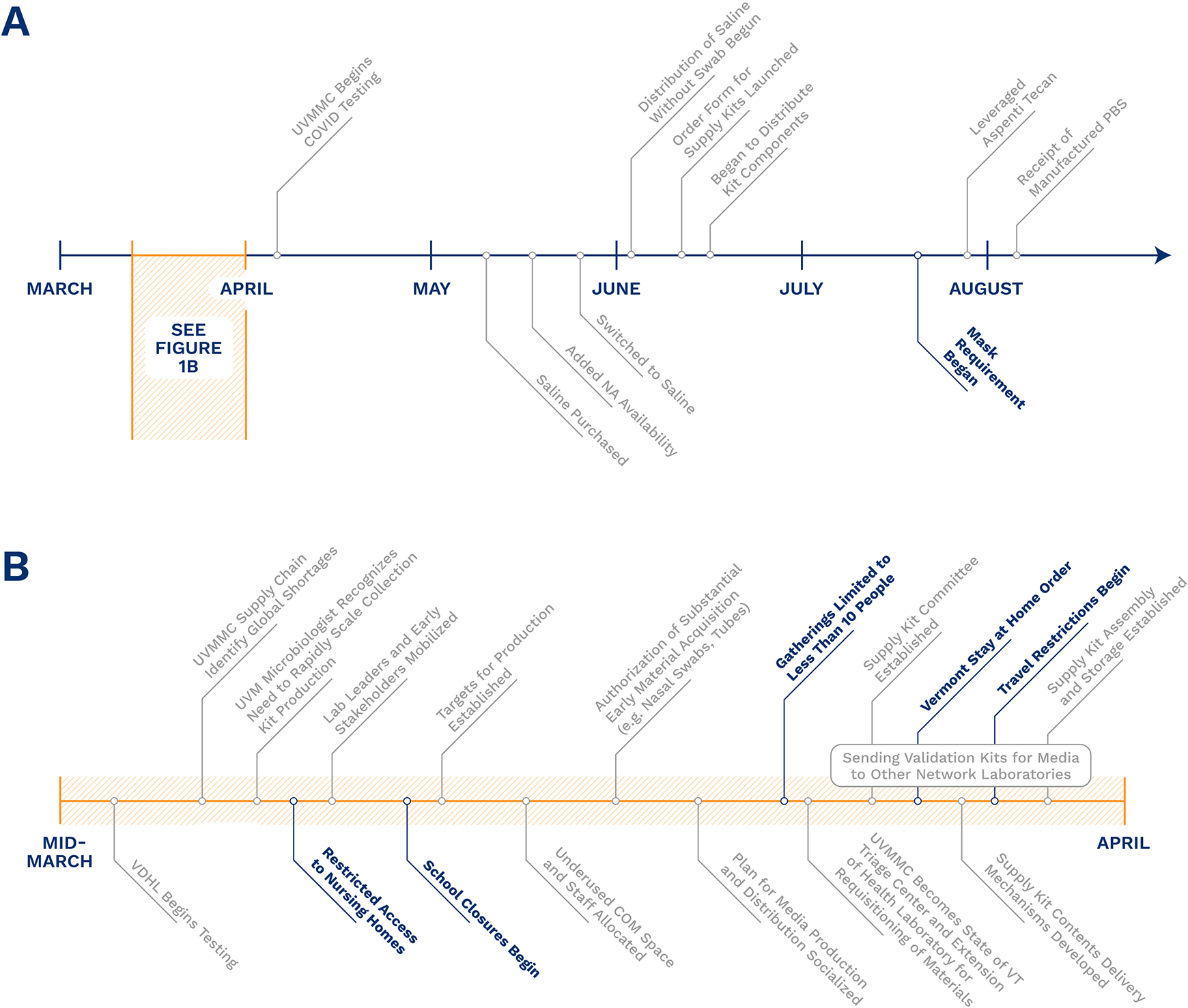
(A) Timeline of specimen collection kit production in response to COVID-19 pandemic: events leading to and resulting in the preparation of COVID-19 specimen collection kits at University of Vermont Medical Center (UVMMC) in conjunction with significant Vermont COVID-19-related events. (B) A focus on events occurring in late March that allowed for the production of COVID-19 specimen collection kits.
This study examined the coordinated work across the UVMMC, the University of Vermont College of Medicine (COM), and Aspenti Health to produce specimen collection kits for COVID-19 viral testing. In addition, we describe the collaboration needed to manage collection kit supplies across the University of Vermont Health Network (UVMHN), an integrated academic health system that includes 6 hospitals (including UVMMC) and a home health and hospice affiliate, serving more than one million residents across Vermont and Northern New York, and 16 area hospitals bridged under the Northeast Community Laboratory Alliance (NECLA), a nonprofit organization supporting community-based hospital laboratories across Vermont and upstate New York 11 and the Vermont Department of Health Laboratory (VDHL).
Operational efforts spanned the clinical operational team, outreach pathology, phlebotomy, microbiology, blood bank, research and commercial laboratories and personnel, pathology leadership, supply chain management, shipping and handling, external couriers, and area hospital laboratory management involving an estimated 70 to 75 individuals, including approximately 3 to 6 individuals per section and approximately 20 individual organizations.
Targeted interviews and follow-up queries (n = 12) reaching each of these sections and/or organizations were conducted from October 6, 2020, to December 17, 2020. As needed, resources used to document events and discussions included individual email communications and digital calendars from these individuals. In addition, a sampling of daily reports (60 total reports, representing an estimated two-thirds of the total) from supply chain management were used to corroborate timeline events, process workflows, and provide insight into collection kit production and distribution. This sampling spanned the full time frame of the study and represented those reports that could be retrieved. This report was disseminated to key leaders and staff across UVMMC daily Monday through Friday beginning in April. Global, national, and statewide statistics were compiled from multiple sources. 1,2,12 -14 Timeline data for Vermont events were retrieved from Vermont’s COVID-19 Response and Resources website. 10
After recognition of global shortages in collection kit availability by the UVMMC Supply Chain, UVMMC microbiology championed the need for rapid scaling of collection kits beginning on March 12, 2020. After mobilization of laboratory leadership the following day, the team developed an early project plan that, with some evolution, would be used from March to June. Iterative modifications of the project continued from June until the end of the study period in August. With the early acquisition of a bulk supply of nasopharyngeal swabs, 15 the production of viral transport media (VTM) and collection kits was the focus of early project plan requirements.
The project was submitted for self-determination as a quality improvement tool through the UVMMC institutional review board. According to the policy defining activities which constitute research at the University of Vermont and UVMMC, this work met criteria for operational improvement activities exempt from ethics review. Analysis was based on available data from inventory management and publicly available datasets. Timelines and processes were mapped during interviews and triangulated with subsequent discussions and data from UVMMC Supply Chain. The study’s time span from March to August was based upon the initial penetration of COVID-19 through a point of relative stabilization of collection kit supplies in our region. Verification of media types was performed on the Panther Fusion Assay (Hologic, Inc). Spiked samples were generated using ATCC strains or SeraCare for SARS-CoV2 to one dilution above the known limit of detection of the assay.
Report data were analyzed in Python using Pandas; figures were created in Matplotlib. COVID-19 pandemic metrics for cases per test were based on previous calculations from The Harvard Global Health Institute and COVID Exit Strategy. 14,16
Results
Early plans to produce specimen collection kits included the need to develop target levels of kit production for project success; recognition that specimen collection kits would need to be produced in-house given early limitations in commercial availability; specification of the product; resource requirements; and the recognition of the need for partnerships (Table 2).
Early Project Determinations to Support Statewide Collection Needs.
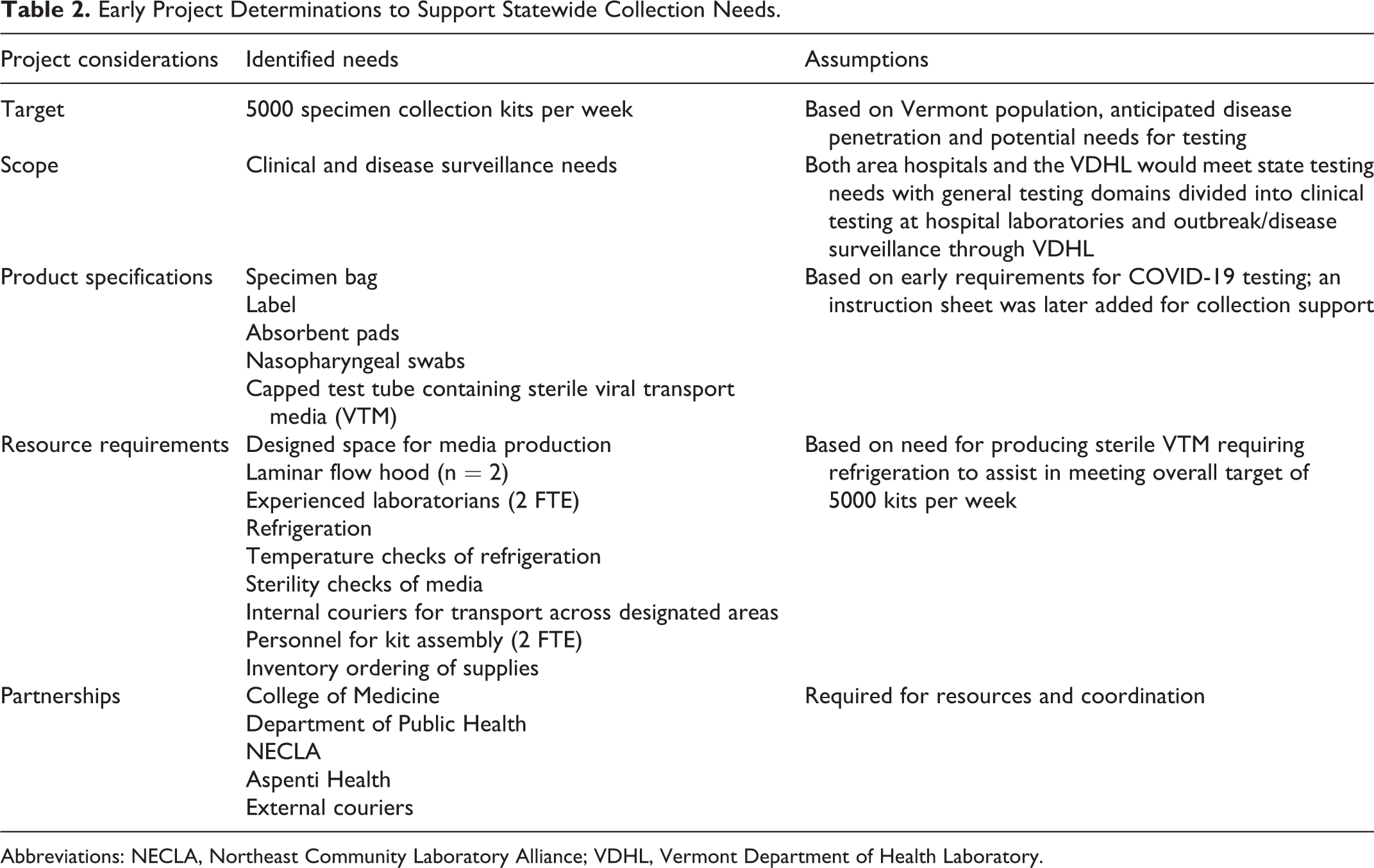
Abbreviations: NECLA, Northeast Community Laboratory Alliance; VDHL, Vermont Department of Health Laboratory.
Due to coinciding social distancing effort in Vermont and at the University of Vermont, available space and personnel within the COM, meeting most of the requirements for the project, was rapidly identified. The specification of quality-controlled refrigeration for storage of VTM necessitated inclusion of the UVMMC blood bank in initial plans where supplies could be stored prior to assembly (Figure 2). The microbiology and phlebotomy teams were required to support demonstration of VTM sterility as well as for kit assembly. Collaborative partnership was as essential for collection kit production and the designation of University of Vermont as a State of Vermont Department of Health Laboratory for the purposes of requisitioning materials (Figure 1) on March 20, 2020, supported this. Locally based external couriers worked collaboratively to modify transport protocols (such as increased refrigerated supply transport for VTM, reducing the number of drivers who used each car, and decontaminating the drivers space before and after each shift) and to shift schedules to accommodate delivery to Vermont Department of Health Laboratory (VDHL), area hospitals, nursing homes, provider offices, mobile sites, and pop-up collection sites throughout the state. Early efforts required an estimated 7.6 full-time equivalent (FTE) for daily kit production. Early production efforts including the transient storage strategy for assembled kits and a poster that provided instructions in lieu of a formal standard operating procedure are shown in Figure 3.
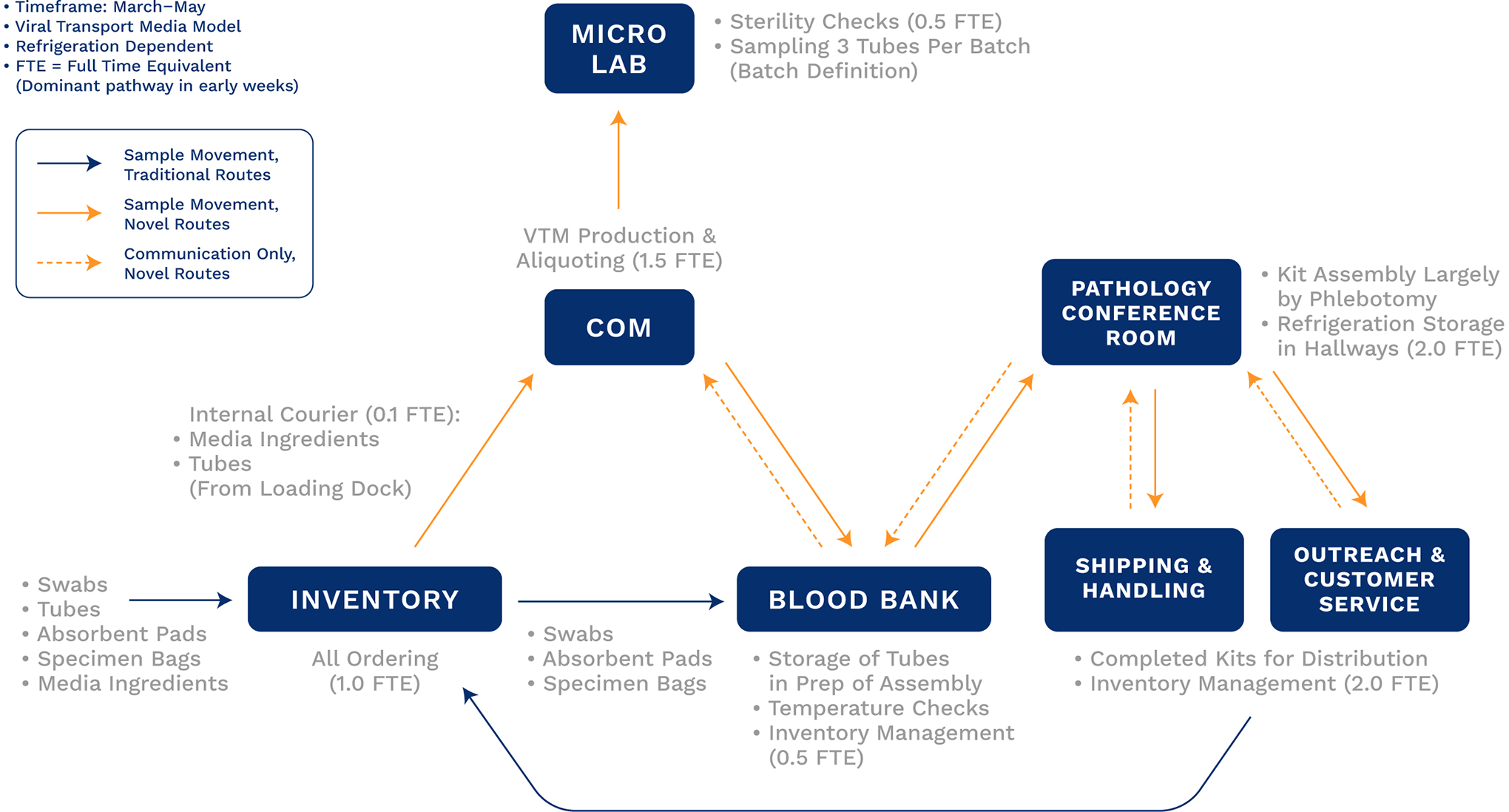
Process maps for specimen collection kit production to include the steps to produce and distribute specimen collection kits during use of viral transport media.

COVID-19 specimen collection kit assembly within the pathology conference room. (A) A photograph of specimen collection supplies and assembled kits within the first few months of COVID-19 pandemic response. (B) A photograph of COVID-19 specimen collection kit assembly instructions. This poster, which was housed at the site of assembly, was used in lieu of protocol development.
Kit production became simpler as the COVID-19 pandemic evolved (Figures 4 and 5). The critical shift was the ability to reduce reliance on VTM and adopt use of sterile phosphate-buffered saline (PBS). The COM continued to dilute and filter PBS when 10× was available (not depicted) but this rapidly was transitioned to the clinical laboratory when 1× PBS was more readily accessible (Figure 4). The ability to rely on PBS due to internal validation of the use of PBS in testing eliminated the need for refrigeration, relieving the dependency on UVMMC blood bank for inventory management and temperature checks and the need for hallway-based refrigeration of kits after assembly. In addition, fewer total FTE were needed (6.5 FTE; Figure 4). In early May, anterior nares swabs (NA) were introduced for use in skilled nursing facility patients. As supplies increased, this was extended to pediatric patients and hematology/oncology patients. Later, restrictions were removed as supplies became more available. Reliance on NA swabs also reduced the need for collection in a negative pressure space, which had been used to limit aerosolization exposure to collectors.
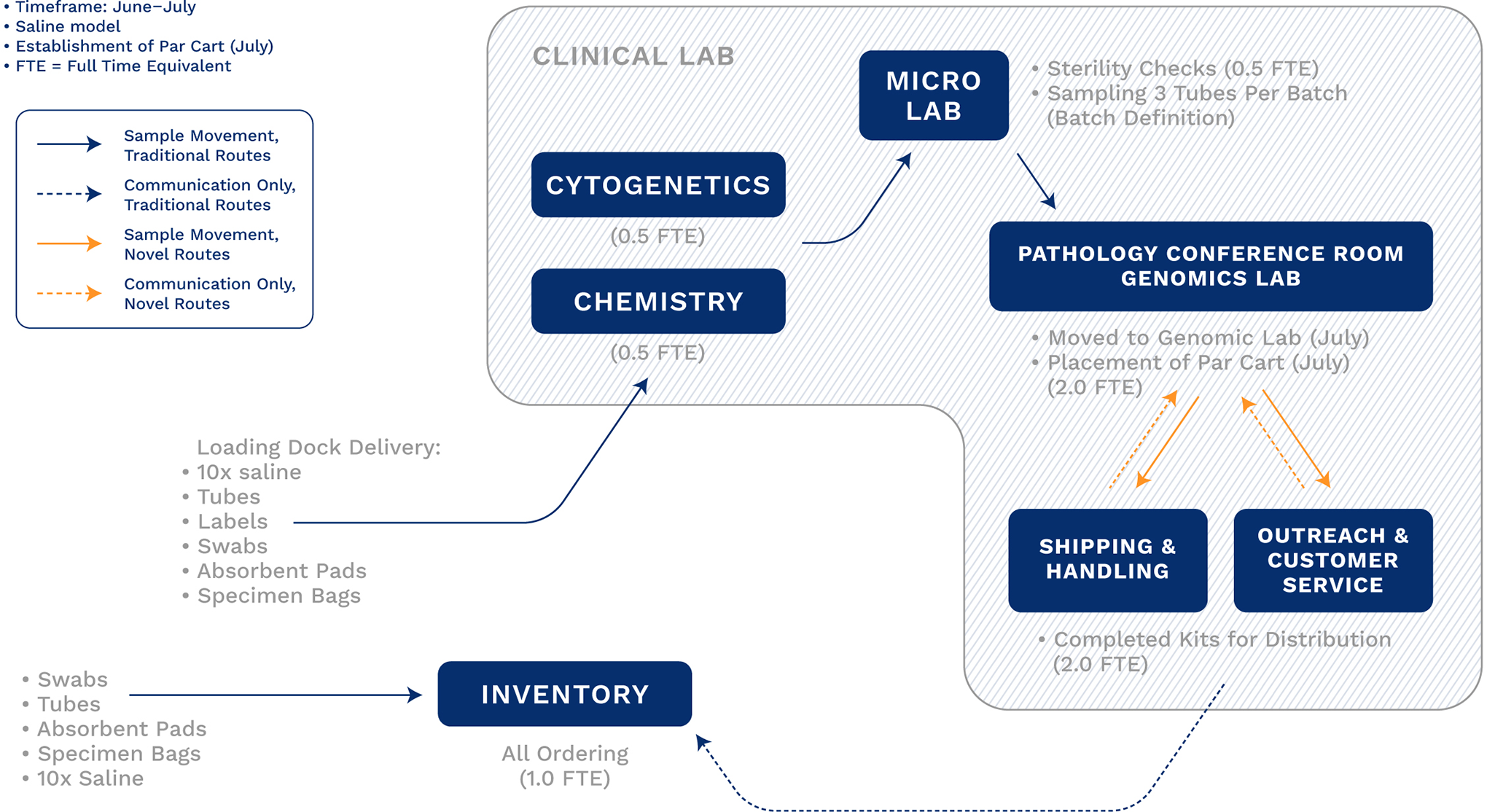
Process maps for specimen collection kit production to include the steps to produce and distribute specimen collection kits during use of saline.
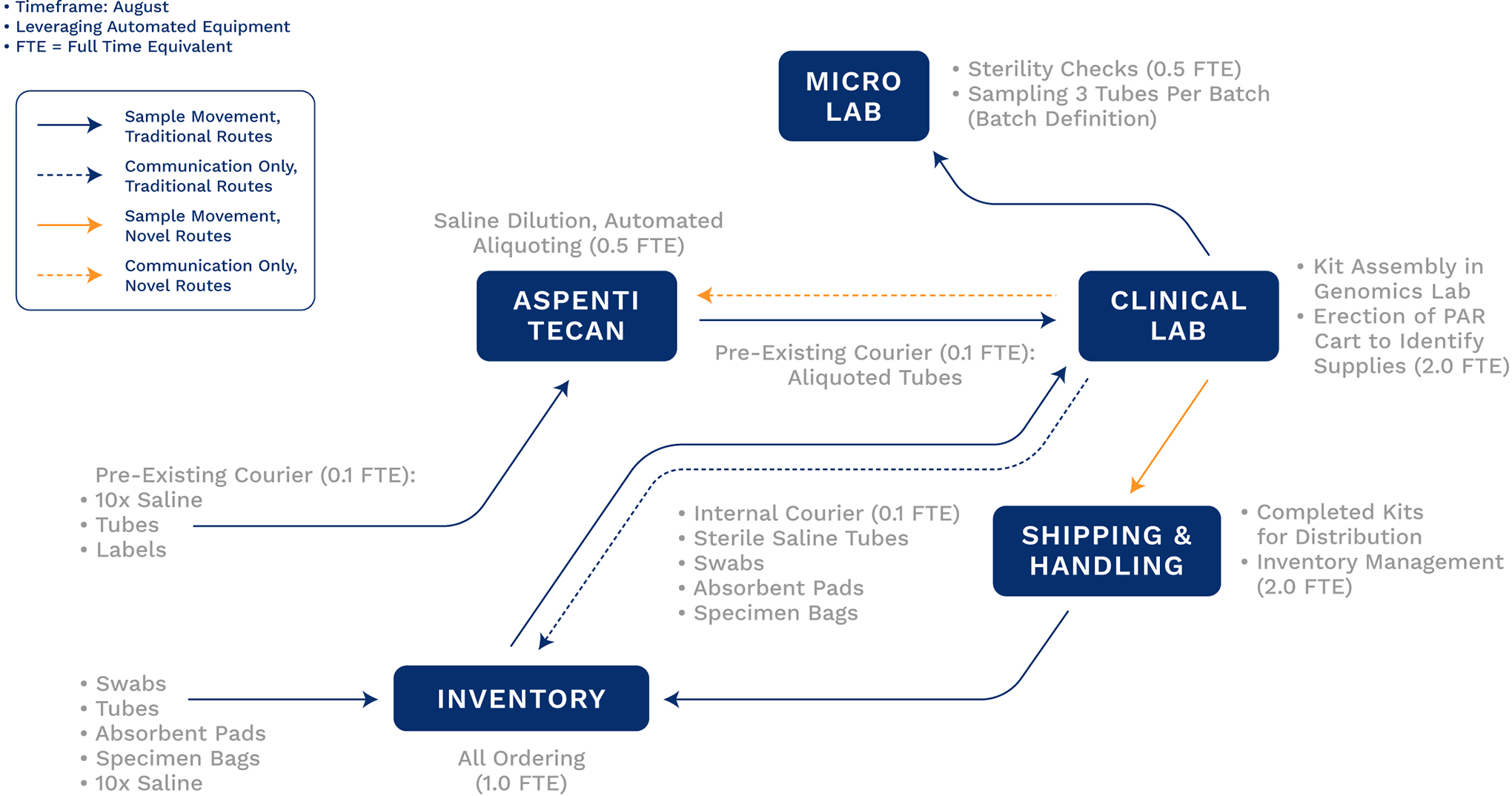
Process maps for specimen collection kit production to include the steps to produce and distribute specimen collection kits during use of externally sourced automated liquid handling system.
Due to the need for expediency, targeted verifications of various media types, including locally produced VTM and PBS, were performed both for the detection of SARS-CoV2 and all other virus types tested in our laboratory near the limit of detection (n = 39, including samples in triplicate for 6 media types spanning SARS-CoV2, herpes simplex virus [HSV], Varicella zoster virus [VZV], influenza, types A and B, respiratory syncytial virus, adenovirus, metapneumovirus, rhinovirus, and parainfluenza viruses types 1-4). Determinations were evaluated qualitatively and all samples matched expected values. Although daily communications among the Collection Kit Committee enabled visibility to the on-hand specimen collection kits and allowed for continuous adjustments to production and purchasing, there is some suggestion that a secondary pathway of distribution through the laboratory and customer service directly may have resulted in an underestimation of distribution efforts during the early months of reporting. A sampling of microbiology sterility checks suggest the target production goals were met (data not shown). This operational constraint was surmounted with the adoption of a par cart in July—an inventory management method to determine a minimum amount of stock that allows for replenishment to a predefined level. With the par cart, UVMMC Supply Chain had improved visibility into supply kit levels were able to support defined minimal and maximum levels and institute bar code scanning more readily.
Shifts in August included support from a local commercial laboratory, Aspenti Health, to aliquot high volumes of sterile saline into tubes using automated liquid handling equipment (Figure 5), which saved substantial time in aliquoting (previously requiring 2 staff 4 hours per day each, reducing the total FTE to 5.3; Figure 5), yielding higher throughput and a more ergonomic solution for staff. In the last 6 weeks of the study period, collection kit supplies were no longer seen as a testing barrier unless a substantial surge were to arise (Figure 6).
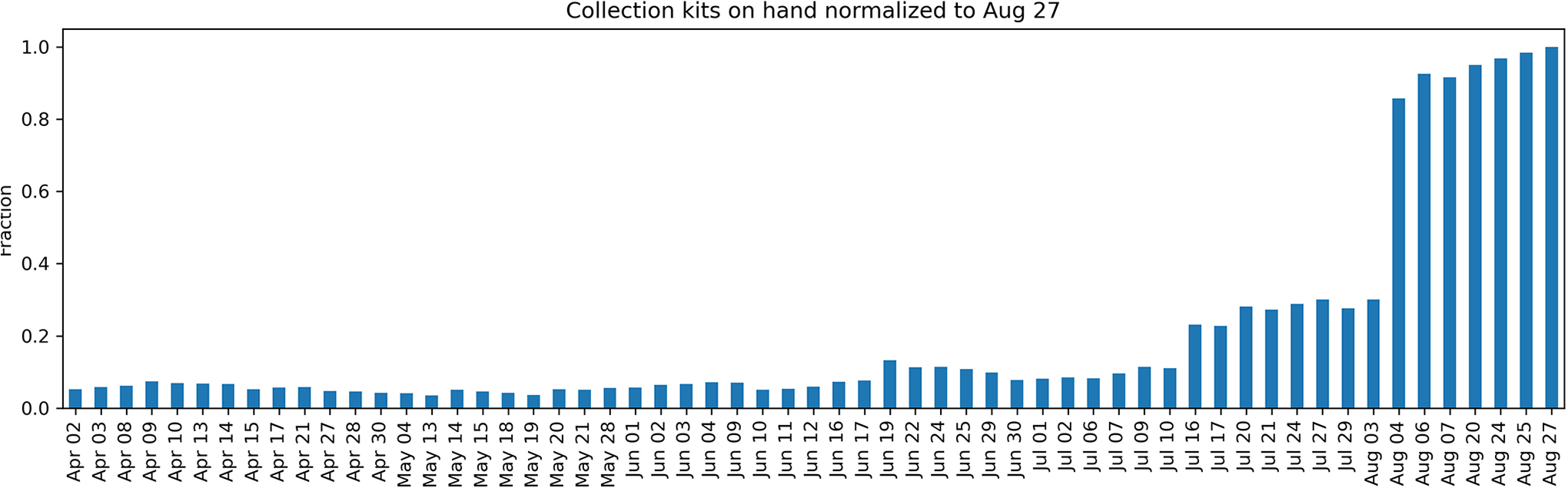
On-hand distribution specimen collection kit normalized by the last study date. The distribution delineates between New York and Vermont hospital distribution. A minor portion of specimen collection kits were housed at affiliated New York hospitals.
On-hand specimen collection kits inventory grew during this study period (Figure 6). The fraction of collection kits not requiring any media production or assembly decreased linearly over the course of the study. As UVMHN supports hospitals throughout Vermont and upstate New York, the median proportion of on-hand New York collection kits represented 7% of the overall number of kits.
Although the initial distribution of specimen collection kits was externally sourced, in-house sources soon became the predominant pathways to supply collection kits across the region (Figure 7). These were initially reliant on VTM and later on saline sources for PBS production. Then, as the summer progressed, externally sourced kits became more readily available and reemerged as the predominant strategy. The shifting reliance of different types of media is reflected in inventory management data with daily updates to on-hand supplies of kits through the Collection Kit Committee (Figure 7).
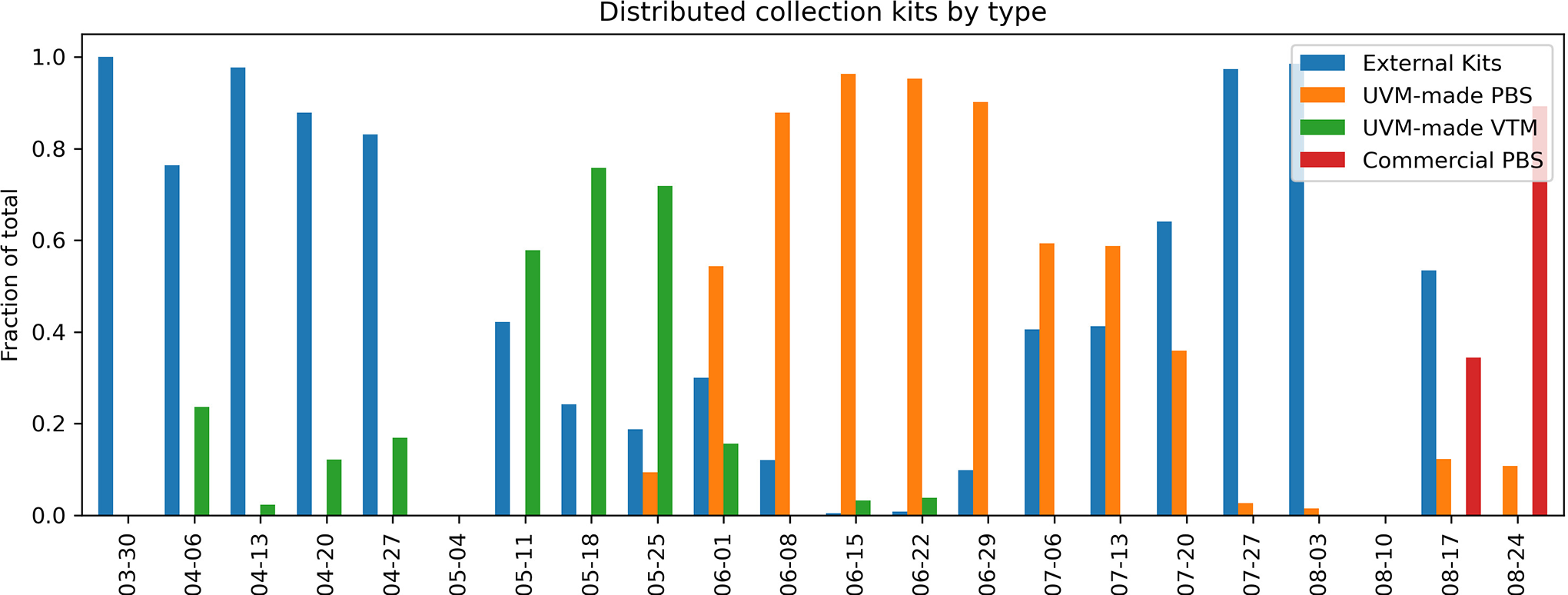
Distributed collection kits by type: external media kits (those with and without swabs; blue); in-house, College of Medicine (COM)-made viral transport media (VTM; green); in house, UVM-made phosphate-buffered saline (PBS; orange); and commercial PBS (red).
To support statewide testing management, the UVMMC was named the State of Vermont triage center for prioritization of COVID-19 testing. The UVMMC, the UVMHN, the VDHL, and 16 area hospitals bridged under NECLA developed a collaborative approach to respond to the COVID-19 pandemic and to surmount supply chain and operational barriers. Ordering providers and hospitals agreed upon just-in-time acquisition of supplies and collection kits, allowing for only 2 to 3 days’ worth of on-site kits and limiting multiple stockpiles across the state. Validation kits for VTM were distributed across the network (Figure 1B) to accommodate the use of in-house VTM development. Active management and coordination were required for judicious use of anterior nasal swabs in particular. Given their limited availability, their distribution was accompanied by instructions on how and when this limited resource could be used. In later months, specimen collection kit components were distributed to larger area hospitals, allowing for on-site, decentralized kit assembly of specimen collection kits (Figure 1A). A laboratory COVID-19 operational team, representing laboratory leadership and including inputs from clinical service needs and the VDHL, informed changes to the approach. In addition, network-wide needs were directed through a consortium of UVMHN Chief Medical Officers.
Ongoing communication efforts were required for coordination of inventory management and utilization. The UVMMC laboratory leadership met once to twice daily with VDHL leadership and, through NECLA, laboratory directors from 16 area hospitals met approximately every 2 to 3 days for the first 2 to 3 months and ad hoc thereafter. The UVMMC-based clinical operations team met multiple times per day in the early weeks, scaling back to once daily including weekends through May. For the remainder of the study period, this team met daily Monday through Friday. Across these teams, topics discussed included reference laboratory choices and specimen triaging, testing volumes and turnaround times, operational obstacles, supply chain management, and validations. Statewide leadership discussions also supported supply chain utilization management and included: laboratory representation on statewide emergency management calls, UVMMC’s pandemic response center’s discussions, and statewide infectious disease committee meetings.
Discussion
By producing in-house collection kits and managing kit distribution and utilization, the UVMMC and collaborating partners across the state were able to surmount a critical barrier to scaling testing during the early months of the COVID-19 pandemic. Limitations in testing options in the first months posed particular challenges including the need for quality-controlled refrigeration and the development of new pathways for internal transports of supplies and product. Decentralized inventory management became an increasing challenge over the following months and necessitated conversion to more traditional inventory management strategies such as par carts, establishment of minimums and maximums, and the use of bar coding. The unique features of the clinical laboratory positioned the laboratory to provide a central role in the preanalytical requirements of COVID-19 testing and included substantial internal collaboration across multiple laboratory divisions and institutions, outreach partnerships, routinely performed quality checks such as for refrigeration and sterility, and visibility to evolving testing requirements and leadership.
As is often observed during disease outbreaks and natural disasters, 15 shifting of resources during the pandemic was a particularly effective operational strategy. This included reassigning staff to COVID-19 pandemic-related activities. Phlebotomists, 1 laboratory educator, 1 cytogenetics laboratorian, and 3 research colleagues were mobilized to support specimen collection kit production. In addition, instrumentation that would have been dormant or underutilized was leveraged, including research laboratory hoods, water filters, and automated liquid handling equipment. Workforce and resource planning are paramount in the setting of disease outbreaks and natural disasters. 17
Leveraging preexisting partnerships both internal to COM’s research laboratories and external organizations (such as NECLA, Aspenti Health, and the VDHL) was essential to the success of this project and the ability to scale COVID-19 testing across the state. Internally, the COM’s research laboratories allowed for scale, available human and equipment resources, as well as thought leadership in response to the changing testing landscape. Externally, the long-standing partnerships through NECLA bridged institutional barriers to drive collaboration in laboratory medicine. The strength of this partnership was foundational both by enabling UVMMC and to leverage preexisting infrastructure such as common ordering and reporting mechanisms but also to build on the trust needed to commit to as-needed supply ordering.
External relationships with NECLA partners, reference laboratories, Aspenti Health, and VDHL have strengthened during these collaborations, bolstering relationships, and trust. Continuous coordination was essential, not only to secure supply chain needs but also to ensure transport medium and swab type would be valid at all testing locations. Continuous communication was essential to ensure that any validations for other viruses, such as HSV, VZV, and other respiratory viruses, could be completed prior to the distribution of these collection kits.
In particular, the partnership with VDHL continues to be a critical collaboration beyond this project. The importance of these types of partnerships have been noticed in prior health emergencies. 18,19 Similar to previous studies, this type of collaborative endeavors is not only useful for resource and logistical management but also for strengthening trust and social capital. 18
Despite the importance of the operational efforts, this study has a few limitations. Because COVID-19 response was the highest priority, concurrent cataloging of events and data were deemphasized. This required a retrospective determination of the sequence and extent of specimen kit production and utilization. For example, while on-hand collection kit data were assiduously tracked through UVMMC Supply Chain and updated daily at the Collection Kit Committee, the secondary pathway of distribution through the laboratory and customer service directly may have resulted in an underestimation of distribution efforts.
The COVID-19 pandemic posed unprecedented supply chain concerns that have compromised response strategies. 7 For example, nasopharyngeal swabs could only be acquired from 2 companies initially. 7 Vermont’s public health leadership identified supply chain barriers as the largest single threat to outbreak response and credited collaborative efforts to produce collection kits as having been a factor to slow viral spread across the state. 20 The need for specimen collection kits was one of the most significant obstacles faced in our region initially. However, as the pandemic progressed, other supply chain constraints, predominated. Supply chain constraints have spanned testing reagents, testing kits, swabs, and other consumables. In addition, both locally and nationally shortages in laboratory expertise to perform appropriate testing have been seen. 21
Due to a rapid laboratory response leveraging resources and partnerships, UVMMC was able to support statewide specimen collection kit needs. The ability to scale testing to support disease surveillance as well as clinical management is an essential component of disease outbreak response. 5 Individuals involved in this effort likened it to creating a new product line for UVMMC. This endeavor has provided substantial insights for future outbreaks that might require in-house solutions.
Although the specific supply chain requirements for future outbreaks or disasters may not be predictable, there are several lessons that can be identified from this effort that may inform future outbreak and natural disaster responses. First, rapidly forecasting needs is critical for the success of any project but particularly during a pandemic where competing priorities can prevail. Second, creative problem-solving to leverage resources outside of traditional hospital-based laboratory settings can be effective. Third, newly developed processes and systems are more vulnerable to operational limitations than reliance on well-honed systems. Innovative strategies may be very effective but should be managed with an awareness of their limitations. As noted previously, 7 redundancy in solutions with contingency plan management is critical for future responses. Finally, the importance of collaboration cannot be overstated. Partnership and collaboration can enhance efforts to problem solve and creatively surmount obstacles.
The clinical laboratory has played a critical role in COVID-19 response both in the state of Vermont and nationally. Broad scale testing has been essential to identify public health threats and contain COVID-19 transmission. 5 In addition, as suggested by this study, laboratory management of preanalytical variables can have a profound impact on our collective ability to response to future outbreaks.
Footnotes
Authors’ Note
Jill Warrington, the corresponding author, drafted the manuscript, coordinated with the Institutional Review Board at her home institution (the University of Vermont Medical Center), and prepared the document for submission. Vanessa Clark, Lauren Risley, Linda Coulombe, Tania Hong, Andrew Goodwin, Jessica Crothers, and Christina Wojewoda led the quality improvement project with development of triaging systems and mechanisms for developing supply kits. All provided input, including review of emails, calendars, project management material, and/or interviews, into the methods and results of this project as well as critically reviewed the manuscript. Michael Lewis and Mark Fung provided material input to the study design and manuscript writing. Gregory Warrington provided substantial contributions to the data abstraction, analysis, and figure generation. The datasets generated and/or analyzed during the current study are not publicly available but are available in an aggregated, de-identified from the corresponding author on reasonable request. According to the policy defining activities which constitute research at the University of Vermont/University of Vermont Medical Center, this work met criteria for operational improvement activities exempt from ethics review.
Acknowledgments
The authors want to thank their respective institutions for recognizing the importance of scholarship and supporting the efforts required to produce this study and in particular Debra Leonard, MD, PhD, and Chris Powell for championing thought leadership. The authors also want to thank the UVM COM staff including Beth Kirkpatrick, MD, Marya Carmolli, Cassandra Ventrone, for their support and dedication in the production of VTM early in the pandemic as well as their input and operational expertise. The authors also want to express gratitude to Andrew Dernavich from BFA Graphic Design for his support in Figure graphical design.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Vanessa Clark and Lauren Risley are salaried by Aspenti Health, a laboratory focused on population health management for Substance Use Disorders. Vanessa Clark holds a small portion of stock options (<$1000 in value) at Aspenti Health. Jill S. Warrington carries a nonsalaried position as Chief Medical Officer at Aspenti Health where she holds a small portion of stock options (<$1000 in value). She also is a member of the community advisory council at Blue Cross Blue Shield of Vermont. Mark Fung serves as an ad hoc consultant for Cerus Corporation and Biocogniv. He also receives honorariums from Grifols Corporation for educational presentations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research for ninth author is partially supported by Simons Foundation Collaboration Grant for Mathematicians 429570. The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
