Abstract
Small cell carcinoma of the bladder (SCCB) is a rare, aggressive malignancy that accounts for less than 1% of all bladder cancers. In this report, we highlight the clinical manifestations of SCCB (including epidemiology, cystoscopic and imaging findings), summarize insights into the molecular mechanisms underlying its pathophysiology, detail current methods of staging, review local and systemic treatment, and explore novel agents currently in clinical development. Most of the regimens used for SCCB treatment are extrapolated from small cell lung cancer, a more common cancer that shares the neuroendocrine and aggressive clinical phenotype of SCCB. Greater preclinical research can help to elucidate pertinent similarities and differences between SCCB and other neuroendocrine cancers as well as reveal new therapeutic targets, while increased participation of patients with SCCB in clinical trials may provide additional treatment options for patients with this aggressive cancer.
Keywords
Introduction
Bladder cancer is the tenth most common cancer worldwide, with an estimated 550,000 new cases diagnosed each year. 1 More than 90% are urothelial carcinoma (UC), some with histologic subtype morphologies (micropapillary, sarcomatoid, glandular, etc.); 2 and some with divergent differentiation including squamous and glandular. Small cell carcinoma of the bladder (SCCB) can exist as a pure small cell phenotype but is most commonly found mixed with UC. SCCB is a rare and aggressive neuroendocrine malignancy accounting for <1% of all bladder cancers. Given its rarity, clinical decisions are largely informed by case reports, retrospective analyses, and extrapolation from related histologies such as small cell lung cancer (SCLC). Here we review current knowledge of the pathogenesis, presentation, workup, and management of SCCB.
Epidemiology
SCCB was first reported in 1981 after neurosecretory granules were identified in an invasive bladder carcinoma. 3 While making up a small proportion of all bladder cancers, incidence of SCCB has been trending upward, increasing from 0.05 cases to 0.14 cases per 100,000 from the 1990s into the 2000s. 4 SCCB affects mainly men (77.8% compared to 22.2% women) with a median age of 73 at time of diagnosis (range 33 to 95 years). The major modifiable risk factor is smoking; 50%–79% of patients report as current or former smokers.5,6
Pathogenesis
SCCB is often mixed with other histologic subtypes, most commonly UC. One report of 81 cases found pure small cell carcinoma in only 1/3 of patients; the remaining patients had a mixture of small cell and other subtypes (Figure 1). 7 This raises the question of SCCB's pathogenesis, specifically whether there is a shared cell of origin. Early genetic studies from bladder tumors with co-existent UC and small cell carcinoma provide a model to assess the clonal origins of each constituent histology. Cheng et al. evaluated 20 patients whose tumors were composed of mixed UC and small cell carcinoma. Microdissection of these histologically distinct areas, followed by analysis of loss of heterozygosity at 5 microsatellite sites and X-chromosome inactivation patterns in female patients, revealed near complete concordance in loss of heterozygosity patterns and X-inactivation between the different histologies within each patient. 8 Other case studies of admixed bladder UC and small cell carcinoma have reported similar results, including concordant TP53 mutations and other microsatellite patterns. 9 These findings of shared genetic alternations across histologies support the theory that small cell carcinoma and UC emerge from a single clone that has acquired genetic mutations that are shared between the UC and small cell components of mixed tumors, with later clonal events (which might be some combination of genetic/epigenetic alterations or transcriptional aberrations) that give rise to an SCCB cell population; this is more likely than the independent and simultaneous emergence of both tumor histologies from different cell types within the epithelium of the genitourinary (GU) tract.
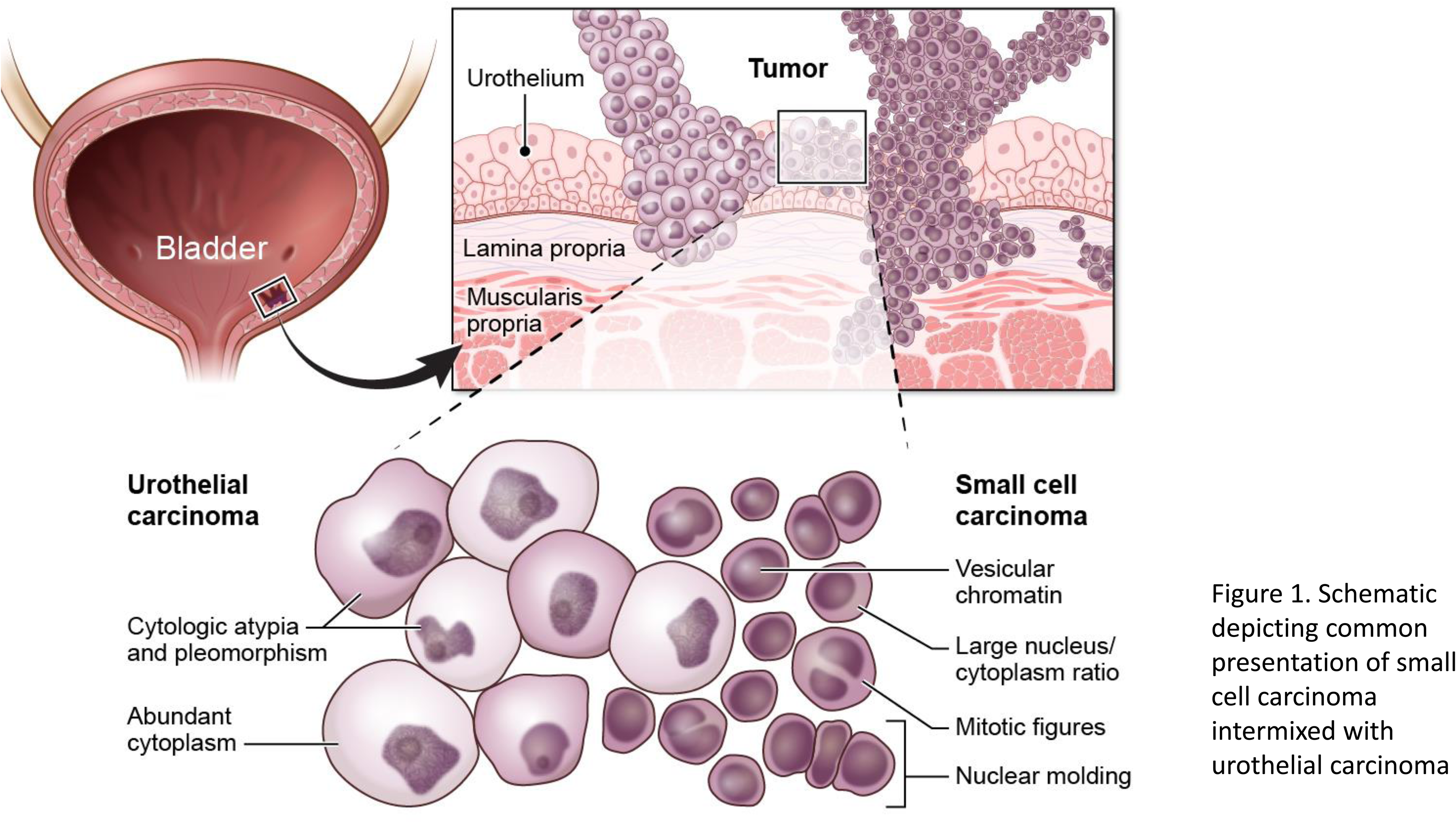
Schematic depicting common presentation of small cell carcinoma intermixed with urothelial carcinoma.
Molecular and genetic characteristics
These early investigations prompted further research into the underlying mechanisms that drive the development of small cell carcinoma and how it differs from UC and other histologies. Genetic studies have repeatedly shown that the most common aberrations in SCCB are the loss of TP53 and RB1, a characteristic shared by small cell carcinomas in other tissues. 10 A recent study by Kamoun et al., which sought to generate a consensus molecular classification and categorization schema for muscle-invasive bladder cancer (MIBC), found 100% TP53 mutations and 67% RB1 mutations among neuroendocrine-like tumors, rates higher than those seen in other MIBC subtypes. 11 Furthermore, a subsequent analysis of an archived database of GU tract malignancies found very high rates of TP53 and RB1 mutations (92% for TP53, 75% for RB1, and 72% for the presence of mutation in both genes) in small cell carcinomas of the GU tract relative to UCs within this database (56% for TP53, 20% for RB1). 12 Additionally, deactivation of TP53 and RB1 in epithelial cells has been found to generate tumors and derivative cell lines with neuroendocrine properties.13,14
While enriched in tumors of small cell/neuroendocrine phenotype vs. other subtypes of UC, pathogenic variation in the TP53 and RB1 pathways is not exclusive for this rare histologic type. The Cancer Genome Atlas (TCGA) and Foundation Medicine database analyses also show that these mutations are neither necessary (given the emergence of a small cell phenotype without both TP53 and/or RB1 loss) nor sufficient for the small cell phenotype (given that tumors can retain a UC morphology despite the presence of these mutations).
In addition to TP53 and RB1 aberrations, mutations in the core promoter region of TERT, the catalytic reverse transcriptase component of the telomerase complex, are commonly seen in cancers of the GU tract. Telomeres become shorter with each round of DNA replication, while telomerase elongates telomeric DNA. The ability to overcome this barrier to unlimited numbers of division, most commonly via reactivation of telomerase, is a well-recognized hallmark of cancer. 15
TERT mutations are commonly observed in GU neuroendocrine tumors. 16 Hoffman-Censits et al. reported a TERT promoter mutation rate of 68% (90/132 samples) among the subset of patients with small cell carcinomas of the bladder/ureter in their query of the Foundation Medicine database and a nearly equal percentage (66%, n = 1113) in urothelial bladder cancers. 12 Other studies of SCCB patients have reported that 55%–100% of tumors tested carried TERT promoter mutations, while no TERT mutations were detected in patients with SCLC or neuroendocrine carcinoma of the prostate.17–19 Increased telomerase activity through TERT promoter mutations likely allows small cell bladder carcinomas to achieve the high replicative capacity, as reflected in its molecular signature of high cell-cycle activity11,20,21 and aggressive clinical behavior.20,21 This further suggests that neuroendocrine tumors that arise from tissues other than the bladder may use other mechanisms to reactivate telomerase and bypass replicative senescence.
In vitro models of neuroendocrine cells demonstrate that expression of neuron-related transcription factors such as ASCL1, NEUROD1, INSM1 are increased. Similar increases in these transcription factors appear to occur in the transition from UC to a small cell bladder phenotype.14,21 Notably, these transcription factors are also activated in neuroendocrine prostate cancer 22 and in SCLC. 23 C-MYC, one of the factors used to induce a neuroendocrine phenotype, is a downstream target of NEUROD1 in lung neuroendocrine lines.23,24 TCGA data support SOX2 and E2F3/SOX4 activation as important events in the neuronal molecular classification within this dataset.20,25 SOX2 has a well-documented role as one of the major transcription factors defining the embryonic stem cell state 26 as well as normal progenitor/multipotent cells, such as neural progenitor cells. 27 SOX2 also contributes to oncogenesis in UC, 28 among many other tumors. 29 A recent publication detailed the ability to subclassify SCCB using the ASCL1, NEUROD1, and POUF2F3 transcription factors in a similar manner as previously published for SCLC. 30 Their analysis highlighted the downstream expression patterns driven by these transcription factors and posited this heterogeneity as a potential contributor to variable responses to therapeutic interventions such as immunotherapy. Furthermore, there was differential expression of targets for antibody-drug conjugate targets NECTIN4, TROP2, CEACAM5, and DLL3.
Mutations in epigenetic regulators are commonly seen in all subtypes of bladder cancer. This underlying epigenetic dysregulation may contribute to the heterogeneity that brings about the neuroendocrine phenotype. 22 UC has high rates of genetic alterations in epigenetic regulators, such as KDM6A, ARID1A, and KMT2C/D, among all molecular subtypes.12,20,31 These findings might represent part of the bladder-specific pathway by which tumors reach a neuroendocrine state. Heterogeneity generated by the complex interplay of epigenetic and small- and large-scale genetic dysregulation may lead to such a phenotype. Tumors that histologically appear as UC but show evidence of neuronal/neuroendocrine transcriptional patterns, such as observed in the initial recognition of a neuronal/neuroendocrine transcriptional program 32 and in subsequent TCGA analysis, 20 are evidence of the intermediate step on the progression to clinically apparent small cell tumors and provide further evidence of their emergence from the same cell of origin as the more prevalent UC.
Spectrum of neuroendocrine disease
Above, we have detailed similarities between SCCB and UC that are classified as neuroendocrine-like. SCCB also exists within the spectrum of neuroendocrine neoplasms (NEN). NENs are heterogeneous malignancies that are divided into well-differentiated neuroendocrine tumors (NETs) and poorly differentiated neuroendocrine carcinomas (NECs), also known as high-grade neuroendocrine carcinomas (HGNECs). 33 Well-differentiated NETs are often slow-growing, and patients can survive many years. Poorly differentiated NECs, such as SCCB, large-cell neuroendocrine bladder cancer, and poorly differentiated NEC not otherwise specified, are high-grade malignances that often progress rapidly and have poor outcomes. 34 Furthermore, NENs are classified histopathologically by the degree of tumor differentiation and the proliferative rate index (%Ki67). There is no well-established grading system for NETs in genitourinary tract. Drawing from the World Health Organization (WHO) classification of gastrointestinal tumors, %Ki67 can be used to subclassify well-differentiated NETs into low-, intermediate-, and high-grade, while NECs are by default high-grade and poorly differentiated (Ki67 index >20% and usually >55%). 35 NECs of the bladder are histomorphologically identical to small cell carcinomas or large cell NECs of the lung and are often rapidly progressive.
While a sizable percentage of NECs originate from the gastrointestinal system (37.4%), a similar number (34.4%) originate from several sites (e.g., lung, head and neck, GU tract). Notably a significant percentage (28.2%) are of unknown primary organ site. 33 Most patients with NECs present with metastatic disease; more than two thirds of gastrointestinal NECs are metastatic at diagnosis.33,36 NECs are characterized by a degree of genomic instability, which is greater in higher-grade disease.37,38
A recent retrospective review of urinary NENs demonstrated the spectrum of neuroendocrine disease within the GU system. 39 A total of 134 patients with urinary NENs were examined, 94 with bladder primaries, 32 with kidney primaries, 2 with ureter primaries, 2 with urethral primaries, and 4 unspecified. Twenty-seven patients (20.2%) had well-differentiated NETs, while 95 patients (70.9%) had poorly differentiated NECs. Notably, 92.6% of the bladder primaries were poorly differentiated while 78.1% of the kidney primaries were well differentiated. The median overall survival (mOS) for the entire cohort was 4.2 years; however, metastatic well-differentiated NETs had a median time to death of 8.2 years (95% CI, 3.5 years–incalculable) compared to 1 year for metastatic poorly differentiated NECs (95% CI, 0.8–1.3 years).
Clinical features
Clinical presentation of SCCB is similar to urothelial bladder cancer, with the majority of patients (68%–90%) having gross or microscopic hematuria.7,40 Other common symptoms include dysuria, urinary frequency, suprapubic discomfort, and urinary tract infections. Patients rarely present with paraneoplastic syndromes such as Cushing's syndrome, which is seen in other endocrine tumors.
A retrospective review of 16 patients with SCCB histopathology evaluated at the National Institutes of Health Clinical Center in the U.S. (Table 1) revealed male gender predominance (88%), a median age of 63 years, and large tumor size at time of diagnosis (1.7–7.4 cm) (unpublished). Tumors were frequently visible on CT imaging at the time of diagnosis, as were nodal or metastatic disease (47%). Lymph nodes, bone, and liver were the most common sites of metastatic disease.
Patient characteristics from a retrospective analysis of 16 SCCB patients treated at the US National Institutes of Health from 2011–2021.

Diagnosis
The diagnostic approach for SCCB is like that of other bladder cancers. Cystoscopy will often reveal fungating or ulcerating lesions mostly located on the lateral and posterior walls. 7 Given the high proportion of patients with metastatic disease at presentation, imaging is also recommended. Staging of SCCB follows the American Joint Committee on Cancer criteria for urothelial and other bladder cancers. 41 Most patients present with locally advanced or metastatic disease, with 36% classified as stage IV at time of diagnosis. 42
Pathology
Diagnosis of small cell carcinoma is based on histopathological examination, as defined by WHO guidelines for SCLC. Microscopically, the tumor cells are usually arranged in sheets, nests, or trabeculae in which nuclear crowding and molding is present. Necrosis can be seen, and mitotic figures are frequently found. The tumor cells have inconspicuous nucleoli, finely stippled chromatin, and scant cytoplasm. Vascular invasion may be present (Figure 2). The majority of SCCB coexists with urothelial carcinoma. Large-cell neuroendocrine carcinoma of the bladder show large polygonal tumor cells with abundant cytoplasm, prominent nucleoli and seen as nests, trabecula or organoid patterns. 43
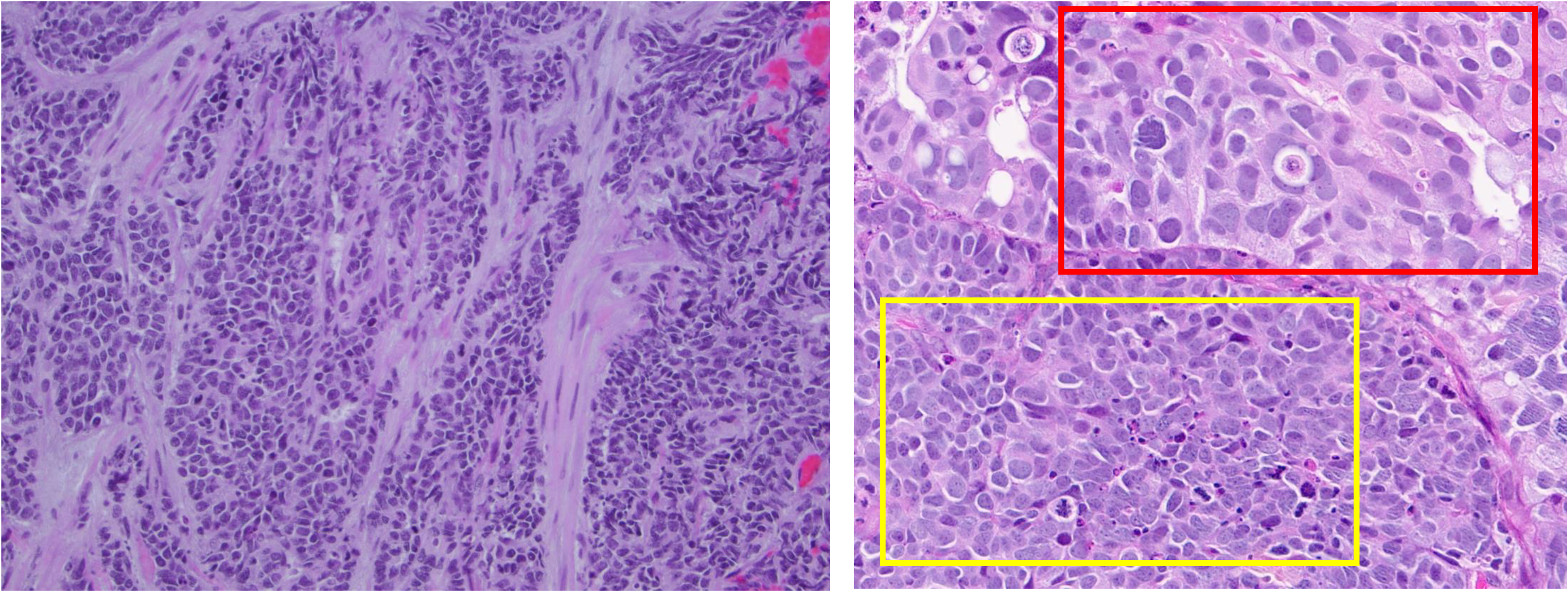
(A) H&E stain of a small cell carcinoma of the bladder. Tumor infiltrates the muscularis propria. Cells are small with scant cytoplasm and prominent nuclei. X200×. (B) H&E stain of tumor with mixed small cell carcinoma (less cytoplasm and high mitotic activity - yellow box) and urothelial carcinoma components (cytologic atypia and abundant cytoplasm - red box) X40×.
Diagnosis of small cell carcinoma is confirmed by immunohistochemistry. The tumor cells are positive for neuroendocrine markers like synaptophysin, chromogranin, INSM1, neuron-specific enolase, and CD56. They are also positive for pancytokeratin, CK8, and CK19, mostly focal and perinuclear dot-like. They demonstrate a high Ki67 proliferation index.
Notably, up to 40% of cases of SCCB stain positive for TTF-1, and the tumor should not be erroneously diagnosed as metastasis from the lung. Although there can be similarities between lung and bladder small cell carcinomas, genomic alterations in small cell carcinoma of the bladder more closely resemble those of UC rather than SCLC.16,18,19 Electron microscopy shows dense neurosecretory granules in the cytoplasm and desmosomes, supporting epithelial differentiation.
There may be significant changes in histological as well as molecular patterns between primary and metastatic sites in SCCB. Tumors often evolve and become predominantly neuroendocrine and poorly differentiated.
Imaging of small cell bladder tumors
The role of imaging in the workup of bladder cancer is to identify MIBC, extravesical involvement, and nodal and distant metastasis. 44 Imaging protocols for the diagnosis and staging of bladder cancer are based on studies primarily evaluating imaging for UC, the most common type of bladder cancer.44–46 CT urography is the most common imaging modality for the diagnosing and staging of UC, with a reported accuracy of 91%. Patients with both UC and SCCB frequently present with hematuria47,48 and therefore undergo an initial evaluation for suspected bladder cancer with cystoscopy and CT urography. At the U.S. National Cancer Institute, CT urography includes non-contrast, and post-intravenous contrast imaging of the urothelial tract and excretory phase images of the abdomen and pelvis. Cystoscopy is crucial for obtaining a tissue diagnosis and for detecting small bladder wall lesions that may be inconspicuous on CT urography. 46
Multiparametric bladder MRI is superior to CT imaging for local bladder tumor staging, specifically for differentiating non-muscle-invasive bladder cancer (NMIBC) from MIBC because of the high soft tissue contrast resolution of MRI.44,45 MRI more reliably distinguishes a bladder tumor from the various layers of the bladder wall needed to determine local staging. 46 Although assessment of bladder wall muscle invasion is important for patient management, assessment of early metastatic disease in SCCB is most critical because of its highly aggressive behavior. 6 CT imaging, preferably with urography, is often preferred for SCCB as it is most widely used in bladder UC. Its accuracy is 40%–92% for detection of perivesicular invasion and 40%–85% for diagnosis of lymph node metastasis. 44
Bladder tumors with small cell carcinoma histopathology cannot be readily distinguished from other forms of bladder cancer based on CT imaging, possibly due to the tumors’ frequently observed mixed histopathology.47,48 As with non-SCCB, an SCCB mass may present as asymmetric or focal bladder wall thickening and as a soft tissue mass protruding into the bladder lumen (Figure 3, Images 1–3).
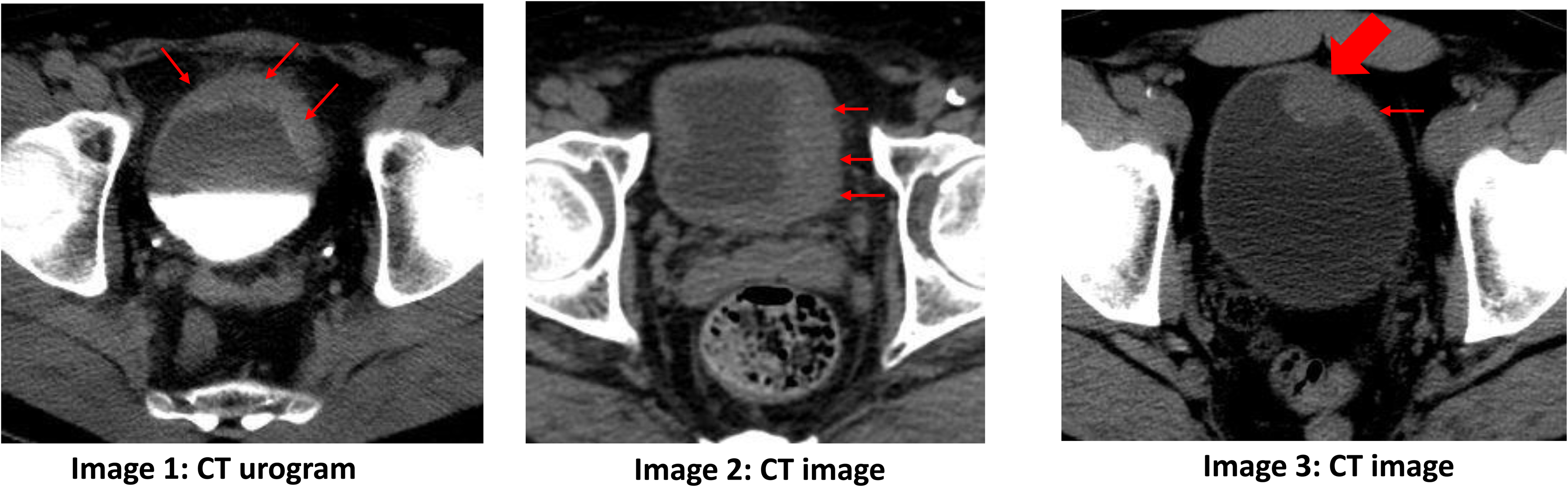
Three different presentations of small cell carcinoma of the bladder. Images 1 and 2 demonstrate asymmetric bladder wall thickening (thin red arrows). Image 3 demonstrates a soft tissue mass (thick red arrow) protruding into the bladder lumen and contiguous asymmetric bladder wall thickening (thin red arrow).
Patients with SCCB are at increased risk for early metastatic disease and rapid progression 6 despite treatment. Therefore, surveillance, restaging, and assessment of therapy response with CT imaging is essential. Imaging can demonstrate tumor characteristics, including initial vigorous response to systemic treatment and/or rapid interval growth without systemic treatment (Figure 4, Images A—D). MRI can evaluate brain metastasis, which is more common with small cell carcinoma of the lung than of the bladder. 49 One series of 409 patients with SCCB found that only 6 (1.5%) had brain metastases at any time during their disease course. 50 However, the incidence may be higher, and clinicians should consider obtaining a baseline brain MRI prior to initiating systemic therapy.
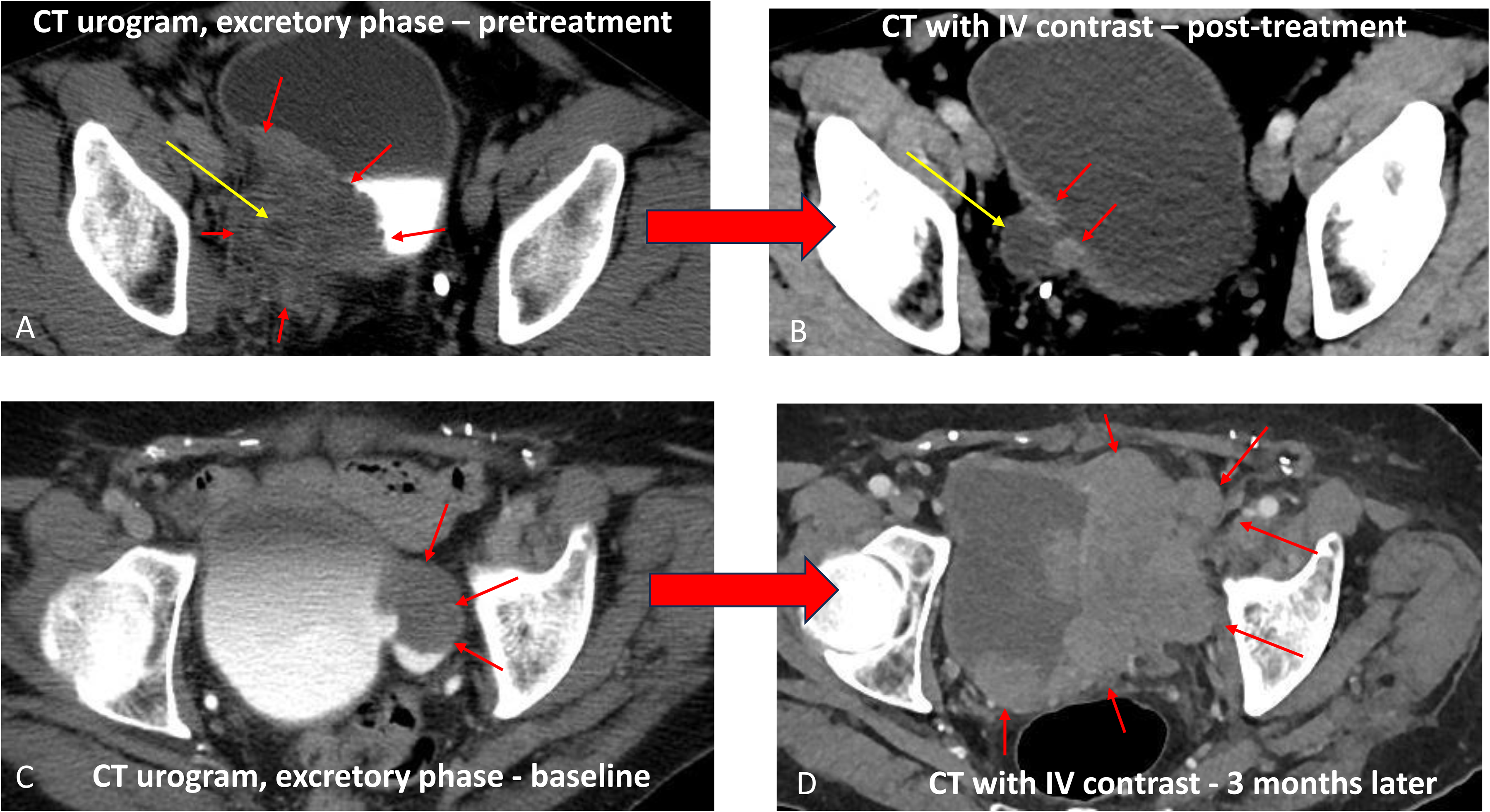
Axial CT images A and B are from a 49-yo male with SCCB. Image A depicts a large tumor mass encasing the right ureterovesical junction (short red arrows), including a right bladder wall diverticulum (long yellow arrow). A significantly decreased tumor burden (short red arrows) is noted on follow-up. Image B, 7 months post-treatment indicating a robust response to initial systemic treatment. Axial CT images C and D are from an 81-yo male with SCCB. Image C demonstrates tumor, manifested as a large filling defect, largely confined to a left lateral wall bladder diverticulum (red arrows). Image D, obtained after initial tumor resection but no interval systemic therapy, depicts a 3-month follow-up CT image demonstrating marked interval tumor growth (red arrows) into the bladder lumen, the bladder wall, and the left pelvis perivesicular soft tissues.
Small cell bladder carcinoma and PET imaging
Positron Emission Tomography (PET)/CT can identify and evaluate functional tumor behavior based on radiotracer properties, with 18F-FDG as the predominant imaging agent. Molecular imaging studies often commingle SCCB with other cancers because of its extreme rarity. There are few publications on PET imaging in bladder cancer, and they rarely include data on imaging of SCCB.51–54 Most SCCB cases present with distant metastasis. 55 Tumors are typically large, broadly polypoid, and located in the lateral bladder wall (54%), posterior wall (20%), trigone (10%), dome (8%), or anterior wall (8%). 6 Characteristic of its tumor class, SCCB is aggressive and poorly differentiated, resulting in high glucose metabolism manifesting as strong 18F-FDG avidity 56 (Figure 5). Because 18F-FDG is excreted through the GU tract, accurate evaluation of the bladder is unreliable but may still be useful.57–59 This complements earlier work on the value of 18F-FDG in the staging and restaging of extrapulmonary small cell carcinoma. 53 Merging different modalities, 18F-FDG PET/MRI also demonstrates potential benefit in detecting SCCB metastasis. 54 Other PET radiotracers such as 11C-choline and 11C-acetate have been investigated in bladder cancer, but with no compelling results and a very short half-life, future clinical applications are limited. 18F-FDG PET has clinical merit in the evaluation of SCCB and could be useful in therapeutic decisions if the technology is available. The rarity of this disease makes robust PET/CT studies difficult to achieve, but existing information is promising. Newer PET radiotracer agents with different tumor targets have additional imaging potential. [68Ga]FAPI binds to cancer-activated fibroblasts in the tumor microenvironment and has demonstrated superiority to [18F]FDG in detecting metastatic lesions in advanced bladder cancers and high-grade UC and, therefore, should be explored in SCCB.60,61 Successful functional imaging paves the way for future targeted radionuclide therapy, encompassing theranostic applications that would be beneficial in this difficult disease.
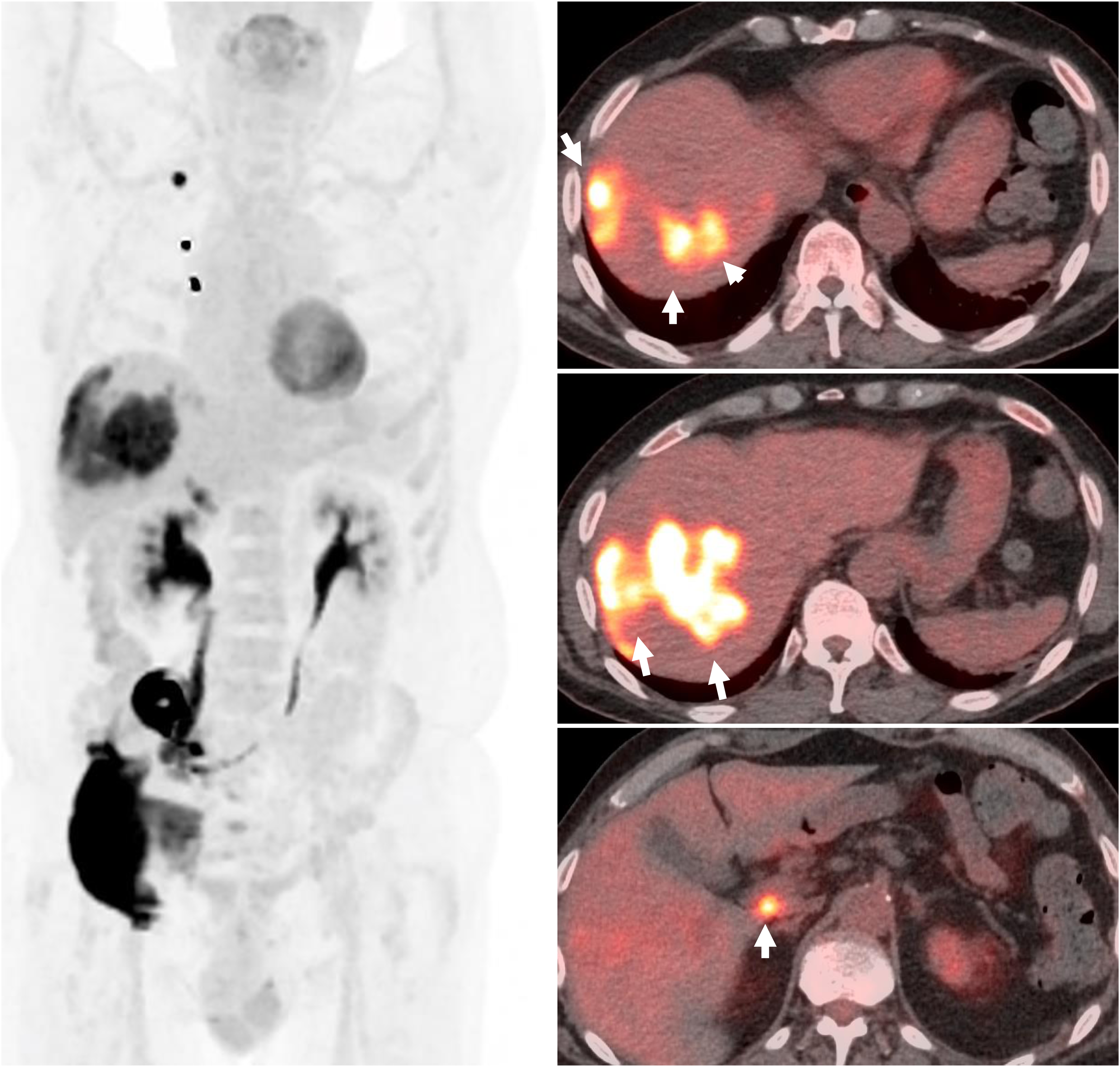
82-yo M with metastatic small cell carcinoma of the bladder. 18F-FDG-PET maximal intensity projection and fused PET/CT images demonstrate multiple hypermetabolic liver metastases and a porta hepatis lymph node (arrows). Tracer contamination is noted at the anterior right chest due to Port-a-Cath injection.
Treatment
SCCB can present as either pure small cell or admixed with other histologic subtypes. While pure vs admixed cases are not always clearly reported, patients with any component of small cell have worse outcomes compared to patients with urothelial carcinoma histology. 62 Data are mixed concerning the prognosis of patients with pure small cell vs small cell admixed with other histologies.7,63–65 Current NCCN guidelines recommend treating pure small cell and admixed small cell similarly. 66 EAU guidelines state that mixed histologies that are predominantly urothelial should follow pure UC treatment recommendations, while patients with predominant non-urothelial differentiation, such as small cell carcinoma, should follow separate treatment algorithms. 67 It is our practice to treat patients with any component of small cell with small cell regimens and not UC regimens.
Localized disease
SCCB, unlike UC, tends to present in the advanced or locally advanced stage; therefore, the reported incidence of SCCB NMIBC is relatively low. Large national datasets, such as the SEER database, report a small number of patients with SCCB. In the SEER database from 1991–2005, 78/642 patients (12%) were reported to have SCCB NMIBC, while more recent data from the National Cancer Database (NCDB) showed that 177/856 patients (21%) had SCCB NMIBC.4,68 Similarly, larger institutional series report that 5%–10% of SCCB patients present with NMIBC; however, these results are rendered unreliable by the fact that most series do not distinguish NMIBC from non-metastatic MIBC.6,7,69,70
Multiple strategies have been used to treat SCCB NMIBC. However, a standard therapy for this disease has not been established due to the paucity of data and clinical trials. Transurethral resection of bladder tumor (TURBT) is the most common surgical strategy as strong data support its use in urothelial and other NMIBC histologies.71,72 However, combination therapy with either adjuvant radiotherapy or neoadjuvant platinum-based chemotherapy has been reported along with use of more radical extirpative techniques such as partial or radical cystectomy (RC) with or without radiotherapy or chemotherapy.49,69,73 This is likely due to the concern for occult higher-stage disease, which has been well-documented in patients with MIBC and ≤cT2 disease.74,75 Given the biologic similarities of SCCB and SCLC, similar therapeutic strategies such as single or combination adjuvant or neoadjuvant radiotherapy or chemotherapy have been employed.6,74 Unfortunately, clinical staging of SCCB NMIBC is a poor indicator of overall pathologic staging, as review of the NCDB data shows that 50% of patients with ≤cT1 disease who underwent RC were understaged, and 21% of these patients were found to have node-positive disease. 75 Though high upstaging may suggest that more radical surgical therapy is appropriate in lower stages of the disease, multiple analyses of the SEER dataset and smaller case series show conflicting evidence of whether RC has improved outcomes over multimodality bladder-sparing approaches.42,69,76
There have been no prospective comparisons of treatment modalities in SCCB NMIBC; therefore practitioners must rely on retrospective datasets, the limitations of which cannot be overstated. Given the lack of data on the benefit of adjuvant therapy or more extirpative therapy (partial or radical cystectomy) in SCCB NMIBC, organ-sparing approaches such as TURBT remain the mainstay of treatment. More directed work studying outcomes only in patients with pathologic NMIBC is needed to develop a standard of care.
As described above, the traditional distinction between NMIBC and MIBC in SCCB is muddied by understaging and poor correlation between stage and metastatic potential. For this reason, SCCB is often categorized as localized or metastatic disease, like SCLC. Current National Comprehensive Cancer Network (NCCN) guidelines recommend some combination of either chemoradiation or neoadjuvant platinum-based chemotherapy followed by local treatment with RC or radiation therapy for localized disease. A recent retrospective analysis reported the outcomes of 147 patients with non-metastatic SCCB, 77 108 of whom underwent RC. Of the 39 who did not undergo RC, 16 (41%) received chemotherapy only, 14 (36%) received definitive chemoradiation, and 9 (23%) received no cancer-directed therapy due to poor performance status. For the 108 who did undergo RC, 71% received neoadjuvant chemotherapy (NAC) while 13% received adjuvant therapy. Those who underwent NAC + RC had the longest mOS (14.5 years), followed by chemoradiation (6.2 years) and RC + adjuvant chemotherapy (5.4 years). For those who underwent RC only the mOS was 1.2 years, for those who had chemotherapy only the mOS was 0.7 years, and for those who underwent no treatment the mOS was 0.6 years. These results, while retrospective, support the use of NAC in localized disease and are similar to other retrospective series with similar findings.74,78–80
When used, NAC is generally a platinum-based chemotherapy regimen, most often a small cell type regimen (i.e., etoposide and platinum) rather than urothelial type regimens (i.e., gemcitabine-platinum or MVAC).77,81 One of the few prospective studies done in SCCB examined the use of an alternating doublet of ifosfamide plus doxorubicin (IA) with etoposide plus cisplatin (EP) 65 in patients with surgically resectable disease. In this prospective phase II study, 18 patients with ≤ cT4aN0M0 received 4 cycles of IA-EP (2 IA and 2 EP) with downstaging seen in 78% and a median OS of 58 months.
Locally advanced and metastatic disease
SCCB's rarity results in a lack of disease-specific clinical trials with substantial power to alter practice-based guidelines. Most data on treatment of locally advanced and metastatic disease come from case reports, retrospective studies, institutional experience, and early-phase trials.82,83 Metastatic SCCB is treated like SCLC with platinum and etoposide chemotherapy regimens. Carboplatin can be substituted for cisplatin where toxicity is a concern.
As noted above, a phase 2 trial explored the use of alternating doublets of IA-EP in the neoadjuvant setting. 65 An additional 12 patients with unresectable or metastatic SCCB were also treated. Eight patients had clinical CRs and 3 patients had PRs, for an ORR of 92% and an mOS of 13 months. A phase 2 trial of ifosfamide, paclitaxel, and cisplatin (TIP) in advanced non-urothelial bladder cancer enrolled one SCCB patient with recurrent perirectal lymph node disease 4 years after radical cystoprostatectomy. 83 The patient achieved a CR but later died from a second primary pancreatic adenocarcinoma about 4 years after completing TIP.
As with most cancers, treatment dogma has evolved with the advent of immunotherapy in SCLC but has not yet been translated to SCCB. There are several case series/reports using various PD-1/PD-L1 checkpoint inhibitors, including atezolizumab, nivolumab, and pembrolizumab.84–87 A phase 2 study of durvalumab and tremelimumab in metastatic non-urothelial urinary tract carcinoma included 7 patients with small cell urinary tract disease. 88 All had progressive disease (PD); however, one patient had PD per RECIST but was perceived to have a clinical benefit (mixed response) with decreasing liver metastasis and remained on treatment 22 months after initiating therapy. A recent phase 1b study looked at the frontline combination of pembrolizumab with cisplatin-based therapy in patients with high grade neuroendocrine tumors of the GU tract, including 7 patients with stage III-IV SCCB. 89 The combination was found to be safe and tolerable, and demonstrated an early efficacy signal with an ORR of 43% (95% CI 0.06–0.80) and a 24-month PFS rate of 86% (0.63–1.00) and 24-month OS rate of 86% (0.63–1.00).
A phase 1 trial of the tyrosine kinase inhibitor cabozantinib and nivolumab alone or with ipilimumab included 3 small cell patients with one responder and one PD. 90 This concept has been brought forward in the ICONIC trial (NCT03866382), which is currently ongoing and includes an SCCB cohort. Several case reports have published the use of lurbinectedin, an alkylating agent approved for SCLC in the post-platinum setting, in a patient with SCCB.91,92 Recently, MD Anderson Cancer Center published an abstract on the use of lurbinectedin in patients with metastatic high-grade GU neuroendocrine tumors, including 7 patients with bladder primaries. Of these 7 patients, there was 1 CR and 2 PRs, with another 2 patients having stable disease. 93 A current trial is exploring the use of lurbinectedin, with or without avelumab, in the second-line setting for patients with SCCB and other HGNECs of the urinary tract (NCT06228066). Other ongoing trials currently enrolling SCCB patients are listed in Table 2.
Ongoing trials enrolling SCCB patients.
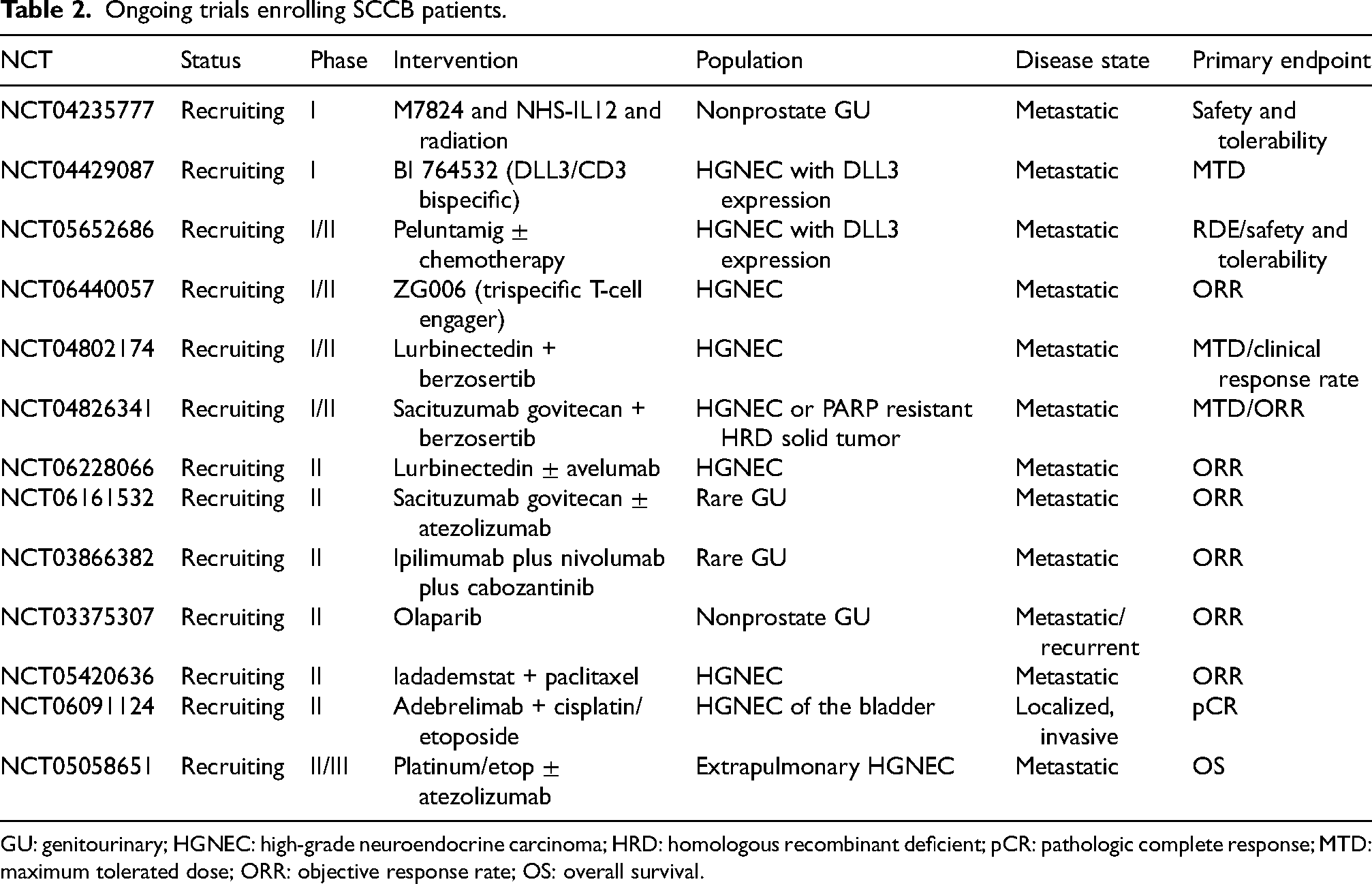
GU: genitourinary; HGNEC: high-grade neuroendocrine carcinoma; HRD: homologous recombinant deficient; pCR: pathologic complete response; MTD: maximum tolerated dose; ORR: objective response rate; OS: overall survival.
Applying current standards and emerging therapies in SCLC to SCCB
As noted above, given the dearth of evidence for the treatment of SCCB, data are often extrapolated from SCLC to guide clinical decision making. SCLC is the most aggressive type of lung cancer, the leading cause of cancer death in men and the second leading cause of cancer death in women worldwide.94,95 Similar to SCCB, SCLC has a poor prognosis, with a 5-year survival rate of just 7%. Although SCLCs are sensitive to first-line therapy with a platinum agent (cisplatin or carboplatin), etoposide, and immune checkpoint inhibitor, most patients relapse or progress within a few weeks to months.96,97 There is a strong biological rationale for the recent addition of immune checkpoint inhibitors to the decades-long use of the platinum-etoposide doublet for SCLC treatment98–100 (Table 3). In SCLC, the addition of atezolizumab, an anti-PD-L1 antibody, to carboplatin and etoposide in the phase 3 IMpower133 trial significantly improved mOS from 10.3 to 12.3 months. 101 In the phase 3 CASPIAN trial, the addition of durvalumab, another anti-PD-L1 antibody, to chemotherapy significantly improved mOS from 10.3 to 13 months. 102 However, in both these studies, only 13%–18% of patients remained progression-free at one year. In contrast to other solid tumors, PD-L1 expression is low in SCLC tumor cells and does not seem to correlate with immunotherapy benefit.103,104
Current FDA approved therapies for small cell lung cancer.
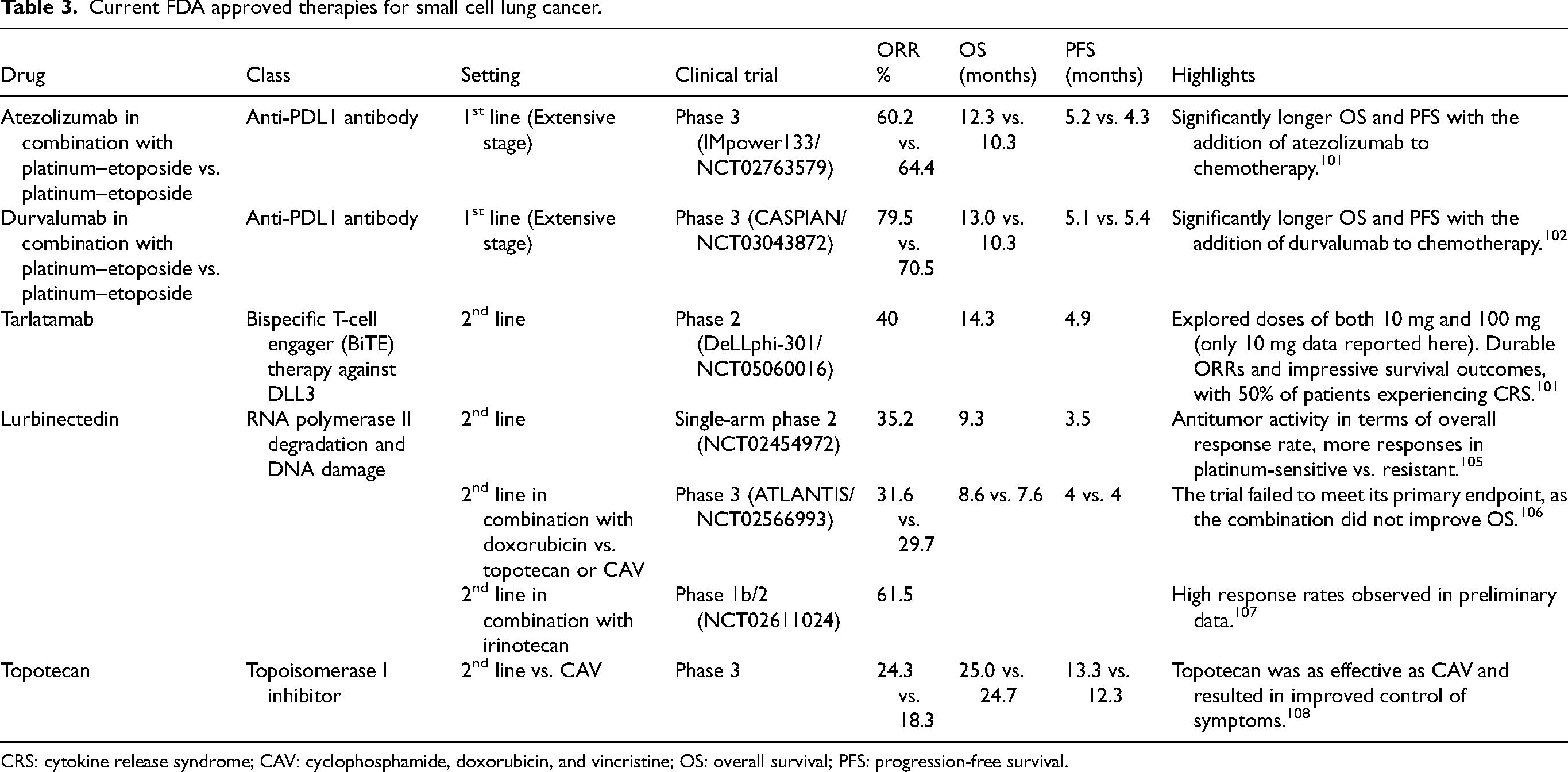
CRS: cytokine release syndrome; CAV: cyclophosphamide, doxorubicin, and vincristine; OS: overall survival; PFS: progression-free survival.
Based upon the results from SCLC, an immune checkpoint inhibitor plus platinum-etoposide chemotherapy is now often used in the first-line metastatic setting in SCCB, although there is little prospective data in this setting. Few options exist for SCLC second-line therapy and there is no standard for SCCB. For SCLC, topotecan and lurbinectedin are the standard second-line treatment options, with tarlatamab also being a new therapeutic option. Topotecan was established as the standard second-line therapy in 1996 based on results of a randomized phase 3 trial comparing topotecan to cyclophosphamide, doxorubicin, and vincristine. Notably, the study only included patients who had relapsed at least 60 days after completion of first-line therapy. Antitumor efficacy was similar across both treatment arms, but patients who received topotecan, administered 1.5 mg/m2 IV daily for 5 days every 21 days, had significant improvement in symptoms such as dyspnea, anorexia, fatigue, and hoarseness. 108 Lurbinectedin was granted accelerated approval by the FDA in 2020 as second-line treatment based on the results of a single-arm, phase 2 basket trial. 105 The study recruited 105 patients with disease progression after treatment with platinum-based chemotherapy and included patients with both platinum-sensitive and platinum-resistant disease (with subgroups based on chemotherapy-free interval less than 90 days versus greater than or equal to 90 days after completion of first-line therapy) who received lurbinectedin IV 3.2 mg/m2 every 3 weeks. The study reported an ORR of 35.2% with median progression-free survival (PFS) of 3.5 months and mOS of 9.3 months. Although responses were observed in both the subgroups, patients with platinum-sensitive tumors had better outcomes. Compared with topotecan, lurbinectedin has an easier schedule of administration, though the efficacy and toxicity of these agents have not been directly compared. Both are options that can be extrapolated from SCLC for use in later lines of therapy for SCCB.
There are a number of new and emerging therapeutic classes being explored for the treatment of SCLC, and these therapies may potentially offer new treatment options to patients with SCCB. Tarlatamab (AMG 757) is a first-in-class, half-life-extended bispecific T-cell engager (BiTE) molecule that promotes intratumoral infiltration of activated T cells and facilitates T cell-mediated killing of DLL3-expressing SCLC tumors. 109 DLL3 is an inhibitory NOTCH ligand selectively expressed in SCLC and other HGNECs.10,110 Unlike rovalpituzumab tesirine (“Rova-T”), an antibody-drug conjugate against DLL3 with a pyrrolobenzodiazepine (PBD) payload that was toxic in clinical studies, tarlatamab has demonstrated an acceptable safety profile.111–113 The efficacy of tarlatamab was explored in a phase 2 study, DeLLphi-301 (NCT05060016), that explored doses of both 10 mg and 100 mg for efficacy and safety. 114 The results of the 10 mg dosing demonstrated an ORR of 40% with an mOS of 14.3 months (95% CI 10.8–NE) and a median PFS of 4.9 months (95% CI 2.9–6.7). These results led to FDA accelerated approval of tarlatamab. Notably, the toxicity profile of this BiTE therapy was different from other second-line therapeutic options, with cytokine release syndrome seen in 51% of patients (grade 3 in 1%) and immune effector cell-associated neurotoxicity syndrome seen in 8% of patients (no grade 3 events). To date there have been no reported cases of the use of tarlatamab in SCCB.
Other therapeutic avenues that have been explored in SCLC have involved targeting DNA damage response (DDR) pathways. SCLC is a transcriptionally active disease, with the majority of tumors having homozygous loss of RB1 and TP53 tumor-suppressor genes, 10 leading to genomic instability, replication stress, and increased dependence on intact DDR pathways. 115 One of the earliest events in DDR is the recruitment of poly (ADP-ribose) polymerases (PARPs), nuclear proteins that contribute to DNA repair and the maintenance of genomic stability. Although SCLC tumors and cell lines have uniquely high expression of PARP and SCLC cell lines are sensitive to PARP inhibitors, PARP inhibitors have failed to show meaningful benefit in patients across multiple clinical trials (Table 4). For example, in randomized trials of the PARP inhibitor veliparib with first- or second-line chemotherapy agents, higher response rates were observed with the combination but did not translate to OS or PFS benefit.116,117 Similarly, combinations of PARP inhibitors with immunotherapy agents have not demonstrated clinical benefit over historical controls of patients treated with immunotherapy alone. 111 However, subsets of SCLCs may benefit from PARP inhibitors, as indicated by prolonged PFS and OS of patients with high SLFN11-expressing tumors treated with temozolomide plus veliparib compared with low SLFN11 tumors. 117 SLFN11 regulates response to DNA damage and replication stress and is a predictive marker of sensitivity to multiple DNA-damaging chemotherapies across many cancers. 118 An ongoing clinical trial is testing maintenance therapy with a combination of immunotherapy and PARP inhibitor in patients with SLFN11-expressing tumors (NCT04334941). Olaparib is currently being tested in a phase 2 study in patients with bladder cancer and rare GU histologies, including SCCB, that harbor DDR alteration (NCT03375307).
Emerging therapies for small cell lung cancer.
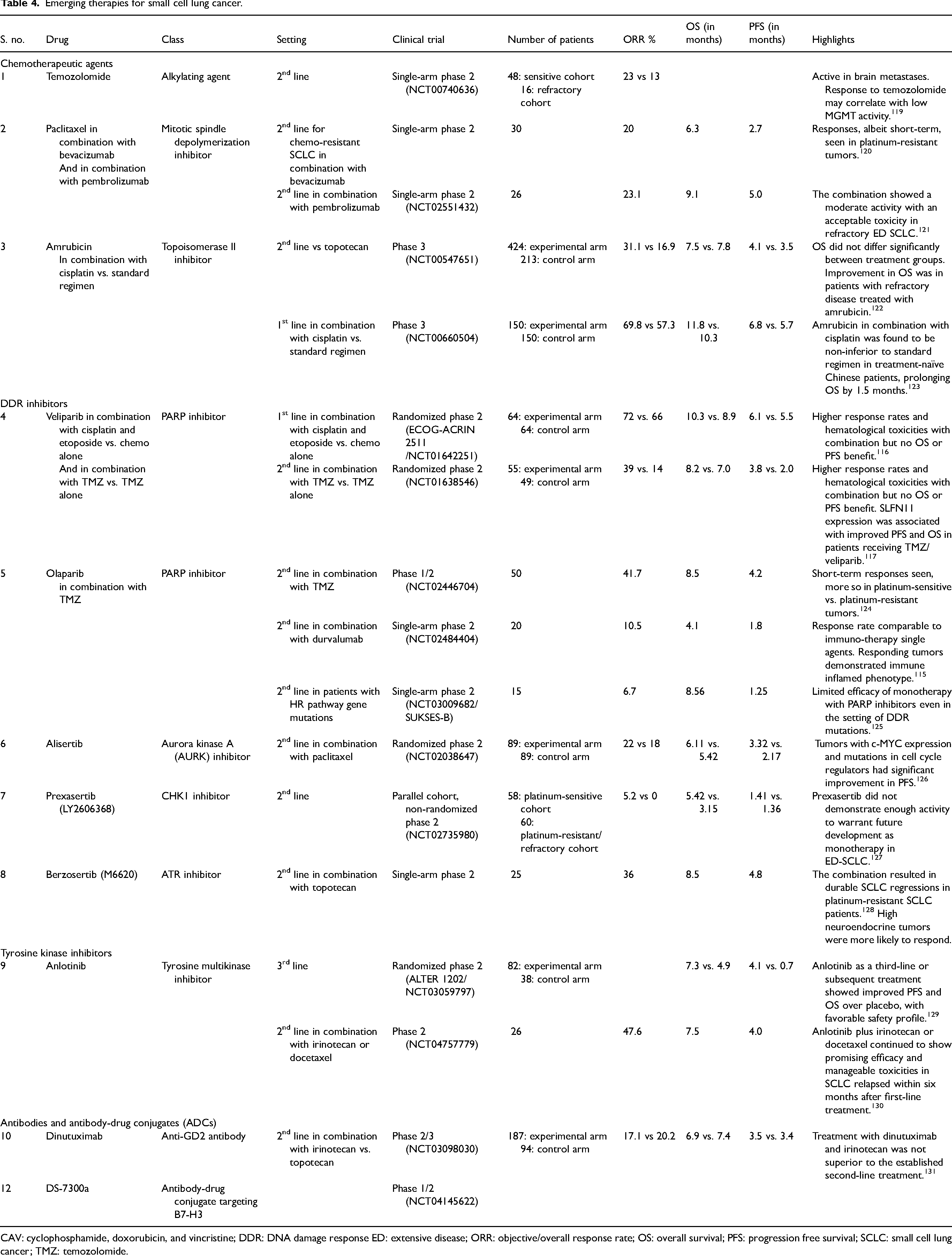
CAV: cyclophosphamide, doxorubicin, and vincristine; DDR: DNA damage response ED: extensive disease; ORR: objective/overall response rate; OS: overall survival; PFS: progression free survival; SCLC: small cell lung cancer; TMZ: temozolomide.
ATR (ataxia-telangiectasia-mutated and rad3-related protein kinase) is a DDR master regulator that plays a key role in stabilizing the genome when DNA replication is compromised. ATR-mediated S-phase arrest prevents cell division and promotes DNA damage repair, thereby avoiding additional DNA damage and maintaining genomic stability. A recent study identified synergistically cytotoxic activity of ATR inhibition and topoisomerase 1 inhibition. The combination of the first-in-class ATR inhibitor berzosertib with topotecan produced an ORR of 36% in SCLC patients. 128 Durable tumor regressions were observed in patients with platinum-resistant SCLCs, typically fatal within weeks of recurrence. Notably, the study also observed responses to the combination in a cohort of patients with extrapulmonary small cell neuroendocrine cancers, including 2 SCCB patients, suggesting that DDR inhibition may be a therapeutic opportunity for small cell cancers independent of their tissue of origin.
Other novel immunotherapy approaches include DS-7300, an antibody-drug conjugate against B7-H3, a transmembrane protein overexpressed in SCLC and associated with poor prognosis. 132 The payload, DXd, is an exatecan derivative that inhibits topoisomerase 1. DS-7300 could provide tumor-targeted delivery of topoisomerase 1 inhibitors, thus minimizing systemic toxicity and enabling more tolerable combinations. There is an ongoing study of sacituzumab govitecan, an antibody-drug conjugate that also has a payload of the topoisomerase 1 inhibitor SN-38, in rare GU tumors that include SCCB and other HGNECs of the urinary tract (NCT06161532).
Given the rarity of SCCB, prospective trials to guide therapy are lacking. Understanding the current and future therapies in SCLC provides insights into strategies that may be extrapolated to SCCB to guide both clinical trial design and clinical management.
Conclusion
SCCB is a rare form of bladder cancer often mixed with other histologies. Recent evidence has indicated a shared cell of origin for SCCB when seen with other histologic subtypes. While sharing some of the same risk factors of UC and a similar presentation, it is a much more aggressive malignancy with a poor prognosis. Most cases show a loss of TP53 and RB1 and commonly have TERT mutations. Initial workup consists of cystoscopy and TURBT with CT imaging and brain MRI to evaluate for metastatic disease. Given its high glucose metabolism, there is also a growing role for PET imaging to assist in staging. Unfortunately, there have been few prospective studies to guide care. Patients are staged as having either localized or metastatic disease, with platinum-based chemotherapy used in both contexts. Local therapy options include TURBT, cystectomy, and radiation therapy sequenced either before or after systemic therapy. For metastatic SCCB, current therapeutic options are often extrapolated based on its similarity to SCLC, which may guide future treatments. Furthermore, greater representation of patients with SCCB in prospective interventional clinical trials (whether tumor-agnostic, specific to neuroendocrine phenotype or neuroendocrine marker positive, or specific for SCCB) may provide additional therapeutic options for patients with this aggressive cancer.
Footnotes
Ethical considerations
Ethics approval has been obtained from the NIH Institutional Review Board (protocol number IRB001054).
Author contributions
Nicholas Simon, Andrea Apolo – conceptualization, project administration, writing. Andre R. Kydd, Dilara Akbulut, David Takeda, Jaydira Del Rivero, Maria Merino, Bernadette Redd, Liza Lindenberg, Esther Mena, Elias Chandran, Sandeep Gurram, Salah Boudjadi, Scot Niglio, Parth Sharma, Anish – writing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute (NCI), National Institutes of Health (NIH), (grant number ZIABC011928 and ZIABC011351).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
