Abstract
Background:
In current clinical practice, the use of switch maintenance avelumab is recommended for patients with locally advanced and metastatic urothelial carcinoma who experience favorable responses to first-line chemotherapy.
Objective:
We aimed to evaluate the potential advantages of platinum-based chemotherapy (Pl-CT) rechallenge after maintenance avelumab.
Methods:
A retrospective analysis involving 383 patients treated with first-line Pl-CT between 2015 and 2023 was conducted. Subsequent treatment strategies included Pl-CT or enfortumab vedotin (EV) following maintenance avelumab or pembrolizumab, and their benefit was evaluated.
Results:
Pl-CT rechallenge following maintenance avelumab did not show significant benefits, demonstrating lower response rates and shorter progression-free survival compared to EV. Conversely, both Pl-CT and EV following pembrolizumab showed similar efficacy.
Conclusions:
These findings suggest that in the current clinical landscape, EV might be a more preferable option than Pl-CT rechallenge subsequent to avelumab maintenance therapy, thereby influencing treatment decisions for metastatic urothelial carcinoma.
Background
The sequence of treatment for locally advanced or metastatic urothelial carcinoma (la/mUC) has dramatically shifted. The current clinical practice guidelines in Japan recommend platinum-based chemotherapy (Pl-CT) as the first-line treatment for patients with la/mUC. 1 In the future, the guidelines also suggest the potential use of enfortumab vedotin (EV) and pembrolizumab as first-line treatments. 2
Subsequent to 1L chemotherapy, patients demonstrating complete response, partial response, or stable disease can receive switch maintenance therapy with avelumab, while those experiencing disease progression are treated with second-line (2L) pembrolizumab. Recent studies have highlighted the potential efficacy of Pl-CT rechallenge following the administration of pembrolizumab in patients with la/mUC.3–5 Clinical evidence regarding the optimal treatment strategy subsequent to immune checkpoint inhibitor (ICI) therapy, specifically the choice between Pl-CT rechallenge and EV, is limited. Furthermore, the efficacy of Pl-CT rechallenge following maintenance avelumab therapy remains unclear.
We hypothesized that a significant benefit might be observed with Pl-CT rechallenge following maintenance avelumab. The rationale behind the anticipated benefit of Pl-CT rechallenge after maintenance avelumab lies in the absence of progressive disease in patients after completion of 1L Pl-CT. This study aimed to evaluate the potential advantages of Pl-CT rechallenge after ICI therapy including maintenance avelumab and pembrolizumab.
Methods
Study design
The study protocol was approved by the Institutional Review Board for Clinical Studies (Medical Ethics Committee ID: 2891) as a multicenter retrospective study. The opt-out method was applied to obtain consent from the participants via posters and/or websites. This study was conducted in compliance with the principles of the Declaration of Helsinki.
A retrospective chart review of 383 consecutive patients who underwent Pl-CT as 1L systemic treatment for la/mUC between 2015 and 2023 was conducted. Among the 383 patients, four groups were identified and included in this study: those who underwent Pl-CT rechallenge following maintenance avelumab (Ave-CT), those who received EV following maintenance avelumab (Ave-EV), those who underwent Pl-CT rechallenge following pembrolizumab (Pem-CT), and those who received EV following pembrolizumab (Pem-EV). The duration from the end of 1L Pl-CT to the start of the Pl-CT rechallenge was not restricted to a specific period. The clinicopathological characteristics and oncological outcomes were compared among the four groups.
Outcomes
The objective response rate (ORR) as per the Response Evaluation Criteria in Solid Tumors (RECIST) ver 1.1 was used to compare outcomes among the four groups. 6 Additionally, the change in ORR was evaluated between 1L chemotherapy and Pl-CT rechallenge in the Ave-CT group and Pem-CT group, respectively. Moreover, we evaluated oncological outcomes, including progression-free survival (PFS) and overall survival (OS), with the observation period defined from 1L chemotherapy or subsequent chemotherapy after ICIs.
Statistical analysis
Quantitative variables are presented as median and interquartile range (IQR), and categorical variables are expressed as proportions. Statistical analyses of patient clinicopathological characteristics were performed using appropriate tests, such as Student's t-test and Fisher's exact test. Prognostic factors for PFS and OS were assessed using Kaplan–Meier survival curves generated using GraphPad Prism 7.0 (GraphPad Software, San Diego, CA, USA). Statistical significance was set at P value of less than 0.05. These analyses were conducted using EZR software (Saitama Medical Center, Jichi Medical University, Saitama, Japan). 7
Results
Patient characteristics
Of the 383 patients, 307 who did not receive subsequent chemotherapy following ICI therapy, 6 who received non-platinum based subsequent chemotherapy following ICIs, and 15 with a lack of clinicopathological information and oncological outcomes, were excluded from the analysis. The remaining 55 patients were assigned to one of four groups: 8 patients (2.1%) in Ave-CT group, 11 (2.9%) in Ave-EV group, 15 (3.9%) in Pem-CT group, and 21 (5.5%) in Pem-EV group (Supplementary Figure). Table 1 summarizes the clinicopathological characteristics at the initiation of the Pl-CT rechallenge or EV. Patients in the Ave-CT group had a significantly lower proportion of lung metastases than other three groups. Patients in the Pem-CT group underwent cystectomy more frequently than those in the other three groups. The numbers of cycles of 1L Pl-CT in Pem-CT group was significantly lower than in the other groups. There was no significant difference in other clinicopathological factors.
The clinicopathological characteristics at the initiation of the Pl-CT rechallenge or EV. Four groups were identified: Ave-CT, Ave-EV, Pem-CT, and Pem-EV groups.
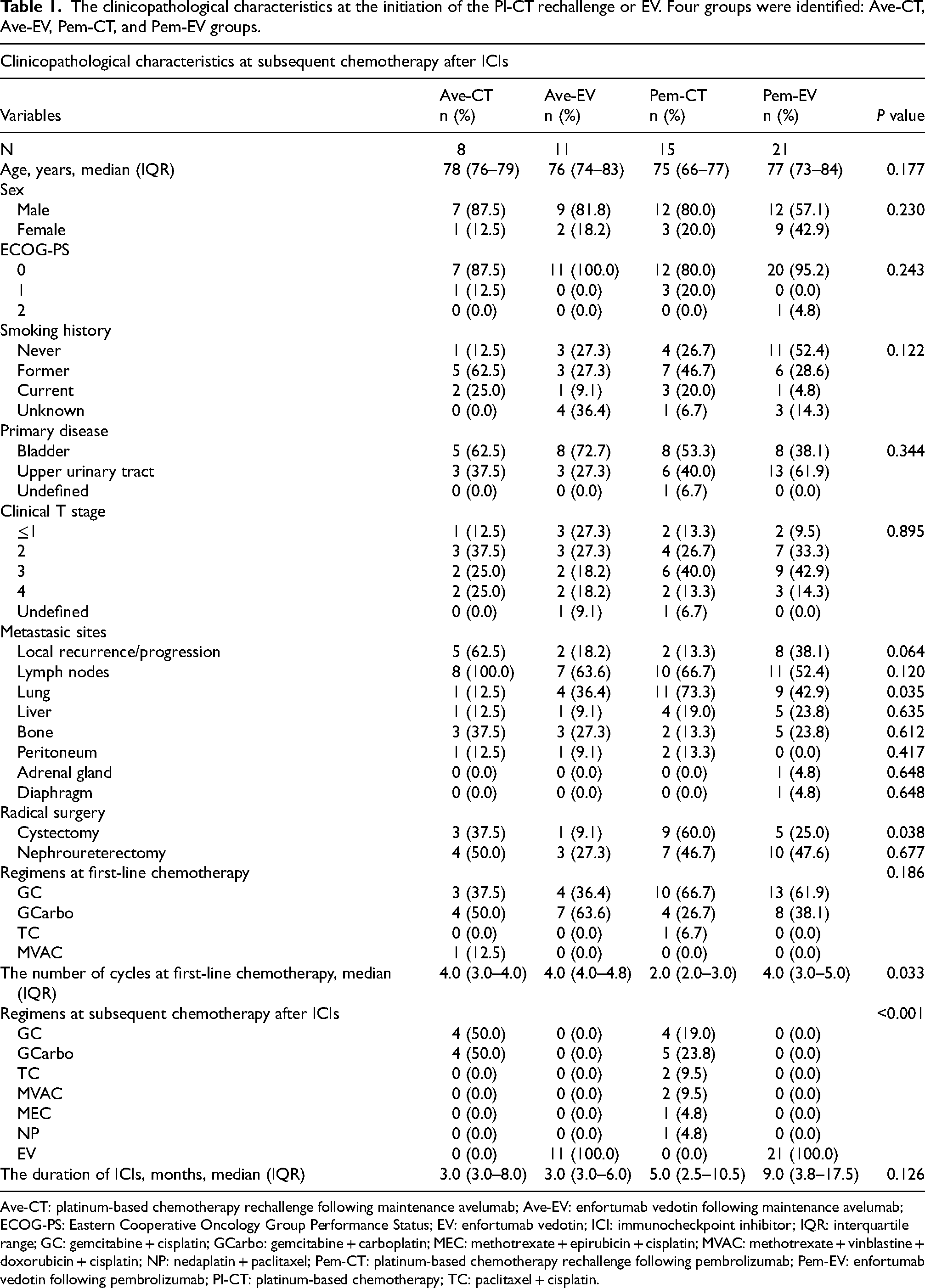
Ave-CT: platinum-based chemotherapy rechallenge following maintenance avelumab; Ave-EV: enfortumab vedotin following maintenance avelumab; ECOG-PS: Eastern Cooperative Oncology Group Performance Status; EV: enfortumab vedotin; ICI: immunocheckpoint inhibitor; IQR: interquartile range; GC: gemcitabine + cisplatin; GCarbo: gemcitabine + carboplatin; MEC: methotrexate + epirubicin + cisplatin; MVAC: methotrexate + vinblastine + doxorubicin + cisplatin; NP: nedaplatin + paclitaxel; Pem-CT: platinum-based chemotherapy rechallenge following pembrolizumab; Pem-EV: enfortumab vedotin following pembrolizumab; Pl-CT: platinum-based chemotherapy; TC: paclitaxel + cisplatin.
Oncological outcomes
A lower ORR was observed in the Ave-CT and Pem-CT groups compared to the Ave-EV and Pem-EV groups, but the difference was not statistically significant (Figure 1(A): 25.0% and 33.3%, respectively). The Sankey diagram shows a shift in the ORR between 1L chemotherapy and Pl-CT rechallenge (Figure 1(B)). In Ave-CT group, high rates of disease progression for Pl-CT rechallenge were observed although the response for the 1L chemotherapy was beyond stable disease. Additionally, in Pem-CT group, the percentage of progression disease for Pl-CT rechallenge increased compared to those for 1L chemotherapy (26.7% to 46.7%). Figure 2 showed the Kaplan-Meier curves of PFS from subsequent chemotherapy after ICIs and OS from 1L chemotherapy or subsequent chemotherapy after ICIs. Figure 2(A) shows that EV was associated with longer PFS than Pl-CT rechallenge, though this difference was not statistically significant. The Ave-CT and Ave-EV groups demonstrated similar OS (Figure 2(C) and (E)). Furthermore, no significant differences were observed in PFS or OS between the Pem-CT and Pem-EV groups (Figure 2(D) and (F)).
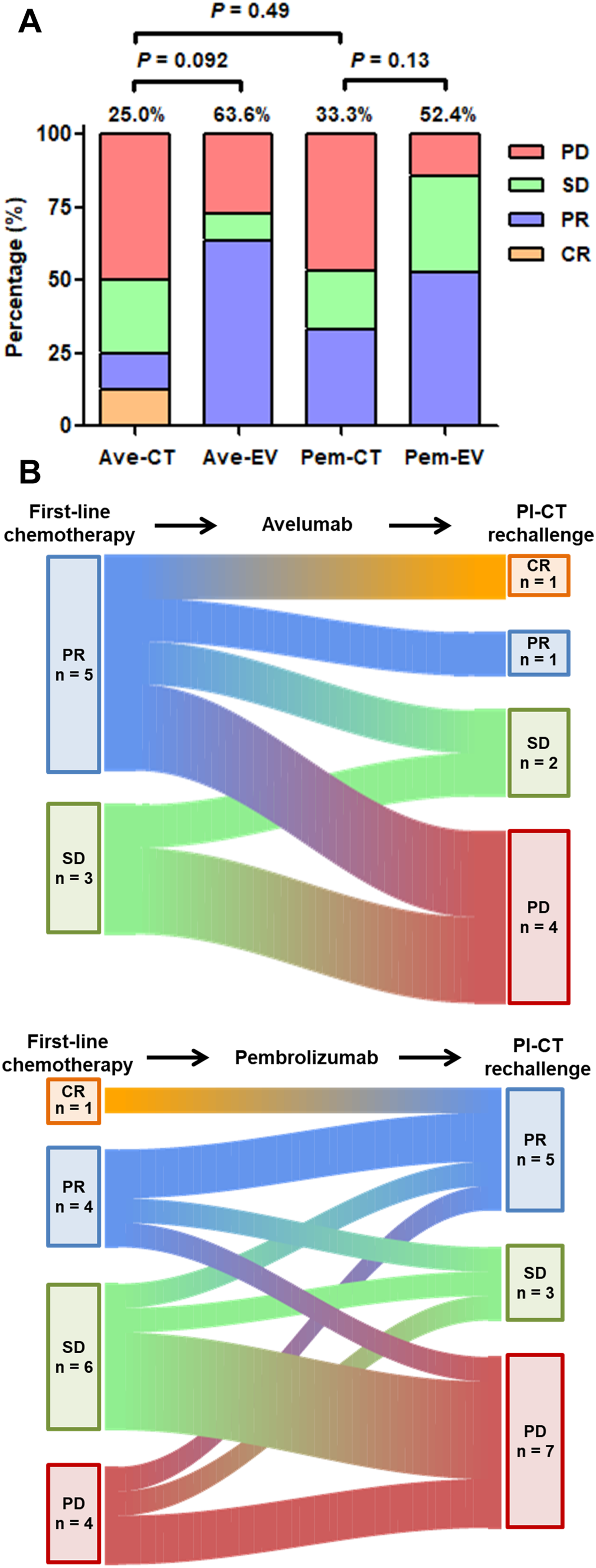
The bar chart of ORR and Sankey diagram between 1L chemotherapy and Pl-CT rechallenge.
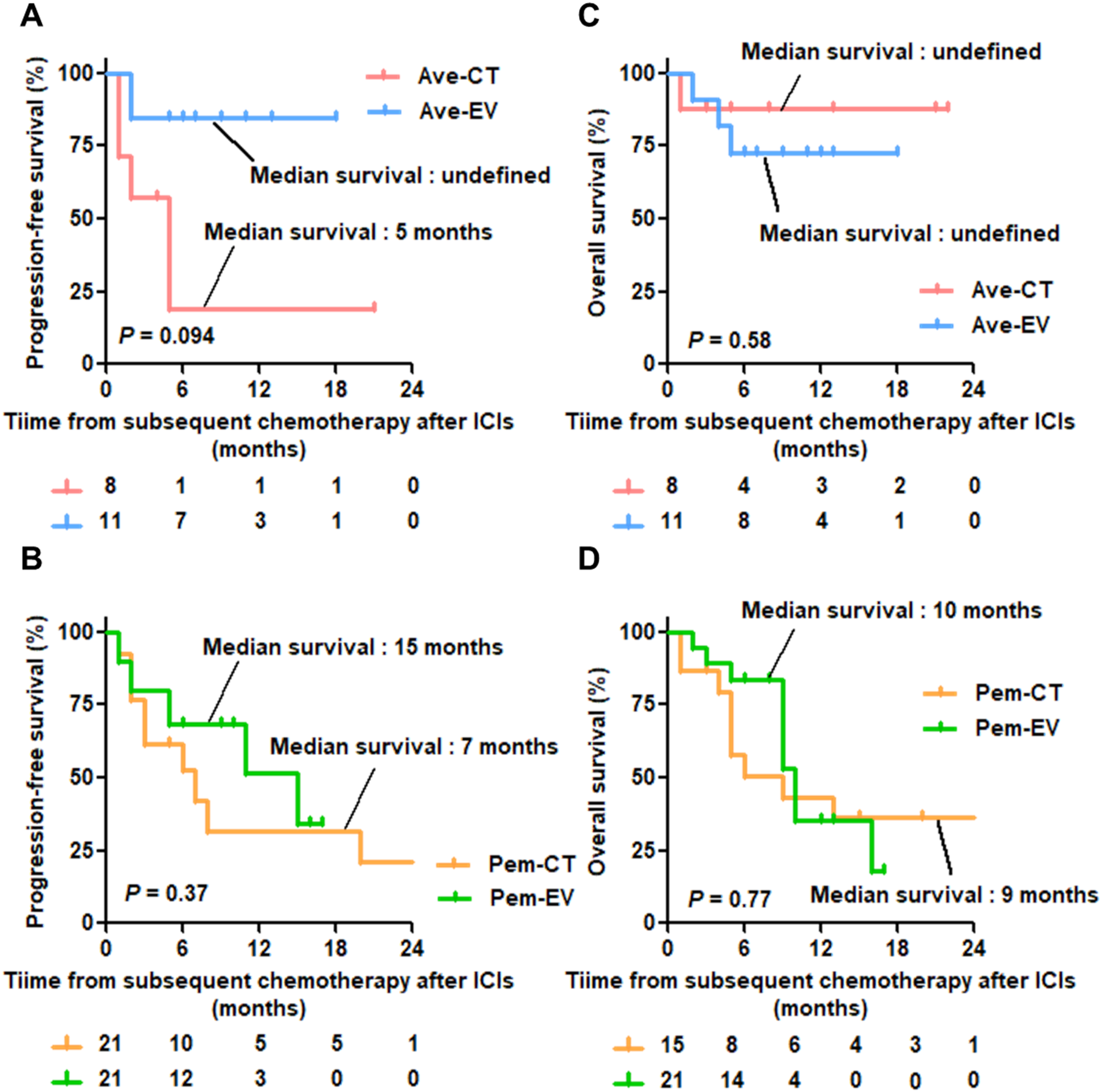
Kaplan–Meier curves of PFS and OS.
Discussion
The efficacy of chemotherapy rechallenge after maintenance avelumab are lacking.8,9 Theoretically, the tumor is anticipated to retain sensitivity to Pl-CT following avelumab maintenance therapy. Saleh et al. described one of the hypotheses to explain the high response of chemotherapy rechallenge after pembrolizumab, involving tumor microenvironment modification induced by ICIs. 10 We also assumed the high efficacy of chemotherapy rechallenge after maintenance avelumab when those after pembrolizumab had the high potential. However, our study did not corroborate this assumption.
Our findings indicate that EV may be a superior choice following ICI therapy, particularly in the maintenance avelumab group. Currently, the treatment sequence for mUC has shifted, and combination therapy with EV and pembrolizumab has recently been. While ICIs can improve resistance to Pl-CT, the efficacy of EV remains superior due to the specific mechanism of the antibody-drug conjugate. 2 Furthermore, it is unlikely that all patients will receive this combination therapy as first-line treatment, with some patients continuing to receive Pl-CT. Therefore, our results will be relevant in the next era when EV and pembrolizumab therapy become more widely approved.
In this study, we selected patients who were treated with Pl-CT rechallenge subsequent to maintenance avelumab. Our results suggest that Pl-CT rechallenge after maintenance avelumab fails to confer a notable benefit in terms of PFS. Although a good response to 1L chemotherapy was observed, it did not universally translate to a similar level of benefit for Pl-CT rechallenge across all patient groups. On the other hand, we did not observe a significant difference in OS between Ave-CT and Ave-EV groups. This is partly due to sequential therapies, including EV, after Pl-CT rechallenge. However, the study is limited by its small sample size and retrospective design. This small size may lead to low reproducibility and significant bias, potentially resulting in misinterpretation of findings. We cannot rule out the possibility that these study results contradict the original effectiveness of chemotherapy rechallenge. Therefore, further accumulation of cases is necessary to validate these findings.
This study had several other limitations that need to be addressed. First, this is the multicentre analysis and thus many physicians are involved in this study. They had each indication to initiate chemotherapy rechallenge after ICIs and this is a big selection bias. Second, data spanning 9 years from 2015 to 2023 was collected retrospectively. Over time, treatment approaches including surgical techniques have changed, and there are the various decisions made by numerous attending physicians. Moreover, this study includes patients who underwent Pl-CT rechallenge before maintenance avelumab was approval. Treatment modalities varied across different eras, potentially leading to the significant selection bias.
Conclusions
The lack of beneficial effects from Pl-CT rechallenge following maintenance avelumab contradicted our hypothesis. Conversely, after pembrolizumab, both Pl-CT rechallenge and EV demonstrated similar benefit. In current clinical setting, EV may be more favorable than PL-CT rechallenge following the maintenance avelumab.
Supplemental Material
sj-docx-1-blc-10.1177_23523735251317423 - Supplemental material for Platinum-based chemotherapy rechallenge or enfortumab vedotin after maintenance avelumab or pembrolizumab for locally advanced or metastatic urothelial carcinoma
Supplemental material, sj-docx-1-blc-10.1177_23523735251317423 for Platinum-based chemotherapy rechallenge or enfortumab vedotin after maintenance avelumab or pembrolizumab for locally advanced or metastatic urothelial carcinoma by Nobutaka Nishimura, Makito Miyake, Norimi Takamatsu, Kosuke Mieda, Kuniaki Inoue, Akira Tachibana, Keichi Sakamoto, Mikiko Onishi, Fumisato Maesaka, Takanosuke Yoshikawa, Mitsuru Tomizawa, Takuto Shimizu, Kenta Onishi, Shunta Hori, Yosuke Morizawa, Daisuke Gotoh, Yasushi Nakai, Nobumichi Tanaka and Kiyohide Fujimoto in Bladder Cancer
Footnotes
Acknowledgment
The authors would like to thank all patients who participated in this study for their important contributions. Clinicopathological statistics are based on the results of contributions from a number of institutions in Japan. We also thank the urologists for their invaluable help in obtaining and summarizing the data used in this study.
Informed consent
The opt-out method was used to obtain consent from participants through the use of posters and/or websites.
Author's contributions
Conception: Nobutaka Nishimura, Makito Miyake, and Kiyohide Fujimoto; Performance: Takuto Shimizu; Interpretation of data: Norimi Takamatsu, Kosuke Mieda, Kuniaki Inoue, Akira Tachibana, Keichi Sakamoto, Mikiko Onishi, Fumisato Maesaka, Takanosuke Yoshikawa, Mitsuru Tomizawa, Kenta Onishi, Shunta Hori, Yosuke Morizawa, Daisuke Gotoh, Yasushi Nakai, and Nobumichi Tanaka.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Makito Miyake received honorarium from Pfizer Inc., Merck Biopharma Co., Ltd, MSD K.K., and Astellas Pharma Inc. Kiyohide Fujimoto received honorarium from Pfizer Inc., Merck Biopharma Co., Ltd, MSD K.K., Ono pharmaceutical co. Ltd, and Astellas Pharma Inc. Other authors declare no conflict of interest.
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
