Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040. 1
Keywords
Primary Objective
Objective H2.5: Evaluation of the Bleeding Patient: Describe how to evaluate a bleeding patient with a hemorrhagic disorder, and explain how the history influences testing, including the uses and limitations of screening PT, PTT, and platelet counts.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic H: Hematology; Learning Goal 2: Diagnosis and Management of Coagulation Disorders.
Patient Presentation
A 17-year-old Bengali American boy was brought into the primary care clinic by his mother for evaluation of a large hematoma. Two days ago the boy had attended soccer try-outs in high school and after heading a soccer ball, the patient developed a “goose-egg” on his forehead that has not decreased in size. The patient reports no loss of consciousness or pain, but reports a slight headache. He also does not report a history of easy bleeding or bruising, taking aspirin or non-steroidal anti-inflammatory drugs (NSAIDs), excessive bleeding after minor cuts or dental care, nosebleeds, swollen joints from trauma, fatigue, shortness of breath, or trouble breathing on exertion.
The patient has no prior surgeries, no allergies, and is currently not taking any medications. The patient’s mother immigrated to the United States from the People’s Republic of Bangladesh in 1996 and he reports a maternal uncle with an unknown bleeding disorder had passed away from an HIV-related tuberculosis infection in the early 1990’s due to a plasma transfusion-related HIV infection.
The patient’s vital signs are within normal limits. On physical examination there is no conjunctival pallor, however a 5-cm × 8-cm raised, tender ecchymosis on the patient’s forehead is noted, and bilaterally warmer than normal, stiff knees, which the patient attributes to his initial day of training. The patient was sent for laboratory testing and discharged with acetaminophen and ice packs as needed.
Diagnostic Findings, Part 1
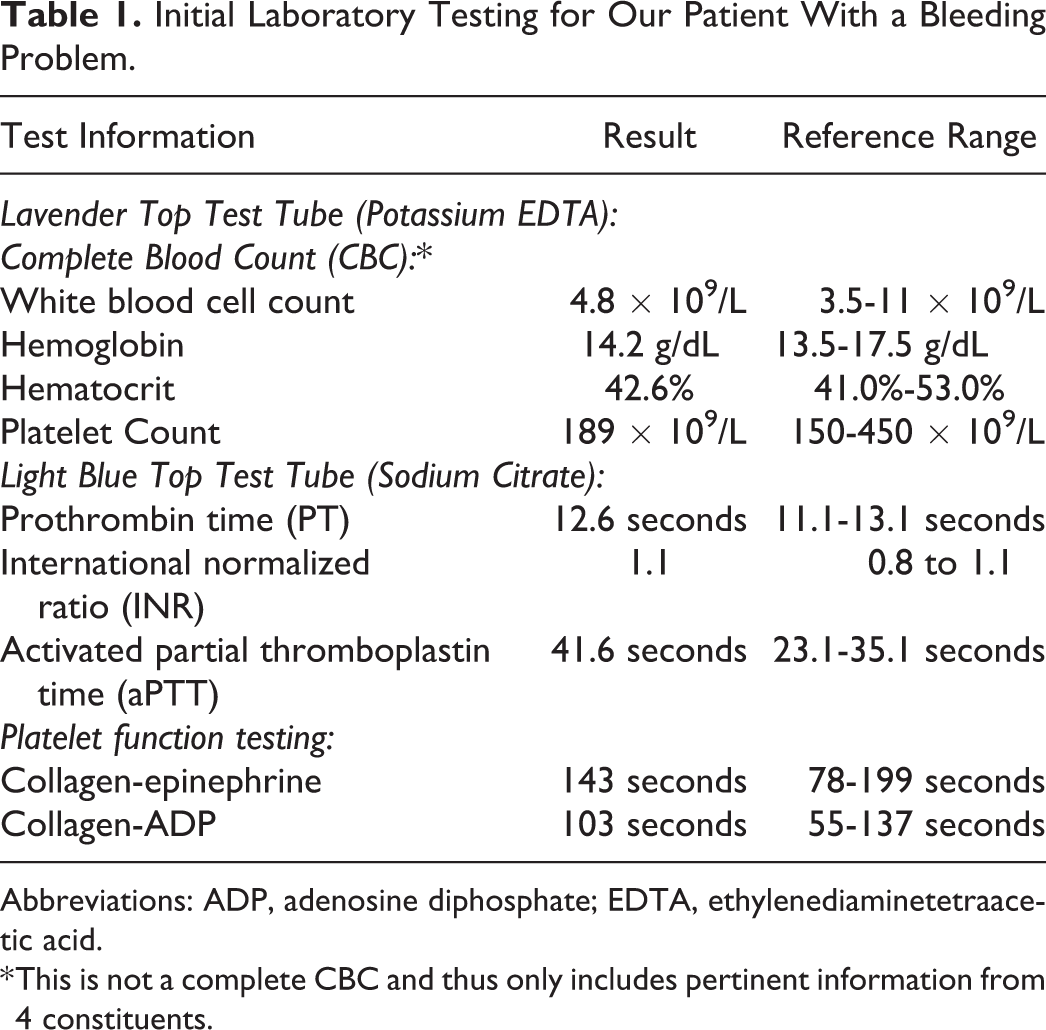
The initial laboratory testing for the patient are given in Table 1. 2 Standard reference ranges are given with the patient’s results.
Initial Laboratory Testing for Our Patient With a Bleeding Problem.
Abbreviations: ADP, adenosine diphosphate; EDTA, ethylenediaminetetraacetic acid.
* This is not a complete CBC and thus only includes pertinent information from 4 constituents.
Questions/Discussion Points, Part 1
Given the Clinical History, What Is a Broad Differential Diagnosis?
The patient has a traumatic hematoma, stiff, warm knees after vigorous activity, and a family history of a maternal uncle with an unspecified bleeding disorder. Laboratory values are all within normal limits with the exception of activated partial thromboplastin time (aPTT; also called PTT), which is prolonged. Since aPTT can be used for screening deficiencies/nonfunctioning proteins of the intrinsic and common pathways of the coagulation cascade, for our male patient, most likely on the differential includes hemophilia A or hemophilia B/christmas disease. Hemophilia C is less likely; hemophilia C is more prevalent in Ashkenazi Jews/Israeli Arabs/Iraqi Jews than in other patient populations. With a negative family history for autosomal dominant inheritance, von Willebrand Disease (vWD) is less likely. If the patient were malnourished, a vitamin K/vitamin C deficiency would be included, and with the patient’s age and medication history, even less likely liver disease or a heparin-induced coagulopathy. 3
What Further Testing Is Indicated in This Patient?
The patient’s aPTT is abnormally prolonged, but prothrombin time (PT) is within normal limits. A prolonged PT illustrates problems with the common and extrinsic pathways—if both PT and aPTT were prolonged, a common pathway problem would be more likely. Thus, to further characterize the patient’s prolonged aPTT, a mixing study would be warranted along with possibly ordering factor assays.
Diagnostic Findings, Part 2
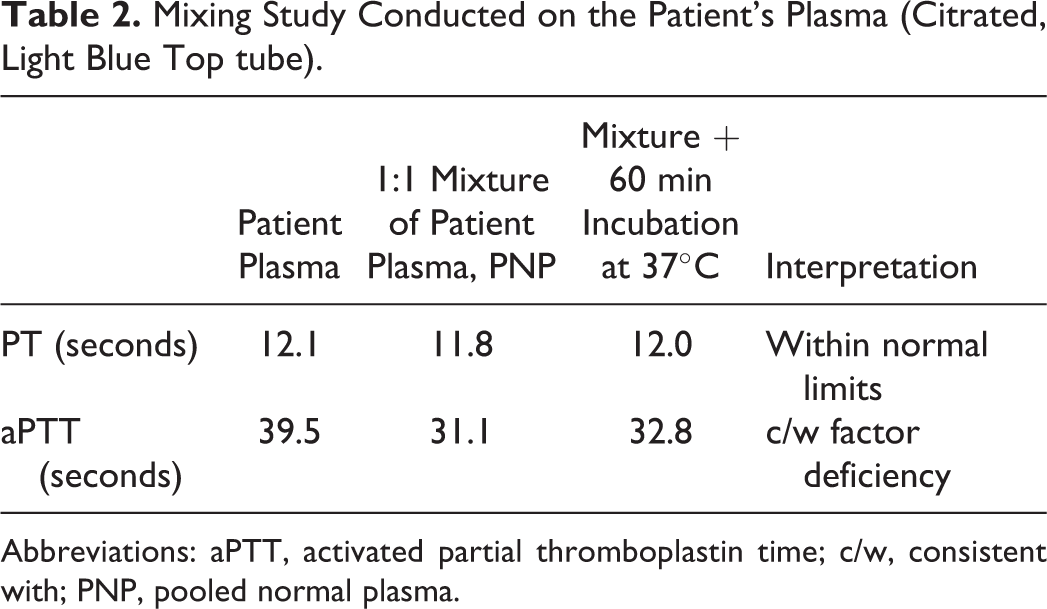
Mixing study and factor assays for patient A are given in Tables 2 and 3, respectfully. 2 Factor assays for factors VIII (most likely), IX, or XI (less likely) would be of concern if the mixing study rules out a lupus anticoagulant/progressive inhibitor.
Mixing Study Conducted on the Patient’s Plasma (Citrated, Light Blue Top tube).
Abbreviations: aPTT, activated partial thromboplastin time; c/w, consistent with; PNP, pooled normal plasma.
Factor Assays Conducted on Citrated Patient Plasma.

The patient was referred to the hematology/oncology clinic and was seen 1 week later where the hematologist drew labs, administered 150 μg of Stimate (DDAVP, or desmopressin) to the patient intranasally in each nostril and redrew labs 120 minutes later. Results are given in Table 4. 2
Hematology/Oncology Clinic Laboratory Findings for Factor VIII Initially and 120 Minutes After DDAVP Administration.

Abbreviations: DDAVP, 1-Deamino-8-D-Arginine Vasopressin.
Questions/Discussion Points, Part 2
What Is the Most Likely Diagnosis in This Patient?
The patient most likely has hemophilia A, which is a qualitative/quantitative deficiency in factor VIII. Hemophilia A is an X-linked recessive disease which affects males and homozygous females. This explains why his mother is asymptomatic, but her brother is reported having had a bleeding disorder. A pedigree is given in Figure 1. Desmopressin can cause the release of von Willebrand Factor (vWF), which indirectly increases factor VIII via stabilization. Factor VIII has a half-life of 2.4 hours when unbound; vWF-bound factor VIII has a half-life of 12 hours. 4
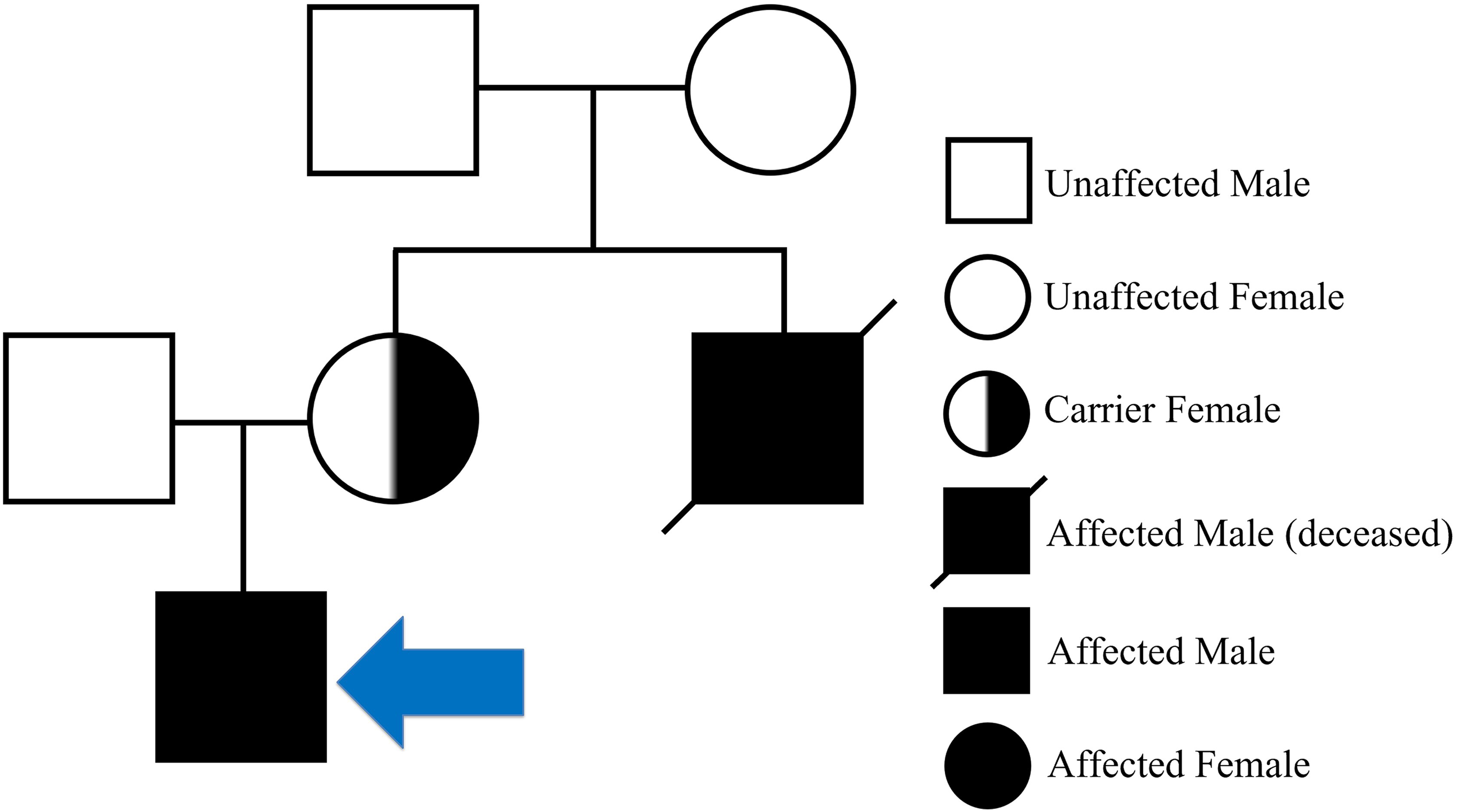
Patient’s pedigree which is typical of a pedigree of a patient with hemophilia. The blue arrow indicates the patient in this case.
Hemophilia patients can present with deep tissue bleeding, including spontaneous hemorrhages in joints, which begin with stiffness, increased temperature, and later decreased range of motion and swelling. Laboratory evaluation yields a normal PT, abnormal aPTT, mixing study that corrects immediately and after 1 hour of incubation, and decreased factor VIII assay activity (<1% in severe, 2%-5% in moderate-severe, and 6%-50% in mild disease). 4 These patients respond to vasopressin, which causes release of factor VIII, and are normally treated with recombinant factor VIII. One possible complication of treatment is the development of antibodies to factor VIII, which appears on a mixing study as a progressive inhibitor (immediate normalization of aPTT but prolongation after 1 hour). 5 Of note, hemophilia patients who received factor VIII concentrates before 1985 were at high risk of parenteral transmission of HIV and developing AIDS, which occurred in the patient’s maternal uncle. 4
What Is the Best Way to Evaluate a Patient With a Possible Bleeding Diathesis?
A detailed history and physical examination are essential in the initial evaluation of a bleeding patient. The major causes of bleeding can be categorized into platelet plug formation disorders and coagulation clotting factor disorders.
Identification of age and sex may clue providers into hemophilia (male, usually presenting at infancy or childhood). A detailed history of present illness should include questions about platelet plug formation and vWD-related symptoms (immediate bleeding/bruising with trauma, small pinpoint bruising, gingival bleeding, heavy menses and iron deficiency anemias, nosebleeds), along with coagulation defect-related symptoms (delayed recurrent bleeding, joint, muscle, and deep tissue bleeding, large bruises). Traumatic events such as surgeries, dental procedures, or other impact-related trauma will help characterize the bleeding. Recent viral infections hint at immune thrombocytopenic purpura. A past medical history of cancer/hematologic malignancy, cardiopulmonary bypass (thrombocytopenia/platelet function disorder) may illicit causes of bleeding. Family history of bleeding may be instrumental in making the diagnosis (males primarily in hemophilia). 3 Review of medications (β-lactam antibiotics, NSAIDs, aspirin, anticoagulants like clopidogrel, coumadin or heparin, corticosteroids, fish oil, valproic acid, or selective serotonin uptake inhibitors) may also help identify a cause of bleeding problems. Social history of malnutrition, heavy alcohol intake, or physical abuse may become key findings as well. 3
On physical exam, visualization of mucocutaneous bleeding and pinpoint petechiae suggest a platelet disorder. Hemarthroses or large hematomas point toward a coagulopathy of clotting factors, with lymphadenopathy and splenomegaly increasing suspicion for malignancy. Splenomegaly after infection may be due to a postviral immune thrombocytopenic purpura. Hepatomegaly and jaundice hint at liver failure. 3,5
How Does the Patient History Influence Ordering Diagnostic Laboratory Testing?
When evaluating patients for possible bleeding disorders, the patient history is key to ordering the most appropriate tests to confirm the suspected diagnosis. In general, PT/international normalized ratio (INR)/PTT, complete blood count (CBC) with differential, and platelet function assays are commonly ordered during the initial evaluation of a bleeding patient. Prothrombin time, aPTT, thrombin time, and fibrinogen are best for screening if history and physical exam are suggestive of a clotting factor disorder/coagulopathy. If a platelet plug formation disorder is suspected, platelet function assays and CBC/diff (which includes platelet count and manual differential) are excellent screening tests. 3,5
What Are the Uses and Limitations of Common Diagnostic Tests Used to Evaluate a Bleeding Patient?
Usage of PT and aPTT as screening tests are key steps in the initial diagnostic laboratory evaluation of a bleeding diathesis. Prothrombin time is used to evaluate the extrinsic pathway (factor VII) and the common pathway (factors X, V, II, and I), with results recorded in seconds. For patients on heparin, this test counters any possible heparin anticoagulant effects via usage of protamine sulfate, a low-molecular-weight compound extracted from salmon sperm that neutralizes heparin. 6 One limitation of PT is the reagent variability, which, by company and region, results in different reference ranges.
International normalized ratio is utilized to standardize PT results worldwide. Different testing platforms use different phospholipid reagents, some of which are better at initiating the coagulation cascade than others. The assay’s tissue is evaluated and compared worldwide among vendors; the resulting ratio calculated is the INR. It is recommended to utilize the INR instead of PT when reviewing results from outside facilities. International normalized ratio is also used to evaluate and adjust anticoagulant dosing of coumadin/warfarin, with an ideal range being between 2 and 3. Coumadin primarily affects coagulation factors made by usage of vitamin K (Factors II, VII, IX, X, protein C, protein S). Since factor VII is the sole constituent in the extrinsic pathway, INR is an excellent test in monitoring warfarin therapy.
Activated partial thromboplastin time is used to evaluate the intrinsic (factors XII, XI, IX, and VIII) and common pathways, and is also reported in seconds. This test is commonly used to evaluate effectiveness of heparin therapy. Activated partial thromboplastin time screening results are prolonged with problems in both common and intrinsic pathways, but cannot definitively identify a cause, warranting further workup via mixing studies/liver enzyme assays. 3
Platelet count via a CBC and manual differential evaluate the quantity of platelets, along with visually inspecting the platelets microscopically. This test can rule out bleeding due to thrombocytopenia but cannot detect pseudothrombocytopenia caused by platelet clumping. The manual differential can be used to identify platelet clumping and abnormally large platelets (seen in Bernard Soulier Syndrome and May-Hegglin Anomaly for example), but large platelets are not diagnostic of any specific disease. 3,5
Thrombin time (TT) evaluates for dysfibrinogenemias/hypofibrinogenemias—essentially, large quantities of factor IIa (thrombin) are added to patient plasma and the resultant time to clot formation is given in seconds. This is an indirect test; if nonfunctional fibrinogen is present in the patient plasma, TT will not be able to differentiate a functional versus a quantitative deficiency. 5 Fibrinogen testing is available as well and gives a direct value of the fibrinogen concentration in mg/dL, but, does not evaluate the qualitative function of the fibrinogen present.
Mixing studies are primarily used to characterize abnormalities detected in PT/aPTT screening, namely factor deficiencies versus lupus anticoagulants versus progressive inhibitors. Patient plasma is mixed with pooled normal plasma of patients with normal PT/aPTT testing, and then evaluated immediately and after 1 hour of incubation at body temperature (37°C). If the prolonged value is corrected immediately and after incubation, it is most likely a factor deficiency. If the value corrects initially then becomes prolonged after 1 hour of incubation, a progressive inhibitor (like a factor VIII antibody) is suspect. If there is no correction immediately or after an hour, a lupus anticoagulant is the most likely cause. Mixing studies are screening tests, and cannot discern which clotting factor is deficient, and further testing is necessary based on which test or tests were prolonged (PT or aPTT). 3
Factor activity assays measure activity by mixing patient plasma with testing plasma specifically deficient for the factor in question. These tests are effective in detecting decreased factor activity but should not be run before a mixing study is evaluated for factor inhibitors, which would confound the factor activity assay results if present. When properly utilized, factor activity assays are key for assisting in the diagnosis of hemophilia A (low factor VIII), hemophilia B/christmas disease (factor IX deficiency), or hemophilia C (deficient in factor XI). 4,7
Platelet function testing is an efficient screening tool used to evaluate the quality of platelets regardless of clotting factor quality/quantity. Closure time is measured in seconds as patient citrated plasma is aspirated across a membrane with collagen and epinephrine (EPI) or collagen and adenosine diphosphate (ADP) in a platelet function analyzer/microscopic aperture. 8 This testing platform has a very high negative predictive value, with some exceptions, meaning that a normal value is indicative of normal primary hemostasis. Closure time values are elevated in patients with Bernard Soulier Syndrome, Glanzmann’s Thrombasthenia, vWD, platelet secretion defects, platelet dense granule deficiencies, ADP inhibitor medications (like clopidogrel), NSAIDs and aspirin, low vWF activity, liver disease, uremia, and thrombocytopenia. 8 This test is a screening test as well; elevated values warrant follow-up laboratory testing. Platelet function testing can be influenced by the presence of NSAID’s and aspirin, giving a falsely elevated value in collagen-EPI, and can also be elevated due to certain types of foods (cocoa elevates both C-EPI and C-ADP), and type O patients with naturally lower concentrations of vWF. 8
Of historical significance is the bleeding time assay. This test was previously performed by a phlebotomist. A sphygmomanometer would be inflated to 40 mm Hg, and the phlebotomist would use a lancet to make 2 superficial incisions in the patient’s ventral forearm. Then, the phlebotomist would blot the incision with filter paper every 30 seconds until the bleeding ceased. Usage of this assay in theory would allow providers to evaluate patient’s ability to control bleeding and thus, platelet quality and function. This test is highly inaccurate, and was unable to be standardized, with sources of variability and error fluctuating with each phlebotomist, along with inability to standardize the exact dimensions/depth of incisions made. Platelet function analyzers have replaced this archaic method. 9
Teaching Points
Patient history and physical exam are very effective tools in determining what diseases belong on the differential diagnosis and what laboratory testing to order.
PT, aPTT, CBC/diff, and platelet function assays are good for initial screening for bleeding diatheses.
The most common bleeding disorder, von Willebrand disease, will often yield normal PT and aPTT results, which is why platelet function assays are intrinsic to screen for this disease.
The most common bleeding disorder in males, hemophilia A, is characterized with a prolonged aPTT, an immediately corrected mixing study, and below normal factor VIII activity assay. Hemophilia B is less common but has the same presentation, and a below normal factor IX activity assay.
Mixing studies that correct immediately indicate a factor deficiency, mixing studies that do not correct are most likely due to lupus anticoagulants, and mixing studies that correct immediately and are then prolonged are most likely due to a progressive inhibitor like a factor VIII antibody.
Laboratory screening to rule in/rule out coagulopathies need to be run in order; the disease must be evaluated initially via patient history and physical exam findings, then consider screening tests like PT/PTT/TT/fibrinogen/platelet count/platelet function assays as needed before ordering for mixing studies or factor activity assays. Ordering coagulation testing without a well-developed differential diagnosis will lead to a delay in diagnosis and therapy as well as poor utilization of laboratory resources.
Bleeding time has poor reproducibility and may miss mild platelet function disorders. Platelet function assays are superior to historical bleeding times in evaluation of platelet function.
Footnotes
Authors’ Note
The opinions expressed herein are those of the authors and are not necessarily representative of the official policy of the Uniformed Services University of the Health Sciences (USUHS), the Department of Defense (DOD), the United States Army/Navy/Air Force, or the US Government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
