Abstract
Background
The practice patterns and efficacy of ddMVAC administered with split-dose cisplatin for patients with muscle-invasive bladder cancer (MIBC) remains largely undefined.
Objective
To characterize the application and overall survival (OS) in patients with MIBC receiving conventional ddMVAC versus split-dosed ddMVAC and to examine the predictive variables in those receiving split-dosed cisplatin.
Methods
Using data from the CancerLinQ Discovery database, we identified 626 patients with bladder cancer between 2000–2023 with receipt of ddMVAC. The primary outcome was OS by receipt of split-dose versus conventional ddMVAC. A secondary outcome of interest assessed predictors of receipt of split-dose ddMVAC. Use of split-dose versus conventional ddMVAC was compared using chi-square tests. Univariate and multivariable OS were estimated using Cox proportional hazards models. Predictors of receipt of split dose versus conventional ddMVAC were estimated using logistic regression models.
Results
Most patients with MIBC are treated with standard dose ddMVAC. In multivariate analysis, no statistically significant difference in OS was observed between split-dose and conventional ddMVAC (HR 1.3, CI 0.78–2.18, p = 0.316). We demonstrate a notable decline in the use of split-dose cisplatin over time. Baseline GFR and performance status were not predictors of split-dosing in this cohort.
Conclusions
Most patients with MIBC received conventional ddMVAC with decreasing frequency of split-dose cisplatin use over time. We did not observe a difference in OS between patients with MIBC who received standard versus split-dose cisplatin.
Introduction
It is estimated that in 2024, 83,190 patients will be diagnosed with bladder cancer with muscle-invasive bladder cancer (MIBC) representing approximately 20–25% of these new diagnoses.1,2 MIBC is defined as localized bladder cancer with invasion into the muscularis propria without disseminated disease, conferring a higher likelihood of distant spread and a five-year overall survival of 71%. 3 Neoadjuvant cisplatin-based combination chemotherapy followed by radical cystectomy is a category 1 recommendation in the National Comprehensive Cancer Network (NCCN) guidelines. 4 Neoadjuvant chemotherapy (NAC) functions to both target micrometastatic disease as well as aid in downstaging chemotherapy-sensitive tumors. Multiple clinical trials have demonstrated a survival benefit of NAC in MIBC.5,6 The combined analyses of the NORDIC 1 and NORDIC 2 trials showed a statistically significant difference in the five-year combined overall survival (OS) in patients receiving NAC (56% versus 48%, p = 0.045). 7 A long-term update of the BA06 30894 trial—one of the largest prospective trials examining NAC in MIBC—demonstrated a 10-year OS benefit in those receiving cisplatin-based NAC (36% vs 30%; p = 0.037). 8
The combination of dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin (ddMVAC) chemotherapies is the preferred NCCN regimen for neoadjuvant chemotherapy. The VESPER trial, an open-label, randomized Phase III trial, investigated outcomes in patients assigned to receive either four cycles of gemcitabine plus cisplatin (GC) or six cycles of ddMVAC. The results revealed that patients receiving ddMVAC in the neoadjuvant setting experienced improved five-year OS and extended time to death compared to those treated with GC. 9
The ddMVAC regimen is given every two weeks for 3–6 cycles with growth factor support. While ddMVAC is a preferred regimen for patients with MIBC, impaired renal function may preclude the use of standard, full-dose cisplatin. The administration of cisplatin given as a split dose (35 mg/m2) on days 1 and 2, is a potential treatment option in those patients with impaired renal function. The practice patterns, tolerance, and efficacy of ddMVAC administered with split-dose cisplatin, however, remains largely undefined in the literature.
We hypothesize no significant difference in OS between patients with MIBC receiving conventional versus split-dose cisplatin as part of a ddMVAC regimen. This hypothesis is explored by comparing OS outcomes between these two groups and by examining predictors for using split-dose cisplatin. The study also aims to understand the trends in practice patterns regarding the use of split-dose cisplatin over time.
Materials and methods
Data source
The data was sourced from CancerLinQ Discovery (www.cancerlinq.org/solutions/researchers): Bladder Cancer Dataset 2013–2023, August 2023 dataset release, American Society of Clinical Oncology's (ASCO's) CancerLinQ LLC. CancerLinQ Discovery® is a clinical database containing patient data aggregated from electronic health record systems of various participating clinical facilities across all 50 states. CancerLinQ® is a subsidiary of the American Society of Clinical Oncology (ASCO) and was developed to support the delivery of high-quality cancer care. Data collected include patient demographics, diagnoses, medications, procedures performed, laboratory tests, radiation, and clinical trial information.
Patient population
This study included patients aged 18 years or older diagnosed with bladder cancer between January 1, 2000, and July 31, 2023 (n = 81,538). Included patients had a record of receiving MVAC (n = 1132) and a second dose within 14 ± 3 days of the first dose (n = 768). Patients with confirmed metastatic disease (M1) were excluded (n = 91). To further exclude patients with M1 disease as well as locally advanced N2 and N3 disease who may have received ddMVAC, any patient treated with one of the following antineoplastic agents—gemcitabine, paclitaxel, docetaxel, carboplatin, atezolizumab, durvalumab, enfortumab vedotin, avelumab, pembrolizumab or erdafitinib—within six months of start of ddMVAC were excluded. Patients who received only adjuvant nivolumab (n = 39) were included in this final cohort. Patients who received more than six cycles of ddMVAC (n = 47) or received split dose ddMVAC on days 1 and 8 (n = 2) were excluded, resulting in a final patient cohort of 626 patients (Figure 1).
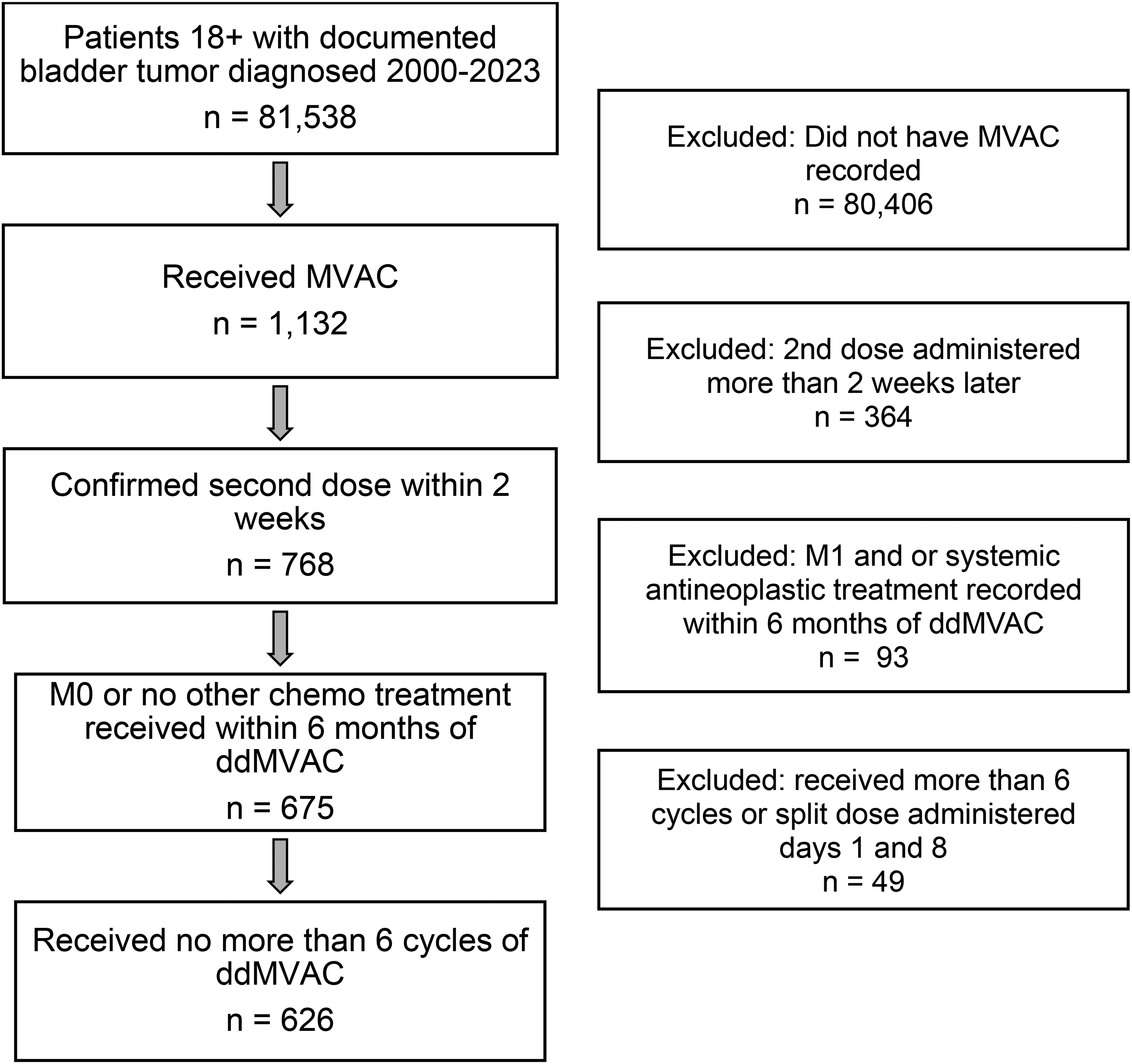
ddMVAC cohort consort.
Defining split dose
Split-dose ddMVAC was defined as receipt of cisplatin on days 1 and 2–3 (n = 60) of the first 14-day ddMVAC cycle. Patients who received split-dose cisplatin following cycle 1 (n = 27) were included in the conventional dosing cohort.
Defining GFR
Baseline and post-treatment glomerular filtration rate (GFR) were defined as GFR one week prior to the start of ddMVAC and GFR six weeks following the last ddMVAC dose.
Outcomes
The primary outcome of this analysis was overall survival by receipt of split-dose versus conventional ddMVAC. A secondary outcome of interest was identifying predictors of receipt of split-dose ddMVAC. Covariates of interest were patient sex, age at diagnosis, race/ethnicity, geographic region, year of diagnosis, functional status at the time of diagnosis (ECOG), Charlson comorbidity index (CCI), baseline and post-treatment glomerular filtration rate (GFR) and were examined in univariate regression models. Covariates that showed statistical significance in the univariate regression estimates were included in the multivariate analysis.
Statistical analysis
Use of split-dose versus conventional ddMVAC was compared by patient demographics using chi-square tests. Univariate and multivariable overall survival were estimated using Cox proportional hazards models. Violations of the proportional hazards assumption were addressed using time-dependent variables. Predictors of receipt of split dose versus conventional ddMVAC were estimated using logistic regression models. All analyses were performed in SAS version 9.4 (SAS Institute Inc., Cary, NC) and were evaluated at a p-value <0.05.
Results
626 patients with MIBC treated with ddMVAC were included in the final analyses (Table 1). The cohort comprised 75.6% men (n = 473) and 22% women (n = 138). Of the 626 patients, 90.4% (n = 566) received conventional ddMVAC while 9.6% (n = 60) received a split-dose regimen. There were no statistically significant differences in gender, age at diagnosis, year of diagnosis, ECOG functional status, CCI, baseline GFR, or number of ddMVAC cycles received between the conventional ddMVAC and split-dose cohorts.
Patient demographics.
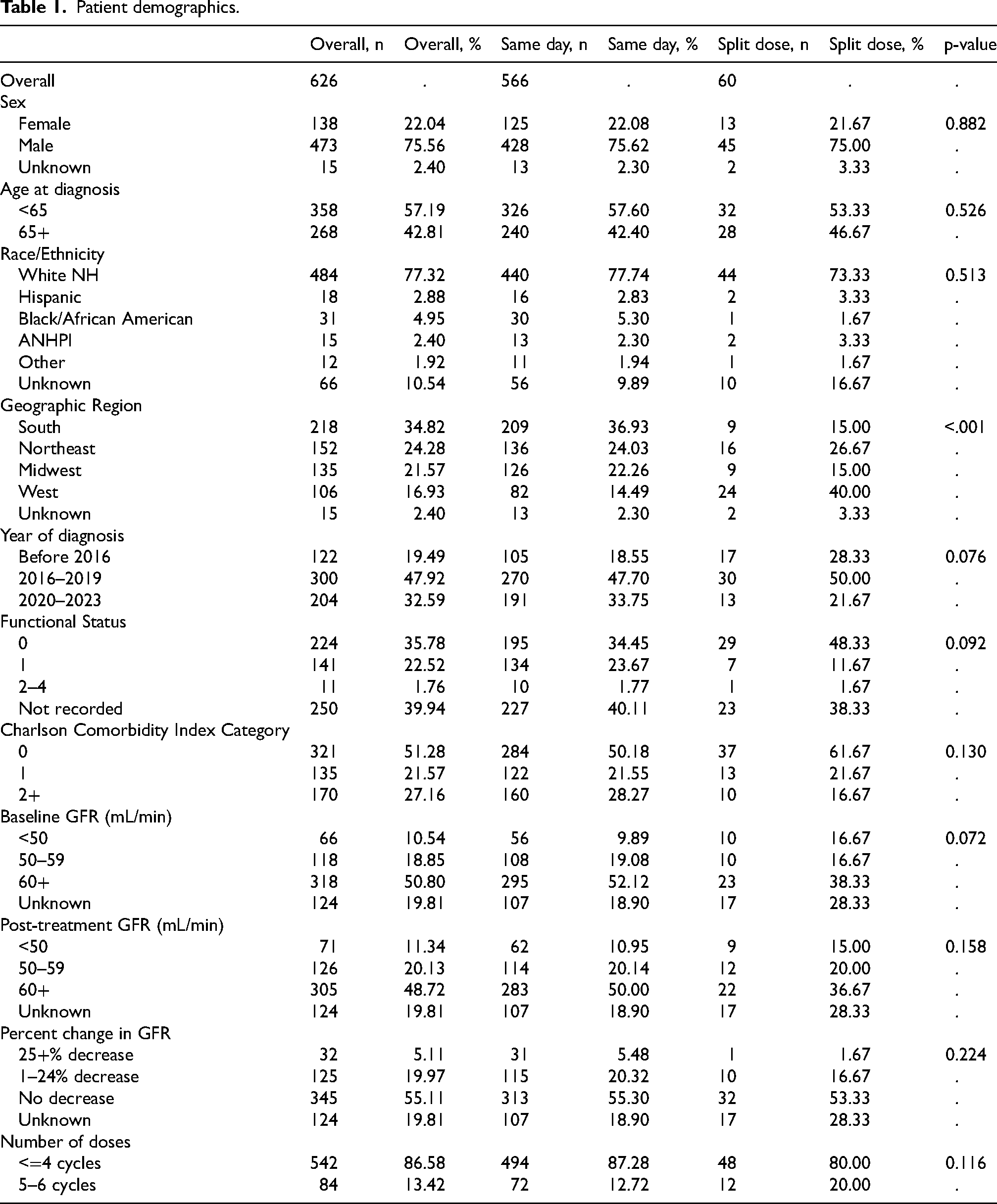
Patients 65 years or older at diagnosis had a similar distribution of conventional versus split-dosing when compared to patients <65 years (90% conventional vs. 10% split; 91% conventional vs. 9% split, respectively). While not statistically significant, the practice of split-dose cisplatin has declined over time (13.9% before 2016, 10.0% 2016–2019, 6.4% 2020–2023) (Table 1 and Figure 2). Only 1.8% (n = 11) of this cohort had a recorded ECOG performance status of 2+, 35.7% of patients having an ECOG of 0 (n = 224), and 22.5% having an ECOG of 1 (n = 141). Notably, 39.9% of this cohort (n = 250) had missing ECOG data (Table 1).
In the overall cohort of 626 patients, 10.5% (n = 66) had a baseline GFR <50 mL/min, 18.5% (n = 118) a baseline GFR of 50–59 mL/min, and 50.7% (n = 318) a baseline GFR =>60 mL/min. Although not statistically significant (p = 0.072), we observed a trend toward increased use of split-dose in patients with impaired renal function. Among those patients with a GFR <50 mL/min, 15.1% (n = 10) received split-dose cisplatin compared to 8.5% (n= 10) of patients with GFR between 50–59 mL/min and 7.2% (n = 23) in those with a GFR of 60 ml/min or higher.
Change in GFR after completion of chemotherapy was also assessed. Of those patients given conventional ddMVAC (n = 566), 55.3% (n = 313) had no decrease in post-treatment GFR, 20.3% (n = 115) had a decline in post-treatment GFR of 1–24% from baseline, and 5.5% (n = 31) demonstrated a 25% or greater decrease in post-treatment GFR (Table 1). Of those patients who received split-dose ddMVAC (n = 60), 55% (n = 32) had no change in post-treatment GFR, 16.6% (n = 10) had a decline of 1–24% from baseline and 1.7% (n = 1) had a 25% or greater decrease in post-treatment GFR.
Many patients (86.5%, n = 542) completed four or fewer cycles of ddMVAC with 7.8% (n = 49) receiving two cycles, 18.8% (n = 115) receiving three cycles, and 60.4% (n = 378) receiving four cycles. Only 13.4% (n = 85) received five or six cycles. In comparison to patients who received four or fewer cycles, patients who received five or six cycles did not have a statistically significant improvement in OS (HR 0.73 CI 0.43–1.24, p = 0.246).
We excluded patients who had received carboplatin or any systemic therapy within six months prior to the initiation of ddMVAC. Importantly, none of the patients in this cohort were administered Carboplatin within 60 days of the first ddMVAC cycle, indicating that a transition from cisplatin to carboplatin by the second or third cycle is unlikely.
Survival analyses
Age at diagnosis (p = 0.011), geographic region (p = 0.010), and ECOG functional status (p = 0.003) were statistically significant in the univariate analyses of OS and included in the multivariate model (Table 2). Additionally, split dosing of cisplatin was also included in multivariate analyses given the clinical associations of interest (Figure 3). In the multivariate OS analysis, there was no statistically significant difference between split-dose and conventional ddMVAC (HR 1.3, CI 0.78–2.18, p = 0.316) when adjusted for age at diagnosis, geographic region, and ECOG functional status (Figure 4).
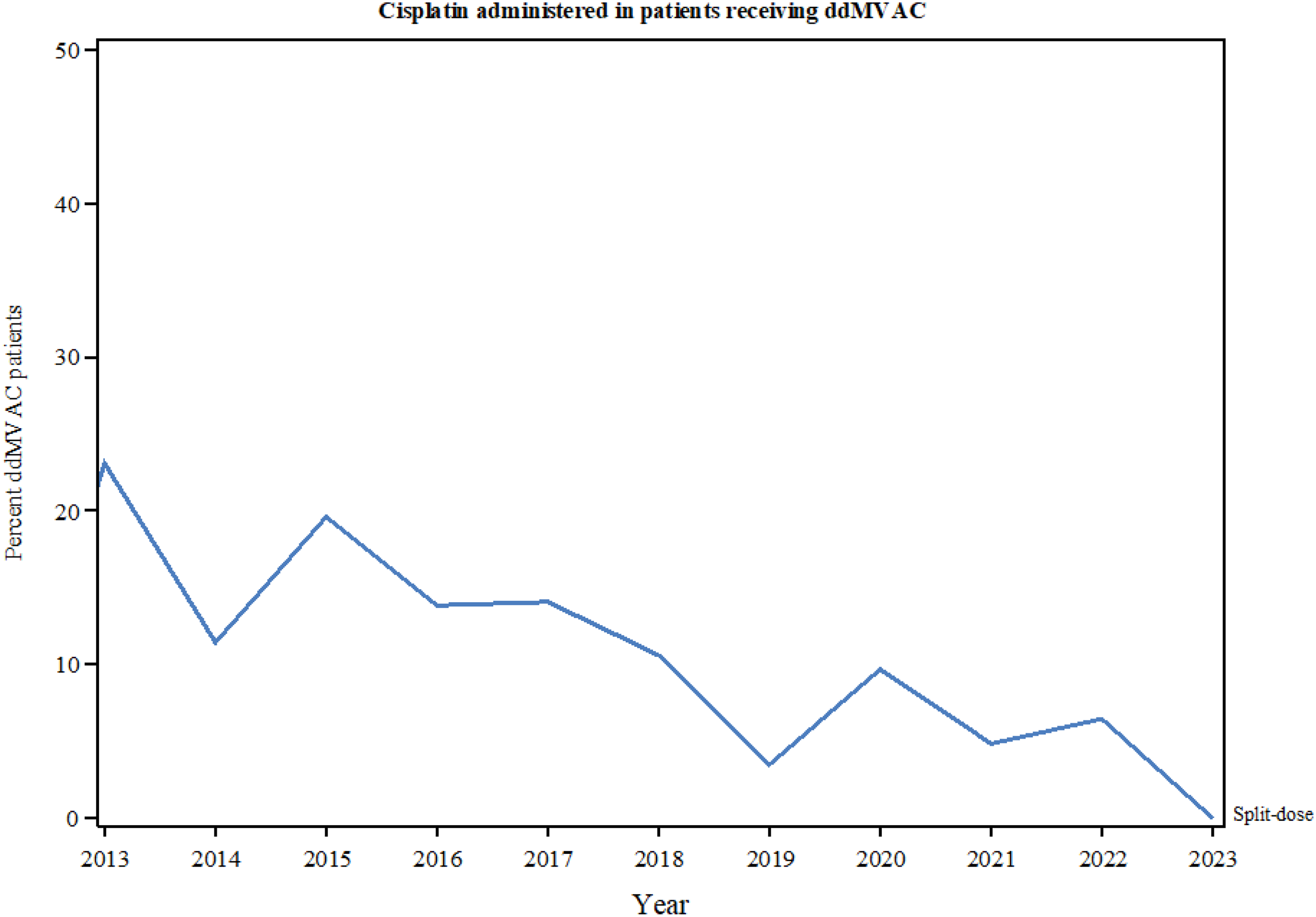
Patterns of use of split-dose cisplatin over time.
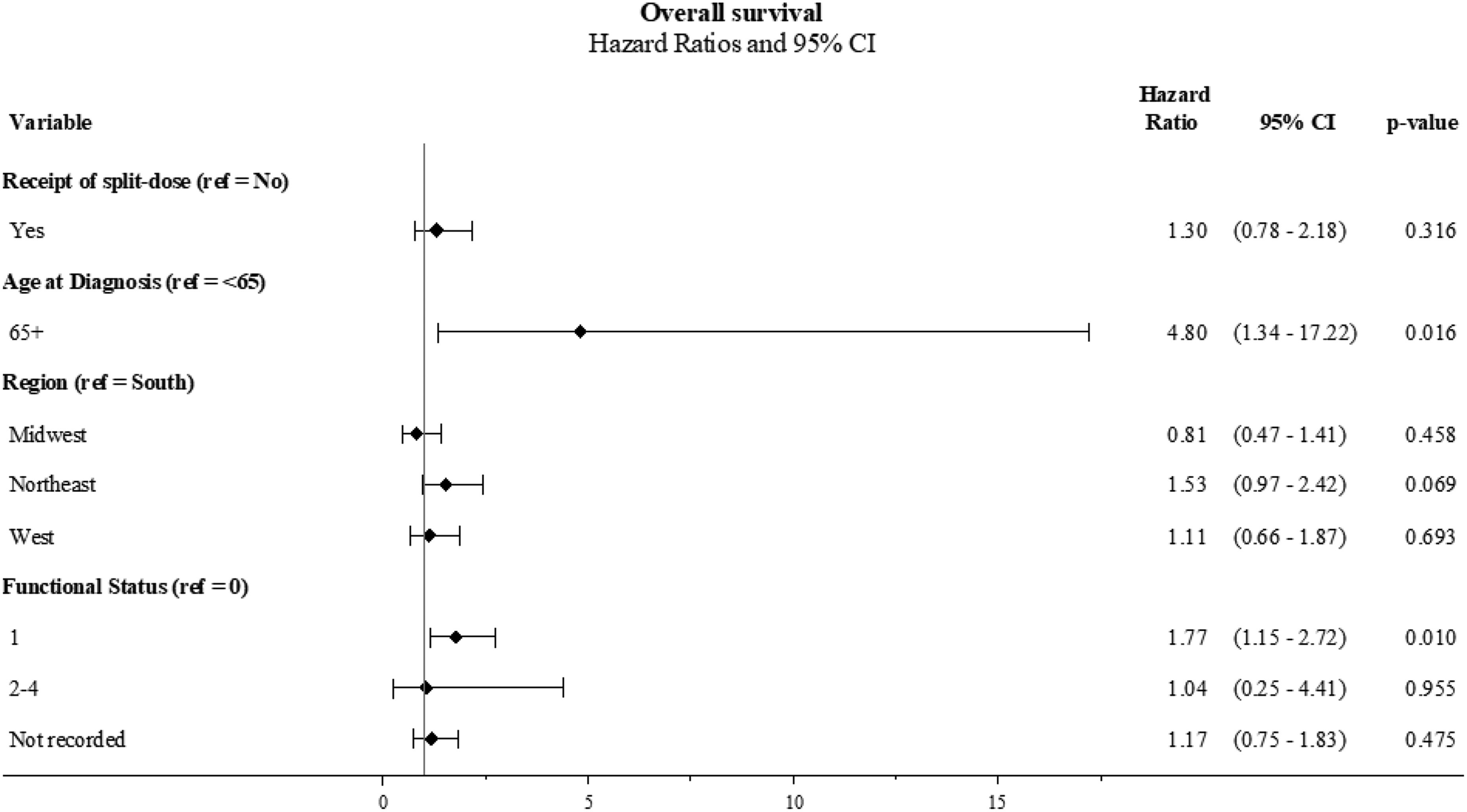
Overall survival, multivariate analysis.
Univariate overall survival.
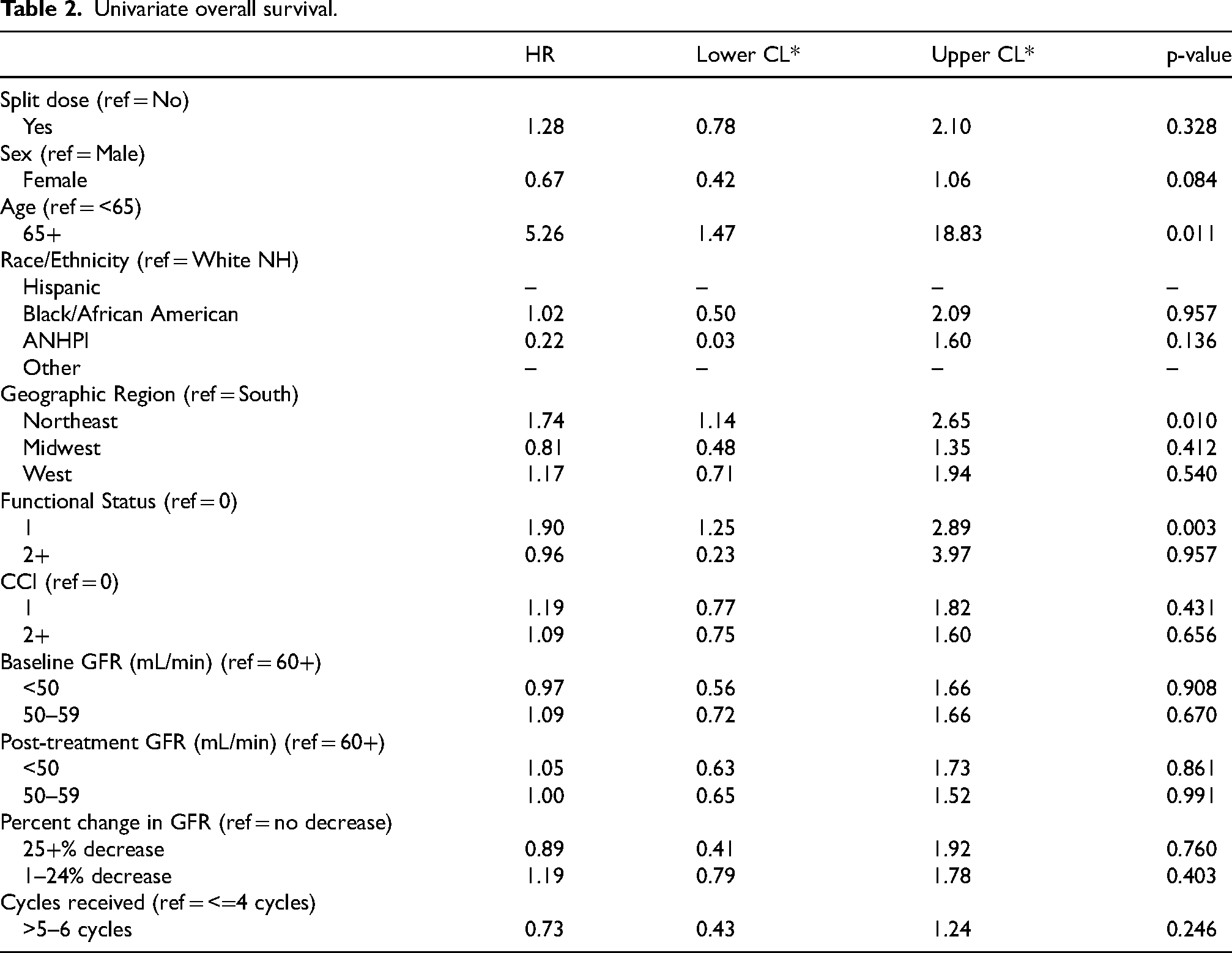
While geographic region was found to be statistically significant in the univariate analysis of OS, this effect was not sustained after adjusting for confounders including the use of split-dose cisplatin, age at diagnosis, and EGOC functional status. Specifically, after adjustment, no significant difference in OS were observed between patients treated in regions outside of the South. Hazard ratios for the Midwest (HR 0.81, p = 0.45), Northeast HR (1.53, p = 0.069), and West (HR 1.11, p = 0.693) regions all indicated a lack of significant regional effects on OS.
Our data suggest that older age at diagnosis (>65+ years) confers a worse OS compared to patients <65 years at diagnosis (HR 4.8, CI 1.34–17.22, p = 0.016). These data also suggest that compared to patients with an ECOG of 0, patients with an ECOG of 1 had statistically significantly worse OS (HR 1.77 CI 1.15–2.72 p = 0.010).
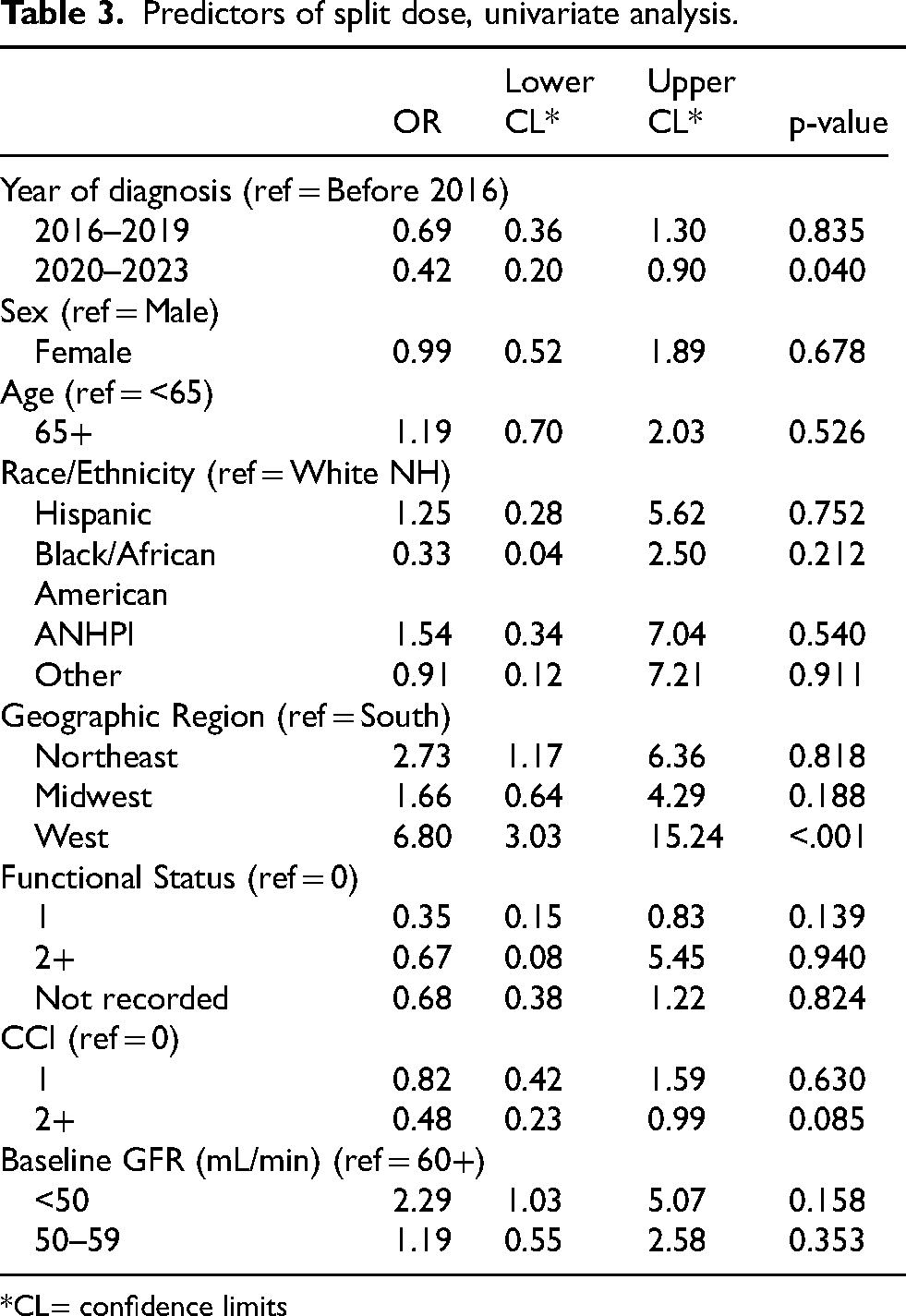
Predictors of split dosing
In univariate analyses, year of diagnosis and geographic region were identified as significant predictors of cisplatin split-dosing (Table 3). However, in the multivariate model, the year of diagnosis was not a statistically significant predictor of receiving split-dose cisplatin (Figure 5). Geographic region, on the other hand remained a significant predictor, with patients treated in the West region being substantially more likely to receive split-dose cisplatin compared to those treated in the South (HR 6.45, p=<0.001) (Figure 5). No significant differences in the likelihood of receiving split-dose cisplatin were observed for patients treated in the Midwest or Northeast compared to South (HR 1.62, P = 0.084 and HR 2.57, p = 0.798, respectively) (Figure 5). Notably, baseline GFR was not a significant predictor of split-dose cisplatin administration and was therefore excluded from the multivariate analysis.
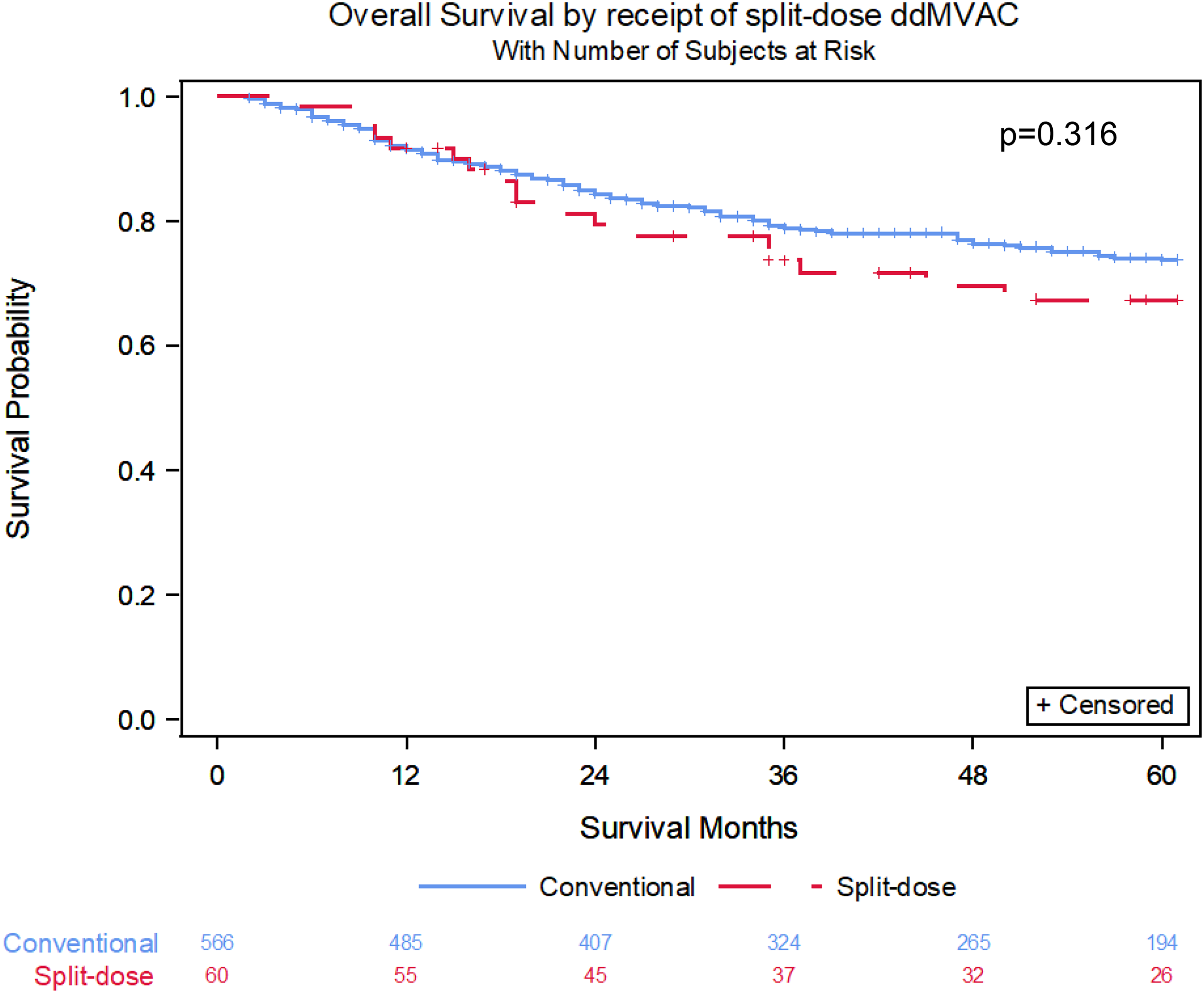
Overall survival of split-dose versus conventional-dose cisplatin.
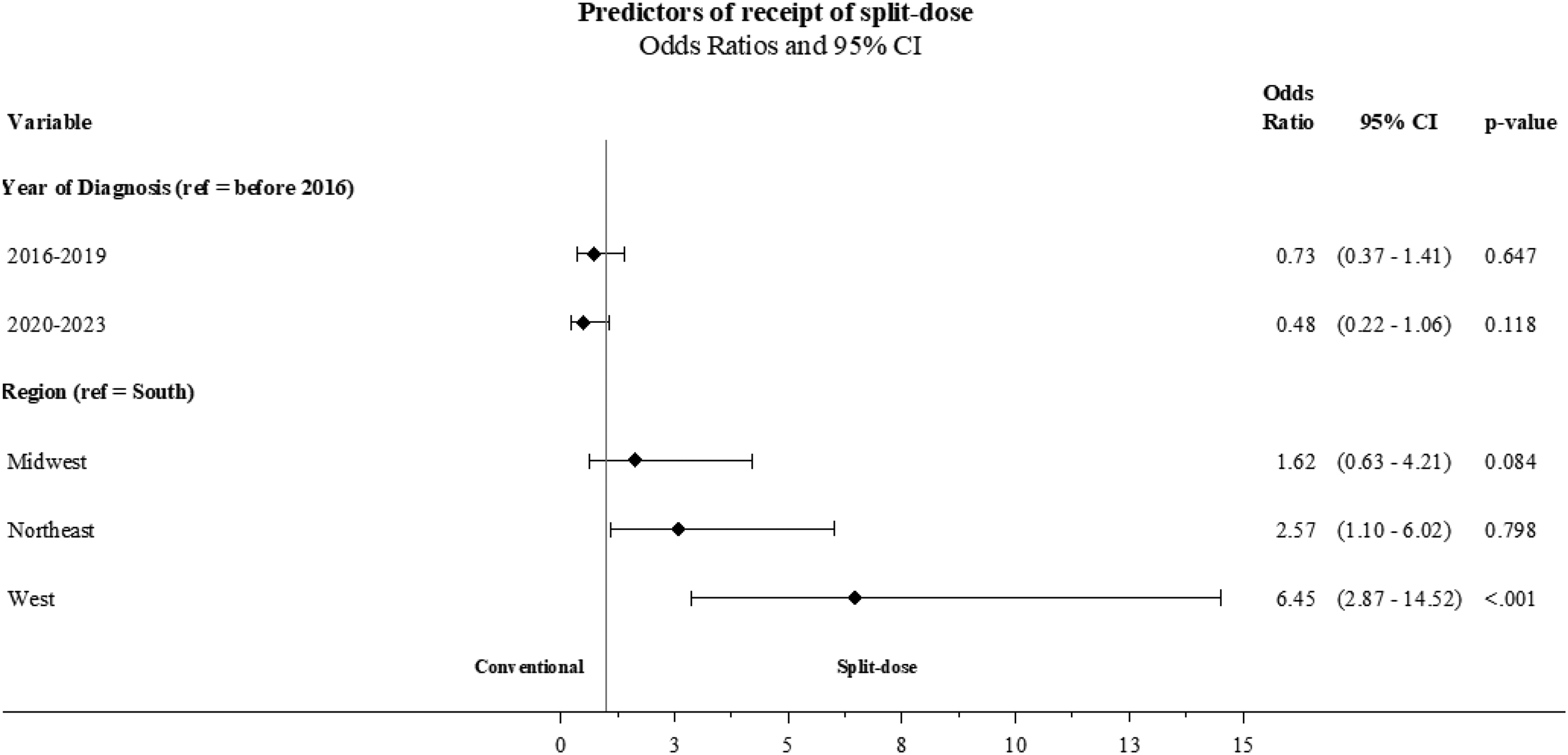
Predictors of split-dose, multivariate analysis.
Predictors of split dose, univariate analysis.
CL= confidence limits
Discussion
NCCN treatment guidelines indicate split-dose cisplatin can be considered for patients with MIBC and borderline renal dysfunction undergoing NAC, classifying it as category 2B recommendation. 4 However, the efficacy and clinical practice patterns for modified cisplatin dosing remain largely undefined. This clinical data from the CancerLinQ Discovery® database revealed a strong preference for conventional cisplatin dosing in the neoadjuvant setting, with 90.4% of the overall ddMVAC-treated MIBC cohort (n = 566) receiving the full cisplatin dose in a single treatment day.
To date, limited studies have compared the outcomes of conventional versus split-dose NAC, with most existing research focusing specifically on split-dose cisplatin in combination with gemcitabine. 10 In a multi-institutional matched cohort study, Osterman et al. observed lower pathologic response rates with split-dose cisplatin compared to conventional dosing. However, this difference was not statistically significant. 11
In our multivariate analysis of OS, we did not find a statistically significant difference in survival outcomes between patients receiving split-dose versus conventional cisplatin dosing (HR 1.30, CI 0.78–2.18, p = 0.316), after adjusting for age at diagnosis geographic region, and ECOG functional status. However, within this model, across all patients, those 65 years and older experienced significantly poorer OS compared to patients younger than 65 years (HR 4.80 CI 1.34–17.22, p = 0.016). Of note, this older population has an increased risk of all-cause mortality.
The current choice of NAC regimen—cisplatin plus gemcitabine versus ddMVAC—often depends on various factors, including a patient's functional status, baseline comorbidities, renal function, goals of care, and provider preference. Notably, only 1.8% of this ddMVAC cohort had an ECOG performance score of 2 or higher, which may reflect real-world practice patterns given the high toxicity associated with neoadjuvant ddMVAC. While functional status can influence the decision to use or avoid ddMVAC, our data suggests that for patients treated with ddMVAC, ECOG performance status did not significantly predict the use of split-dose cisplatin in multivariate analysis. Furthermore, in the multivariate analysis of OS, an ECOG score of 0 was associated with improved survival compared to an ECOG score of 1 (OR 1.77, p = 0.010). Although we did not observe a significant difference in OS between ECOG performance status 2+ and 0 (HR 1.04, 0.25–4.41, p = 0.955), our limited sample size and the number of missing data points hinder our ability to draw definitive conclusions for this group.
Although our analysis suggests no regional effects on overall survival, it does indicate notable regional variation in the use of split-dose cisplatin. Specifically, patients treated in the West were significantly more likely to receive split-dose cisplatin compared to those treated in the Southern region (HR 6.45, p=<0.001). This observed regional difference may reflect true variations in clinical practice patterns across different geographic settings. However, it is also important to consider the potential biases inherent in this data. As a convenience sample of practices and cancer centers that have opted to participate in the CancerLinQ database, these findings may not fully represent broader, national trends and may therefore be influenced by the specific characteristics of the practices that chose to join the registry, potentially limiting generalizability of these observations.
The VESPER trial indicated that patients who achieved a pathologic complete response (pCR) received a higher dose of cisplatin. This study also found that the proportion of patients achieving pCR was comparable among those receiving four, five, and six cycles of treatment; however, undergoing surgery before completing four cycles was associated with worse outcomes. In the GC arm, similar findings were observed, as pCR rates were lower for patients who underwent cystectomy prior to completing four cycles. 12 Notably, we did not observe a statistically significant improvement in OS among patients receiving five and six cycles of ddMVAC compared to those who received four or fewer cycles (HR 0.73, CI 0.43–1.24, p = 0.246). It is also worth noting that only 13.5% (n = 85) of this cohort received the higher dose of cisplatin in the real-world practice setting. However, the limited sample size of those patients in this higher dosing group means that we are limited in our ability to draw large conclusions from this data.
These data demonstrate a notable decline in the use of split-dose cisplatin over time. Specifically, 13.9% of patients received split-dose before 2016, compared to 10.0% between 2016 and 2019, and 6.3% between 2020 and 2023. The year of diagnosis was a statistically significant predictor of split-dose cisplatin administration, with patients diagnosed and treated between 2020 and 2023 being significantly less likely to receive split-dose compared to those diagnosed before 2016 (OR 0.42, CI 0.20–0.90, P = 0.040). This observed decreasing trend may reflect the evolving criteria for cisplatin eligibility over time. Early guidelines, such as the Galsky criteria, classified patients as cisplatin-ineligible if their GFR was <60 mL or creatinine clearance (CrCl) was <60 mL/min. These criteria also considered factors such as ECOG performance status, hearing impairment, heart failure, and peripheral neuropathy. Additionally, shifts in the definitions of adequate renal function and variations in methods for calculating GFR—such as CKD-EPI, Cockcroft-Gault, and combined creatinine-cystatin C equation—may have also contributed to this trend.13–15 Recent findings from the NIAGARA study in which patients with a GFR of 40–60 mL/min were deemed eligible for cisplatin highlight the ongoing discrepancies in the definition of cisplatin eligibility based on renal function, both in the clinical trial contexts and likely in real-world practice.
As only approximately 10% of this cohort had documented CrCl data, we relied on GFR to define renal function. In this cohort of 626 patients, approximately half (n = 318) had a pre-treatment GFR of ≥60 mL/min, 18.1% (n = 118) a baseline GFR between 50–59 mL/min and 11.3% (n = 71) a baseline of <50 mL/min. Interestingly, only 10.9% (n = 20) of those patients with GFR <60 mL/min received cisplatin over consecutive days. Additionally, in these analyses, compared to those with a GFR >60 mL/min, we did not find baseline GFR to be a significant univariate predictor of split-dose cisplatin (GFR mL/min <50 HR 2.29, p = 0.158; GFR 50–50 mL/min HR 1.19, p = 0.353). While we must be careful not to overinterpret these findings given the limitations of a retrospective analysis, these findings may also suggest that the administration of ddMVAC in real-world oncology practice may often deviate from NCCN guidelines.
In the evolving systemic treatment landscape for urothelial carcinoma, immuno-oncology, targeted therapy and antibody-drug conjugates are being actively evaluated. Despite these developments, the application of split-dose cisplatin may still hold relevance in real-world clinical practice. The recently published Phase 3 NIAGARA study which investigated the combination of durvalamab and GC in the neoadjuvant setting, 16 suggests that split-dose cisplatin could expand treatment options for a broader population. Furthermore, administering split-dose cisplatin in patients who may not be eligible for immunotherapy, as well as its potential applications in later stages of disease and the palliative intent setting, could provide valuable options for treating providers.
Given the retrospective nature of this study and the reliance on real-world data, we acknowledge potential gaps in data collection and reporting. We excluded patients with M1 disease and those who received systemic therapies other than nivolumab within six months of ddMVAC, to best define the cohort of patients with MIBC and to exclude those with locally advanced disease or receiving palliative treatment. Nonetheless, the limitation inherent in using a retrospective dataset, coupled with the small sample size in our analyses, restricts our ability to draw robust conclusions about the impact of split-dose cisplatin on clinical outcomes.
In summary, many patients with MIBC are treated with standard dose ddMVAC with decreasing frequency of split-dose ddMVAC use over the observation period. Neither baseline GFR nor performance status were predictors of split dosing in this cohort. There was also no difference noted in overall survival between patients with MIBC who received standard versus split-dosing.
Footnotes
Acknowledgments
The authors have no acknowledgments.
Author contributions
EBC- Interpretation of data, writing the article. CJE- Conception, interpretation of data, writing the article. TWF: Conception, interpretation of data, writing the article. EMK- Performance of data, interpretation of data, writing the article. ERK-Conception, interpretation of data. All authors had access to this data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NCI grant P30CA046934. Urologic Research Fund, CU Anschutz. Kathleen Allison in memory of Tom Allison. Additional funding was provided by the Schramm Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study were sourced from CancerLinQ Discovery® (![]() ): Bladder Cancer Dataset 2013–2023, August 2023 dataset release, American Society of Clinical Oncology's (ASCO's) CancerLinQ LLC and can be made available upon request and are subject to a license agreement with CancerLinQ.
): Bladder Cancer Dataset 2013–2023, August 2023 dataset release, American Society of Clinical Oncology's (ASCO's) CancerLinQ LLC and can be made available upon request and are subject to a license agreement with CancerLinQ.
