Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.
Keywords
Primary Pathology Learning Objective
Competency 2: Organ System Pathology, Topic: Head and Neck (HN), Learning Goal 2: Head and Neck Neoplasia
Secondary Pathology Learning Objective
Competency 3: Diagnostic Medicine and Laboratory Diagnosis; Topic CYP: Cytopathology; Learning Goal I: Cytologic Diagnosis
Patient Presentation
A 62-year-old obese, hypertensive, Caucasian male presents to his primary care physician with a slowly growing left cheek mass for 1-year duration. He has a 40 pack-year smoking history. He has no family or personal history of carcinoma. He is HIV-negative and has not experienced any dysphagia, odynophagia, B-symptoms, trismus, or facial weakness.
Diagnostic Findings, Part 1
On physical examination, his primary care physician notes a palpable, 2 cm, “doughy,” mobile mass in his left parotid gland. There appears to be mild erythema and slight puckering of the skin overlying the mass. The contralateral parotid gland has no palpable masses and there is no cervical lymphadenopathy. Tongue sensation and facial nerve function are intact. The patient undergoes imaging studies and fine needle aspiration (FNA) biopsy of his lesion.
Questions and Discussion Points, Part 1
Given the Clinical History, What Are the Most Likely Salivary Gland Neoplasms This Patient Might Have, and What Clinical Features Would Suggest Each Diagnosis?
Salivary gland tumors are rare, making up less than 2% of all tumors. Up to 80% of salivary gland tumors appear in the parotid gland, and 70% of parotid gland tumors are benign. Parotid gland neoplasms are more likely to be benign than submandibular gland neoplasms, which are more likely to be benign than minor salivary gland neoplasms, which are more likely to be benign than sublingual gland neoplasms. Although certain clinical features can be suggestive, they are unreliable to definitively differentiate between benign or malignant. 1,2
The most common salivary gland tumor is pleomorphic adenoma and makes up approximately 50% of all salivary gland tumors and 80% of all benign salivary gland tumors. They typically are painless, mobile, and slow growing and occur most frequently in the parotid gland—features all seen in our patient. However, pleomorphic adenoma occurs more frequently in females, and the average age of presentation is in the mid-40s—unlike our patient. 1 -3
Warthin tumor is the second most common salivary gland neoplasm and occurs almost exclusively in the parotid gland. It is seen more frequently in male patients, the average age of presentation is in the mid-60s, and it has a strong correlation with smoking history. In addition, the “doughy” texture of the mass is a frequent finding in Warthin tumor. Up to 10% of Warthin tumors are multifocal, and up to 15% of Warthin tumors occur bilaterally, so either of these features would also raise the suspicion for this diagnosis. 1,2 However, our patient’s lack of a contralateral mass on physical examination certainly does not rule out this diagnosis.
Mucoepidermoid carcinoma is the most common malignant salivary gland neoplasm. It affects a broad age range and is the most common primary malignant salivary gland tumor in both adults and children. It is likely to demonstrate symptoms related to its invasive nature, including rapid growth, pain, immobility, skin changes and facial asymmetry, cervical lymphadenopathy, and loss of nerve function. 1,2 The patient demonstrated few of these concerning symptoms (mild erythema and slight puckering overlying the mass) and the diagnosis is lower on the differential.
In summary, the differential diagnosis includes but is certainly not limited to pleomorphic adenoma, Warthin tumor, and mucoepidermoid carcinoma. The skin erythema and puckering may be seen in a malignant process (mucoepidermoid carcinoma); however, the mobility, painlessness, slow growth, and absent lymphadenopathy suggest a benign tumor (Warthin tumor or pleomorphic adenoma). These features combined with the “doughy” texture, patient’s smoking history, male sex, and patient age suggest a Warthin tumor.
What Risk Factors Predispose a Patient to Develop Salivary Gland Neoplasms?
Ionizing radiation, in the form of atomic bomb exposure, previous head and neck radiation therapy, and radioactive iodine treatment for thyroid disease, have all shown an increased association with the development of salivary gland neoplasms. There is also a strong association with smoking and the development of Warthin tumor—smokers are 8 times more likely to have a Warthin tumor than the general population. Certain genetic alterations occur more frequently in certain salivary gland tumors but familial aggregation has not yet been observed in salivary gland tumors. 1
What Diagnostic Testing Is Available for This Patient?
Although the clinical history and certain physical examination findings, such as mobility of the mass or overlying skin changes, can suggest a benign or malignant process, computed tomography, magnetic resonance imaging (MRI), and ultrasonography are much more accurate. Imaging is primarily used to assess malignant features, rather than to ascertain a specific diagnosis. Imaging studies can determine if a neoplasm is contained within the salivary gland or if it has invaded into adjacent structures. It can also assess regional lymph node and distant metastases. However, a tissue biopsy is ultimately required to make a diagnosis in salivary gland tumors. Tissue biopsy can be performed via fine needle aspiration biopsy or core needle biopsy. 2
What Are the Advantages and Limitations of Fine Needle Aspiration in Presurgical Assessment of Salivary Gland Tumors?
Salivary gland tumors are a heterogeneous and morphologically diverse group of benign and malignant neoplasms. They are often sampled by fine needle aspiration and/or core needle biopsy, but given the significant overlap in morphology and immunohistochemical staining between salivary gland tumors, tissue biopsy still has limitations in its ability to confer a definitive diagnosis. Accuracy of FNA diagnosis varies across studies and practitioners. Generally, cytologic sampling has approximately 96% to 98% sensitivity in identifying a salivary gland neoplasm, with 79% sensitivity and 96% specificity in distinguishing benign from malignant tumors. Even in the absence of a definitive diagnosis, however, tissue biopsy can still help narrow down the differential, making it a valuable tool in triaging patients for medical or surgical management. Core biopsy may offer better accuracy than FNA biopsy, but it also causes more discomfort to the patient and has greater risk of complications, such as nerve damage and tumor seeding along the needle tract. 4
Diagnostic Findings, Part 2
Magnetic Resonance Imaging
The patient undergoes MRI of the neck with and without contrast, which demonstrates 2 enhancing parotid masses located in the superficial lobe of the left parotid gland. The masses measure 1.2 × 0.8 × 0.9 cm and 0.7 × 0.6 × 0.5 cm. The deep parotid lobes are not involved. The right parotid gland is unremarkable. No infiltrative features or cervical lymphadenopathy are seen.
Cytologic Assessment
Upon biopsy of the larger lesion, thick, green-brown fluid is aspirated, resembling motor oil. A representative photomicrograph of the FNA biopsy of the patient’s larger lesion is shown in Figure 1A.
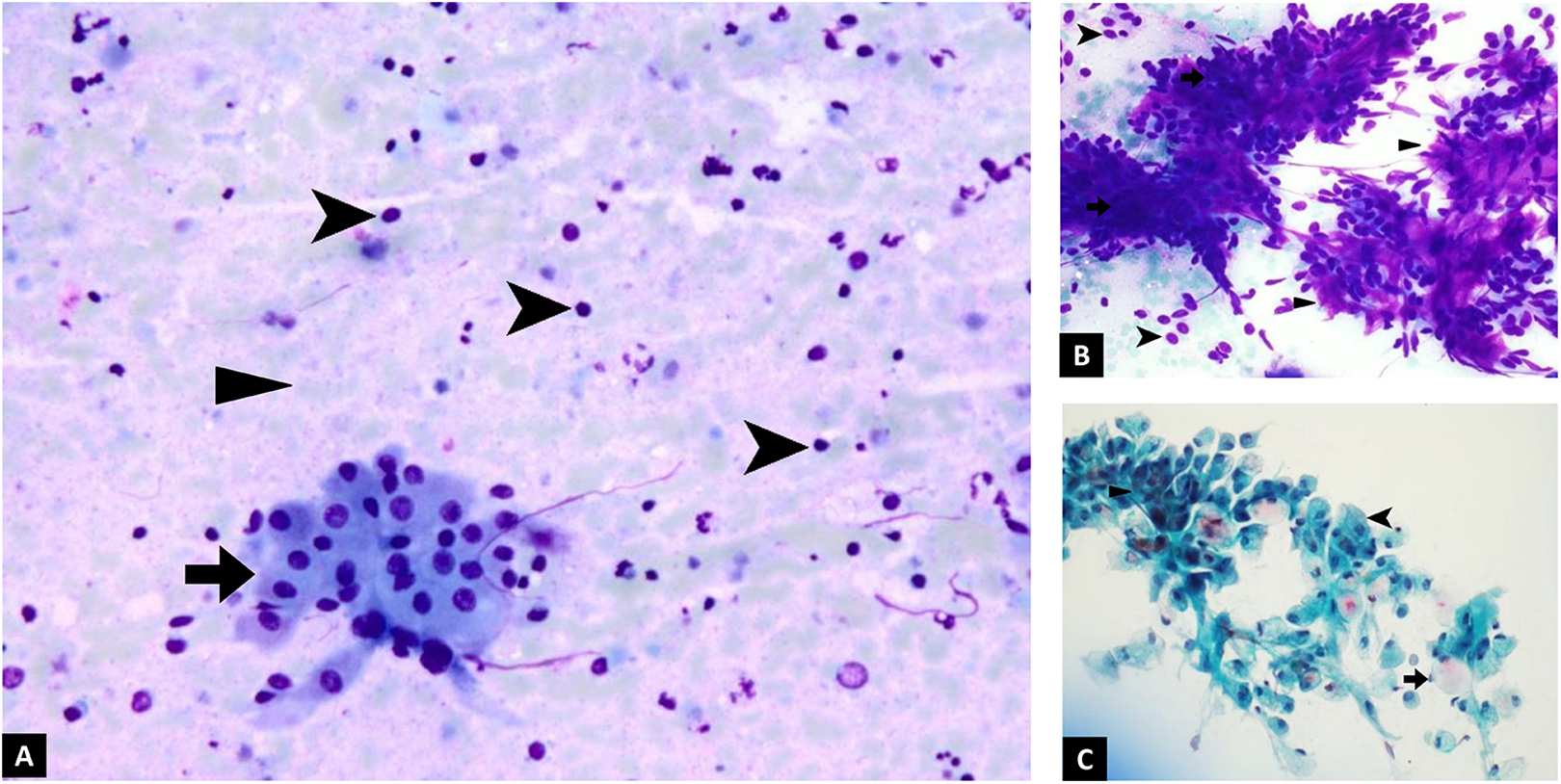
Cytologic features. A, FNA biopsy of the patient’s lesion, showing Warthin tumor with sheets and clusters of oncocytic cells (arrow), lymphocytes (arrowhead), and granular debris in the background (triangle). B, Cytology of pleomorphic adenoma for comparison, with cohesive sheets and clusters of ductal cells (arrow), individual myoepithelial cells (arrowhead), and metachromatic, magenta-colored, fibrillary stroma (triangle). C, Cytology of mucoepidermoid carcinoma for comparison, with signet-ring type mucin producing cells (arrow), polygonal squamoid cells with dense blue cytoplasm (arrowhead), and smaller intermediate cells (triangle).
Questions and Discussion Points, Part 2
How Do the Findings on MRI Help Narrow Down the Differential?
Since no infiltrative features or cervical lymphadenopathy are seen, a benign neoplasm is favored. On imaging, the tumor was found to be 2 separate nodules, rather than a solitary lesion that was noted on physical examination. The clinical presentation plus the features seen on radiology (multiple lesions with no infiltrative features) make a diagnosis of Warthin tumor likely.
Discuss the Cytologic Features of the Fine Needle Aspirate Biopsy in Figure 1A
The “motor oil” fluid that is aspirated from the lesion is a common feature of Warthin tumor. The biopsy of the tumor has 3 distinct features, which are diagnostic of Warthin tumor: sheets and clusters of oncocytic cells (arrow), lymphocytes (arrowhead), and granular debris in the background (triangle).
The Cytologic Features of the 2 Other Neoplasms Considered in the Differential Diagnosis Are Shown in Figure 1B and C for Comparison. What Features Distinguish the Patient’s Warthin Tumor From Pleomorphic Adenoma and Mucoepidermoid Carcinoma?
Figure 1B: Pleomorphic adenoma: This tumor has cohesive sheets and clusters of ductal cells (arrow), individual myoepithelial cells (arrowhead), and metachromatic, magenta-colored, fibrillary stroma (triangle) when stained with a modified Wright Giemsa (Diff Quik) stain.
Figure 1C: Mucoepidermoid carcinoma: This tumor has a mixed population of cells, with 3 distinct cell types. Signet-ring type mucin producing cells with pink mucin (arrow), polygonal squamoid cells with dense blue cytoplasm (arrowhead), and smaller intermediate cells (triangle) when stained with a Papanicolaou stain.
What Are the Treatment Options for the Patient?
Treatment recommendations are currently based on retrospective reviews and not randomized clinical trials. Complete surgical resection with negative margins is the gold standard, and patients with benign tumors (such as Warthin tumor and pleomorphic adenoma) and low-grade tumors can be treated by surgery alone. High-grade tumors, tumors with positive resection margins, and tumors with high-risk features are usually treated with surgery and adjuvant radiation therapy. Because he has a benign diagnosis of Warthin tumor, he can be treated with surgery alone. 5
Diagnostic Findings, Part 3
Histologic Assessment
The patient undergoes superficial parotidectomy to remove the 2 masses. A representative photomicrograph of the patient’s lesions is shown in Figure 2A.
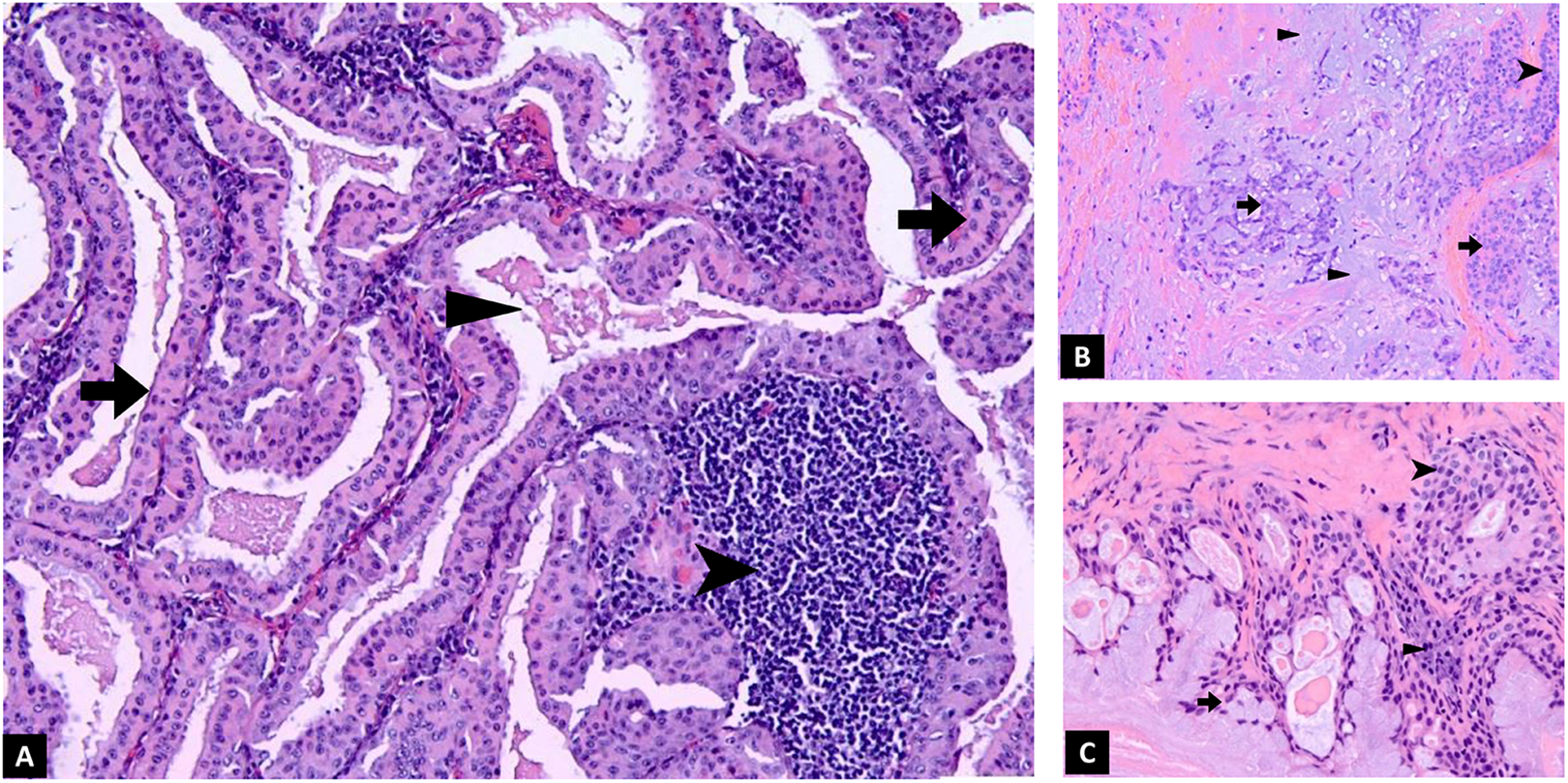
Histologic features. A, Surgical excision of the patient’s lesion showing Warthin tumor with oncocytic cells (arrow) and lymphoid stroma (arrowhead). Granular debris fills the cyst spaces (triangle). B, Histology of pleomorphic adenoma for comparison, with ductal cells (arrow) surrounded by a layer of myoepithelial cells (arrowhead), arranged in a chondromyxoid stroma (triangle). C, Histology of mucoepidermoid carcinoma for comparison, with mucin producing cells (arrow), squamoid cells (arrowhead), and intermediate cells (triangle).
Questions and Discussion Points, Part 3
Discuss the Histologic Features of the Surgical Excision in Figure 2A
The cystic lesion is lined by oncocytic cells (arrow) with a lymphoid stroma (arrowhead). Granular debris fills the cyst spaces (triangle). These are diagnostic of and consistent with the initial biopsy diagnosis of Warthin tumor.
The Histologic Features of the 2 Other Neoplasms Considered in the Differential Diagnosis Are Shown in Figure 2B and C for Comparison. What Features Distinguish the Patient’s Warthin Tumor From Pleomorphic Adenoma and Mucoepidermoid Carcinoma?
Figure 2B: Pleomorphic adenoma: This tumor has ductal cells (arrow) surrounded by a layer of myoepithelial cells (arrowhead), arranged in a chondromyxoid stroma (triangle).
Figure 2C: Mucoepidermoid carcinoma: This cystic tumor has a mixed population of cells, with 3 distinct cell types—mucin producing cells (arrow), squamoid cells (arrowhead), and intermediate cells (triangle).
What Is the Expected Outcome After Superficial Parotidectomy to Excise the Patient’s Warthin Tumor?
Surgical excision of Warthin tumor is curative, and we expect the patient to have a good clinical outcome. The risk of local recurrence after complete surgical excision is less than 2%.
The other tumors on our initial differential behave quite differently. Pleomorphic adenoma is also benign but has a higher local recurrence rate of up to 5% if treated by parotidectomy, and up to 25% if treated with simple enucleation. The behavior of mucoepidermoid carcinoma depends on the grade of the neoplasm. Local recurrence rates range from 15% to 30%, and 5-year survival ranges from 50% to 90%. 1,2
Teaching Points
Salivary gland neoplasms are rare, accounting for less than 2% of all tumors. Up to 80% of salivary gland neoplasms occur in the parotid gland, and up to 70% of parotid gland tumors are benign. The likelihood of malignancy in each salivary gland in increasing order is: parotid gland, submandibular gland, minor salivary gland, and sublingual gland. Clinical features are not definitively diagnostic of a benign or malignant process. Malignant tumors are suggested by rapid growth, pain, immobility, skin changes and facial asymmetry, cervical lymphadenopathy, and loss of nerve function. Benign tumors are suggested by absence of pain, mobility, slow growth, absence of nerve involvement. Radiologic studies can provide important clues to the nature of a salivary gland tumor, including invasion beyond salivary gland parenchyma into adjacent structures and local lymphatic spread. Fine needle aspiration biopsy of salivary gland tumors is an important initial diagnostic test that can frequently give an accurate diagnosis. When a specific diagnosis cannot be rendered, cytologic analysis can give a differential diagnosis that helps guide clinical/surgical management. The 3 most common salivary gland tumors (pleomorphic adenoma, Warthin tumor, and mucoepidermoid carcinoma) have distinct cytologic and histologic appearances. The most common salivary gland neoplasm is pleomorphic adenoma, a benign tumor that typically presents in the mid-40s and has a female predilection. The second most common benign salivary gland neoplasm is Warthin tumor, which commonly presents in male smokers in their mid-60s. On physical examination, it has a “doughy” texture and can be multifocal or occur bilaterally. On aspiration, it produces a “motor oil” like fluid. The most common malignant salivary gland neoplasm is mucoepidermoid carcinoma. It occurs over a broad age range and is the most common salivary gland malignancy in adults and children. Pleomorphic adenoma and Warthin tumor can both be treated by surgical excision alone and have less than 5% risk of local recurrence. The treatment and outcome of mucoepidermoid carcinoma is dependent on the tumor grade. High-grade tumors may require surgery and adjuvant radiation therapy, with local recurrence rate as high as 30%, and 5-year survival of 50%.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
