Abstract
Background
Neighborhood Deprivation (ND) indices serve as indicators of socioeconomic status and are recognized determinant of survival across multiple cancer types.
Objective
To determine the potential effect of ND on urothelial bladder cancer survival outcomes using a California-specific measure, the Healthy Places Index (HPI).
Methods
We queried our institutional database for patients who underwent radical cystectomy from 2013 to 2019. Patients were categorized into HPI quartiles based on their residential areas, with Q1 representing the most disadvantaged and Q4 the least disadvantaged. Multivariable logistic regression was used to identify predictors of adverse pathological features. Multivariable cox regression was utilized to evaluate the association of HPI with overall survival (OS) and recurrence free survival (RFS).
Results
A total of 729 patients were included in our analysis. Belonging to the most disadvantaged quartile (Q1) was associated with higher odds of lymphovascular invasion (HR 1.94, p < 0.001), and extravesical disease (HR 1.87, p < 0.001) at presentation, compared to the least deprived quartile (Q4). Survival analyses showed that worse ND was independently associated with worse OS (Q1 vs Q4: HR: 1.76, 95% CI: 1.18–2.62, p = 0.006) after adjusting for age, extravesical disease, node-positivity, and lymphovascular invasion. OS was positively associated with the education, economic, and transportation subdomains of HPI in univariable analyses.
Conclusions
Greater ND is associated with adverse pathology on presentation and worse OS following radical cystectomy for UBC. Larger prospective studies are warranted to further assess the influence of social disparities on survival outcomes following RC.
Introduction
Socioeconomic status (SES) has been established as a source of ongoing disparities in cancer survival, with urothelial bladder cancer (UBC) being no exception.1–3 SES can influence health through multiple mechanisms, including higher exposure to risk factors such as smoking and limited physical activity, 4 as well as delays in seeking care, resulting in higher stage disease at the time of diagnosis. 5 This potentially higher stage at presentation accentuates the devastating influence of cost of care, particularly for those with lower SES, as it has been previously shown that treating metastatic bladder cancer incurs an extra lifetime cost of $150,012 compared to newly diagnosed organ confined disease. 6 Therefore, an understanding of how social determinants of health (SDoH) affect UBC diagnosis and outcomes is critical to improve care and reduce costs for both patients and healthcare systems. However, individual-level SES depends on variables such as income and education levels which can be challenging to use as it requires information that is not routinely gathered in patient visits. As a result, most studies rely on community-level neighborhood deprivation (ND) measurements as a surrogate for individual-level SES.7–8
ND is an attempt to quantify various factors of disparities in SDoH which are related to the place of residence by utilizing a deprivation index. A deprivation index is an ordinal scoring system to rank levels of ND in one geographic location compared to others.9–11 Previous studies have investigated the possible role of ND in surgical complications, demonstrating an association between greater deprivation and worse outcomes.12,13 However, national-level indices may overlook important regional modifiers, potentially under- or overestimating the effects of individual determinants that directly influence health - such as systematic bias in calculating relative wealth urban populations.14,15 Therefore, it is essential to validate and implement state-level indices to more accurately assess the impact of SDoH on oncological outcomes.
In this study, we used a state-specific deprivation index based on regional census data to evaluate our single institutional cohort, while controlling for longitudinal shifts in demographics due to major economic events. We hypothesized that greater ND, as measured by the California Healthy Places Index (HPI), is independently associated with adverse pathological features at the time of cystectomy (i.e., extravesical disease, presence of lymphovascular invasion, and lymph node positivity), higher short-term complication and readmission rates, and ultimately worse recurrence-free survival (RFS) and overall survival (OS). To the best of our knowledge, this is the first study to utilize the California-specific HPI to evaluate the impact of SDoH on survival outcomes in patients with UBC within a Californian cohort.
Methods
Study design and population
Our prospectively maintained, IRB-approved radical cystectomy (RC) database was queried to identify patients who underwent an open or robotic RC with curative intent between 2013 and 2019. We selected this study period because HPI values could be reliably calculated using the 2015–2019 census data, which also coincided with the increased adoption of robotic assisted radical cystectomy and the stabilization of its learning curve and procedural volume. This timeframe allowed us to maximize follow-up, ensuring that a reasonable proportion of patients reached the 5-year mark. Additionally, while socioeconomic indices have been widely studied in the context of COVID-19 outcomes, the pandemic itself likely introduced substantial short-term fluctuations in the subcategories of these indices such as insurance coverage, housing, and transportation, thereby altering HPI values. To minimize this confounding effect, we limited our cohort to the pre-COVID-19 era (2013–2019). Exclusion criteria included patients with distant metastatic disease (M1), RC for reasons other than UBC, concurrent malignancies, and residence outside of California. The rationale for excluding patients with M1 disease was that RC in this population is not performed with curative intent but rather as a salvage procedure to manage symptoms such as severe hematuria or pain.
Demographic and clinicopathologic variables were collected, including age, sex, race, smoking status (never vs. present or previous), Charlson Comorbidity Index (CCI), surgical approach (open vs. robotic), diversion type (orthotopic neobladder vs. ileal conduit vs. continent cutaneous diversion), receipt of neoadjuvant and adjuvant chemotherapy, pathological staging (organ confined [<(y)pT2N0], extravesical [> (y)pT2N0], and lymph node positive [pN + ]), and lymphovascular invasion (LVI). A total of seven surgeons performed the surgeries - two exclusively open, four solely robotic, and one performing both open and robotic RCs.
Neighborhood deprivation index
The HPI (Supplementary Table 1) is a California-specific composite index that combines 23 SDoH measures with regional environmental data, such as the proportion of green space and presence of water contamination. 16 The HPI is subcategorized into eight domains: economy, education, healthcare access, housing, neighborhood conditions, clean environment, social environment, and transportation. The index generates a score from 1 to 100 for each domain, with lower scores indicating greater socioeconomic deprivation in each area compared to other regions of the state, based on census tracts, ZIP codes, and other regional or county-level divisions. The HPI was developed by the California Public Health Initiative to better quantify SDoH across the state of California, inform policy adjustments, and guide resource allocation. The data used to calculate HPI measures are primarily derived from the 2015–2019 American Community Survey of the US Census. Patients’ ZIP codes were extracted from the institutional database, matched to corresponding HPI scores, and categorized into quartiles, with Q1 representing the most disadvantaged and Q4 the least disadvantaged. Prior census data from the American Community Survey have demonstrated that neighborhood socioeconomic conditions exhibit strong decadal persistence, as reflected by the “Year Householder Moved into Unit” metric. This is particularly evident in Southern California, where 50-year tract analyses reveal relatively stable socioeconomic patterns with median tenures of 12 years. Therefore, the association of HPI with zip codes collected at the time of surgery likely remained stable throughout the patient's course of care.17,18
Outcomes
The primary outcomes were overall survival (OS) and recurrence-free survival (RFS), defined as the time in months from diagnosis to death and recurrence, respectively. Secondary outcomes were adverse pathological features on cystectomy including extravesical disease (EV), pN+, and LVI.
Statistical analysis
Categorical and continuous variables were reported as frequency (percentage) and median (interquartile range), respectively. Chi-squared tests were utilized for between-group comparisons of categorical variables. Multivariable (MVA) logistic regressions were used to investigate the predictors of adverse pathology on presentation (i.e., EV disease, pN+, and LVI). Survival analyses were performed using Kaplan-Meier (KM) curves and Log-rank tests. MVA Cox regressions were utilized to adjust for potential confounders. Reverse KM curves were used to estimate the median follow up duration. All statistical analyses were conducted using SAS Version 9.4 (SAS Institute Inc., Cary, NC, USA). A p-value of less than 0.05 was considered statistically significant in all analyses.
Results
In total, we identified 729 patients who underwent RC with curative intent for UBC between 2013 and 2019 with at least 2 years of follow-up (Table 1). The median follow-up time was 3.8 years (IQR: 3.0–5.2 years). The proportion of patients who were followed at three, four, and five years were 77.3%, 45.6%, and 27.6%, respectively. The median age at diagnosis was 71.0 years (IQR: 64–77 years), and most patients (N = 605,83.0%) identified as Caucasian. The distribution of patients across HPI quartiles were: Q1 (most disadvantaged): 180 (24.7%), Q2: 186 (25.5%), Q3: 184 (25.2%), and Q4 (least disadvantaged): 179 (24.6%). As shown in Table 1, there were significant differences regarding the distributions of disease stage, urinary diversion type, and adjuvant chemotherapy administration among the different quartiles.
Demographics and clinicopathological characteristics stratified by HPI quartile.
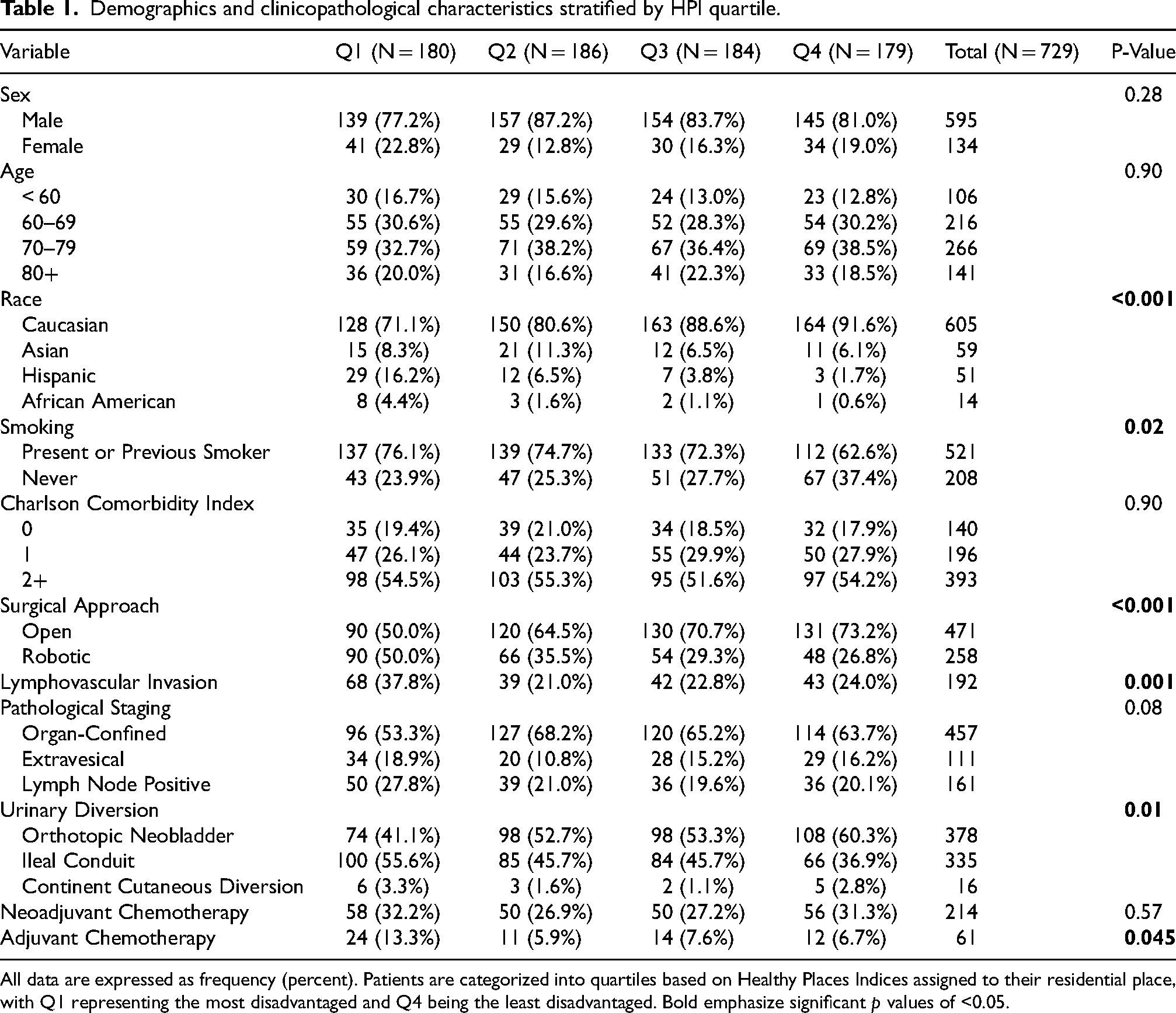
All data are expressed as frequency (percent). Patients are categorized into quartiles based on Healthy Places Indices assigned to their residential place, with Q1 representing the most disadvantaged and Q4 being the least disadvantaged. Bold emphasize significant p values of <0.05.
Our multivariate logistic regressions showed that after adjusting for the potential confounding effects of age, gender, and smoking status, belonging to the most deprived quartile (Q1) was associated with increased odds of EV disease (OR: 1.88, 95% CI: 1.87–2.97, p < 0.001), and LVI (OS: 1.94, 95% CI: 1.21–3.15, p < 0.001) compared to the least deprived quartile (Q4). Additionally, belonging to the second to most deprived (Q2) quartile was also associated with increased odds of EV disease on final pathology compared to the Q4 (Supplementary Table 2). However, the cumulative 30- and 90-day complication and readmission rates were comparable across the quartiles (Table 2).
Postoperative complications stratified by HPI quartile.
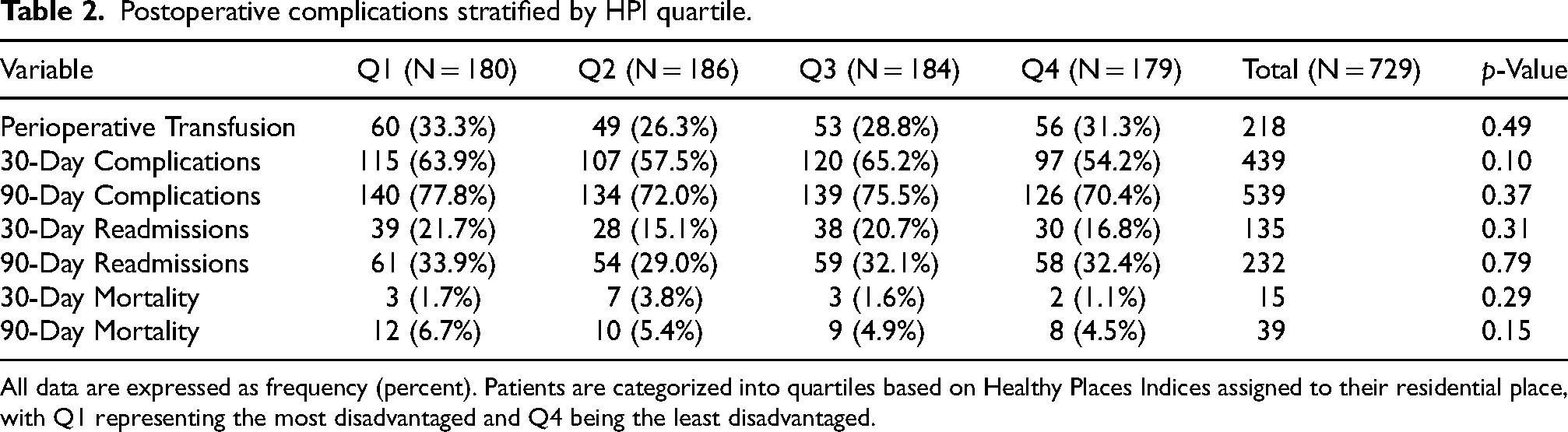
All data are expressed as frequency (percent). Patients are categorized into quartiles based on Healthy Places Indices assigned to their residential place, with Q1 representing the most disadvantaged and Q4 being the least disadvantaged.
The log rank analyses (Figures 1 and 2) showed that the RFS and OS were significantly different among the quartiles with a significant trend towards worse survival in more deprived quartiles (p = 0.012, and 0.011, respectively). On MVA, patients in the most deprived quartile (Q1) had significantly worse OS, with an approximately 76% higher hazards of death compared to those in the least deprived quartile (Q4) (p = 0.006). No significant between-group difference was observed in RFS. Common predictive factors of worse OS and RFS were EV disease, pN+, and LVI on final pathology. Older age and non-orthotopic urinary diversions (i.e., ileal conduit and continent cutaneous diversion) were associated with worse OS (p = 0.003 and p = 0.024, respectively), but not worse RFS (Table 3).
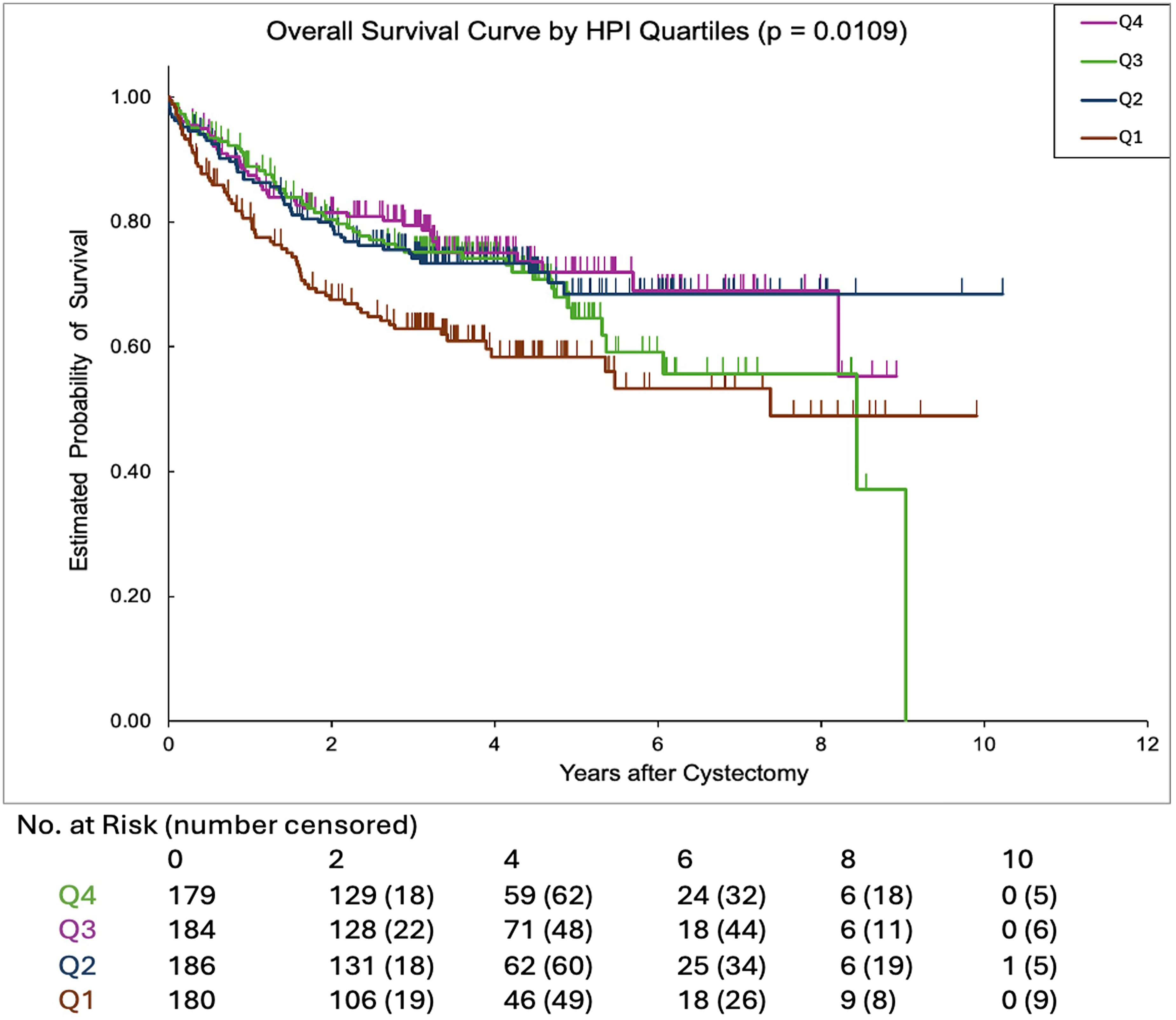
Overall survival in bladder cancer patients stratified by HPI quartiles.
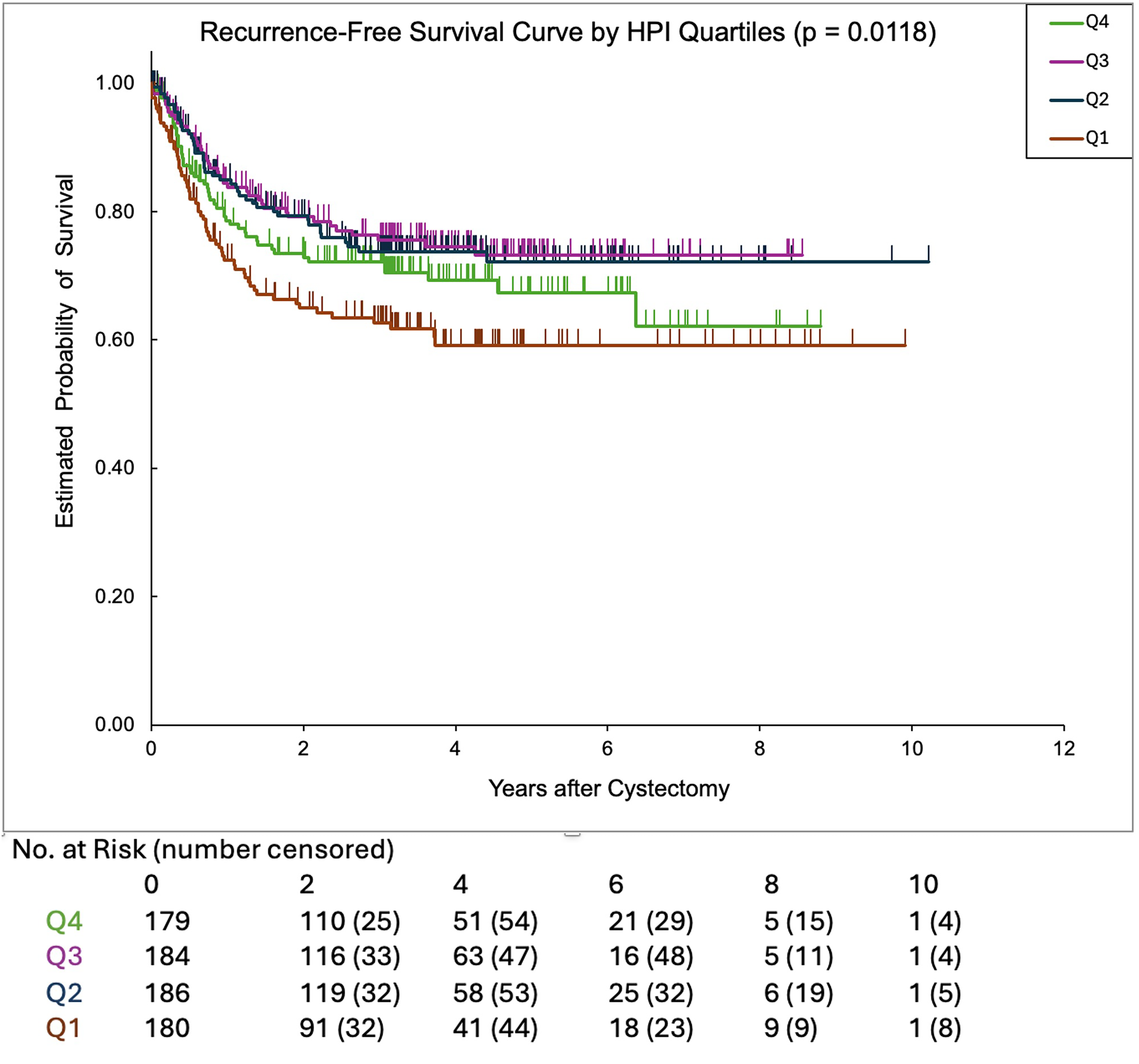
Recurrence-Free survival in bladder cancer patients stratified by HPI quartiles.
Multivariate analysis of variables impacting overall and recurrence-free survival.
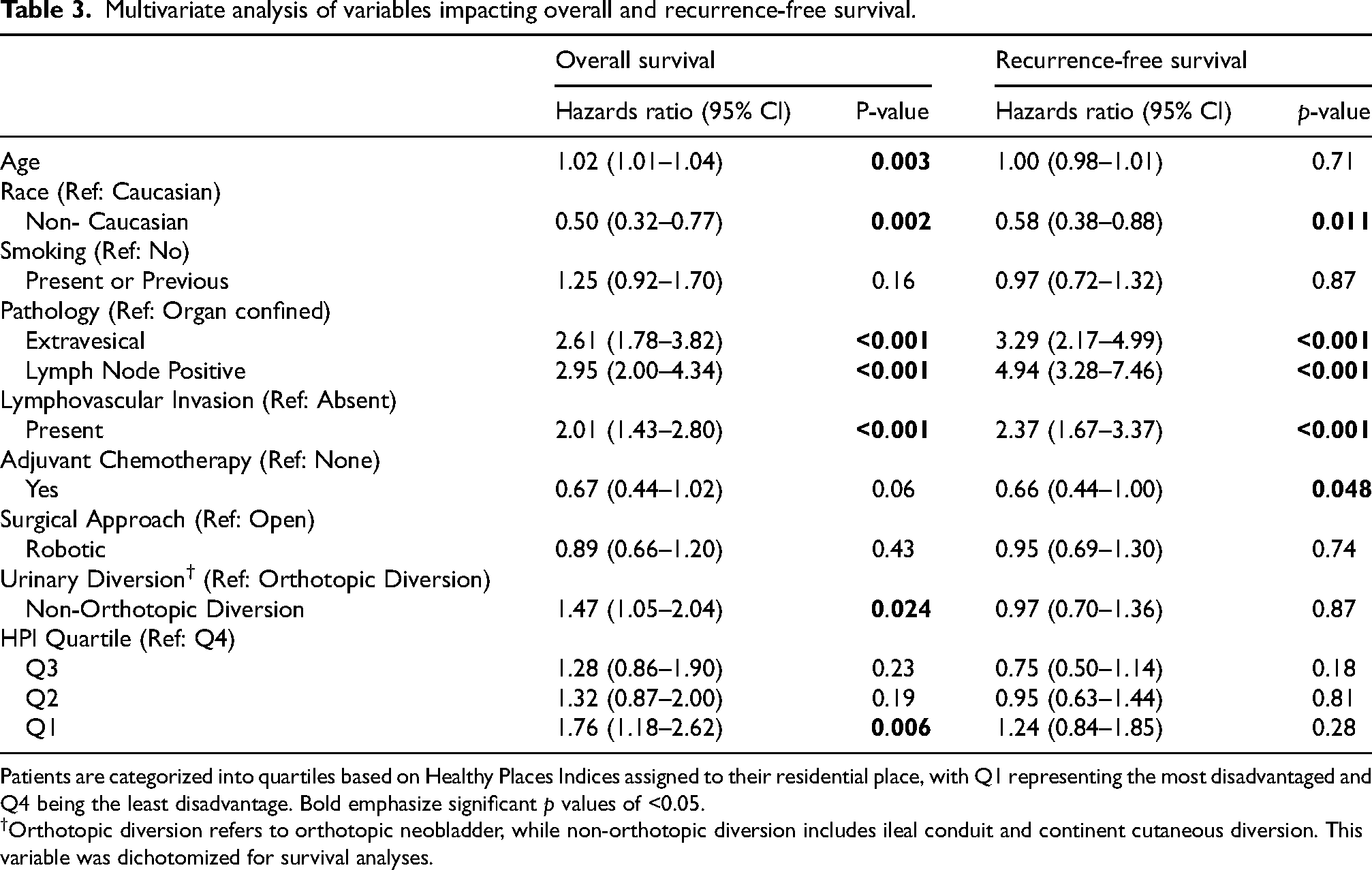
Patients are categorized into quartiles based on Healthy Places Indices assigned to their residential place, with Q1 representing the most disadvantaged and Q4 being the least disadvantage. Bold emphasize significant p values of <0.05.
†Orthotopic diversion refers to orthotopic neobladder, while non-orthotopic diversion includes ileal conduit and continent cutaneous diversion. This variable was dichotomized for survival analyses.
We then assessed the association of the 8 major subdomains of HPI on RFS and OS using univariate cox regressions. As observed in Supplementary Table 3, none of the subdomains showed any statistically significant association with RFS. However, education, economic and transportation subdomains were shown to be significantly associated with OS; with patients in the most deprived quartiles showing poorer OS compared to the least deprived quartiles.
Discussion
Bladder cancer imposes a significant clinical and financial burden on patients and healthcare systems. Socioeconomically disadvantaged patients face additional barriers that may delay timely intervention, hinder follow-up care, and affect outcomes. Prior studies have correlated lower income, educational attainment, occupational status, and Medicaid insurance coverage with more advanced disease at diagnosis and poorer survival outcomes.19–23 In this single institutional effort, we found that greater ND was associated with more advanced disease and adverse pathological features at the time of diagnosis. While the 30- and 90-day complication, readmission, and mortality rates were comparable between the ND quartiles, greater deprivation remained significantly associated with worse OS following RC, after adjusting for potential confounders.
Patients in the most socioeconomically deprived quartile exhibited a 1.76-fold increase in hazards of mortality following RC, consistent with previous studies demonstrating decreased life expectancy among lower SES populations with UBC.22,24 The association between better survival outcomes and higher SES can be attributed to multiple factors including better access to optimal treatment, lower stage of UBC at presentation, less treatment delays, and lower burden of comorbidities.20,22–24 Previous studies have demonstrated that improved survival due to receipt of chemotherapy was governed by SES, with patients of lower SES having less access to neoadjuvant chemotherapy (NAC) or longer time to therapy initiation.19,25,26 In the current study, 29% of patients received NAC, with similar distribution across HPI quartiles, suggesting a paradigm shift in access to neoadjuvant chemotherapy in recent times. However, patients in the most deprived quartile still had worse OS despite supposedly standard treatment at the same facility. These results may reflect other neglected barriers to care, including lower rates of private transportation ownership and education, as suggested by our findings. Additionally, neighborhoods inhabited by lower SES quartiles, are more likely to have a higher proportion of immigrants, a population that may face language barriers and limited access to necessary educational resources beyond economic obstacles. 27 The strong association between HPI and OS, alongside the lack of an association with RFS, suggests that contextual factor (such as access to primary care and transportation, adherence to surveillance, and management of comorbidities) may affect mortality through mechanisms not directly related to cancer biology or recurrence.
Our results also demonstrated that worse SES as measured by the HPI index was associated with higher odds of presenting with advanced stage disease or showing more aggressive features on pathology, after adjusting for potential confounders. Previous studies have demonstrated that there is an association between lower SES with higher stage disease and postulated that this can be a predictor of worse prognosis. 23 Despite the established association between lower SES and higher stage disease, no correlation has been identified between SES and disease recurrence, as also confirmed by our findings.19–21,28 This seemingly paradoxical finding further supports our postulation that although patients with lower SES may present with more advanced disease – likely due to delays - this does not necessarily translate to a higher risk of recurrence or more aggressive tumor biology. Rather, their poorer OS may reflect the broader adverse influences of socioeconomic disadvantages and its downstream consequences.
Our analyses of the 8 HPI subdomains served as further testament to this hypothesis. Although none of the HPI components were associated with RFS, more favorable conditions in the education, economic, and transportation subdomains were significantly associated with improved OS. While the role of economic advantage is relatively clear in better OS, the possible contribution of better education and easier access to transportation may not be easily discernible. However, some evidence suggests that higher educational status correlates with better understanding of the severity of disease and hence better adherence to health advice.29–30 With regards to transportation, a study on all cancer types has shown that better access to transportation is associated with better survival, presumably due to easier commute to the medical center. 31 The practical implication of these findings may lie in the feasibility of mapping a patient's place of residence to their HPI to guide targeted interventions and provide personalized support, such as transportation vouchers or rideshare services, patient navigation and language-concordant education beyond the clinic, proactive primary-care co-management after RC, and social-work outreach for financial or benefits assistance.
This study has some limitations. The retrospective, single-institutional design restricts the generalizability of our study. Although index surgeries predated the COVID-19 pandemic, follow-up for the more recent patients overlapped with it. Reliable data on the short-term effects of COVID-19 on various components of the HPI – such as insurance, housing, economic, and transportation factors – were unavailable and could have influenced our findings. Additionally, location-based HPI values are assigned based on a single time point. While prior evidence suggests that SES remains largely stable over decades, longitudinal changes in the patients’ SES during follow-up are still possible. The median follow-up of 3.7 years supports the adequacy and robustness of follow-up in our cohort; however, the pandemic may have caused some delays. Certain potential confounders, such as smoking intensity (in pack-years) and occupational exposures, were not captured in our database and thus could not be evaluated. Furthermore, previous studies have identified discrepancies between reported ND indices and individual-level social risk factors. 32 Therefore, our results should be interpreted in the context of these limitations. Larger prospective studies are encouraged to further assess the impact of ND on survival in bladder cancer, particularly through the development of individual-level ND indices and inclusion of supplemental insurance status (e.g., Medicare with supplemental Medi-Cal).
Conclusion
The current study underscored the critical impact of SES and ND on the outcomes of UBC patients following RC. Our results indicated that patients from the most deprived quartiles, as defined by the HPI, were significantly more likely to present with advanced stage disease and had significantly worse OS. Our findings emphasize the necessity for more precise and individualized measures of SES in cancer research to accurately capture the multifaceted impacts of social and economic disparities and develop targeted interventions that can mitigate the detrimental effects of socioeconomic deprivation on health.
Supplemental Material
sj-docx-1-blc-10.1177_23523735251406621 - Supplemental material for Impact of neighborhood deprivation on bladder cancer outcomes: A regional analysis
Supplemental material, sj-docx-1-blc-10.1177_23523735251406621 for Impact of neighborhood deprivation on bladder cancer outcomes: A regional analysis by Anosh Dadabhoy, Chirag Doshi, Mazyar Zahir, Farshad Sheybaee Moghaddam, Luis Santos Molina, Sejal Mehta, Erika L Wood, Leilei Xia, Gus Miranda, Jie Cai, Hooman Djaladat, Anne Schuckman and Siamak Daneshmand in Bladder Cancer
Footnotes
Acknowledgements
None.
Ethical approval and informed consent statements
This study was conducted under the Helsinki and Istanbul declarations. It has been ethically reviewed and approved by the IRB of the University of Southern California (IRB No. HS-01B014). All patients had provided written consent prior to undergoing radical cystectomy for their data to be included in the USC Radical Cystectomy Database.
Author contributions
Conceptualization: SD, AD, FSM. Methodology: SD, AD, FSM. Data Curation: GM, JC, SM. Formal Analysis: JC. Resources: SD, HD, AS, ELW, LX. Writing, original draft preparation: AD, CD, SM. Writing: review and editing: MZ, LSM, ELW, LX, HD, AS. Supervision: SD
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interest
Siamak Daneshmand is an Editorial Board Member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
