Abstract
Background
During surveillance of high-risk non-muscle invasive bladder cancer (HR-NMIBC), occult disease can be missed by standard cystoscopy.
Objective
To determine the utility of enhanced restaging procedures.
Methods
We retrospectively reviewed 297 patients with HR-NMIBC who underwent enhanced restaging procedures during the first surveillance following induction intravesical therapy between 2010–2021. Patients were stratified by number of induction treatments with unique agents (161, 63, and 73 patients with 1, 2, and 3+ treatments) and analyzed using exact logistic regression models. Enhanced restaging procedures included standard cystoscopy (white-light cystoscopy with bladder wash cytology) plus additional components including blue-light cystoscopy, mapping bladder biopsies, retrograde pyelograms, upper tract cytologies, and prostatic urethral biopsies.
Results
When standard cystoscopy was negative, blue light cystoscopy detected occult bladder cancer in 6.0%, 7.4%, and 19% of patients in the 1, 2, and 3+ treatment groups. History of CIS was associated with increased detection with blue light (p = 0.03). Extravesical (upper tract or prostatic urethral) cancer was detected by additional restaging components in 0.6%, 1.7%, and 15% of patients with 1, 2, and 3+ intravesical treatments. On multivariable analysis, receipt of 3+ intravesical inductions increased the odds of having at least one additional restaging component identify cancer (HR 3.76; p < .01).
Conclusions
Blue light cystoscopy improves surveillance of HR-NMIBC, particularly in those with CIS. Additional restaging procedures improved detection of extravesical disease in patients with heavier pre-treatment history. Risk-adapted utilization of enhanced restaging procedures requires further study.
Introduction
Following induction therapy, the guideline-recommended surveillance protocol for high-risk non-muscle invasive bladder cancer (NMIBC) is standard cystoscopy, entailing white-light office cystoscopy and urine cytology.1–3 Enhanced cystoscopy with blue light or narrow band imaging is recommended when available, although there are challenges in incorporating this technology in the office.1,4–6 However, it is unclear if standard cystoscopy is sufficient for contemporary surveillance of all patients with NMIBC.7,8 Historical series show a mixed role of bladder mapping biopsies.9–11 Furthermore, extravesical disease can develop in other parts of the urinary tract such as the upper tracts (UTs) and prostatic urethra; particularly after extended duration of disease and prolonged intravesical treatment(s).1,12,13
Blue light cystoscopy (BLC), which utilizes intravesical hexaminolevulinate to enhance tumor visualization, has been shown to improve detection rates for residual and recurrent disease, particularly CIS.4–6 Given these advantages, multiple studies have demonstrated that BLC-guided TURBT reduces recurrence rates compared to standard cystoscopy alone, and its incorporation into surveillance strategies has been increasingly recognized in clinical practice. Given these advantages, clinical practice guidelines, including the American Urological Association (AUA), European Association of Urology (EAU), and National Comprehensive Cancer Network (NCCN), recommend the use of BLC during the initial TURBT in patients with high-risk disease or suspected CIS.2,3,14 The EAU guidelines further support its use in repeat TURBT for high-risk cases, while an expert consensus statement advocates for its broader implementation.2,15 Despite these recommendations, BLC is not yet universally adopted, largely due to cost and resource constraints, leading to variability in practice.6,16 Furthermore, there are no evidence-based tailored recommendations for its use in select patients during surveillance.
There has been a surge in development of effective salvage intravesical therapies, with recent FDA approvals of pembrolizumab, nadofaragene firadenovec, and nogapendekin alfa inbakicept (NAI), in addition to promising data and ongoing trials supporting CG0070, TAR-200, sequential intravesical gemcitabine and docetaxel (Gem/Doce), and hyperthermic Mitomycin C.7,17–20 As effective salvage options increase, the timely initiation of subsequent therapy will maximize efficacy and oncologic outcomes.21,22 Thus, there is growing rationale to perform a more thorough evaluation than standard cystoscopy, with consideration of early operative evaluation.12,23,24
Our institution routinely employs an enhanced operative restaging procedure during the first surveillance cystoscopy following induction therapy for patients with high-risk NMIBC. Therefore, herein, we aimed to evaluate the effectiveness of enhanced restaging in detecting disease recurrence when standard cystoscopy is negative.
Materials and methods
Study design and population
After obtaining IRB approval, we retrospectively reviewed patients at the University of Iowa between 2009 and 2021 with high-risk NMIBC who underwent a 3-month post-induction enhanced operative restaging procedure with one or more restaging components completed. The review was assessed for transparency via STROBE reporting guidelines. 25 To explore potential patterns in cancer detection across treatment groups and to assess the utility of additional surveillance components in different stages of disease management, cohorts were grouped based upon number of unique intravesical induction treatments received: 1, 2, and 3+. A treatment was defined as ≥1 induction courses with a unique intravesical therapy and did not account for any maintenance therapy received. All patients had received at least 1 prior induction course of intravesical BCG. Thus, immediately prior to operative restaging, patients in the 1 prior treatment group had only received induction BCG with or without IFN. Subsequent induction regimens following BCG included Gem/Doce, sequential valrubicin and docetaxel (Val/Doce), quadruple chemotherapy (Quad Chemo), sequential gemcitabine and cabazitaxel with pembrolizumab (GCP), and BCG + IFN.23,26–29
While the departmental protocol was to perform initial surveillance as an operative restaging procedure, a minority of patients underwent standard cystoscopy in office due to preference to avoid anesthesia or were managed by urologists who preferred standard cystoscopy; these patients were excluded.
Enhanced operative restaging procedures
Standard cystoscopy was defined as white light cystoscopy plus urine cytology. Enhanced operative restaging procedures included standard cystoscopy as well as additional restaging components including BLC, bladder fluorescence in-situ hybridization (FISH), bilateral retrograde pyelograms, bilateral UT wash cytologies, mapping bladder biopsies, and prostatic urethral biopsies. 1 Components were deemed positive if pathologically corroborated via tissue biopsy or urine cytology. FISH data was not included in this analysis. Routine UT imaging was performed per AUA guidelines. 1
Restaging procedures began with a thorough evaluation under white light and collection of urine cytology via the cystoscope. Next, BLC was performed as previously described to improve the detection of CIS and small papillary tumors. 5 Ureteral orifices were then identified and cannulated using a 5-French open-ended catheter to the level of the ureteropelvic junction under fluoroscopic guidance. To minimize UT contamination from the bladder, positive pressure saline was instilled into the ureteral catheter up until entrance into the ureteral orifice and separate catheters and wires were utilized for each upper tract. 12 Upper tract washings were obtained twice in the renal pelvis and at 5-cm intervals down the ureter and pooled to help identify cancerous cells shed from the ureters and renal pelvis. Retrograde pyelograms were then performed to visualize the ureters and renal pelvis and detect any abnormalities not visible via standard imaging techniques (i.e., CT Urogram). Patients received 5 mapping bladder biopsies of the midline dome, midline high posterior wall, midline low posterior wall, and right and left lateral wall biopsies to assess for occult disease which may not be immediately visible on cryptoscopic evaluation. Lastly, standard cystoscopy may miss abnormalities in the prostatic urethra which is an important site of disease spread in high-risk patients. Thus, patients with prostates received mapping right and left prostatic urethral biopsies in the peri-verumontanum area.
Patient-specific circumstances dictated changes in the enhanced restaging procedure protocol. First, if no tumor was seen by white or blue light then all additional components were executed except when a component had already been performed and was negative within 6 months. Second, if gross tumor or areas suspicious for CIS were identified during the restaging procedure, manipulation of the UTs (UT cytology, retrograde pyelograms) were deferred to minimize the risk of extravesical cancer seeding. In this scenario, mapping bladder biopsies were generally deferred since the detection of occult CIS was felt to be less impactful. Additionally, if an upper tract component was deferred due to suspicion of concurrent lower tract disease, it was performed during the subsequent “indicated” restaging once any suspicion for lower tract disease was resolved via negative tissue biopsy.
Analysis
Patient clinicopathologic features, treatment history, and oncologic outcomes were analyzed. Time groups of 2009–2015 and 2015–2021 were used to analyze temporal differences within the cohort and because 2015 marked increased utilization of Gem/Doce and other subsequent salvage regimens.23,26,29–31 Risk stratification was defined per AUA guidelines. For each of the restaging components, a positive result indicated occult cancer was detected on the restaging component only when standard cystoscopy (white light cystoscopy and bladder cytology) did not detect cancer. Exact logistic regression models were used to evaluate the effect of patient clinicopathologic features and treatment history on the odds of having a positive result for the restaging components. Estimated effects of predictors are reported as odds ratios (OR) along with 95% confidence intervals. All statistical testing was two-sided and assessed for significance at the 5% level using SAS v9.4 (SAS Institute, Cary, NC).
Results
Demographics
The final cohort included 297 patients with the following intravesical treatment history: 161 with 1 treatment, 63 with 2 treatments, and 73 with 3+ treatments (Table 1). Prior to advanced restaging procedures, 43%, 70%, and 81% of patients in the 1, 2, and 3+ treatment groups had prior CIS-containing disease, respectively.
Clinicopathological characteristics.
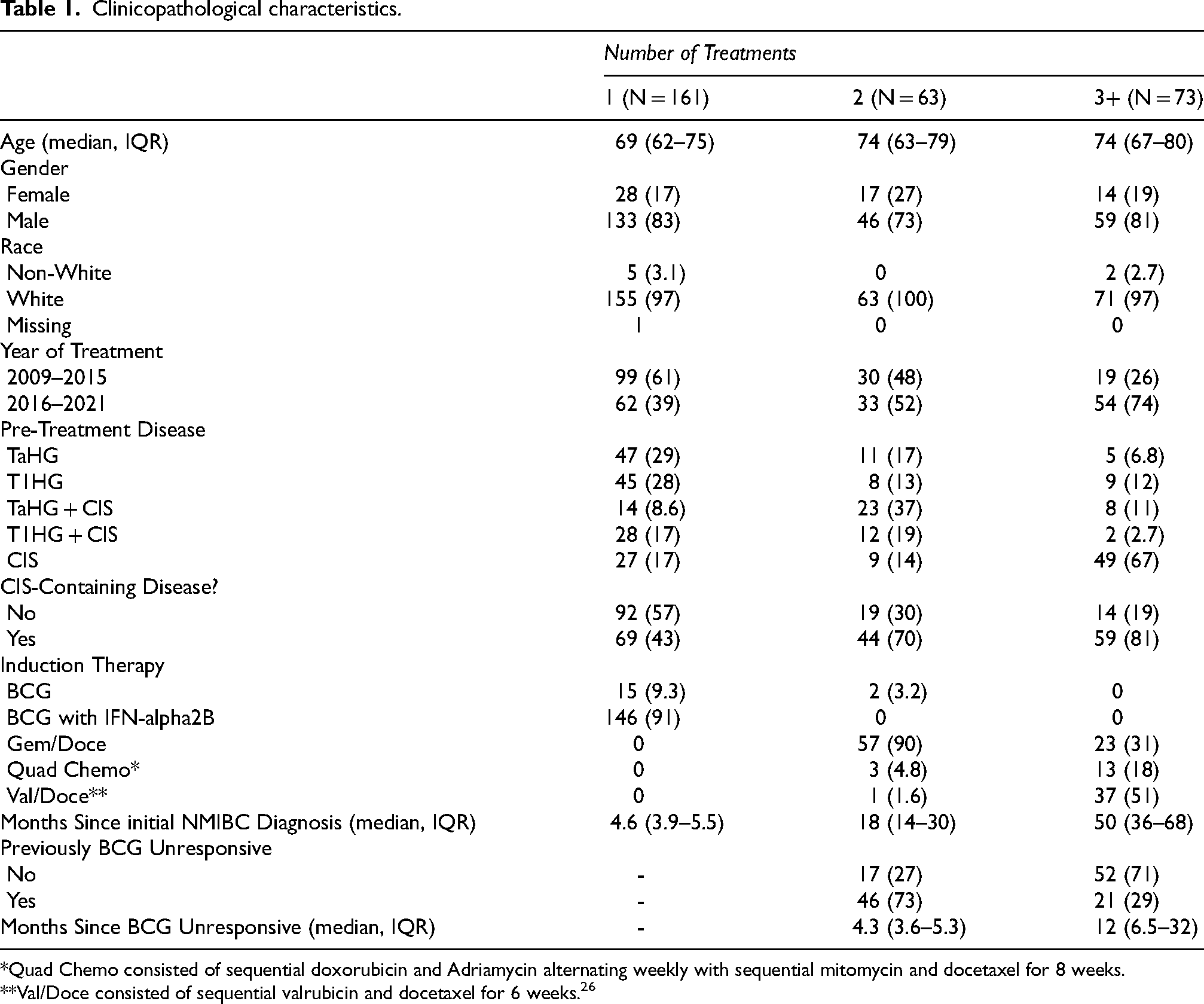
*Quad Chemo consisted of sequential doxorubicin and Adriamycin alternating weekly with sequential mitomycin and docetaxel for 8 weeks.
**Val/Doce consisted of sequential valrubicin and docetaxel for 6 weeks. 26
Cancer detection
Overall, 202 (68%) patients had no cancer and 95 (32%) had cancer detected (Figure 1). Of the 95 with cancer detected, 55 (19%) had a positive standard cystoscopy and 40 (13%) had a negative standard cystoscopy with occult cancer detected using restaging components. There were 40 patients with negative standard cystoscopy but positive restaging components (Supp. Table 1). There were a total of 47 positive components (7 cases of overlap): 24 additional cancers were identified by BLC, 8 by UT cytological washings, 11 by mapping bladder biopsy, 2 by prostatic urethral biopsy, and 2 by retrograde pyelograms. Stratified by treatment history, additional restaging components identified cancer in 6.8%, 9.5%, and 32% of patients with 1, 2, and 3+ intravesical treatments, respectively. When standard cystoscopy was negative, additional restaging components detected occult bladder cancer in 6.9%, 8.1%, and 21% of patients in the 1, 2, and 3+ treatment groups, respectively. Additional restaging components identified non-bladder (UT or prostatic urethral) cancer in 0.6%, 1.7%, and 15% of patients with 1, 2, and 3+ intravesical treatments, respectively. The bulk of occult non-bladder cancers were detected in the UT, with only 2 cases of occult prostatic urethral cancers found in patients with 3+ treatments. Extravesical cancer was rarely found in patients in the 1 and 2+ treatment groups with only 2 patients experiencing UT recurrences that were not in the 3+ intravesical treatment group.
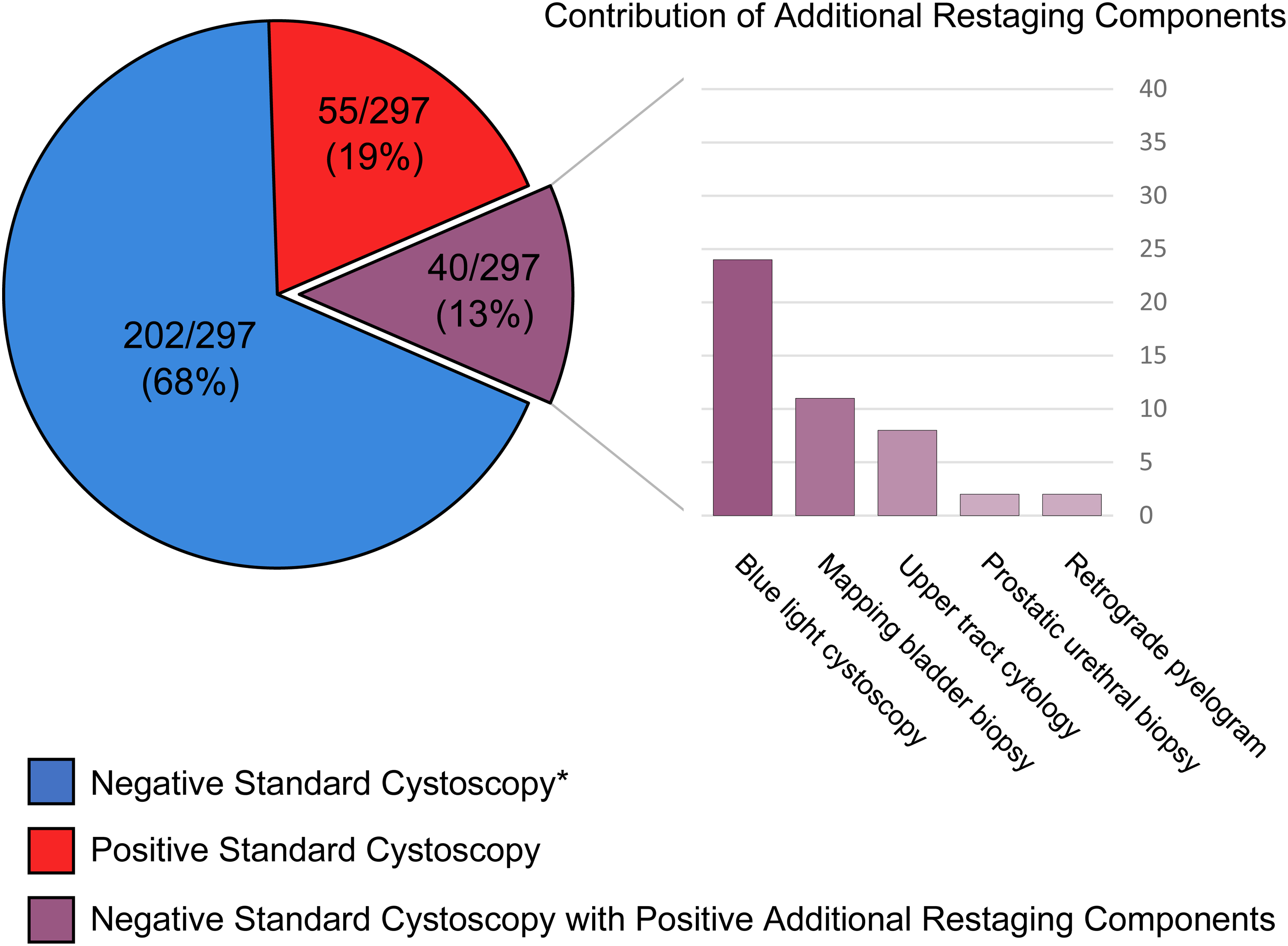
Cancer detection rates of 297 restaging procedures by component.
Cancer detection Among individual restaging components
BLC identified 24 additional cases of cancer in the absence of a positive standard cystoscopy; 7 (6%), 4 (7%), and 13 (19%) in the 1, 2, and 3+ treatment groups, respectively (Figure 2). Pathology of these 24 recurrences included 21 cases of CIS, 1 case of T1HG, 1 case of TaHG, and 1 case of TaHG + CIS. Thus, when standard cystoscopy was negative, BLC detected an additional 20 (14%) patients with CIS and 20/24 (83%) recurrences detected by BLC had some component of CIS. CIS-containing disease (p = 0.03), an increasing number of prior treatments (p = 0.01), and increasing year of treatment (p = 0.03) were associated with increased rate of cancer detection with BLC (Table 2).
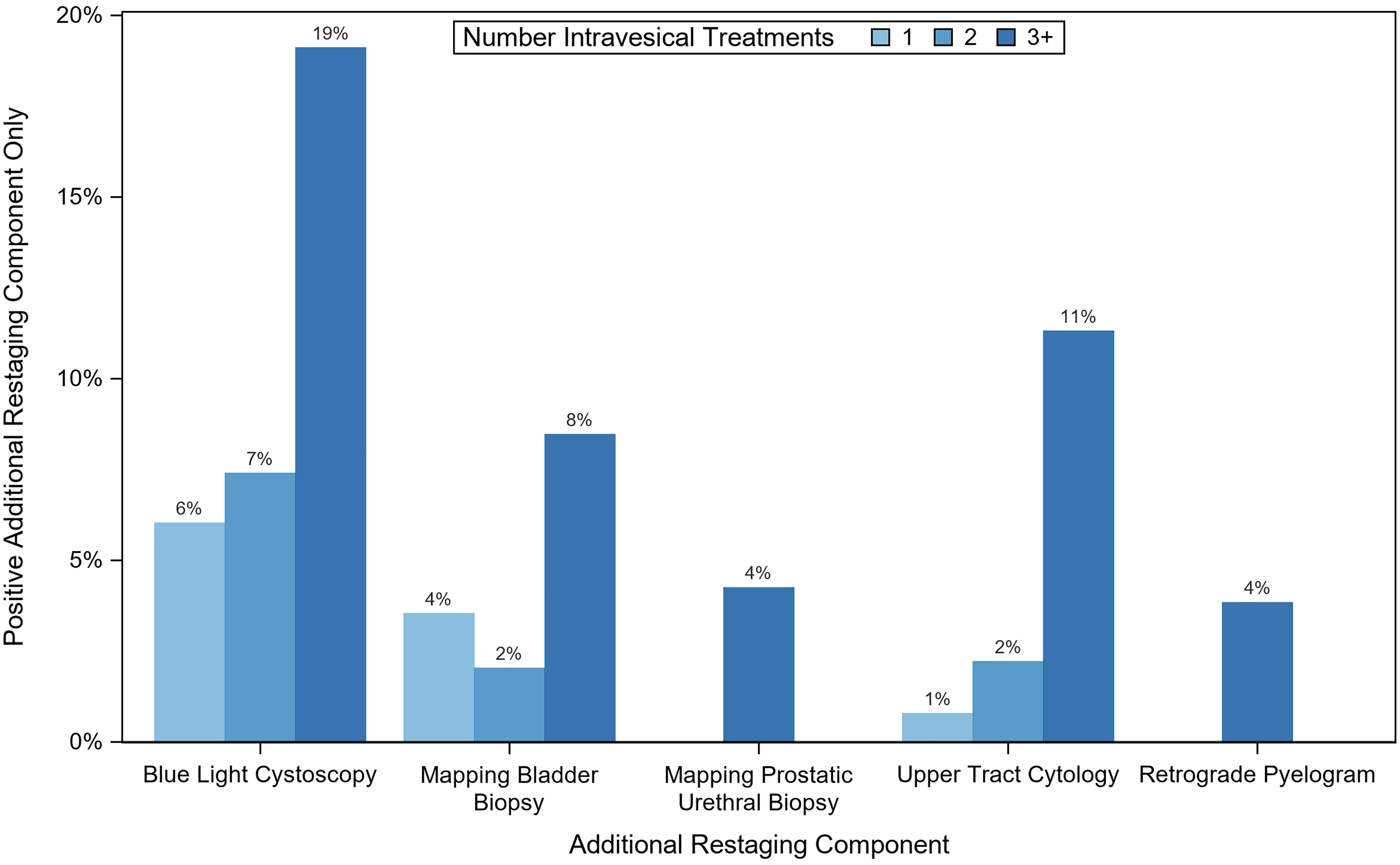
Cancer detection rates of additional restaging components among treatment groups.
Univariable results associated with blue light positivity.
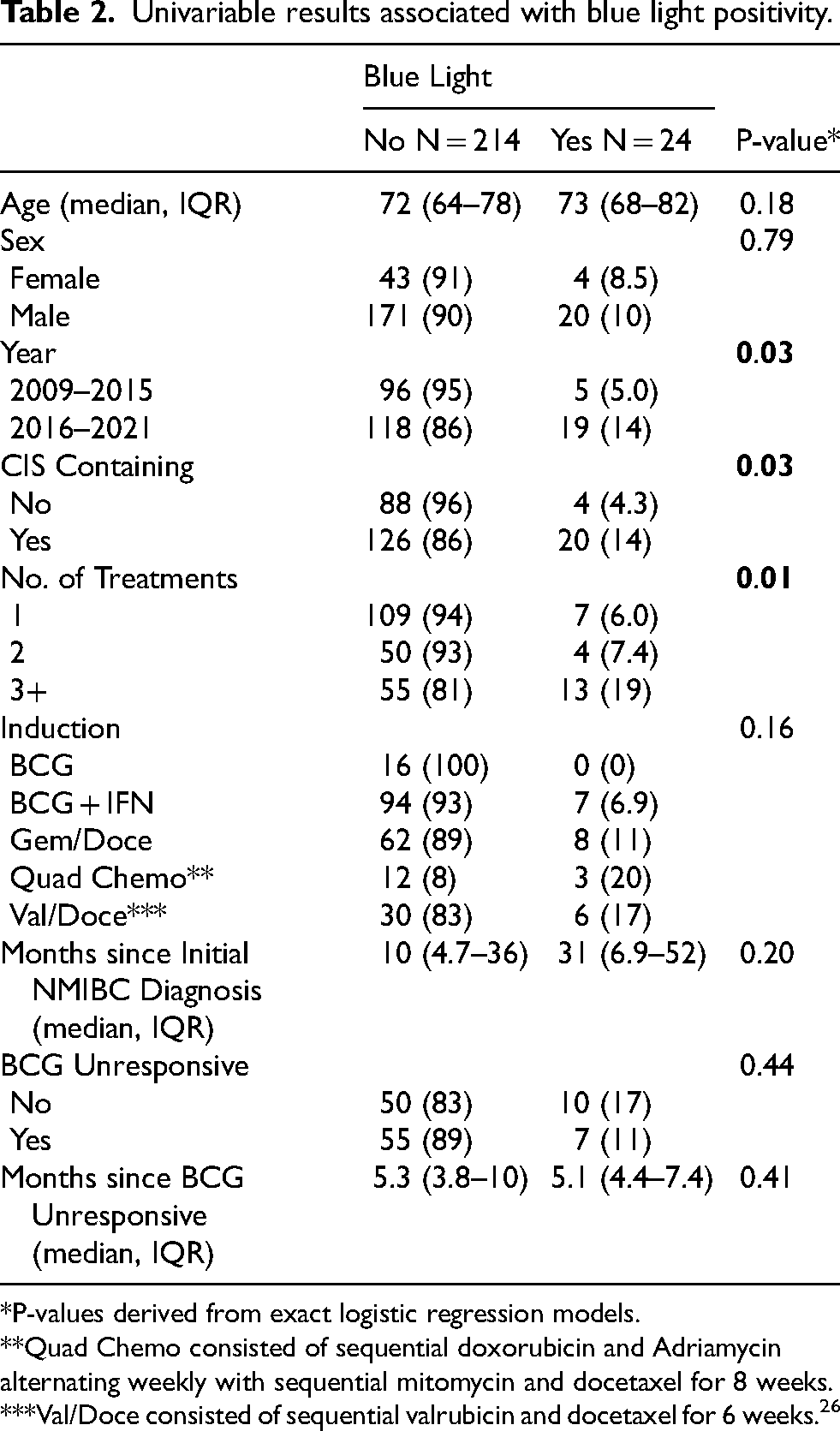
*P-values derived from exact logistic regression models.
**Quad Chemo consisted of sequential doxorubicin and Adriamycin alternating weekly with sequential mitomycin and docetaxel for 8 weeks.
***Val/Doce consisted of sequential valrubicin and docetaxel for 6 weeks. 26
Mapping bladder biopsy identified 11 additional cases of cancer in the absence of a positive standard cystoscopy (Supp. Table 2). Pathology of these recurrences included 9 cases of CIS and 2 cases of TaHG.
A mapping prostatic urethral biopsy identified occult CIS in 2 patients in the 3+ treatment group that had otherwise had a negative evaluation (Supp. Table 3).
UT cytology identified 8 additional cases of cancer via positive UT cytological washings (Supp. Table 4). Increasing number of intravesical treatments (p < .01), increasing year of treatment (p = 0.03), and intravesical therapy used (p < .01) were associated with increased odds of identifying cancer with UT cytological washes.
Retrograde pyelograms identified occult cancer in 2 patients in the 3+ treatment group that had otherwise had a negative evaluation.
Cancer detection Among one or more restaging components
To determine the overall added value of the restaging components, we analyzed whether any additional component was positive when the white light cystoscopy and bladder cytology were negative. On univariable analysis, CIS-containing disease (p = 0.02), an increasing number of prior treatments (p < .01), months since initial NMIBC diagnosis (p = 0.04), an increasing year of treatment (p < .01), intravesical treatment regimen (p < .01), and patient age (p = 0.01) were all associated with increased odds of cancer detection on at least one of the restaging components. Year of treatment and number of treatments were significant across multiple restaging procedures while CIS-containing disease and induction treatment used were more strongly associated with specific procedures. Upon multivariable analysis, number of prior treatments (p < .01) and an increasing year of treatment (p = 0.04) remained significantly associated with the odds of having ≥1 extra surveillance measure identify cancer (Table 3). Patients with 3+ prior treatments were at 3.76 times increased odds of having at least one restaging component identify cancer (p < .01).
Multivariable results associated with one or more surveillance component detecting cancer.
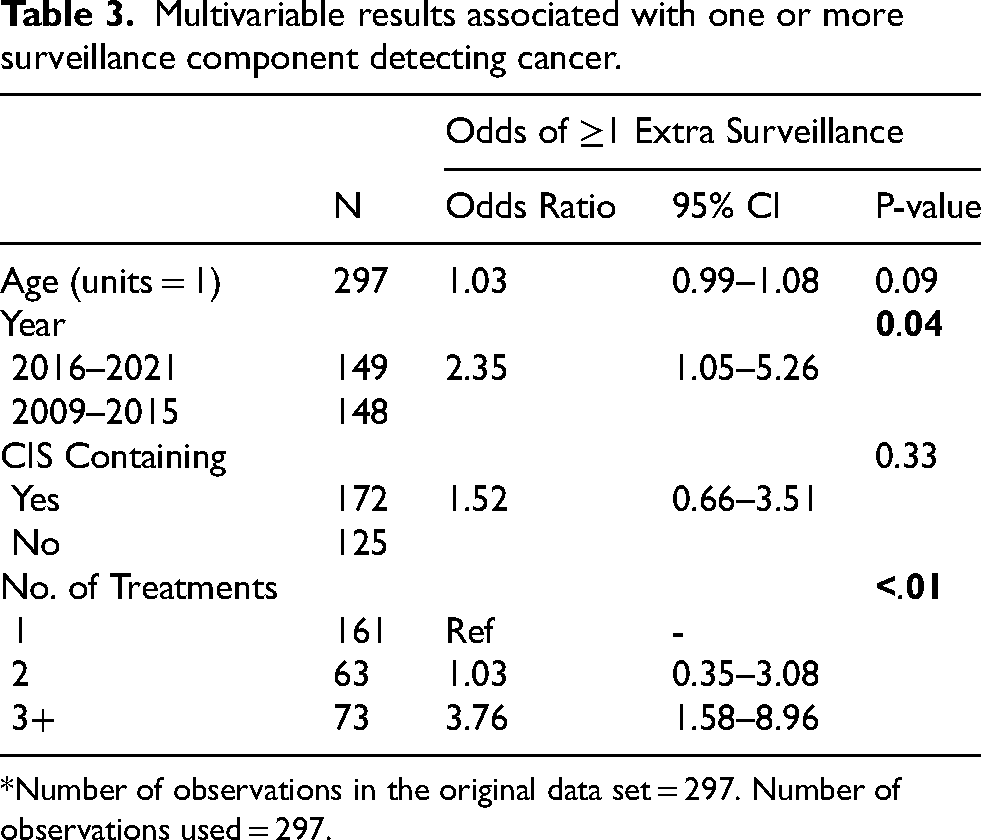
*Number of observations in the original data set = 297. Number of observations used = 297.
Discussion
Our study has several key findings (Table 4). First, BLC identified additional cancer in 10% of patients with a negative standard cystoscopy. Both history of CIS and increasing prior treatment increased the rate of BLC detecting occult bladder cancer, which was predominantly CIS. Next, extravesical cancers were increasingly detected in patients with greater prior treatment, with 15% of patients with 3+ treatments having prostatic urethral and UT disease. Finally, there was increasing utility of any restaging component as prior treatment history increased; multivariable analysis demonstrated that patients with 3 prior treatments were at 3.8 times increased odds of having restaging components identify cancer. In summary, these results suggest that patients with history of CIS benefit from BLC regardless of number of prior inductions, and that patients with significant treatment history, particularly 3+ intravesical regimens, benefit from utilizing all enhanced operative restaging components.
Summary of key recommendations.

Multiple studies have shown improved cancer detection rates when implementing BLC. A meta-analysis found that BLC identified 41% more patients with occult CIS than white light alone. 6 In a Phase III trial of blue-light flexible cystoscopy (BLFC), 13/63 (21%) patients with recurrent NMIBC were detected by BLFC only. 5 In light of these results, an expert consensus statement recommended that BLFC may be implemented at 3-month surveillance for patients with high or intermediate risk disease. 15 However, BLFC is not widely available and many urologists are understandably reluctant to proceed with operative BLC. Within our current study, we confirmed an association between history of CIS and increased detection of occult cancer with BLC, regardless of pre-treatment history. We also found that BLC identified an additional 19% of cancers in the 3+ treatment group. Thus, our results would suggest universally utilizing BLC in patients with high-risk disease, and particularly in those with history of CIS or increased number of prior inductions.
Our results corroborate existing literature demonstrating high rates of extravesical disease recurrence after multiple prior intravesical therapies. Herr has described progression of extravesical tumor relapse in patients with NMIBC, identifying 78/307 (25%) developing UT disease and 61/307 (24%) developing prostatic urethral disease at a median of 56 and 11 months, respectively. 32 A more recent study found that over half of all patients with high-risk NMIBC failing 2+ courses of BCG developed urethral and/or UT disease at a median of 3.5 years. 13 These authors advocated for a detailed work-up of the UT and prostatic urethra following intravesical BCG. Beyond BCG, a recent Phase I trial investigating salvage intravesical cabazitaxel, gemcitabine, and cisplatin found that 3/18 patients recurred only in the prostatic urethra. 33 Furthermore, of the 5 patients who underwent cystectomy, 4 had residual disease of the prostatic urethra and/or UTs. This evidence demonstrates the propensity for disease to develop in extra-vesical “sanctuary sites” in patients with multiple prior intravesical treatments.
There is limited evidence on optimal surveillance strategy and timing for adjunctive procedures for HR-NMIBC.32,34 This has led to heterogenous protocols with standard cystoscopy alone, mandatory 1 year mapping bladder biopsies, and mandatory 3- month bladder biopsies in recent clinical trials.17,18,35 Given these ongoing discrepancies, a recent guidance document (SITC-IBCG) was developed, although it did not include risk-stratified disease evaluation recommendations. 36 Based upon our data, we recommend a risk-adapted surveillance approach with increasing attention directed to patients with history of CIS or more extensive treatment history. In patients receiving their first or second intravesical treatment, BLC should be utilized, especially if there is history of CIS. However, there does not appear to be strong support for additional restaging components in this cohort. While an argument can be made that occult bladder CIS at initial post-BCG surveillance can often be successfully treated with continued BCG, there are still merits to understanding the presence and volume of disease. 1 This information can guide future surveillance approach and BLC utilization. Furthermore, alternative first-line agents may not have the same ability as BCG to eradicate CIS from 3 to 6 months,37,38 and thus effective alternative salvage therapies can be initiated sooner. In patients with heavier pre-treatment history, after 3 courses of intravesical therapy and beyond, it appears crucial to utilize enhanced operative restaging after induction given the high rate of UT (13%) and prostatic urethral (4.3%) disease, and thus these patients should routinely undergo all components of enhanced operative restaging at first evaluation.
Our study found that CIS-containing disease was significantly associated with cancer detection in the univariable analysis but not in the multivariable analysis. This discrepancy suggests that the effect of CIS-containing disease on cancer detection may be influenced by other confounding factors such as number of prior treatments, which could explain the attenuation of CIS's significance in the multivariable model.
While enhanced restaging components, such as blue light cystoscopy, may increase the detection of cancers, the clinical significance of these findings remains to be fully established. However, we believe it is logical that earlier detection of minimal disease can optimize oncologic outcomes, especially as increasing effective bladder sparing options emerge. Furthermore, importantly, false positives can occur when benign or normal tissue fluoresces red under blue light, which could lead to unnecessary biopsies and potentially patient discomfort, complications, and increased healthcare costs. Future large studies are needed to weigh pros and cons of this more pro-active approach.
There are limitations to the current study. The retrospective nature of this study allows for case selection bias. Comparison between treatment groups were limited by the smaller number of patients with an increasing number of prior intravesical treatments. We observed a trend in the prevalence of CIS-containing disease across treatment groups, with prevalence increasing from 43% in the 1-treatment group to 81% in the 3+ treatment group. More pre-treatment CIS (as seen in the 3+ treatment group) could lead to higher detection rates. Though CIS may theoretically potentiate extravesical recurrences, when controlling for CIS-containing disease of the bladder on multivariable analysis, there was still significantly increased utility of restaging components for patients with increasing number of prior intravesical treatments. Additionally, variation in the specific prior treatment regimens could introduce confounding factors. Different intravesical therapies, including BCG or chemotherapy, may influence disease biology and detection outcomes differently and ultimately affect the results. This is a tertiary-care high-volume bladder cancer center, and thus, the generalizability of these results outside academic institutions may be limited given resource availability and the innate patient selection bias. Despite this, the findings presented here offer a risk-adapted approach that can be applied broadly. For instance, clinicians in lower-resource settings could prioritize BLC for high-risk patients, particularly those with CIS or multiple prior intravesical therapies, and consider referral to a tertiary center for more comprehensive evaluations when necessary.
Conclusions
In evaluation of patients undergoing enhanced restaging procedures for high-risk NMIBC, blue light cystoscopy identified additional recurrences missed by standard cystoscopy, particularly in patients with history of CIS. Other additional restaging components became increasingly useful as the degree of prior treatment increased, particularly to detect extravesical disease. These results support consideration of risk-adapted use of enhanced operative restaging procedures in the real-world surveillance of high-risk NMIBC and prospective clinical trials.
Standard Abbreviations: NMIBC – non-muscle invasive bladder cancer; Gem/Doce – gemcitabine and docetaxel; BCG – bacillus Calmette-Guerin; TURBT – trans-urethral resection of bladder tumor; RFS – recurrence-free survival; FISH – fluorescence in-situ hybridization; CIS – carcinoma in situ
Supplemental Material
sj-docx-1-blc-10.1177_23523735251324318 - Supplemental material for The role of blue light cystoscopy and additional operative evaluations during first surveillance after induction therapy for high-risk NMIBC
Supplemental material, sj-docx-1-blc-10.1177_23523735251324318 for The role of blue light cystoscopy and additional operative evaluations during first surveillance after induction therapy for high-risk NMIBC by Ian M McElree, Ryan L Steinberg, Sarah L Mott, Helen Y Hougen, Michael A O'Donnell and Vignesh T Packiam in Bladder Cancer
Footnotes
Acknowledgments
The authors have no acknowledgments.
Author contributions
Conceptualization, MO, VP; Methodology, MO, VP; Software, IM, SB; Validation, MO, VP; Formal Analysis, SB; Investigation, MO, VP, RS, IM; Resources, MO, SB; Data Curation, IM, VP; Writing – Original Draft Preparation, IM, SB; Writing – Review & Editing, MO, HH, VP, RS; Visualization, IM; Supervision, MO, VP; Project Administration, MO, VP; Funding Acquisition, MO, SB. All authors had access to the data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the John & Carol Walter Family Foundation and the Cancer Center Support Grant.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IM, RS, SM, and HH have no disclosures. VP has the following consulting disclosures: Valar Labs, Urogen, Photocure, Veracyte. MO has the following consulting disclosures: Abbott, Photocure, Fidia, Merck, Theralase, Urogen, Vaxiion. Additionally, MO serves on the editorial board for the Bladder Cancer journal.
Data availability
The participants of this study did not give written consent for their data to be shared publicly, so due to the sensitive nature of the research supporting data is not available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
