Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.
Keywords
Primary Objective
Objective CBV2.3: Abdominal Aortic Aneurysm: Describe the clinical consequences of an abdominal aortic aneurysm.
Competency 2: Organ System Pathology; Topic: Cardiovascular—Blood Vessels (CBV); Learning Goal 2: Vascular Damage and Thrombosis.
Secondary Objective
Objective CBV2.2: Aortic Aneurysm and Dissection: Compare and contrast aortic aneurysms and aortic dissections in terms of their predisposing factors, the sites of involvement, and patient populations likely to be affected.
Competency 2: Organ System Pathology; Topic: Cardiovascular—Blood Vessels (CBV); Learning Goal 2: Vascular Damage and Thrombosis.
Patient Presentation
A 68-year-old male patient presents to the emergency department with nausea and a pulsating pain in his right groin. He reports intermittent bouts of syncope in the past week, which he never had previously. He also has chronic lower back pain but states that the pain is now more severe and radiates from belly to back and is very different from his usual back pain. Further interview reveals that the patient has smoked approximately 10 cigarettes per day for the past 30 years. He is in visible distress, although alert and oriented. His vital signs are remarkable for hypertension, with a blood pressure of 170/115.
Diagnostic Findings, Part I
Physical examination of the right groin reveals a small pulsatile mass. Abdominal palpation is suboptimal because of the patient’s size. Auscultation of the abdomen reveals an intermittent bruit, audible during systole, and dependent on heart rate.
Question/Discussion Points, Part I
Discuss the Differential Based on the Clinical Presentation
The patient presents with nausea, hypertension, syncope, increasing lower back pain, and pulsing unilateral groin pain. With his obesity, age, and history of hypertension, the patient likely suffers from atherosclerosis as well. His alertness and orientation suggest he is hemodynamically stable. These factors and symptoms suggest a possible abdominal aortic aneurysm (AAA). The particularly important risk factors in this patient’s history are as follows: (1) his advanced age, as AAA is rare in persons younger than 60 years 1 ; (2) his hypertension, as high blood pressure can induce small tears in the extracellular matrix (ECM), leading to smooth muscle cell (SMC) recruitment, vascular thickening, reduced vascular compliance, and ultimately aneurysm 2 ; (3) his smoking history, as smoking is the dominant behavioral risk factor for AAA 3 ; and (4) the likelihood that he also suffers from atherosclerosis, which is the dominant physiological risk factor for AAA. This is because atherosclerotic plaques weaken the arterial wall through a combination of chronic inflammatory response and ischemia of the underlying media. 2
The most important signs and symptoms from his clinical presentation are (1) the patient’s novel and increasing lower back pain, (2) his pulsing unilateral groin pain and palpable mass, and (3) his absence of hypovolemic shock symptoms (eg, hypotension, decreased alertness), suggesting that any presumptive aneurysm has not ruptured. 2,4
However, the differential diagnosis still includes the following: aortic dissection (AD), pancreatitis (particularly because of the radiating lower back pain), ischemic bowel, renal colic, gastrointestinal hemorrhage, pyelonephritis, and peptic ulcer disease.
The physical examination does little to narrow this differential, as obesity makes a thorough abdominal examination difficult. Nonetheless, given the history and clinical presentation—smoking, hypertension, age, obesity, back and groin pain, nausea—AAA is the most prominent concern. The unilateral groin pain could result from an associated aneurysm of the right common iliac artery—a regular finding in AAA—adding isolated iliac aneurysm to the differential. 4 Therefore, the most likely diagnosis is an unruptured AAA with associated unruptured isolated right common iliac aneurysm. The patient is clearly symptomatic and should be evaluated for possible or imminent rupture. One should note that the diagnosis, to this point, has been based on clinical findings.
What Further Testing Is Indicated for This Patient?
In hemodynamically unstable patients, timely diagnoses might preferably be reached via ultrasound. However, computed tomography (CT) imaging is the preferred method for this hemodynamically stable patient because (1) CT provides a more detailed and accurate assessment of the internal abdominal structures, while also showing relevant vessels and organs that may be involved with the aneurysm. This is one reason why the Society for Vascular Surgery (SVS) recommends CT for the assessment of maximum AAA diameter. 5 (2) Computed tomography imaging offers less variability in technician skill and methodology. (3) There are certain regions of the body where ultrasound is affected by overlying tissue—for instance, the iliac arteries, where overlying bowel can interfere with the imaging process. 6
Therefore, CT scans should be performed to evaluate dilation of the patient’s aorta and/or other major arteries. The thorax, abdomen, and lower limbs should all be evaluated to ensure full coverage of possible aneurysms, dissections, or embolisms.
Diagnostic Findings, Part II
Figure 1 Depicts the CT findings for this patient. Describe the CT scan and your assessment for the next step in care of the patient
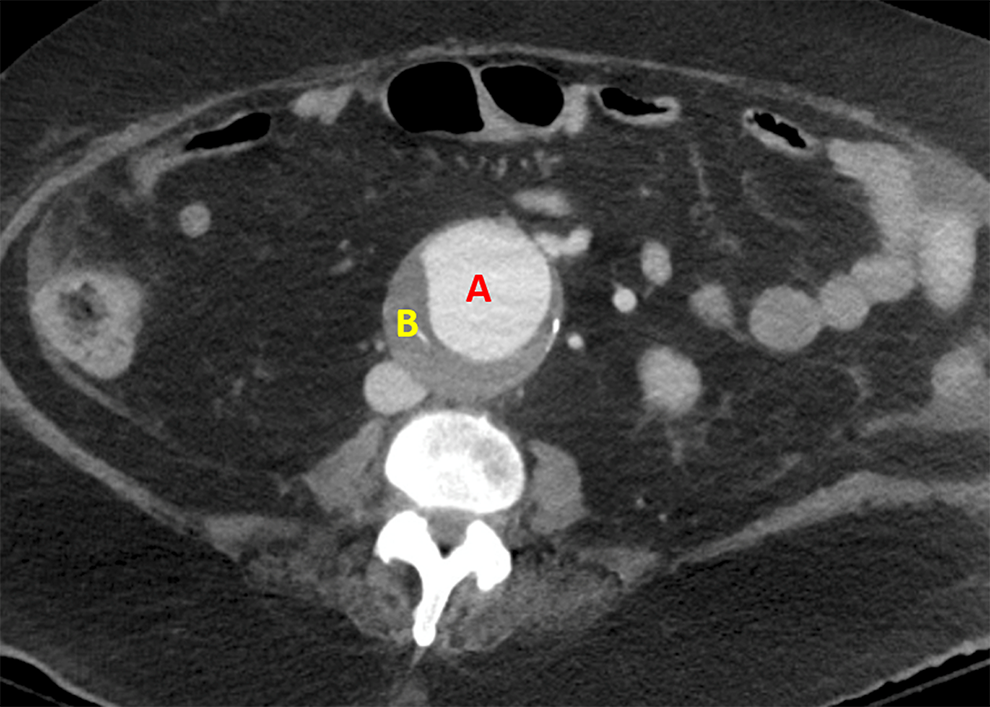
Abdominal CT image of the patient. A = Aortic lumen, highlighted by intravenous contrast material. B = Mural thrombus of aortic aneurysm. Computed tomography imaging was done with intravenous contrast material. Note the inferior vena cava (IVC) posterior and to the right of the mural thrombus. In a ruptured AAA, this would likely be obliterated. AAA indicates abdominal aortic aneurysm; CT computed tomography.
The abdominal CT scan shows a widely dilated area of intravenously injected contrast material, revealing a widened aortic lumen. The mural thrombus can also be seen surrounding this contrast material. These 2 facts confirm an aortic aneurysm. The scan was taken distal to the renal arteries, therefore specifying the diagnosis to AAA. The peritoneal cavity does not contain any extravasation of contrast material, indicating an unruptured AAA. The SVS recommends elective surgical repair for aneurysms larger than 5.5 cm, and the figure caption notes that this aneurysm is 5.8 cm at maximum diameter. As such, this patient should likely undergo surgical repair of the AAA.
Question/Discussion Points, Part II
What Clinical Sequelae Might Coincide With an Unruptured Abdominal Aortic Aneurysm?
Depending on the specific patient and aneurysm, an unruptured AAA may elicit a diverse set of clinical sequelae. For example, most patients with AAA are asymptomatic, and their aneurysms are discovered incidentally during physical examination or unrelated imaging. 4 For patients who are symptomatic—as the patient in this case—AAA may elicit abdominal or lower back pain, fever, syncope, a pulsatile mass in the abdomen or groin, and/or ischemia of the lower limb due to embolism. 4 However, these symptoms are not specific to AAA, and must be corroborated with imaging or further diagnostic testing. In essence, a patient with unruptured AAA may present with wide-ranging clinical sequelae depending on blood pressure, severity of atherosclerosis, and other confounding factors. If one patient has a stable AAA—for example, only 2.5 cm in diameter—clinical signs may be absent. Then again, if an aneurysm is large or aggressive, a patient might present with several of the characteristic symptoms.
Figure 2 Demonstrates the Gross Anatomy of an Unruptured AAA. Briefly Describe the Anatomy and its Clinical Implications
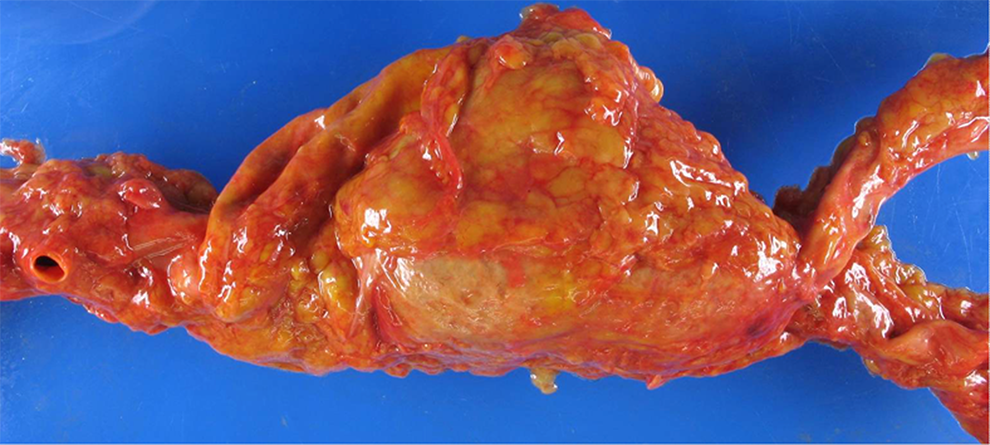
Gross anatomy of abdominal aortic aneurysm (AAA). Note the superior mesenteric artery superior (to the left in this image) to the aneurysm, and the bifurcation of the common iliac arteries inferiorly (to the right). Maximum diameter of aneurysm = 8.0 cm, well beyond the standard for surgical intervention.
Figure 2 shows an infrarenal, unruptured saccular AAA, in the classic location just superior to the common iliac bifurcation. A saccular aneurysm is one which bulges in a distinct, unilateral manner, creating a discrete pouch of dilated vascular wall. In contrast, fusiform aneurysms bulge circumferentially and are thus somewhat symmetrical. Both the saccular and fusiform types are “true” aneurysms because, unless ruptured, all 3 layers of the arterial wall remain intact (These layers are the intima, media, and adventitia.). Blood is therefore retained as a mural thrombus adherent to the arterial lumen, as opposed to in an extravascular hematoma.
The true aneurysm in Figure 2 is 8.0 cm in maximum diameter, making it a remarkably large aneurysm. Such an aneurysm would be at very high risk for rupture, likely necessitating surgical intervention. Some AAAs reach up to 20 cm in diameter, although this is uncommon. 2 An associated atherosclerotic plaque might obstruct blood flow to and through the iliac arteries, causing lower limb ischemia, which could also occur through thromboembolism.
Given the asymmetric, unilateral appearance of the referenced AAA, one might confuse this specimen for a “pseudoaneurysm.” Pseudoaneurysms, like true aneurysms, result from defects in the vascular wall. However, there is a key difference between the two. In true aneurysms, all 3 layers of the arterial wall become dilated, creating an intravascular bulge that has the potential for rupture. On the other hand, pseudoaneurysms occur when blood ruptures through all 3 arterial layers but is contained within the extravascular tissue. 2 This forms an often asymmetrical hematoma outside the artery, still contiguous with the intravascular space. True aneurysms—until they rupture—retain blood completely within the intravascular space.
What Risk Factors Might Influence the Likelihood of Abdominal Aortic Aneurysm Rupture in This Patient?
Maximum AAA diameter is highly indicative of rupture risk. There is <1% yearly risk of rupture for aneurysms smaller than 3.9 cm in maximum diameter, increasing to 1% at 4.0 to 4.9 cm; 1% to 11% at 5.0 to 5.9 cm; 10% to 22% at 6.0 to 6.9 cm; and 30% to 33% at 7.0 cm or larger. 1,5 Rapid aneurysm expansion—defined as a baseline diameter increase of 5 mm or more over a 6-month period, or 10 mm over a 12-month period—also increases risk of rupture. 5 For comparison, the average rate of AAA expansion is approximately 2.6 mm per 12 months. 7 Larger aneurysms tend to expand faster than smaller ones, although expansion is nonlinear and can be quite stochastic. 8
Other strong risk factors for rupture include tobacco use, hypertension, and female sex. 5,9 It is difficult to characterize the exact contributions of tobacco use, because the many components of tobacco smoke have wide-ranging, interrelated bioactivities in vivo. Nonetheless, studies suggest possible pathways in which T-cells and matrix-degrading enzymes cause inflammatory damage to the aortic SMCs and ECM, while also reducing expression of enzymes such as prolyl-4-hydroxylase. 3 These enzymes are critical to collagen synthesis, and their suppression might inhibit repair of aortic tissue. As the aortic tissue incurs further damage and is unable to repair itself, aneurysmal dilation can continue up to rupture.
Studies indicate that females with AAA are more likely than males to suffer from AAA rupture—regardless of aneurysm size interval—with a 4-fold greater risk of rupture in aneurysms less than 5.5 cm wide. 10 Obviously, this does not impact the present patient, but the discrepancy is important to consider nonetheless. Some explanation may be found in the sex-neutral practices of AAA management and classification. For example, the SVS currently recommends observation for asymptomatic aneurysms <5.5 cm in diameter, regardless of patient sex. 5 However, studies also suggest that females have smaller relative aortic diameters, indicating that if a female and a male of similar size have AAAs of equal diameter, the female has undergone a greater proportional dilation of the aneurysm. 11 Therefore, female patients with aneurysm diameters of 5.5 cm may exceed the proper minimum threshold for recommendation of elective surgery. This means that females may incur greater risk of AAA rupture, because their current sex-neutral thresholds for elective surgical intervention are too high.
Finally, uncontrolled hypertension can increase risk of AAA rupture—although the relationship between hypertension and AAA expansion is less clear—as high blood pressures propagate stress upon an already-dilated arterial wall. 1 In order to prevent further damage to the aorta, initial treatment of AAA typically involves antihypertensive medication.
The present patient—given his history of smoking, hypertension, and aneurysm size—is certainly at high risk of AAA rupture. Rupture carries significant potential for mortality, and the patient should thus be carefully monitored and treated moving forward.
Discuss the Mortality Rates Associated With Ruptured Abdominal Aortic Aneurysm
A ruptured AAA is a serious, often fatal surgical emergency. Upon rupture, patients can present with 3 major symptoms: pain, pulsatile mass, and hypotension. However, less than half of patients will present with all three. This can lead to misdiagnosis, contributing to the 90% overall mortality rate in AAA. 12 There are 4 main sites for AAA rupture—intraperitoneal, retroperitoneal, aortocaval fistula, and primary aortoduodenal fistula—and each one presents with distinct and nuanced clinical features. 12
If This Patient Suffered From a Ruptured Abdominal Aortic Aneurysm, How Might His Computed Tomography Scan Differ From the Original?
One would expect extravasation of the contrast material, leading to visible retroperitoneal hematoma and loss of the fat plane between the aorta and its surrounding tissue. In symptomatic but unruptured AAA, the CT often reveals several distinct findings in lieu of such extravasation: irregular aortic wall, draping of the aorta over vertebral bodies, and/or the crescent shape of a layering hematoma. 4
Figure 3 Presents the CT Scan From a Different Patient. Describe the Figure and Diagnose This New Patient
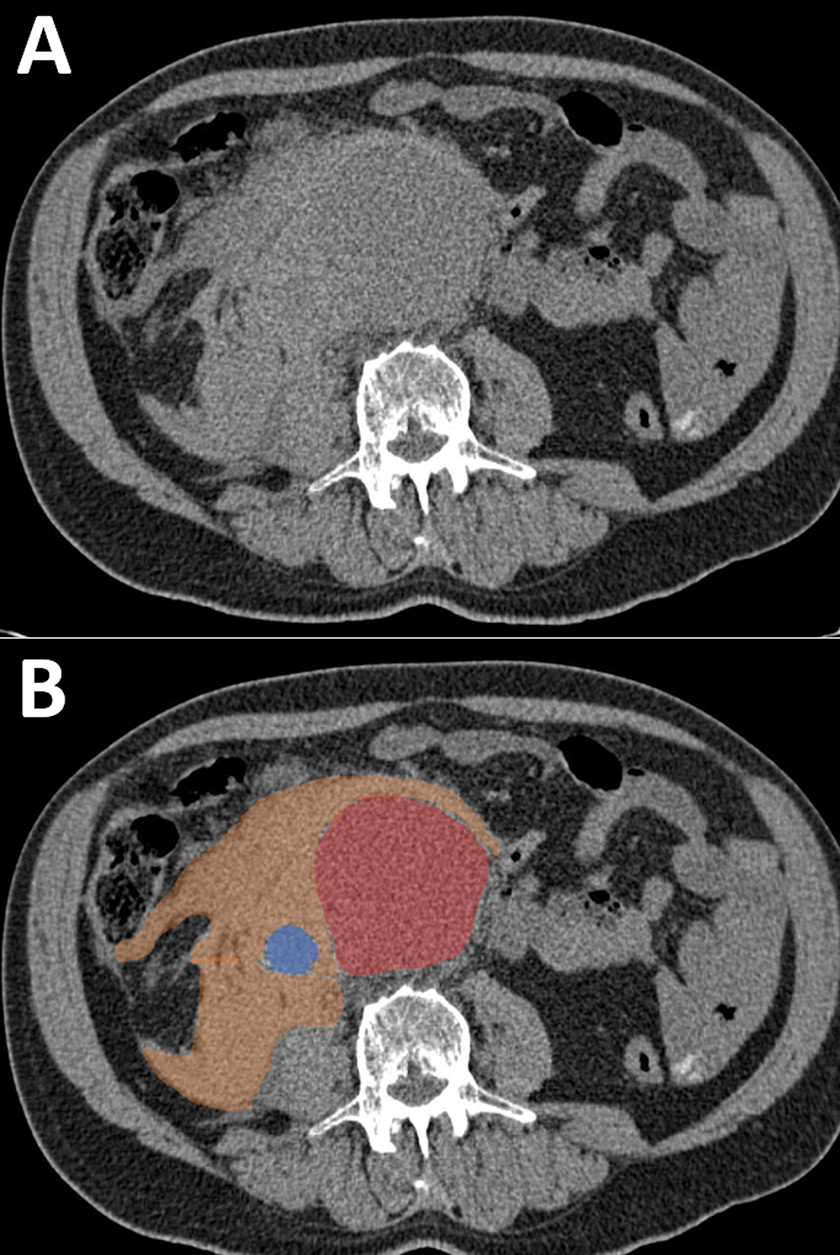
Abdominal computed tomography image of a ruptured AAA (A) alongside the same image where the dilated aorta is highlighted in red, the inferior vena cava in blue, and hemorrhage in orange (B). Maximum aneurysm diameter = 5.8 cm. Computed tomography imaging was done with intravenous contrast material. AAA indicates abdominal aortic aneurysm.
In Figure 3A, we see a dilated aortic lumen, with high density of blood in lieu of the periaortic fat plane. This blood appears to have dissected into the retroperitoneum and obliterated the fat plane around the inferior vena cava (IVC) and right psoas muscle. Figure 3B shows a colored version of the same scan, giving appreciation of the acute blood density surrounding the periaortic fat plane, obliterated IVC, retroperitoneum, and psoas muscle. These images are consistent with a ruptured AAA, as the extravasation almost certainly originated from an aneurysm directly adjacent to this abdominal region.
Compare and Contrast the Etiology, Clinical Description, Risk Factors, and Epidemiology of Aortic Aneurysm and Aortic Dissection
An aortic aneurysm involves dilation of all 3 layers of the arterial wall—the intima, the media, and the adventitia. In contrast, AD occurs when blood enters the aortic wall through an intimal tear and then dissects progressively through the media. 13 A “dissection” then continues as blood pulses through the tissue plane, splitting the attenuated media. Histological data from ADs contrast starkly with those of normal aortic specimens. For instance, Figure 4A depicts a normal, healthy media identified by numerous elastin fibers colored black by the Elastic Van Gieson (EVG) stain. (EVG forms a variety of strong bonds with elastin, staining the elastic fibers black. As such, EVG is typically used to visualize normal or pathologic elastic fibers.) In contrast, Figure 4B depicts a medial layer split by dissecting blood, alongside atherosclerosis between the intima and attenuated media. The pathological term for this attenuation of the media is cystic medial necrosis.
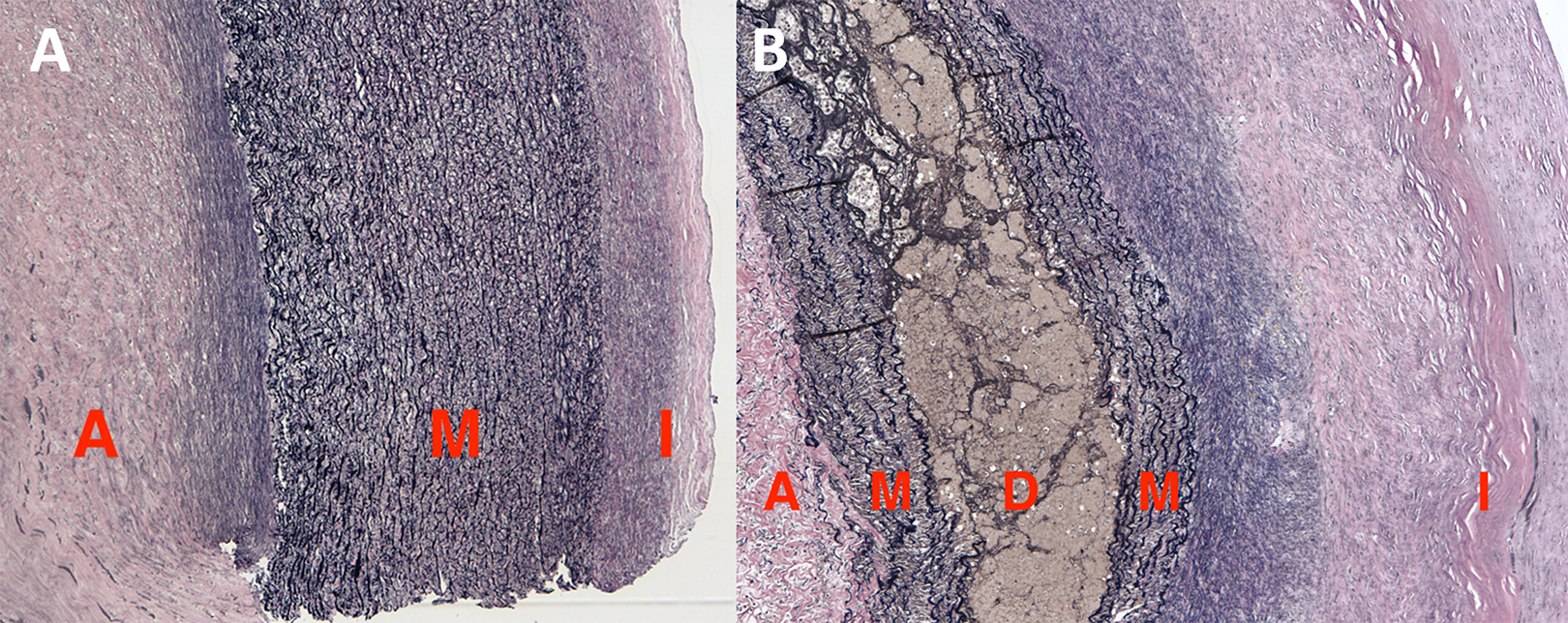
Photomicrograph of normal aorta with intact, elastic fibers of tunica media (A), contrasted with photomicrograph of blood dissecting through attenuated and weakened tunica media (B). Photomicrographs were taken with EVG stain and at 40× magnification. Note how in the dissection specimen, the tunica intima is thickened with an atherosclerotic plaque. A = tunica adventitia; M = tunica media; D = dissecting blood; I = tunica intima. EVG indicates Elastic Van Gieson.
Aortic dissection is therefore characterized by a proximal intimal tear—such as in the ascending aorta—and a medial dissection which can proceed quite distally. This dissection need not involve the adventitia. Nonetheless, adventitial rupture can still occur, necessitating immediate emergency action.
Both AD and aortic aneurysm result from some form of structural vascular weakening in the SMCs and ECM. However, there are distinct differences between the two, beginning with clinical presentation. In AD, patients typically present with a “tearing” pain which begins in the chest and then radiates toward the back. 13 This symptom is often very acute, with 85% of patients describing a sudden onset of unique, piercing, and severe pain. The pain can also migrate to the abdomen as dissection progresses. Syncope presents in 5% to 10% of acute patients, while a pulse deficit occurs in 9% to 30% of thoracic dissections. If a dissection progresses proximally from the ascending aorta to the aortic valve, a heart murmur can also be heard due to aortic valve regurgitation. These clinical signs may vary depending on the site—or type, as mentioned below—of a given AD.
The Stanford classification system names 2 categories for AD: type A and type B. Type A dissections involve the ascending aorta, and may involve the aortic arch or descending aorta. Type B dissections begin distal to the ascending aorta. Ascending dissections occur nearly twice as often as descending dissections, and isolated abdominal dissections are not regularly reported. 13 Aortic dissections are also classified as acute (symptomatic for 2 weeks or less) or chronic (symptomatic for more than 2 weeks).
High blood pressure is the most prominent risk factor for AD, with 1 review finding that over 70% of acute patients have an antecedent history of hypertension. 2,14 Preexisting aneurysms increase risk of AD, being present in over 20% of patients with descending dissections and over 12% of patients with ascending dissections. 13 Family history, male sex, congenital connective tissue disorders, and advanced age can also increase risk for the disease. 13,14 For instance, one review found that over 50% of AD patients younger than 40 years also suffered from Marfan Syndrome, compared to 2% in those older than 40 years. 14 The same review indicated that those with a bicuspid aortic valve are at 9 times greater risk of the disease. Few studies exist to suggest any racial or ethnic predisposition to AD. In the United States, estimated incidence ranges from 2.6 to 3.5 cases per 100 000 person-years, with a mortality rate of 25% to 30%. 13
Aortic aneurysms, like dissections, are diverse and can occur along any point of the aorta. Because this case is concerned with AAA—and because AAA is the most common form of aortic aneurysm—the clinical sequelae alluded to earlier will suffice for clinical comparisons with AD. However, it is important to reiterate that most patients with AAA do not present with any symptoms and that their aneurysms are discovered incidentally or after rupture. 4 Given the aforementioned classifications of AD, it may also help to reference the various classes of AAA mentioned earlier—saccular versus fusiform, true aneurysm versus pseudoaneurysm, and ruptured versus unruptured.
Hypertension is thought to contribute to the onset and/or rupture of some aortic anuerysms. 2,15 Other predisposing factors for aortic aneurysm include atherosclerotic disease, preexisting aneurysms outside the aorta (given that atherosclerosis is a systemic disease), obesity, smoking, and high salt intake. 16 Certain inherited connective tissue disorders, such as Marfan Syndrome, can lead to aortic aneurysm by weakening the ECM and/or SMCs and decreasing structural integrity of the arterial wall. 1,13 However, such diseases are rare. Advanced age and caucasian ethnicity are more likely risk factors: Males between 65 to 80 years of age face up to 6 times higher risk of AAA development than their female counterparts, and caucasian populations have the highest rates of AAA in the United States—up to 10-fold higher than Asian American populations and twice as high as African American groups. 1 In Western countries, the incidence of AAA is approximately 2.5 to 6.5 cases per 1000 person-years, with a prevalence of 4% to 8% based on screening studies. 1 The incidence of AAA rupture is much lower, with one Swedish study suggesting a rate of 11 cases per 100 000 person-years. 17
In essence, AD and aortic aneurysm both involve (1) a weakening or degradation of SMCs or ECM in the arterial wall, (2) potential for rupture, which carries high mortality rates, and (3) hypertension as a risk factor, alongside existing aneurysms, advanced age, and inherited connective tissue disorders.
At the same time, differences between the two include (1) their effects on the intima, media, and adventitia of the arterial wall, (2) their various clinical presentations described above, (3) their systems of classification and categorization, and (4) their presence—or lack thereof—of ethnic, racial, and/or sex-based dispositions.
There are numerous other comparisons to be made based on dissection type, aneurysm classification, and other factors. Table 1 broadly summarizes the similarities and differences between AAA and AD.
Comparison of Abdominal Aortic Aneurysm and Aortic Dissection.
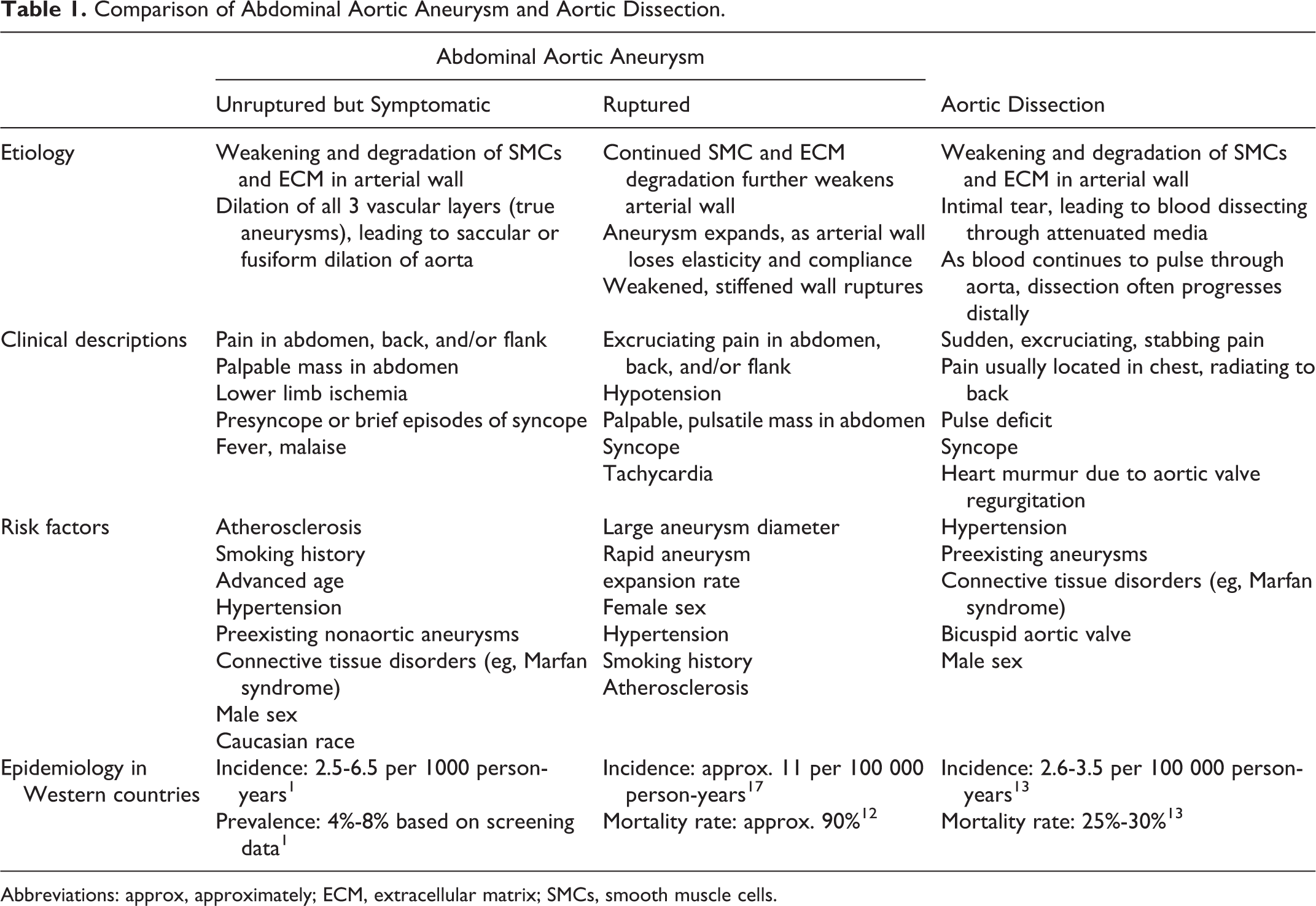
Abbreviations: approx, approximately; ECM, extracellular matrix; SMCs, smooth muscle cells.
Teaching Points
– Although most AAA cases are asymptomatic, clinical features include abdominal and/or back pain, pulsatile masses, nausea, presyncope or brief episodes of syncope, and lower-limb ischemia. – True aneurysms involve dilation of the intima, media, and adventitia and can be fusiform or saccular. – Risk factors for AAA development include atherosclerosis, hypertension, smoking, male sex, advanced age, presence of other non-aortic aneurysms, and caucasian race. – Atherosclerosis and hypertension are particularly important to the etiology of AAA. – Abdominal aortic aneurysm rupture carries a 90% mortality rate and should always be ruled out in patients suspected of AAA. – Risk factors for AAA rupture include large aneurysm diameter, atherosclerosis, rapid aneurysm expansion, tobacco use, hypertension, and female sex. – In AD, blood enters through an intimal tear and dissects through the media, without necessarily involving the adventitia. – Clinical features of AD may include sudden radiating chest pain, pulse deficit, syncope, and heart murmur due to aortic valve regurgitation. – In pseudoaneurysms, blood ruptures through all 3 arterial layers but is contained within extravascular tissue. – In the Stanford classification system, type A aortic dissections involve the ascending aorta, while type B dissections begin distal to the ascending aorta.
Footnotes
Acknowledgments
The authors would like to thank the Stanford University Department of Pathology, for the diagnostic, histologic, and gross images used in this education case.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
