Abstract
Background
Metabolomic research and metabolomics-based biomarkers predicting treatment outcomes in bladder cancer remain limited.
Objective
We explored the serum metabolites potentially associated with the risk of recurrence after intravesical Bacillus Calmette–Guérin (BCG) therapy.
Methods
Two independent cohorts, a discovery cohort (n = 23) and a validation cohort (n = 40), were included in this study. Blood was collected before the induction of BCG therapy (pre-BCG blood; both discovery and validation cohorts) and after six doses of BCG (post-BCG blood; only discovery cohort). Metabolome analysis of serum samples was conducted using capillary electrophoresis time-of-flight mass spectrometry. The endpoint was intravesical recurrence-free survival, which was analysed using Kaplan–Meier estimates, the log-rank test, and the Cox proportional hazard model.
Results
Of the 353 metabolites quantified, nine (2.5%) and four (1.1%) were significantly upregulated and downregulated, respectively. The heatmap of hierarchical clustering analysis and principal coordinate analysis for the fold changes and in serum metabolites differentiated 10 recurrent cases and 13 non-recurrent cases in the discovery cohort. A metabolome response-based scoring model using 16 metabolites, including threonine and N6,N6,N6-trimethyl-lysine effectively stratified the risk of post-BCG recurrence. Additionally, pre-BCG metabolome-based score models using six metabolites, octanoylcarnitine, S-methylcysteine-S-oxide, theobromine, carnitine, indole-3-acetic acid, and valeric acid, were developed from the discovery cohort. Univariate and multivariate analyses confirmed a high predictive accuracy in the validation and combination cohorts.
Conclusions
We demonstrated that numerous types of serum metabolites were altered in response to intravesical BCG and developed high-performance score models which might effectively differentiated the risk of post-BCG tumour recurrence.
Keywords
Introduction
Non-muscle-invasive bladder cancer (NMIBC) is a clinically and biologically heterogeneous disease that accounts for 70–80% of primary bladder cancers (BCa). The most recently updated European Association of Urology (EAU) guidelines stratify patients with NMIBC into low-, intermediate-, high-, and highest-risk groups. 1 The clinical practice guidelines recommend intravesical instillation of Bacillus Calmette–Guérin (BCG) as a standard therapeutic and adjuvant option following transurethral resection of bladder tumour (TURBT) for high-risk disease with or without carcinoma in situ (CIS).2–4 Induction therapy with full-dose BCG instillations followed by one to three-year maintenance therapy is the recommended therapy for high-risk patients. Immediate radical cystectomy (RC) should be offered to very high-risk patients. After careful consideration and discussion, intravesical BCG can be an alternative option for very high-risk patients who decline or are unfit for RC. A nationwide large-scale retrospective study demonstrated that the 2-year intravesical recurrence-free survival (RFS) and progression-free survival (PFS) rates were 83% and 94%, respectively, and the 5-year RFS and PFS rates were 74% and 89%, respectively. 5 A substantial number of patients treated with intravesical BCG experience long-lasting severe toxicity such as chronic cystitis, lower urinary tract dysfunction, and ladder contracture. 6 Accurate and noninvasive biomarkers for predicting post-BCG outcomes are still limited in real-world clinical practice.
Metabolomics, defined as the quantitative study of metabolites, has been extensively utilised in oncological research using mass spectrometry (MS) and nuclear magnetic resonance spectroscopy to explore differences in several materials (tissue, blood, and urine) between patients with and without cancer. 7 Metabolites are molecules smaller than 1500 kDa in biological systems, and alterations in their concentrations are involved in genetic and/or environmental effects. Based on the theory that metabolites are dynamic functional outputs of multiple organs and organisms, one of the major advantages of metabolomics is the efficient monitoring of biochemical alterations in the human body. 8
Several studies have demonstrated that various metabolic pathways are altered in BCa, providing potential biomarkers for improved tumour detection and possible therapeutic interventions.7,9 Metabolomics studies in the field of BCa aim for its early detection,6,10 accurate staging and grading,11,12 and survival prediction.13,14 However, there is no evidence regarding the potential of metabolomics in patients with NMIBC treated with intravesical BCG. Here, we explored differential metabolites in pre-BCG and post-BCG serum samples that were associated with the risk of intravesical recurrence after the induction of BCG therapy.
Materials and methods
Two study cohorts: discovery cohort and validation cohort
The Ethics Committee of Nara Medical University approved this study (reference protocol ID: 3698). Written informed consent was obtained from all the participants. All data used in this study are stored anonymized at the Department of Urology, Nara Medical University.
A flowchart depicting the study overview, inclusion criteria, sample processing, and analysis is presented in Figure 1. A total of 63 patients with intermediate/high-/ highest-risk NMIBC treated with intravesical BCG at Nara Medical University Hospital were included in this study with two independent cohorts: (1) discovery cohort (n = 23; August 2016–July 2018) and (2) validation cohort (n = 40; August 2018–April 2021). The clinicopathological characteristics included age, sex, history of NMIBC, tumour multiplicity, tumour size, T category, tumour grade, presence of carcinoma in situ (CIS), implementation of a second TUR, maintenance BCG, and treatment outcomes (intravesical recurrence). The patients were stratified depending on their clinicopathological characteristics into low-, intermediate-, and high-risk groups according to the European Association of Urology (EAU) guidelines updated 2019. 15 According to the guidelines, patients were categorised as the highest-risk disease, when those had at least one of the following aggressive factors: i) T1 high-grade UC with concomitant bladder CIS or prostate-involving CIS; ii) multiple and/or large and/or recurrent T1 high-grade UC; iii) lymphovascular invasion (LVI); iv) non-UC histological subtype, or v) BCG-unresponsive NMIBC. Our cohort did not include those with BCG-unresponsive NMIBC. Supplementary Table S1 summarises the characteristics of the two cohorts. Patients with past history of BCa, Ta low-grade tumour, and intermediate-risk disease were more in the discovery cohort than validation cohort. No patients received maintenance BCG in the discovery cohort, whereas 37% of patients received in the validation cohort. No patients with low-risk NMIBC were included in this study because intravesical BCG is not recommended for this disease subset. Of 63 patients included in this study, 9 (14%) had a history of NMIBC before induction BCG, but any of cases did not receive intravesical BCG or maintenance intravesical chemotherapy.
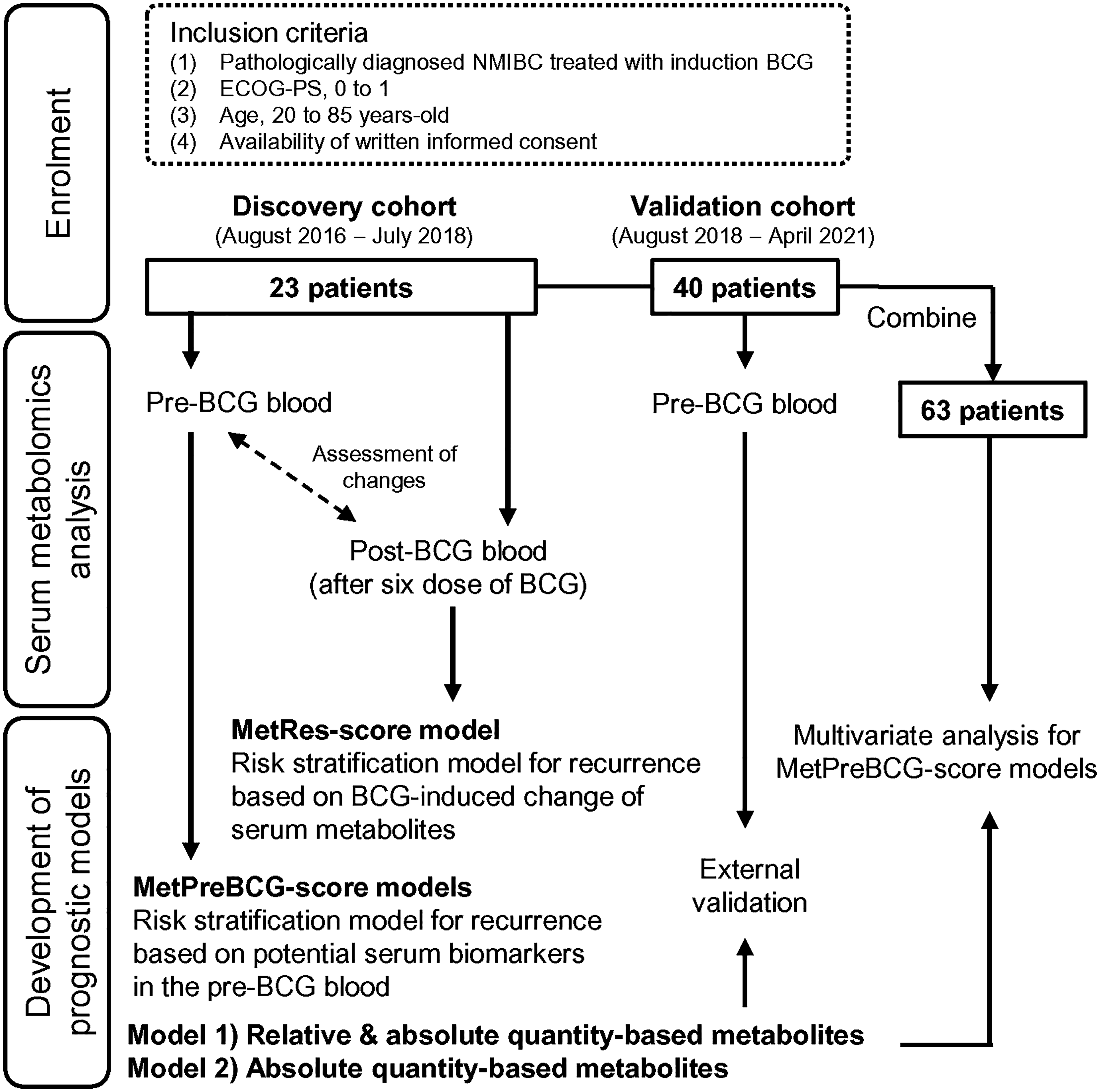
Study overview of the patient cohorts and analysis. This study consisted of three phases: enrolment, serum metabolomic analysis, and the development of prognostic models. Two cohorts, the discovery cohort and the validation cohorts were enrolled in this study. NMIBC, non-muscle invasive bladder cancer; BCG, Bacillus Calmette–Guérin; MetRes score, Metabolite response score; MetPreBCG score, Metabolite Pre-BCG score.
Intravesical BCG therapy and surveillance
The criteria, dose, and schedule for induction BCG (iBCG) and maintenance BCG (mBCG) were inconsistent and administered at the physician's discretion. The intravesical BCG schedule included weekly instillations of the immunobladder (80 mg of Tokyo-172 strain) for 6–8 consecutive weeks, with or without subsequent mBCG. mBCG was administered once a week for 3 weeks at 3, 6, 12, 18, 24, 30, and 36 months after iBCG initiation or until discontinuation due to tumour recurrence, intolerable adverse events, or patient refusal of treatment. 16 At least two out of three doses in the first mBCG round at three months were considered mBCG. Patients were generally followed up after TURBT using white-light cystoscopy and urinary cytology every three months for two years, then every six months in the third and fourth years, and annually thereafter. Recurrence was defined as recurrent tumours of pathologically proven UC of the bladder and prostatic urethra. Neither recurrence in the upper urinary tract nor a positive result on urinary cytology without pathologically proven UC was considered a recurrence. In 23 patients of the discovery set, two patients underwent RC with T1 high-grade UC of the pathological diagnosis and other patients did not experience progression. In 40 patients of the validation set, one patient developed muscle invasive disease and other patients did not experience progression. Because only a few patients experienced disease progression in our cohort, we did not evaluate progression risk in this study.
Metabolite extraction
Blood was drawn from fasting participants into 5-mL vacuum blood collecting tubes before induction of BCG therapy (pre-BCG blood; both discovery and validation cohorts) and after six doses of BCG (post-BCG blood; only discovery cohort). Serum samples were stored in aliquots at −80 °C until further analysis. Fifty µL of serum was added to 200 µL of methanol containing internal standards (H3304-1002, Human Metabolome Technologies, Inc. (HMT), Tsuruoka, Yamagata, Japan) at 0 °C to suppress enzymatic activity. The extract solution was thoroughly mixed with 150 µL of Milli-Q water, after which 300 µL of the mixture were centrifugally filtered through a 5-kDa cutoff filter (ULTRAFREE MC PLHCC, HMT) at 9100 × g, 4 °C to remove macromolecules. The filtrate was then evaporated to dryness under vacuum and reconstituted in 50 µL of Milli-Q water for metabolome analysis.
Metabolomics profiling of serum samples
Metabolome analysis was conducted according to the HMT's Cohort Study Pack using capillary electrophoresis time-of-flight mass spectrometry (CE-TOFMS) based on previously described methods.17,18 Briefly, CE-TOF-MS analysis was performed using an Agilent CE capillary electrophoresis system equipped with an Agilent 6230 TOF-MS (Agilent Technologies, Inc., Santa Clara, CA, USA). 19 The systems were controlled by Agilent MassHunter Workstation Data Acquisition ver.B.09.00 (Agilent Technologies) and connected by a fused silica capillary (50 μm i.d. × 80 cm total length; cat.# 106815, Polymicro Technologies, Phoenix, AZ, USA) with commercial electrophoresis buffer (H3301-1001 and I3302-1023 for cation and anion analyses, respectively, HMT) as the electrolyte. The spectrometer was scanned from m/z 50 to 1,000, and peaks were extracted using MasterHands automatic integration software ver.2.19.0.2 (Keio University, Tsuruoka, Yamagata, Japan) to obtain information including m/z, peak area, and migration time. 20 Signal peaks corresponding to isotopomers, adduct ions, and other product ions of known metabolites were excluded and the remaining peaks were annotated according to the HMT metabolite database based on their m/z values and migration times. 17 The areas of the annotated peaks were normalised to internal standards and sample amounts to obtain the relative levels of each metabolite. Primary 353 metabolites were quantified based on a one-point calibration using their respective standard compounds. Hierarchical cluster analysis (HCA) was performed using the HMT proprietary software, PeakStat ver.3.20. 21
Statistical analyses
Clinicopathological characteristics were compared using the Mann–Whitney U, chi-square, and Fisher's exact tests, as appropriate. Differences in serum metabolites between pre- and post-BCG serum samples were expressed as fold change (FC) and analyzed using the Wilcoxon signed-rank test. Recurrence-free survival (RFS) from the date of administration of the iBCG dose was estimated using the Kaplan–Meier method and compared using the log-rank test. Multivariate Statistical analyses were performed using GraphPad Prism version 10 (GraphPad Software, San Diego, CA, USA). Statistical significance was set at p < 0.05 and 0.05 ≤ p < 1.0 was considered tendency toward statistical significance.
Results
BCG-induced response of serum metabolites
Metabolome analysis of pre- and post-BCG blood matched in the discovery cohort was conducted to identify significant BCG-induced changes in serum metabolites. Of the 353 metabolites quantified in this analysis, nine (2.5%) and four (1.1%) were upregulated and downregulated, respectively. Detailed metabolite characteristics were retrieved from the Human Metabolome Database (HMDB) and are listed in Supplementary Table S2. Four amino acids (arginine, leucine, lysine, and valine), urea, 4-oxopyrrolidine-2-carboxylic acid, cyclohexylamine, N-acetylneuraminic acid, and γ-carboxyglutamic acid were significantly upregulated by six doses of intravesical BCG, whereas glycerol, myristic acid, 2-methylthiazolidine-4-carboxylic acid, and octanoylcarnitine were significantly downregulated (Table 1 and Supplementary Figure S1).
Serum metabolite showing significant change before and after induction BCG therapy in patients with NMIBC.
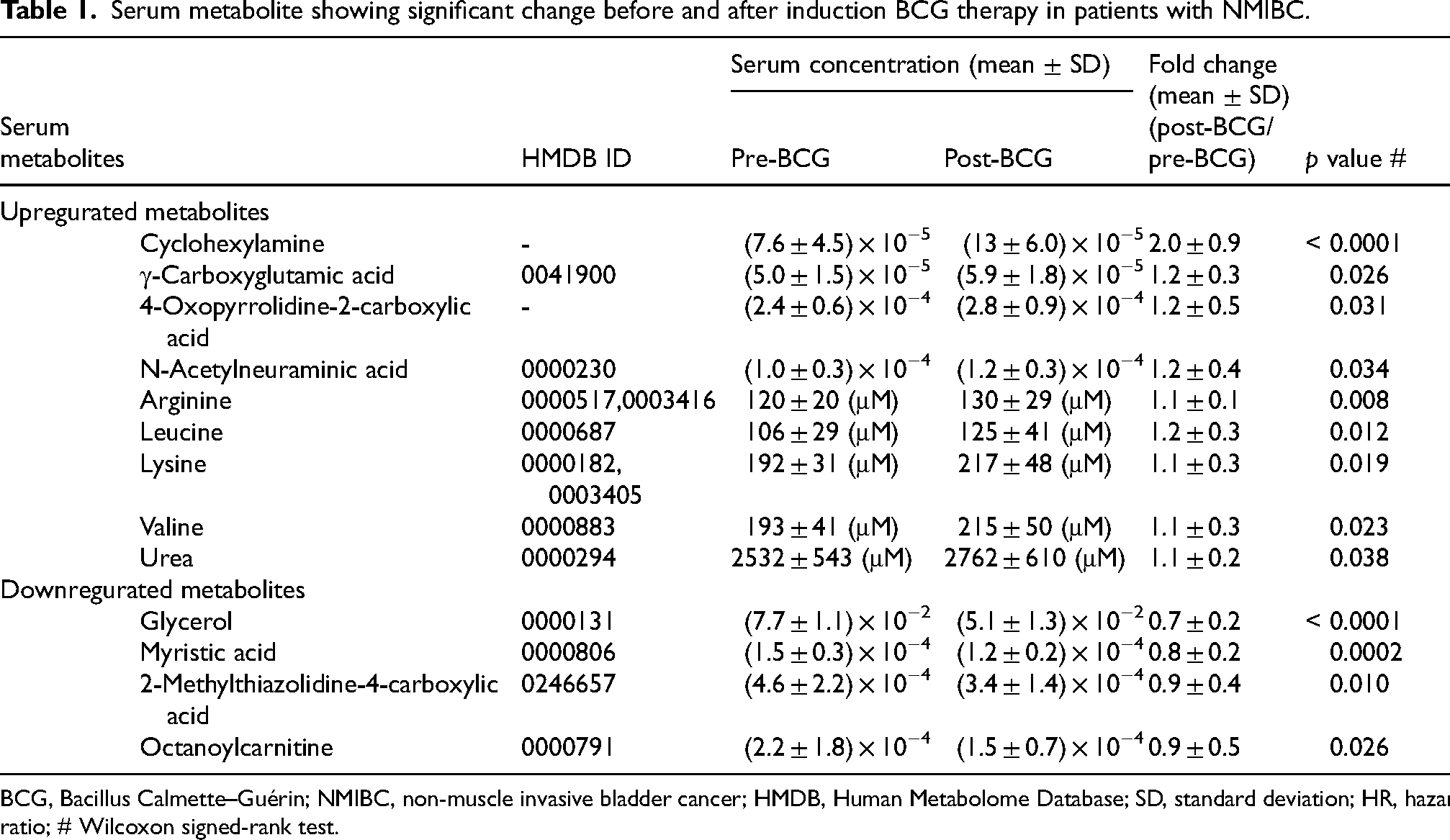
BCG, Bacillus Calmette–Guérin; NMIBC, non-muscle invasive bladder cancer; HMDB, Human Metabolome Database; SD, standard deviation; HR, hazard ratio; # Wilcoxon signed-rank test.
BCG-induced change of serum metabolites and risk of post-BCG recurrence
The FCs of serum metabolites were calculated using the following formula: (serum concentration of a metabolite in post-BCG blood)/(serum concentration of a metabolite in pre-BCG blood). Of the 23 patients in the discovery cohort with a median follow-up of 70 months after BCG induction, 10 (43%) experienced intravesical recurrence. Heatmap of hierarchical clustering analysis for the FCs of serum metabolites differentiating 10 recurrent and 13 non-recurrent cases (Figure 2A, upper). A higher number of metabolites showing high FC were observed in non-recurrent cases than in recurrent cases. This suggests that a metabolite-based systemic response to intravesical BCG therapy is associated with better clinical outcomes. Differences in metabolic response profiles between the non-recurrent and recurrent groups were visualised using unsupervised principal coordinate analysis (Figure 2B). Of the 353 metabolites studied, the FCs of 16 metabolites were associated with the outcome after induction of BCG therapy: Low FC was associated with a higher recurrence risk in 14 metabolites, such as threonine and N6,N6,N6-trimethyl-lysine; High FC was associated with the risk of two metabolites, S-methylcysteine-S-oxide and octanoylcarnitine (Figure 2A, lower). Detailed characteristics of the metabolites are listed in Supplementary Table S2. Table 2 depicts the changes in 16 metabolites, risk of recurrence, and allocated risk scores based on regression coefficients. The Metabolite Response score (MetRes score; full score = 100) was calculated as the sum of the risk scores. The median MetRes score was 43 (range, 4–100; intertertile range, 23–66). Only one patient out of 10 recurrent cases showed a MetRes score ≤ 43. Of note, no patients with a MetRes score ≤ 23 had recurrence, and all patients with a MetRes score > 66 had recurrence after the induction of BCG (Figure 2C). The cut-off values differentiated the risk of recurrence in patients treated with intravesical BCG (Figure 2D).
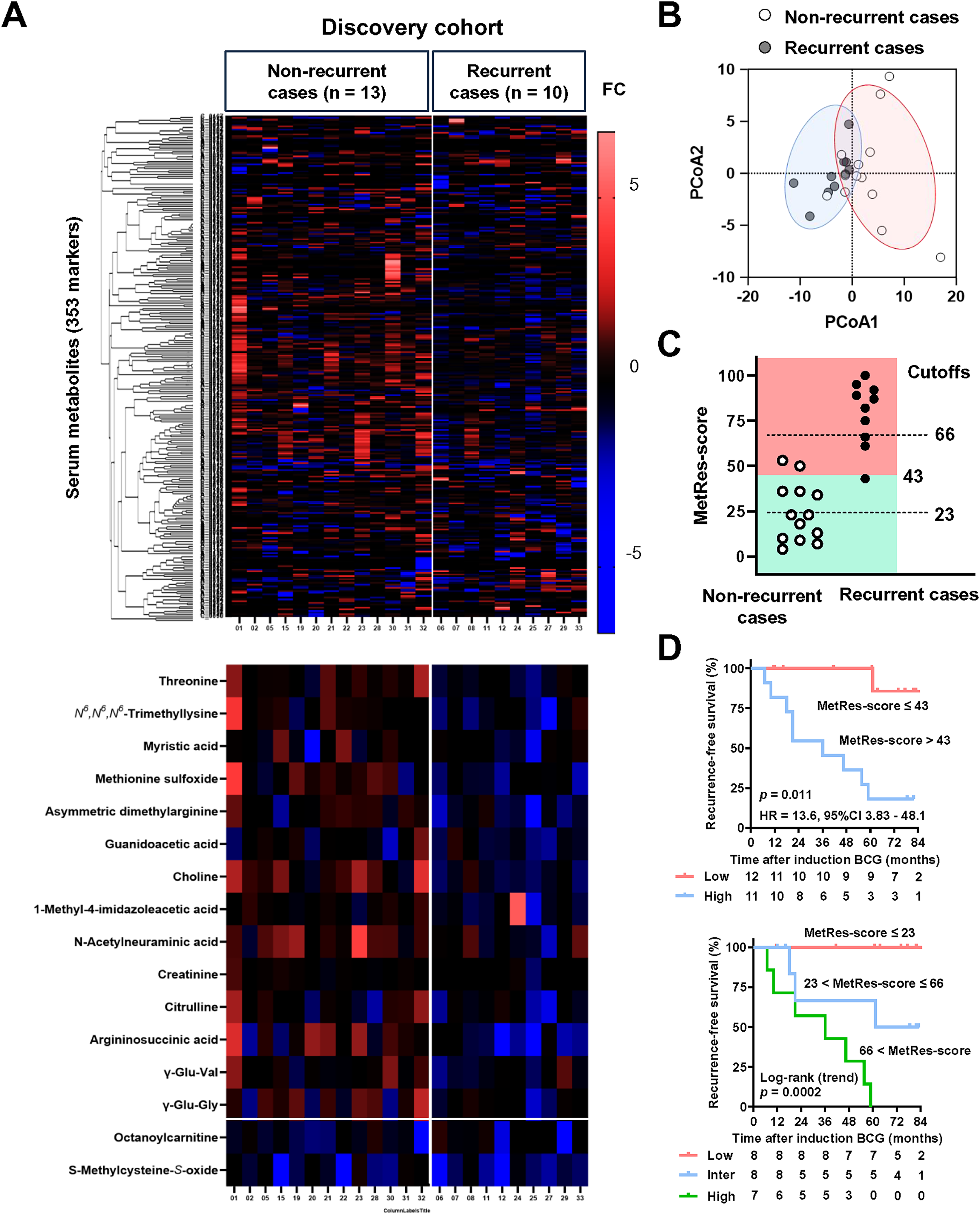
BCG-induced change of serum metabolites and risk of post-BCG recurrence. A, The upper heatmap visualisation of hierarchical clustering analysis of changes in serum metabolites before and after intravesical BCG therapy. Lower heatmap visualisation of serum metabolites in which FC was associated with outcome after induction of BCG therapy. Sixteen of the 353 metabolites were identified as potential prognostic factors. In the upper 14 metabolites, a low FC was associated with a higher risk of recurrence. Among the two lowest metabolites, high FC was associated with a higher risk of recurrence. The red and blue blocks indicate upregulation and downregulation in the post-BCG group compared to the pre-BCG group, respectively.
BCG-induced changes of serum metabolite and recurrence-free survival in patients with NMIBC treated with intravesical BCG.
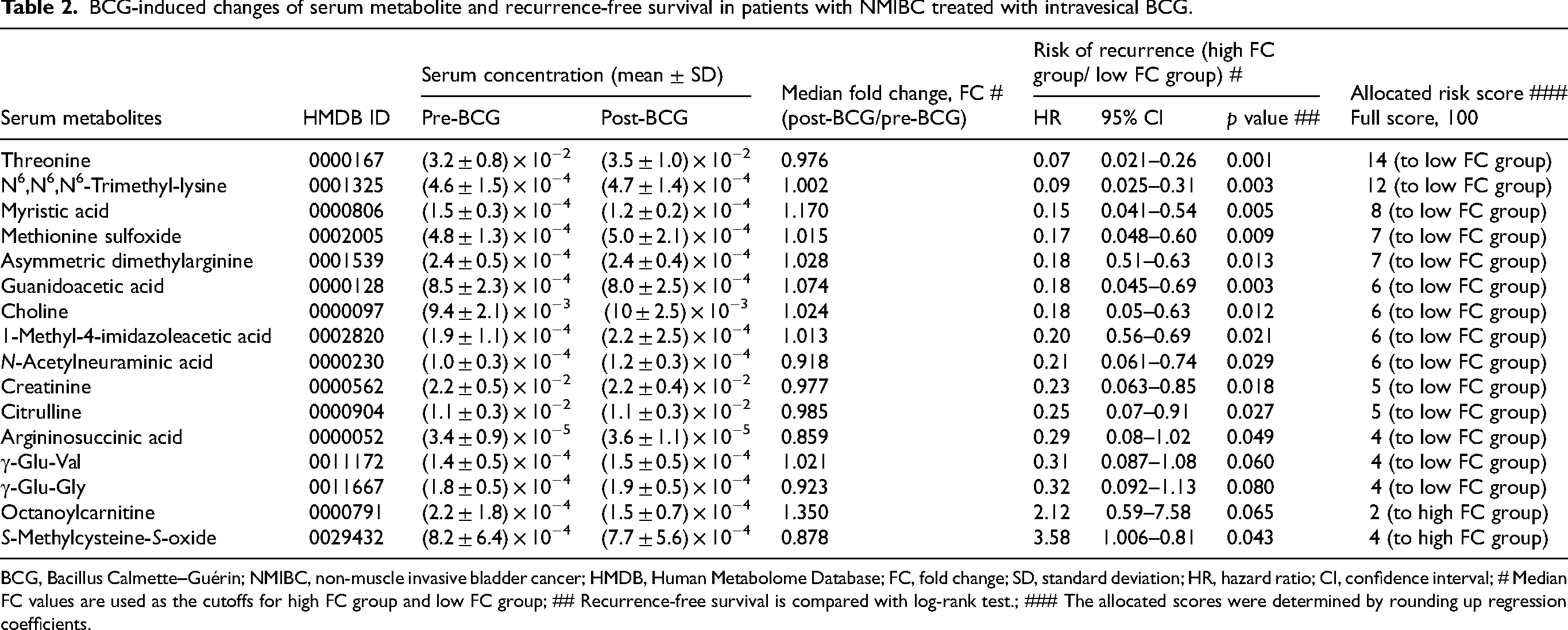
BCG, Bacillus Calmette–Guérin; NMIBC, non-muscle invasive bladder cancer; HMDB, Human Metabolome Database; FC, fold change; SD, standard deviation; HR, hazard ratio; CI, confidence interval; # Median FC values are used as the cutoffs for high FC group and low FC group; ## Recurrence-free survival is compared with log-rank test.; ### The allocated scores were determined by rounding up regression coefficients.
Concentration of pre-BCG serum metabolites and recurrence-free survival in patients with NMIBC treated with intravesical BCG.
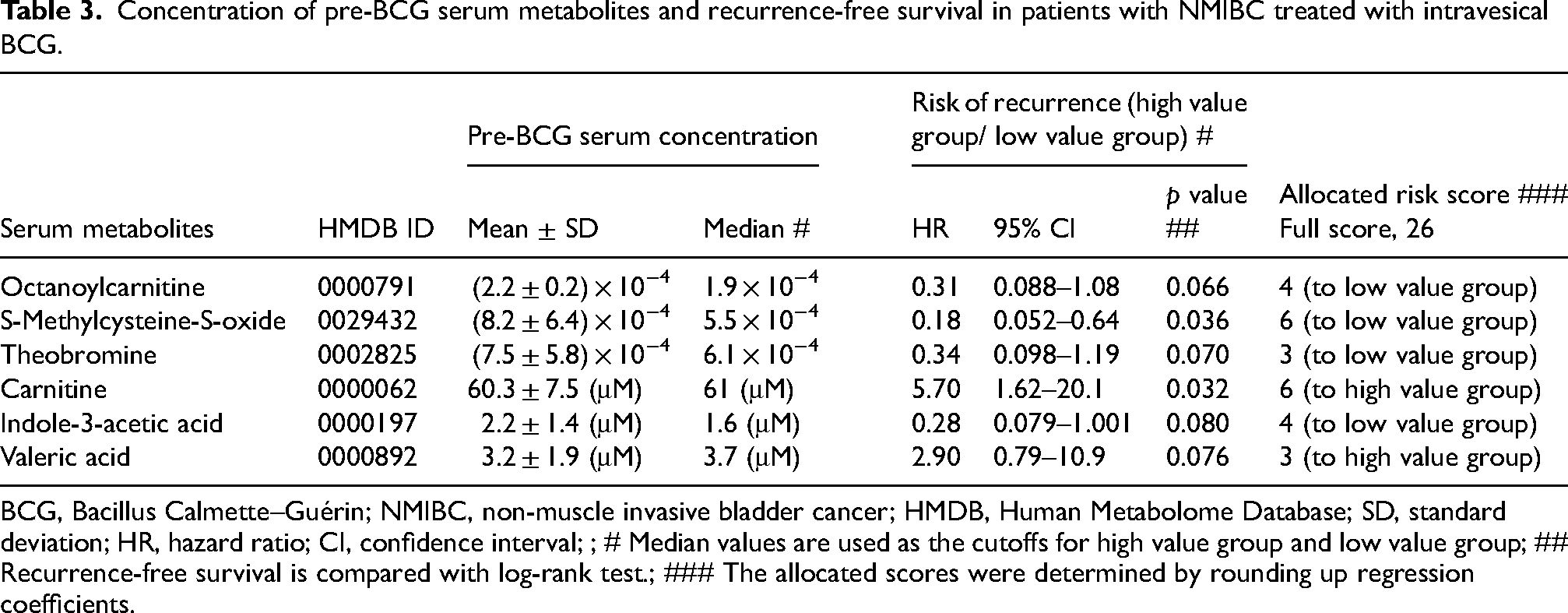
BCG, Bacillus Calmette–Guérin; NMIBC, non-muscle invasive bladder cancer; HMDB, Human Metabolome Database; SD, standard deviation; HR, hazard ratio; CI, confidence interval; ; # Median values are used as the cutoffs for high value group and low value group; ## Recurrence-free survival is compared with log-rank test.; ### The allocated scores were determined by rounding up regression coefficients.
Of 10 recurrent tumours, five (50%) were low-grade UC recurrences and the remaining five (50%) were high-grade UC recurrences. There was no clear variations and significant difference in BCG-induced change of serum metabolites between patients with low-grade UC recurrences and those with high-grade UC recurrences.
Pre-BCG serum metabolites and risk of post-BCG recurrence
We explored serum metabolites in pre-BCG blood that were potentially associated with the risk of post-BCG intravesical recurrence. Based on univariate survival analysis, the pre-BCG serum concentrations of six metabolites out of the 353 tested in this study were potential risk factors with p value < 0.1. Octanoylcarnitine, S-methylcysteine-S-oxide, and theobromine were measured relative to specific standard materials, whereas carnitine, indole-3-acetic acid, and valeric acid were measured as absolute quantities (units, μM). The detailed characteristics of the metabolites are listed in Supplementary Table S2. Table 3 depicts the pre-BCG serum concentrations of the six metabolites, risk of recurrence after BCG induction, and allocated risk scores based on regression coefficients. The metabolite pre-BCG score (MetPreBCG-score) model 1 (full score = 26) was calculated as the sum of the six risk scores. The median MetPreBCG score for Model 1 was 17 (range, 0–26; interquartile range, 12–18). Both groups (low or high, cutoff 17) and three groups (low, intermediate, or high, cutoffs 12 and 18) effectively stratified the risk of intravesical recurrence after the induction of BCG in the discovery cohort (Figure 3A). Next, we applied MetPreBCG-score of Model 1 to the validation cohort with identical risk score calculations and cutoffs. Of the 40 patients in the discovery cohort with a median follow-up of 41 months after BCG induction, 12 (30%) experienced at least one intravesical recurrence. The two-group classification showed modest stratification with HR of 1.90 and p-value of 0.39, whereas the three-group classification did not effectively stratify the risks, with 0.53 of p value (log-rank test for trend).
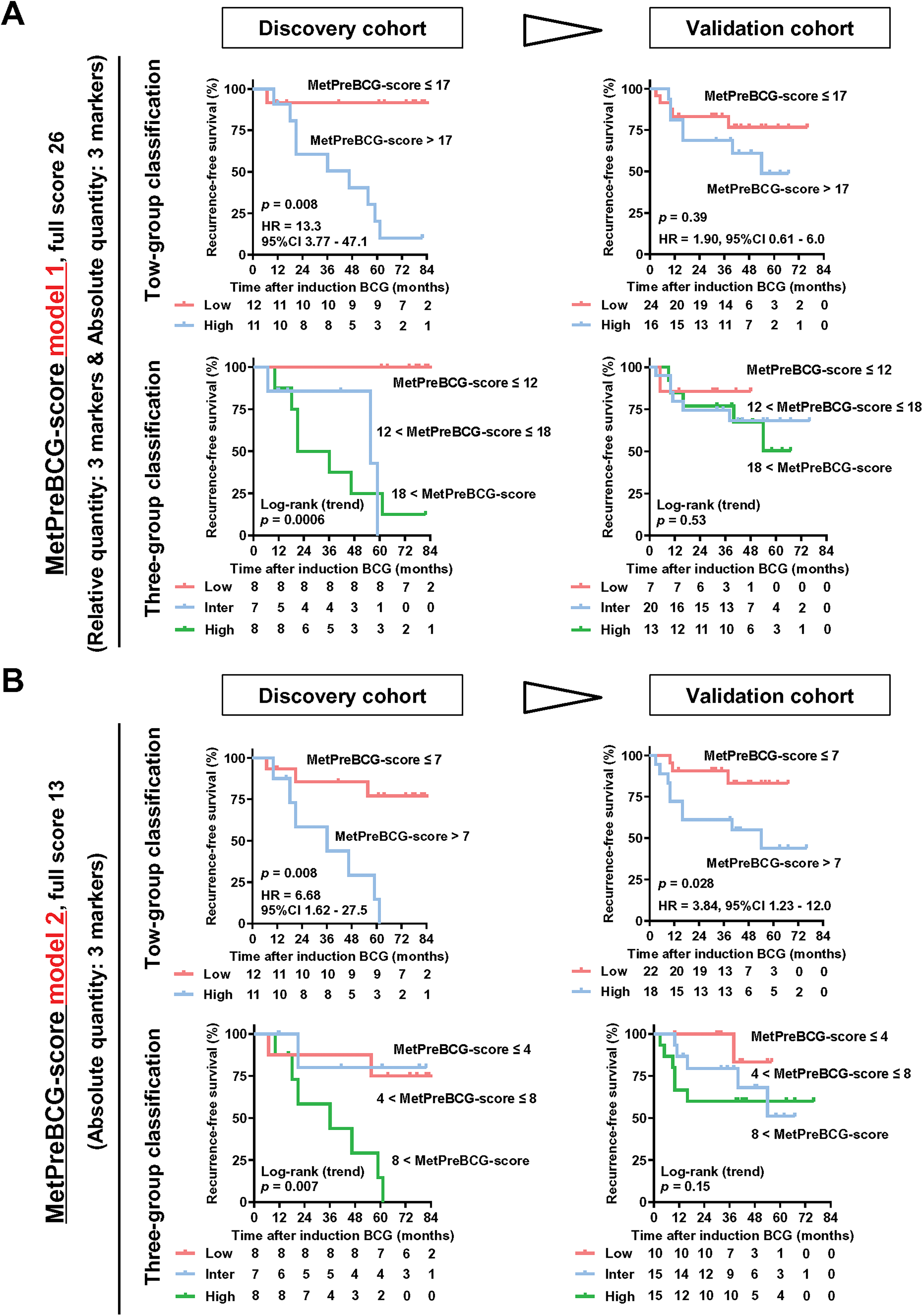
External validation of the developed MetPreBCG-score models.
To develop better clinically useful testing, we tested another risk model that included only three metabolites measured in absolute quantity and excluded those measured in relative quantity. The MetPreBCG score for Model 2 (full score = 13) was calculated by adding the risk scores of carnitine, indole-3-acetic acid, and valeric acid. The median MetPreBCG score for Model 1 was 7 (range, 0–13; interquartile range, 4–8). with identical risk score calculations and cutoff values. The median MetPreBCG score for Model 2 was 7 (range, 0–13). The two-group (low or high, cutoff 7) and three-group classification (low, intermediate, or high, cutoffs 4 and 8) better stratified the risk of post-BCG recurrence, with an HR of 3.84 and a p value of 0.028 for the two-group classification, and a p value of 0.15 for three-group classification (log-rank test for trend), respectively, as compared to the two-group classification by the MetPreBCG-score model 1 (Figure 3B).
In the univariate analysis of the combined cohorts (n = 63), among baseline factors, only a history of urothelial carcinoma was significantly associated with a high risk of post-BCG recurrence (HR 3.66, p value 0.0022; Supplementary Table S3). We confirmed the accurate risk stratification of the MetPreBCG score models in the unadjusted univariate analysis (Supplementary Figure S2). Lastly, we performed a multivariate Cox proportional hazard model to adjust for several confounding variables, including history of urothelial carcinoma and treatment factors (second TUR and mBCG). All the MetPreBCG score models developed in this study effectively differentiated the risk of recurrence in patients treated with intravesical BCG (Table 4).
Univariate and multivariate analysis of recurrence-free survival in patients with NMIBC treated with intravesical BCG.
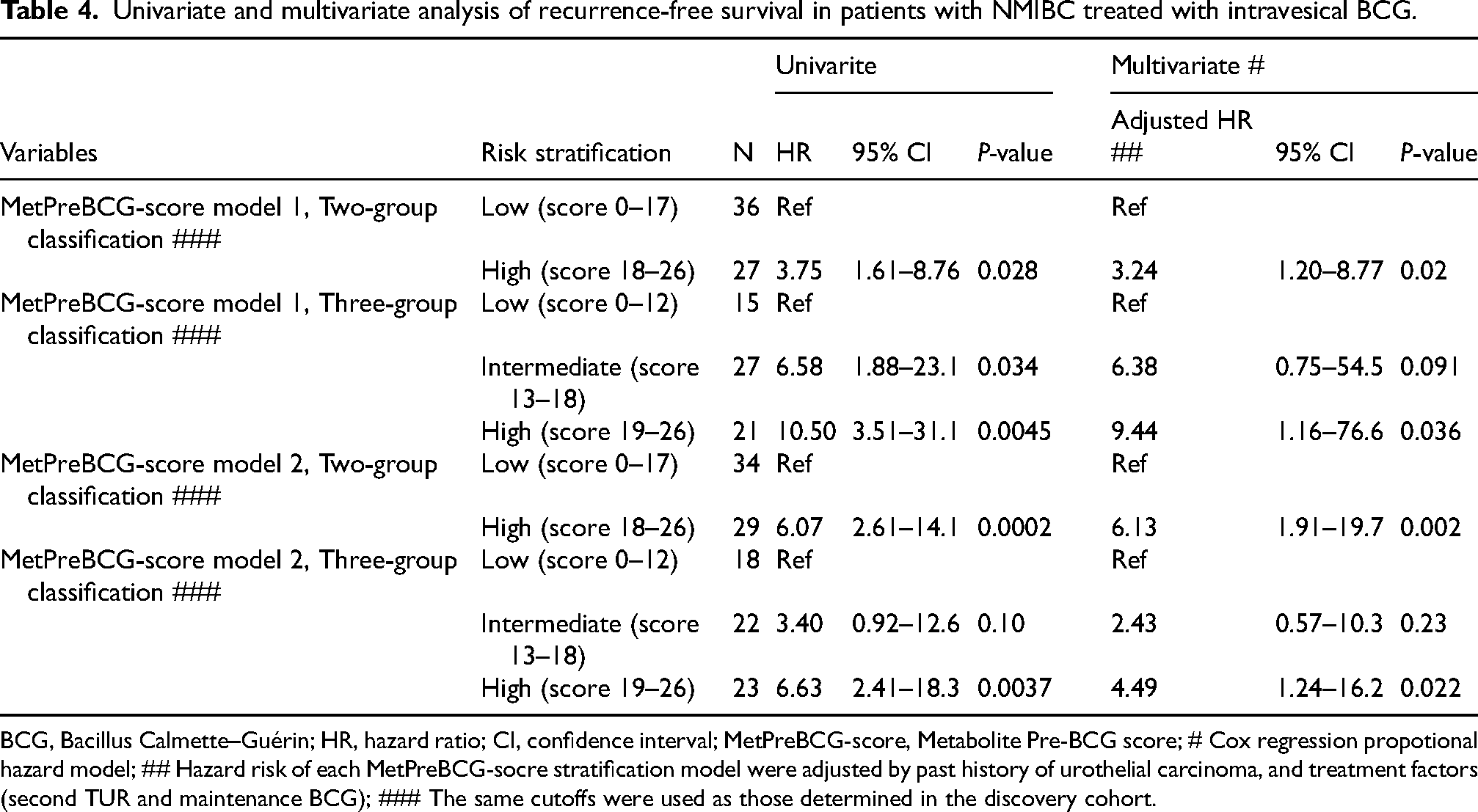
BCG, Bacillus Calmette–Guérin; HR, hazard ratio; CI, confidence interval; MetPreBCG-score, Metabolite Pre-BCG score; # Cox regression propotional hazard model; ## Hazard risk of each MetPreBCG-socre stratification model were adjusted by past history of urothelial carcinoma, and treatment factors (second TUR and maintenance BCG); ### The same cutoffs were used as those determined in the discovery cohort.
Discussion
Metabolomics research in the oncology field establishes promising opportunities for identifying cancer detection markers, risk factors for cancer incidence and disease progression, and predictive markers for anti-tumour drugs. Of these, predictive tools for the response to treatment are clinically vital in the decision-making process of treatment strategies because multiple options can be selected in most situations. 22 For example, NAC provides preoperative benefits through tumour shrinkage, downstaging, and downgrading, although there are possible disadvantages, such as delayed surgical intervention and unnecessary side effects in chemotherapy-resistant tumours. A previous report demonstrated that the metabolomics of blood samples was possible in patients with muscle-invasive BCa 23 or breast cancer 24 who were likely to benefit from NAC. In our pilot study, several serum metabolites potentially associated with the risk of intravesical recurrence after the initiation of BCG therapy were identified using detailed clinicopathological information, long-term follow-up data, and comprehensive metabolome analysis using CE-TOFMS.
The mechanisms underlying BCG-induced anticancer activity and the relevant molecules have been well studied, but their biological effects have not been fully elucidated.25,26 The major biological antitumour effect depends on the pre-BCG baseline and post-BCG status of the Th1/Th2 balance, degree of recruitment of key immunosuppressive cells such as regulatory T cells and myeloid-derived suppressor cells, and polarisation of tumour-associated macrophages in the tumour microenvironment.27,28 We evaluated BCG-induced changes in serum metabolites and confirmed that 13 metabolites were significantly altered after six doses of intravesical BCG. The upregulated metabolites include several organic compounds and three amino acids (arginine, lysine, and valine) that are known to have anti-inflammatory function.29–31 These anti-inflammatory amino acids may be upregulated to inhibit overinflammation induced by intravesical BCG therapy. Of the 13 metabolites showing significant changes after intravesical BCG therapy, only three metabolites, N-acetylneuraminic acid, myristic acid, and octanoylcarnitine, were associated with the risk of post-BCG recurrence. Heatmap visualisation demonstrated a clear association between the serum metabolite-based systemic response to intravesical BCG therapy and better clinical outcomes. We detected 14 metabolites for which failure of upregulation after intravesical BCG therapy led to a higher risk of tumour recurrence. The MetRes score model, which consisted of 16 metabolites, stratified the risk of recurrence in the discovery cohort.
A delay in RC leads to a shortened cancer-specific survival time when compared to immediate cystectomy, at the time of NMIBC. 32 Pre-BCG biomarkers are clinically more useful than response-based biomarkers for predicting patient outcomes after intravesical BCG. In this study, we developed two MetPreBCG scoring models based on selected serum metabolites. MetPreBCG-score model 2, using pre-BCG serum concentrations of three metabolites with absolute quantities (carnitine, indole-3-acetic acid, and valeric acid), would be more easily applied than MetPreBCG-score model 1, using a combination of pre-BCG serum concentrations of three metabolites with absolute quantities and three metabolites with relative quantities (Octanoylcarnitine, S-Methylcysteine-S-oxide, and theobromine). Analysis of unadjusted and adjusted HR demonstrated similar predictive accuracy for MetPreBCG score models 1 and 2 (Table 4). Notably, high serum carnitine levels were associated with a higher risk of post-BCG recurrence (HR: 5.70, vs. patients with low serum levels; p = 0.032). Carnitine is naturally and widely present in many foods, especially foods of animal origin such as red meat, and is synthesised endogenously in the liver, kidneys, and brain from the amino acids lysine and methionine. 33 Carnitine plays a vital role in energy production in the form of adenosine triphosphate by facilitating the transport of long-chain fatty acids to mitochondria. Carnitine supplementation or inadequacy has been reported to be associated with several disorders, such as Alzheimer's disease and dementia, cardiovascular disease (CVD), peripheral artery disease, insulin resistance, diabetes, infertility, osteoarthritis, athletic performance enhancement, and weight loss. 34 A meta-analysis revealed that oral supplementation of L-carnitine can inhibit the inflammatory state through increase in the level of antioxidant capacity and decrease in the levels of CRP, IL-6, TNF-α and malondialdehyde in the serum. 35 Moreover, the anti-oxidant action of L-carnitine in the organs of aged animals and deficiency of L-carnitine are related to a higher rate of infection. 36 Previous evidence and our findings suggest that the anti-inflammatory, anti-oxidant, and anti-infectious actions of high levels of carnitine inhibit Mycoplasma bovis infection of the urothelial mucosa and anti-tumour immunity, leading to a high risk of post-BCG recurrence.
This study has several limitations. First, our study cohort comprised patients who met the inclusion criteria, indicating an inherent potential for selection bias. The enrolled patients will be treated between 2016 and 2021. During this period, the treatment strategy, modality, and surgical skill changed over time, which could influence the outcomes. Second, patients’ backgrounds differed between the discovery and validation cohorts. No patients in the discovery cohort received maintenance BCG, whereas approximately one-third of patients in the validation cohort received maintenance BCG. This is because our hospital began actively considering the maintenance of BCG in early 2018. Third, transurethral biopsies after iBCG were not performed routinely in patients with CIS at our hospital. Blue-light cystoscopy has not been approved in Japan as of 2025. There was a potential risk of invalidation of the cohort of patients with CIS because follow-up cystoscopy accompanied with urine cytology might missed persistent CIS. Fourth, the validation cohort did not have post-BCG blood samples; therefore, the predictive accuracy of the MetRes-score model could not be validated. Fourth, the metabolic profiles of the biological specimens were affected by diet and food intake. However, we did not survey the daily food intake of participants in this study. In addition, we did not evaluate the inter-measurement variation, diurnal variation, or inter-day variation of these metabolomes. The variations could affect the results of this study. Fifth, other events, including progression and mortality, were not evaluated because the number of events was relatively low. Further research with larger sample size would be needed to evaluated the association between the metabolic profiles and risk of progression or cancer mortality.
Conclusions
Evidence from metabolomics research and metabolomics-based biomarkers for predicting treatment outcomes remains limited in BCa. We demonstrated that numerous serum metabolites were altered in response to intravesical BCG therapy and developed three high-performance and non-invasive score models which might effectively differentiated the risk of post-BCG recurrence. Large-scale prospective validation across different countries and ethnicities is needed to incorporate these biomarkers into clinical care.
Supplemental Material
sj-docx-1-blc-10.1177_23523735251325100 - Supplemental material for Serum metabolomic analysis identified serum biomarkers predicting tumour recurrence after Bacillus Calmette–Guérin therapy in patients with non-muscle invasive bladder cancer
Supplemental material, sj-docx-1-blc-10.1177_23523735251325100 for Serum metabolomic analysis identified serum biomarkers predicting tumour recurrence after Bacillus Calmette–Guérin therapy in patients with non-muscle invasive bladder cancer by Makito Miyake, Kota Iida, Nobutaka Nishimura, Sayuri Ohnishi, Takuya Owari, Tomomi Fujii, Yuki Oda, Tatsuki Miyamoto, Takuto Shimizu, Kenta Ohnishi, Shunta Hori, Yosuke Morizawa, Daisuke Gotoh, Yasushi Nakai, Nobumichi Tanaka and Kiyohide Fujimoto in Bladder Cancer
Footnotes
Acknowledgments
The authors have no acknowledgments.
Authors’ contributions
Makito Miyake and Kiyohide Fujimoto had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Miyake, Fujimoto.
Acquisition of data: Nishimura, Owari, Oda, Miyamoto, Shimizu, Ohnishi K, Hori, Morizawa, Gotoh, Nakai.
Analysis and interpretation of data: Miyake, Iida.
Drafting of the manuscript: Miyake, Iida.
Critical revision of the manuscript for important intellectual content: Fujimoto.
Statistical analysis: Miyake, Iida.
Obtaining funding: Miyake, Fujimoto.
Administrative, technical, or material support: Ohnishi S, Fujii.
Supervision: Tanaka.
Other: None.
Funding
This study was supported by KAKENHI grant 16K20159, 23K08720 (M.M.); 15K10605 (K.F.)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
