Abstract
Structured Abstract
Background
In vivo modeling is essential to study bladder cancer biology. Orthotopic bladder tumor models involve direct introduction of tumor cells into the bladder of a laboratory animal. Common techniques for orthotopic tumor introduction include transurethral, ultrasound-guided, or surgical approaches.
Objective
We systematically collected data from published studies that have utilized orthotopic bladder tumor models in mice or rats to identify trends and outcomes across techniques. We used these data to optimize a surgical orthotopic technique.
Methods
A PubMed search was performed to identify articles involving orthotopic implantation of tumor cells into mouse or rat bladders for research purposes. Results were individually reviewed and filtered. All studies reporting preclinical models established via a surgical, transurethral, and/or ultrasound-guided approach were included. Surgical orthotopic tumor implantation experiments were performed and data collected.
Results
A total of 254 studies were identified, of which 187 met criteria and were included in the analysis. Data regarding each orthotopic technique was reviewed and trends were identified. Transurethral installation was the most commonly used method but had the lowest tumor take rate. The surgical approach had the highest metastatic rate. These data were used to inform optimization of the surgical orthotopic approach in our laboratory. The study is limited by its retrospective design and heterogeneity of data reporting across studies.
Conclusions
Tumor take rates vary across orthotopic implantation techniques. Optimization of a surgical implantation approach is feasible. These findings can inform best practices for orthotopic bladder cancer models.
Introduction
Bladder cancer accounts for approximately 80,000 new cancer diagnoses and nearly 20,000 deaths annually in the United States. 1 Preclinical models of bladder cancer are essential for understanding tumor biology and testing novel therapies, and in vivo modeling of bladder cancer in laboratory animals is critical for bridging the gap between in vitro experiments and human clinical studies. 2
Rodent bladder cancer models can be broadly classified into autochthonous (developing de novo within the bladder) or transplanted tumor models. Autochthonous bladder tumor models in rodents can be induced via carcinogenic exposure or via genetic engineering. Carcinogen-induced bladder tumors arise following exposure to specific carcinogens or injurious agents, such as following surgical implantation of foreign bodies, urinary calculus formation, or irradiation.3–6 Since the 1960s, the chemical carcinogen N-butyl-N-(4-hydroxybutyl)-nitrosamine (BBN) has become one of the most reliable methods for carcinogen-induced autochthonous bladder cancer induction in rodent models. 7 In addition, the development of genetic engineering techniques in the 1990s and 2000s enabled the creation of transgenic mouse models, including those using the Cre-Lox or adenovirus-Cre systems for targeted gene manipulation. 8 Advances in these techniques continue to expand the catalogue of available genetically engineered mouse models (GEMMs) of bladder cancer. 9
Transplanted tumor models of bladder cancer are created by implantation of tumor cells or tissue directly into bladders (orthotopic transplantation) or other anatomic sites (heterotopic transplantation) of recipient animals. The increasing number of molecularly annotated human and rodent bladder cancer cell lines has contributed to the widespread use of transplanted tumor models. Many human and rodent urinary bladder cancer cell lines are commercially available and represent different origins, grades, and stages of urothelial carcinoma. Human bladder cancer cell lines can be transplanted into immunodeficient mice to enable study of clinically relevant human tumor genetic features and therapies, as recently exemplified by a study by Mokkapati and colleagues. 10 However, certain aspects of tumor biology are lost when human-derived bladder tumors are grown in an immunodeficient host; for example, the absence of a native immune microenvironment limits investigation into tumor-immune interactions. Conversely, implanting rodent-derived tumor models into syngeneic immunocompetent recipient rodents preserves tumor-immune dynamics and allows for study of immune-directed therapies. However, the translational relevance of these rodent-derived models may be limited by species differences in tumor biology and uncertainty in evaluating drugs that are designed to specifically target human proteins.11,12
Human and rodent transplanted bladder tumor models can be established via heterotopic or orthotopic introduction. Subcutaneous models, whereby tumor cells are injected directly under the skin, are the most common heterotopic models and are widely utilized given their simplicity, reproducibility, and cost-effectiveness. These models allow for tumor monitoring through external measurements and imaging; however, they fail to recapitulate the bladder's unique microenvironment, limiting the ability to study bladder-specific tumor growth, progression, and metastases. Other heterotopic sites include more physiologically relevant sites such as the renal capsule or peritoneum. While these models provide richer tumor-stroma interactions compared to subcutaneous models, they still do not fully recapitulate bladder-specific biology. 13 Orthotopic models involve tumor establishment directly within the recipient rodent bladder and therefore more faithfully represent the relevant tumor environment. Nevertheless, the technical complexity, variability in tumor establishment rates, reliance on advanced imaging for monitoring, and higher costs of orthotopic tumor models underscore the necessity of selecting the tumor transplant approach that best aligns with the specific research objectives.
Orthoptic bladder tumor models can be established surgically, transurethrally, or via ultrasound-guided injection into the bladder. The surgical approach is the most invasive, requiring a lower abdominal incision to directly expose the bladder, but allows for precise injection of tumor cells into the bladder wall under direct visualization.14,15 In contrast, the transurethral method involves catheter insertion through the urethra into the bladder lumen, through which a preconditioning agent is typically first administered prior to introduction of tumor cells into the bladder lumen.16,17 The ultrasound-guided approach employs real-time bladder imaging via ultrasound to visualize and direct transabdominal injection of tumor cells into the bladder wall. 18
In this review, we evaluate these common orthotopic bladder tumor implantation techniques, summarize trends over time, and identify associations among implantation technique, implanted models, and experimental outcomes. Furthermore, based on key findings and limitations identified by our analyses, we optimized a surgical implantation approach to improve efficiency and reproducibility of an orthotopic preclinical model of bladder cancer.
Evidence acquisition
Study selection
PubMed was utilized to search the available published literature over a period spanning 2013 through 2023 for articles pertaining to orthotopic implantation of tumors into mice or rat bladders for research purposes. The search was performed using the query “(bladder cancer) AND (orthotopic) AND (mouse model)”. A total of 254 studies were identified using this approach. Two authors (TH and VD) independently reviewed each study and filtered using the PRISMA approach (Figure 1). 19 Studies were included if they reported data on orthotopic tumor implantation rate (Supplementary Table 1). Other data including metastatic rate, overall survival in implanted mice and/or rat, the primary aim of the study, strain of mouse and/or rat, and bladder cancer cell line details were collected when available.
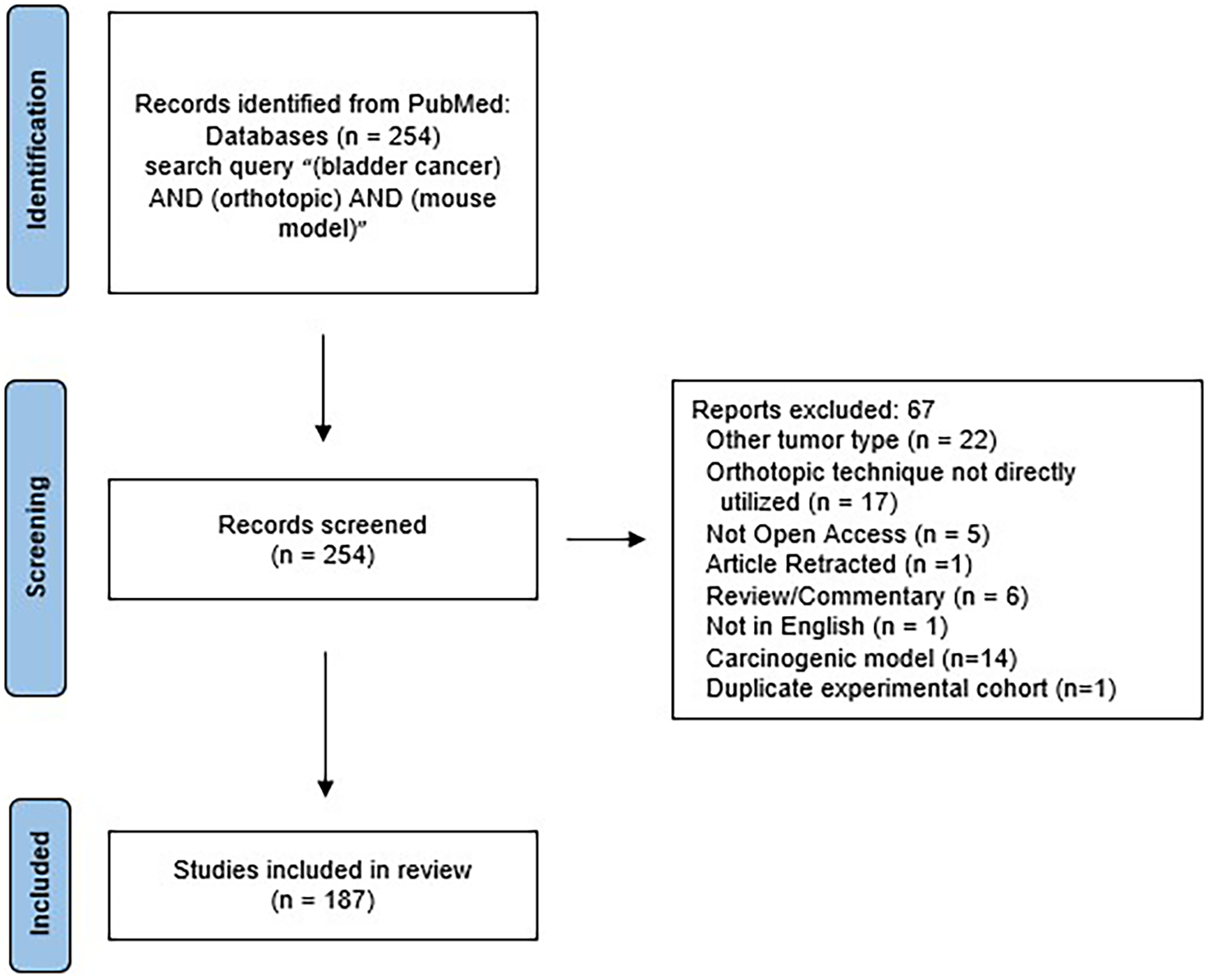
PRISMA study outline.
Orthotopic surgical implantation
All experiments were performed in accordance with the Dana-Farber Cancer Institute (DFCI) IACUC guidelines at the DFCI Longwood Center Animal Resource Facility under an approved protocol. Six-week-old female C57BL/6 (JAX 000664-B6) and NuJ (JAX 002019) mice were purchased from Jackson Laboratories. Mice were anesthetized using isoflurane. The fur overlying the lower abdomen was clipped and the surgical field was prepared with Betadine and alcohol swabs. The mouse was then moved to the sterile surgical field and placed in a supine position on a surgical chuck with heat support provided via a warming pad. The anesthesia nose cone was securely placed, and limbs were secured with surgical tape. The skin and abdominal wall were incised through a ∼1 cm lower midline incision, and forceps were used to expose the bladder. Urine was expelled from the bladder by gentle pressure. A 1:1 mixture of Matrigel (Corning) and bladder cancer cell line suspension in PBS was created with a final concentration of ∼2 × 106 cells in 10 microliters. Cell lines tested were BBN963, 20 a mouse bladder cancer cell line and KU19-19, 21 a commercially available human bladder cancer cell line which has been modified to stably express luciferase. 22 Curved forceps were used to grasp the edge of the bladder and guide a 30 gauge needle into the muscle wall and the cell mixture was gently injected. The bladder was then placed back into the abdomen and the abdominal wall was closed with 4-0 Vicryl sutures and the outer skin was closed with wound clips. Mice were administered subcutaneous Ethiqa XR in the neck tuft for analgesia following surgery and then placed in a clean cage with a heating pad and monitored until fully awake. Following tumor cell injection, bladders were imaged every three days by ultrasound using a Vevo 3100 instrument as well as by bioluminenscence (BLI) using an IVIS Spectrum 2 In Vivo Imaging System (Revvity; for KU19-19 model) . Tumor growth data was analyzed using GraphPad PRISM v10. Unpaired Student's t-tests were used to compare groups and p < 0.05 was considered to be statistically significant. At necropsy, bladders containing orthotopic tumors were harvested and fixed as 5 µm formalin-fixed paraffin embedded (FFPE) tissue sections mounted on glass slides. Sections were stained with hemotoxylin and eosin (H&E) using standard protocols and evaluated at the Dana-Farber Cancer Institute (DFCI) pathology core facility.
Evidence synthesis
Orthotopic tumor Introduction techniques
Transurethral tumor instillation was the most common orthotopic tumor introduction method reported between 2013 and 2023 and was used in 72% of studies (Figure 2A). Surgical implantation and ultrasound-guided injection were used in 22% and 6% of reported studies, respectively (Figure 2B). The tumor take rate was significantly lower in studies using the transurethral instillation approach compared to the ultrasound-guided approach (p = 0.016), but not the surgical approach (Figure 2C). Female C57BL/6 and female athymic mice were the most commonly used recipient strains for orthotopically transplanted mouse and human cell lines, respectively (Figure 2D).
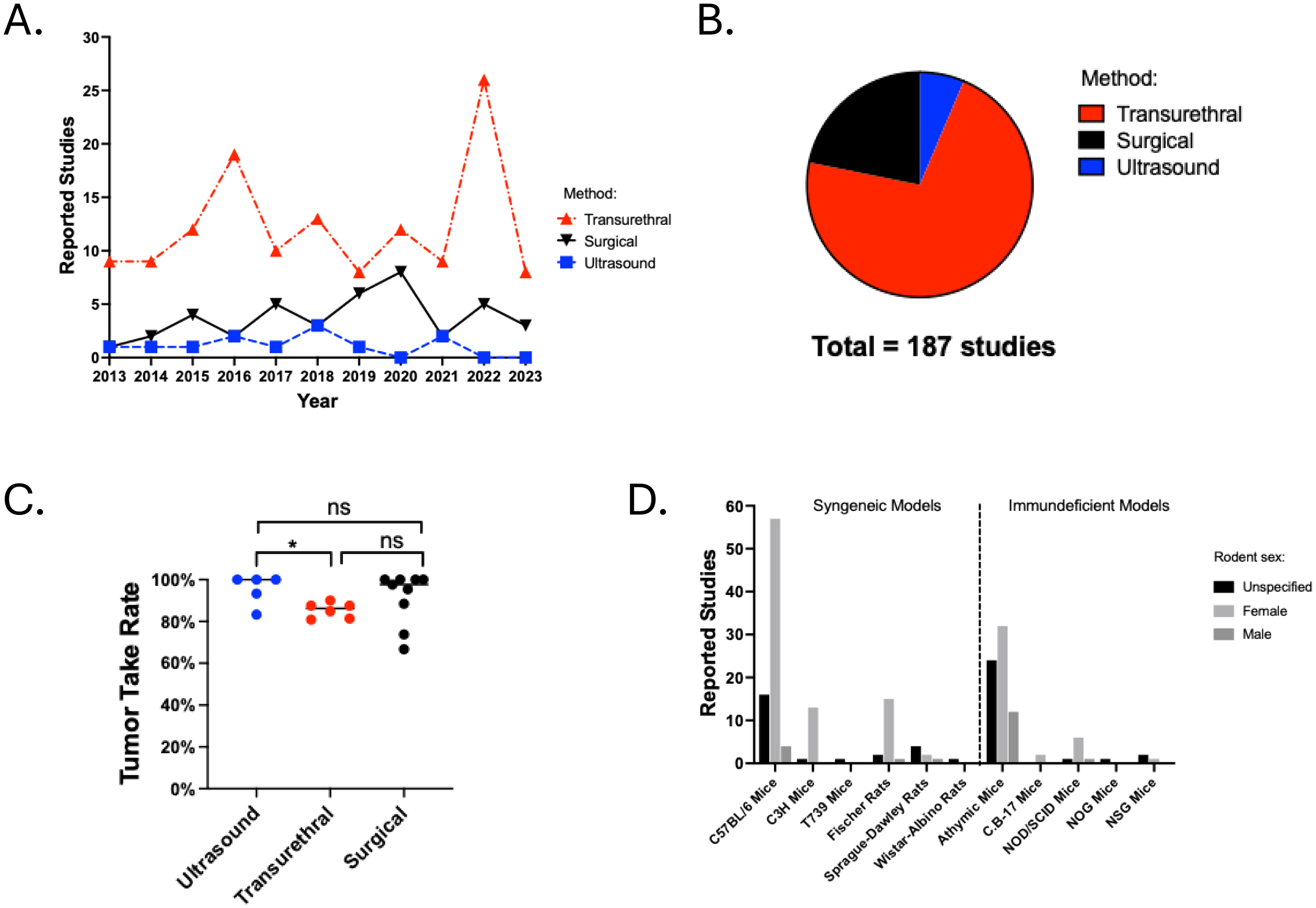
Orthotopic techniques reported during the study period. A. Number of published studies using each orthotopic technique across the study timeline. B. Relative frequency of each orthotopic technique reported in the study. C. Tumor take rate for each orthotopic technique. D. Mouse and rat strains used for orthotopic tumor studies. *p < 0.05; ns, non-significant.
Cell line models
UMUC-3 was the most frequently used human bladder cancer cell line across all orthotopic methods (Figure 3A). Take rates were greater than 75% across methods for all reported cell lines except UMUC-3 using the surgical approach (Figure 3B). UMUC-3 exhibited a notable difference in tumor take rate across orthotopic methods: 100% with ultrasound, 81% with transurethral implantation, and 67% with surgical implantation, highlighting that take rate for a given model may depend on orthotopic technique (Figure 3B). MB49 was the most commonly used mouse bladder cancer cell line across all three orthotopic methods and demonstrated take rates of greater than 80% using both the transurethral and surgical approaches (Figure 3A, B). Tham et al. have reported an optimized protocol for establishing MB49 orthotopic tumors via a transurethral technique. 23
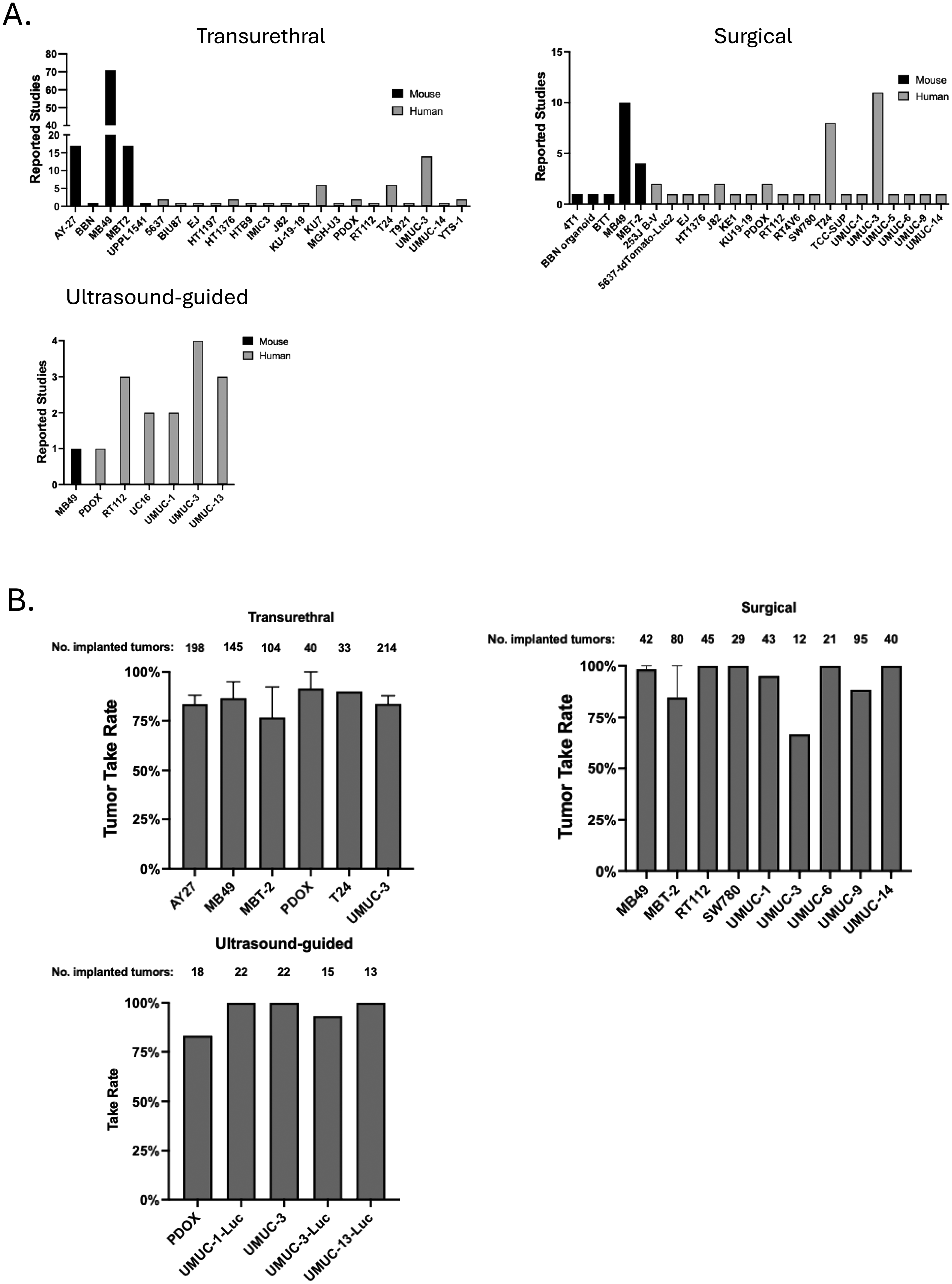
Usage and take rates for orthotopic techniques. A. Number of reported studies across models and techniques. B. Average tumor take rates for each model delivered via each orthotopic technique. Error bars represent standard deviation when results from more than one study was available.
While UMUC-3 and MB49 were the most commonly used human and mouse cell line models, respectively, UMUC-3 had more than twice the number of studies reporting metastatic spread compared to MB49 (Figure 4A). In total, 18 of 29 studies utilizing UMUC-3 report metastases, a higher percentage than any other cell line. Across all cell line models, the most frequent sites of metastases were the lymph nodes, lungs, and liver (Fig. 4B; Supplementary Table 2).
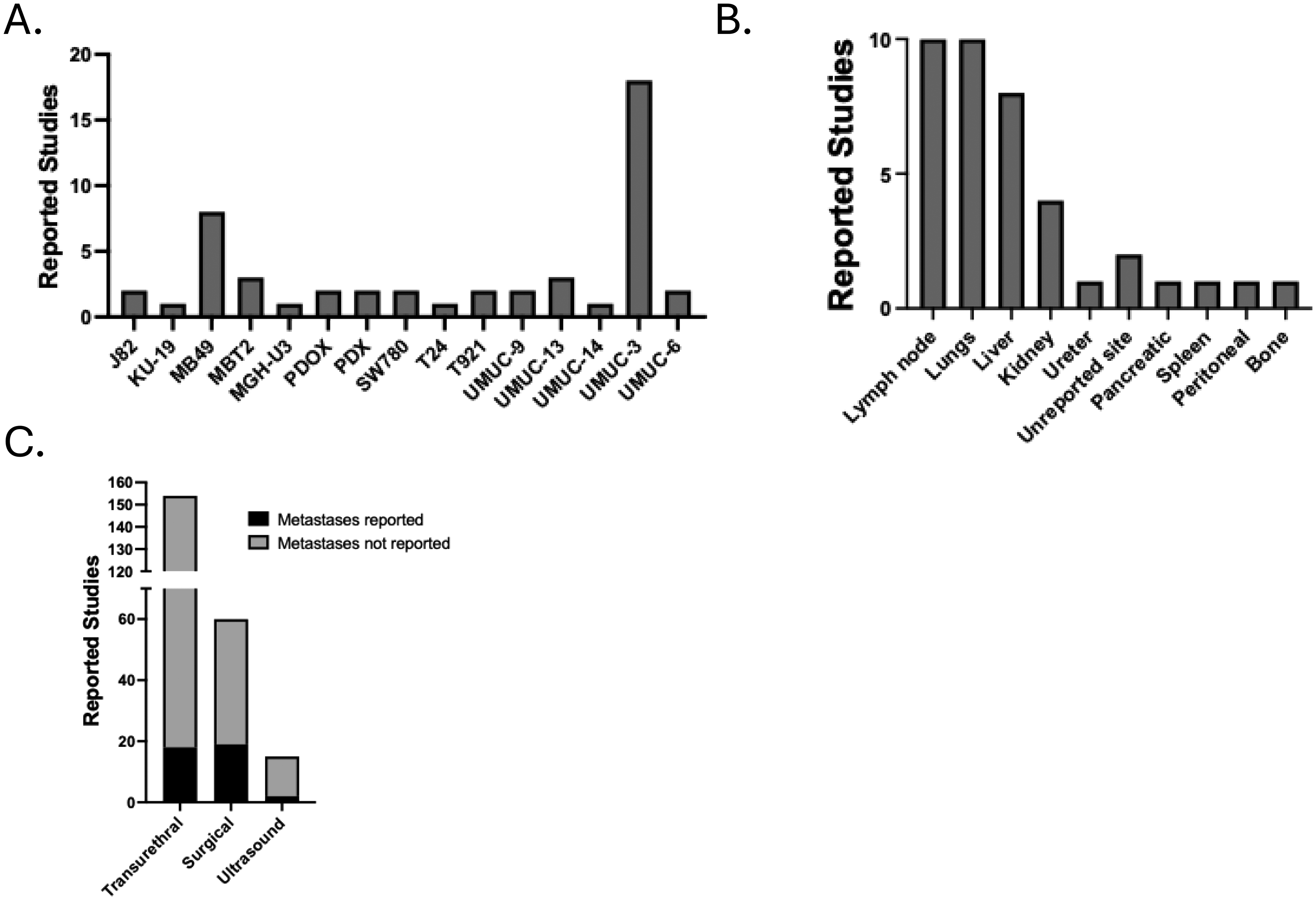
Metastatic properties of orthotopic techniques. A. Number of studies reporting metastasic spread across models. B. Anatomic location of metastases across reported studies. C. Fraction of studies reporting metastases across orthotopic techniques.
Metastatic spread was reported in 12% of studies using the transurethral installation technique, 32% of studies using the surgical implantation technique, and 13% of studies using the ultrasound-guided implantation technique (Figure 4C). The surgical implantation approach disrupts the native tissue planes which delinate the abdominal compartments and lymphatic drainage system to a greater extent than the ultrasound-guided or transurethral methods, perhaps leading to greater potential for development of metastatic disease.
Transurethral instillation preconditioning methods
To establish a bladder tumor via transurethral introduction, a preconditioning agent is typically used to induce bladder inflammation and promote tumor cell attachment and invasion into the bladder wall. Poly-L-Lysine (PLL), as described by Ninalga and colleagues in 2005, 24 was the most common agent used to induce inflammation for the transurethral instillation approach, followed by trypsin and other approaches (Figure 5). Miyazaki et al. investigated the impact of these preconditioning injury methods on tumor characteristics in rat models using the AY-27 cell line and found that PBS or acid-base conditioning is more likely to induce the formation of large, highly invasive tumors, whereas PLL tends to produce smaller, more superficial tumors. 17
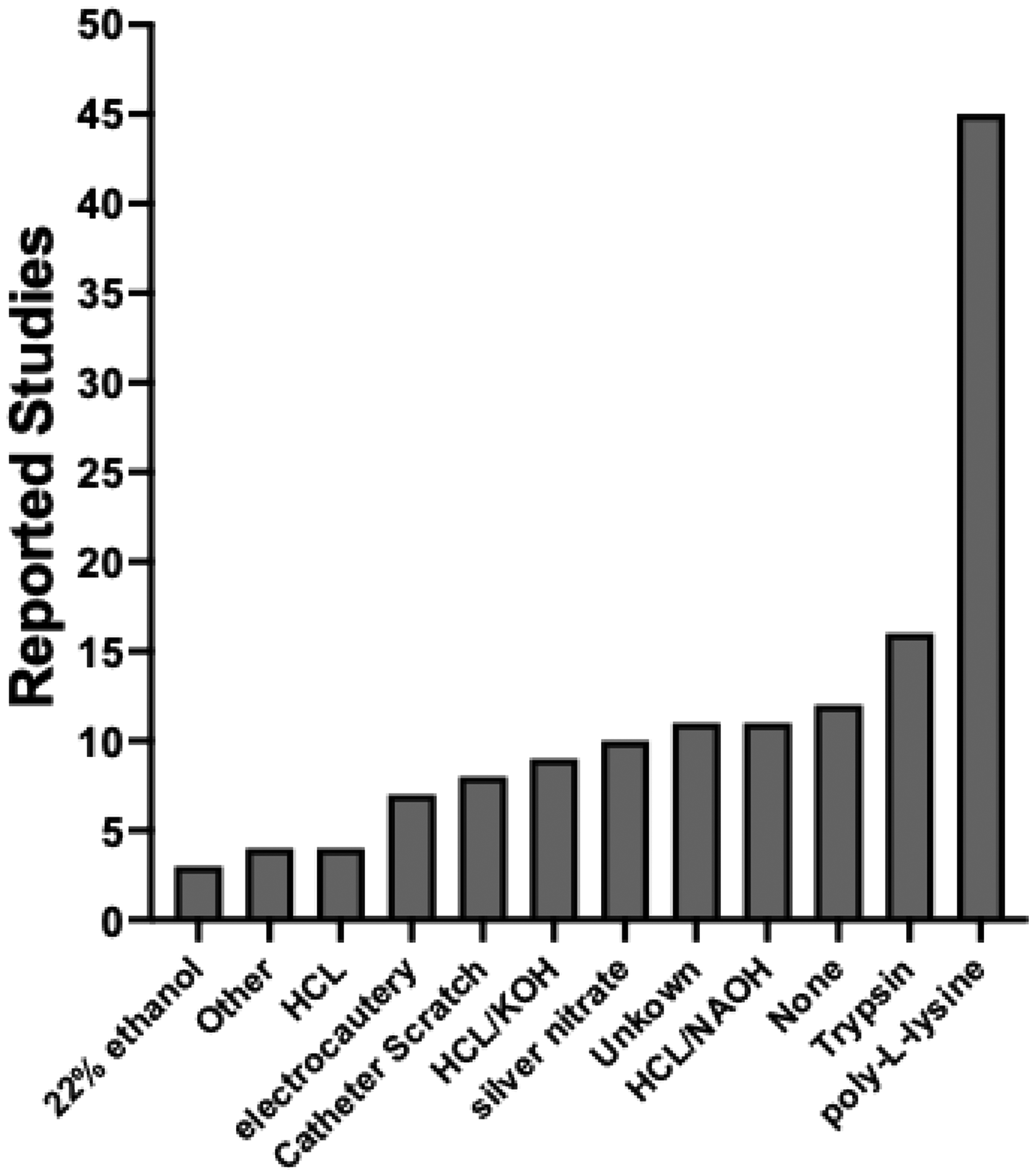
Bladder preconditioning methods used in reported studies of the transurethral instillation approach.
Optimization of a surgical orthotopic tumor implantation method
We initially sought to optimize both the ultrasound-guided and surgical orthotopic tumor implantation techniques for bladder cancer models within our laboratory. Although the ultrasound-guided approach demonstrates a high tumor take rate in published studies, we were less reliably able to establish tumors using this approach and therefore elected to focus the majority of our efforts on the surgical approach.
In optimization of the surgical approach, we initially attempted injections into the bladder wall with fully distended bladders by using a transurethral angiocatheter to control intraluminal volume. Initial attempts were made to perform the procedure with male and female mice; however, female mice were preferentially selected to minimize technical complexity during protocol optimization, given that catheterization in males is procedurally more challenging. However, we ultimately found that injections were easier without a catheter in the lumen and with an empty bladder, as this allowed for injection into a thicker bladder wall without interference from the catheter (Figure 6A). Additionally, we determined that injection volumes greaer than 10 microliters were more challenging due to the small size of the mouse bladder. We also began using curved forceps rather than traditional straight forceps as we found the curvature provided a more natural path for guiding the needle during injections. As we became more proficient with the technique, we were also able to decrease the average surgical time per animal from approximately 19 min to 10 min (Figure 6B). A step-by-step overview of our surgical orthotopic tumor implantation approach is provided in Supplementary Movie 1.
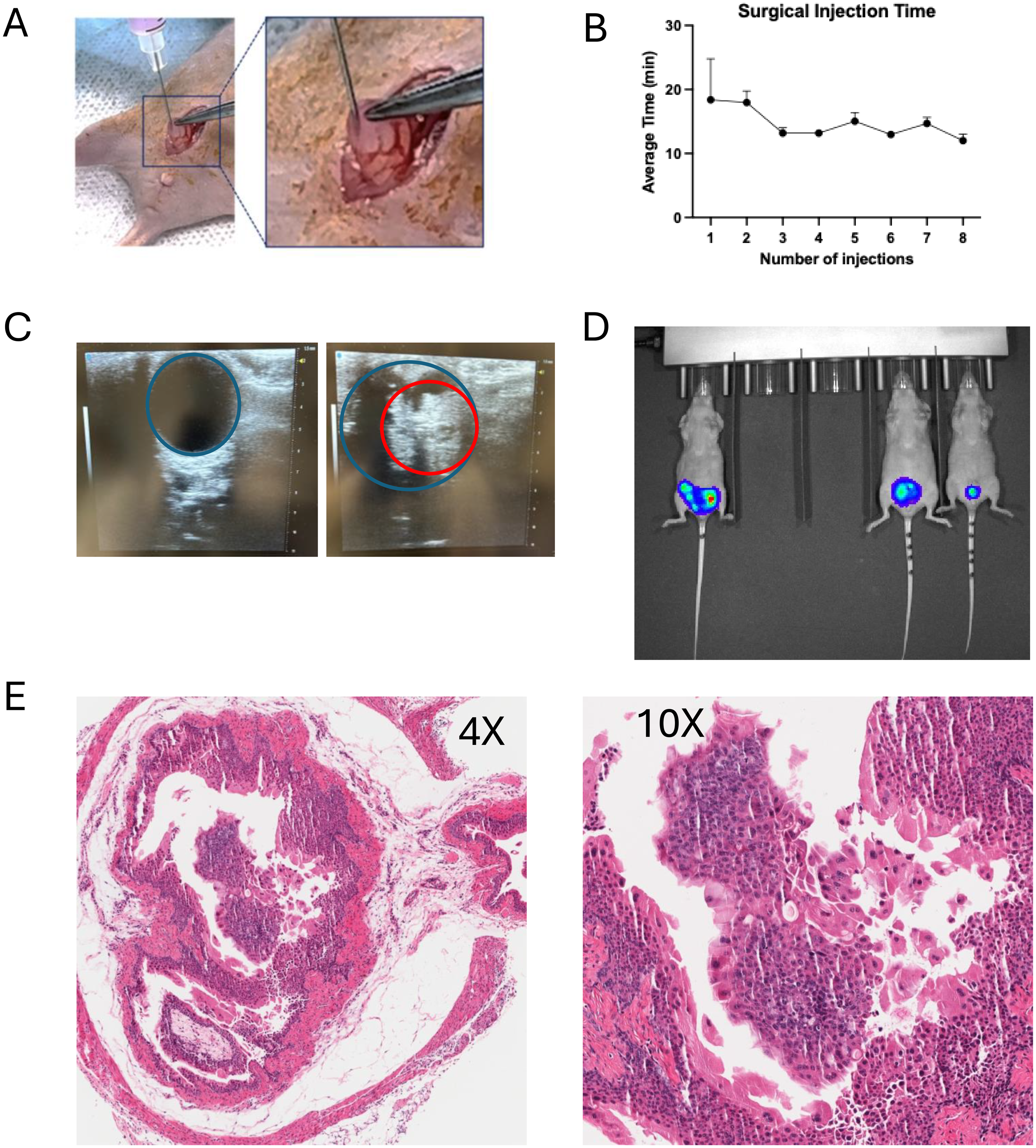
Optimization of a surgical orthotopic tumor implantation approach. A. The tumor cell suspension is injected into the bladder wall under direct visualization. B. Average surgical time for two surgeons (TH and VD) across surgery (injection) number. C. Ultrasound images of mouse bladder (blue) prior to tumor cell injection (left) and following tumor (red) establishment (right). D. Bioluminescence imaging of luciferase-tagged KU19-19 orthotopic tumors. E. Hemotoxylin and eosin (H&E) stained 4X (left) and 10X (right) images of tumors from mouse bladders harvested 23 days following surgical orthotopic implantation of KU19-19 cells.
Using our optimized approach, the survival rate following orthotopic surgical implantation was 98%: 38 of 39 C57BL/6 mice injected with BBN963 cells and 14 of 14 NuJ mice injected with KU19-19 cells completely recovered from surgery and survived at least 1 week following surgery. KU-19-19 tumors were monitored using BLI and BBN963 were monitored using ultrasound (Figure 6C, D). Mice were sacrificed when they reached a morbidity endpoint or at 12 weeks following surgery. At necropsy, 18 of 25 (72%) BBN963-implanted mice and 11 of 14 (79%) KU19-19-implanted mice had gross tumor present within the bladder. H&E staining of harvested tumors demonstrated tumor cells within the bladder wall and lumen (Figure 6E).
Conclusions
Preclinical models are critical tools for understanding tumor biology and testing new therapeutic agents in oncology. In bladder cancer, discoveries made using preclinical models have directly contributed to the remarkable advances in therapy options and clinical outcomes over the past decade. 25 Orthotopic bladder tumor models play an important role in preclinical development as they provide a platform for studying bladder cancer within its native environment. To better understand the landscape of orthotopic models and techniques, we performed a comprehensive review of the published literature from 2013 through 2023. We identified trends in usage as well as differences in relevant features such as tumor take rate and metastatic potential.
Each approach to orthotopic bladder tumor models carries distinct advantages and disadvantages. For example, the transurethral method is minimally invasive, but can be technically challenging in male mice due to the longer urethra. In contrast, surgical implantation allows for precise control of tumor location but is more invasive and requires close recovery monitoring. Finally, the ultrasound-guided injection enables real-time visualization yet depends on specialized imaging equipment and operator proficiency.
The orthotopic technique must be tailored to the specific objectives of the research study. The transurethral approach may be best-suited to study non-muscle invasive bladder cancer (NMIBC) biology and therapies given that tumor cells are introduced within the bladder lumen and thus initially form non-invasive tumors, whereas the surgical and ultrasound-guided approaches involve direct injection of tumor cells into the muscular wall of the bladder and therefore may be more appropriate for modeling muscle-invasive bladder cancer (MIBC). Models with greater propensity for metastatic spread (such as UMUC3) may be favored for studies focusing on tumor migration and dissemination.
Our work has several important limitations. We chose to focus on a contemporary period and therefore changes in technique usage or outcomes prior to 2013 were not captured. Additionally, the reported studies varied in design and reported endpoints, and therefore not all data types were available for all studies. Finally, our experimental efforts focused on the surgical approach, but it is possible that similar (or superior) outcomes may be achievable using either the transurethral or ultrasound-guided approach. Continued innovation in orthotopic blader tumor modeling will contribute to advancing bladder cancer research and therapeutic development.
Supplemental Material
sj-xlsx-1-blc-10.1177_23523735251409720 - Supplemental material for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization
Supplemental material, sj-xlsx-1-blc-10.1177_23523735251409720 for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization by Timothy Hanlon, Vincent D. D’Andrea, Isabella Stelter, Raie Bekele, Yuzhen Zhou and Kent W Mouw in Bladder Cancer
Supplemental Material
sj-pptx-2-blc-10.1177_23523735251409720 - Supplemental material for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization
Supplemental material, sj-pptx-2-blc-10.1177_23523735251409720 for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization by Timothy Hanlon, Vincent D. D’Andrea, Isabella Stelter, Raie Bekele, Yuzhen Zhou and Kent W Mouw in Bladder Cancer
Supplemental Material
sj-docx-3-blc-10.1177_23523735251409720 - Supplemental material for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization
Supplemental material, sj-docx-3-blc-10.1177_23523735251409720 for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization by Timothy Hanlon, Vincent D. D’Andrea, Isabella Stelter, Raie Bekele, Yuzhen Zhou and Kent W Mouw in Bladder Cancer
Supplemental Material
sj-mp4-4-blc-10.1177_23523735251409720 - Supplemental material for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization
Supplemental material, sj-mp4-4-blc-10.1177_23523735251409720 for Orthotopic bladder cancer preclinical models: Comprehensive review and technique optimization by Timothy Hanlon, Vincent D. D’Andrea, Isabella Stelter, Raie Bekele, Yuzhen Zhou and Kent W Mouw in Bladder Cancer
Footnotes
Acknowledgements
We thank the staff at the Dana-Farber Cancer Institute Animal Resource Facility for their assistance and expertise, William Kim (University of North Carolina, Chapel Hill) for the BBN963 cell line, and Inna Khodos (Memorial Sloan Kettering Cancer Center) for helpful discussions regarding orthotopic techniques.
Ethics statement
All experiments were performed in accordance with the Dana-Farber Cancer Institute (DFCI) IACUC guidelines at the DFCI Longwood Center Animal Resource Facility under an approved protocol.
Consent to participate
N/A
Consent for publication
N/A
Author contributions
Made a significant contribution to the concept: TH, VDD, KWM
Made a significant contribution to the design: TH, VDD, RTB, YZ, KWM
Made a significant contribution to the acquisition: TH, VDD, IS, YZ
Made a significant contribution to the analysis: TH, VDD, KWM
Made a significant contribution to the interpretation of data: TH, VDD, IS, RTB, YZ, KWM
Drafted the article: TH, VDD
Revised article critically for important intellectual content: TH, VDD, IS, RTB, YZ, KWM
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the United States National Cancer Institute (NCI) grant R01CA289304, R01CA279221, and R01CA272657 to KWM.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Please contact the corresponding author Dr Kent Mouw (kent_mouw@dfci.harvard.edu) for questions related to the manuscript or to request data, samples, or models that were not included in the published article or supplementary material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
