Abstract
Background
The presence of lymph node metastases in patients with urothelial carcinoma (UC) plays a pivotal role in disease management and prognosis. Patients with locally advanced irresectable or clinically node-positive UC are often considered ineligible for surgery due to the extent of affected lymph nodes. Long term remission or even cure may be achieved in a subset of patients who experience response to systemic induction therapy and consolidative locoregional treatment.
Objective
To assess the pathological response to preoperative systemic induction therapy followed by radical surgery in patients with locally advanced irresectable or clinically node-positive UC.
Methods
Searches were performed until September 2023 in 5 databases (EMBASE, MEDLINE, Web-of-Science, Cochrane and Pubmed Publisher). Studies including patients with cT4bNxM0/cTxN1-3M0 UC, treated with induction chemotherapy or non-chemo induction therapy followed by radical surgery, were selected. The primary outcome was the pathological complete response (pCR) rate, i.e., the proportion of patients without residual disease in the surgical resection specimen (ypT0N0). Secondary outcomes included overall and cancer-specific survival (OS, CSS).
Results
Fourteen studies were included, representing 5715 patients. Two studies reported on non-chemo induction therapy. The reported pCR rate in patients receiving induction chemotherapy varied from 9% to 27% compared to 25% after induction immunotherapy. The 5-year OS and CSS after induction chemotherapy and radical surgery ranged from 25 to 34% and 30 to 49%.
Conclusion
Systemic induction therapy in patients with locally advanced irresectable or clinically node-positive UC resulted in modest pCR rates. Due to considerable heterogeneity between studies, no direct comparison on the efficacy of induction therapy regimens was possible.
Background
Urothelial cancer (UC) is the 10th most common cancer worldwide. 1 The urinary bladder is the predominant site of origin, but the renal pelvis, ureters and urethra can also be affected. UC of the urinary bladder is categorised into non-muscle invasive and muscle-invasive disease, with key differences in disease management and prognosis. 2
Adequate clinical staging of regional lymph nodes is important to guide disease management and select appropriate treatment strategies. However, the sensitivity of cross-sectional imaging to detect lymph node metastases is limited in non-enlarged lymph nodes. This can lead to discrepancies in clinical-pathological staging, which occurs in up to 20% of UC patients.3,4 Consequently, patients initially staged as cN0 may ultimately be diagnosed as pN + . Accurate clinical staging is important as lymph node metastases are associated with poor patient outcome, which emphasizes the pivotal role of lymphatic involvement in UC patients.5–7
The current standard of care in patients with non-metastatic muscle-invasive bladder cancer (MIBC) is cisplatin-based neoadjuvant chemotherapy followed by radical cystectomy (RC) and pelvic lymph node dissection (PLND). 2 In patients with locally advanced irresectable or clinically node-positive disease (cT4bNxM0 or cTxN1-N3M0), treatment options are limited. Patients are often ineligible to undergo surgery due to the extent of affected lymph nodes. However, curative treatment is still possible, provided patients experience adequate tumour response to systemic induction therapy and thereafter undergo radical resection of the primary tumour and locoregional lymph nodes.8–10 The absence of residual tumour in the surgical resection specimen, i.e., pathological complete response (pCR), corresponds with a good prognosis with a 5-year overall survival rate of 70–80%. However, only 15% of locally advanced irresectable or clinically node-positive patients achieve a pCR after systemic induction therapy. 6 Patients with residual muscle-invasive disease (≥ ypT2) or nodal metastasis (>ypN0) have a 5-year overall survival rate of only 15–20%. 11 Therefore, there is an urgent need to improve therapeutic strategies in this patient category.
The literature on the outcome of UC patients receiving preoperative systemic induction therapy is scarce. Clinical trials on chemotherapy or new treatment regimens, such as immunotherapy, recruit patients in either the neoadjuvant or the metastatic setting (stage cT2-T4aN0-1M0 or cM1, respectively), which represent different patient populations. This review aimed to provide an overview of the current literature on the efficacy of systemic induction therapy in patients with locally advanced irresectable or clinically node-positive UC, stratified by chemotherapy versus non-chemotherapy regimens.
Methods
Evidence synthesis
The EMBASE, MEDLINE (OvidSP), Web-of-Science, Cochrane and Pubmed Publisher databases were searched till 30 September 2023. The full search syntax (see Appendix A) was provided by a biomedical information specialist. Articles were independently screened by two authors (VR, JM), first on title and abstract and subsequently on full-text record. Articles were considered relevant if they included adult patients with UC of the bladder, upper urinary tract or urethra, clinical stage cT4bNxM0 or cTxN1-3M0, who were treated with either platinum-based induction chemotherapy, non-chemotherapy based induction therapy or combined regimens, followed by radical surgery. Articles published before 2003 were excluded. Articles including cM1a patients were excluded, except if outcomes were presented separately for cN + versus cM + disease. Studies including only cN1 patients were also excluded, since stage cN1 UC represents neoadjuvant and not induction setting. Additional manual cross-referencing was performed on the included studies. Any disagreements between reviewers were discussed with a third reviewer (JB) until mutual consensus was reached. Studies with non-English language were excluded, as well as case reports, animal studies, conference abstracts and reviews.
Data extraction
Data was manually extracted from the included articles. The primary outcome was the pCR rate, defined as the proportion of patients without residual disease in the surgical resection specimen (ypT0N0). Secondary outcomes included overall survival (OS) and cancer-specific survival (CSS). OS was defined as the first date of diagnosis 12 or the date of surgery13,14 to the date of last follow-up or death. Several studies did not provide a definition of OS.6,15–18 CSS was defined as the start of induction therapy11,19 or the date of surgery14,16,20 to the date of cancer-related death. Relevant baseline characteristics were extracted, including the eligible patient population, whether histological confirmation of cN + disease was mandatory, sub-stratification based on the proportion of cN1, cN2 and cN3 disease or treatment groups (if applicable), treatment regimen and the number of cycles administered. From studies that analysed multiple treatment regimens, such as chemotherapy followed by radical surgery versus surgery alone versus chemotherapy alone, only the results of the chemotherapy followed by radical surgery subgroup were included.
Results
Literature search results
The PRISMA flow diagram depicts the results of the search strategy and the selection of relevant articles (Figure 1). Fourteen studies were included, representing 5715 patients in total. Initially, we aimed to systematically assess and compare chemotherapy with non-chemotherapy treatment alternatives. However, literature on non-chemotherapy regimens was scarce with only 2 out of 14 included articles. Additionally, reported outcomes on OS and CSS were not uniform and the number of studies per survival outcome was limited. Consequently, we decided to conduct a narrative review.
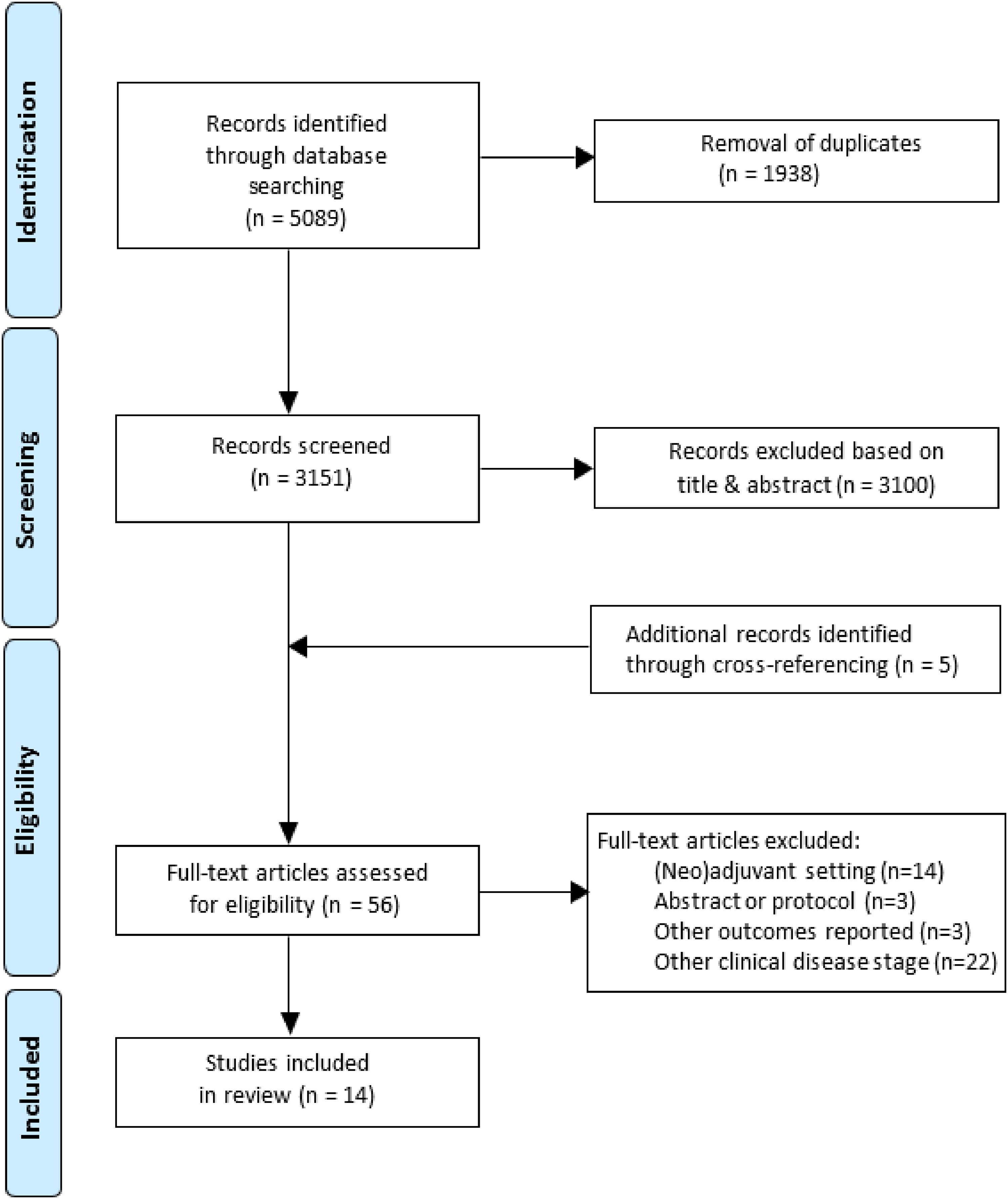
Prisma flowchart illustrating the outcomes of the search strategy and evidence synthesis for patients with cT4bNxM0 or cTxN1-3M0 urothelial carcinoma treated with platinum-based induction chemotherapy, non-chemotherapy based induction therapy or combined regimens followed by radical surgery.
Baseline characteristics
Included studies were either retrospective or prospective cohort studies on UC or UC of the bladder specifically. None of the studies included patients with UC of the upper tract or urethra exclusively. Two studies on induction immunotherapy included one patient with UC of the bladder with upper tract involvement.21,22 All studies utilised radiological imaging for clinical staging but only one study required histological confirmation of lymph node metastases. 12 The included number of patients per study varied from 24 to 1739 patients.15,21 The proportion of cN1 versus cN2 versus cN3 patients ranged from 29% to 70% (cN1), 9% to 45% (cN2) and 5% to 26% (cN3). Some studies combined patients with stage cN2 and cN3 disease into one subgroup,11,12,17 while other series did not provide information on the clinical staging of the patients included. 6 Six studies compared several treatment regimens, such as chemotherapy followed by RC, RC followed by adjuvant chemotherapy, RC alone and/or chemotherapy alone.12,13,15–17,23 In two studies, patients undergoing pre-operative and adjuvant treatment were grouped together.13,16 Specifically, cN + patients received induction chemotherapy, while patients with persistent lymph node positive disease after surgery (ypN+) were treated with adjuvant chemotherapy. 13 Reported results represent the group treated with IC + RC only. 13 It is noteworthy that, in the patient population considered by Stanik et al. (2019), only 8% received induction chemotherapy whereas 92% received adjuvant chemotherapy. Data on the induction chemotherapy subgroup alone was not provided. 16 Four studies did not report detailed information on chemotherapy, such as the regimen and the number of cycles administered.13,15–17 Median follow-up time ranged from 13 months to 57 months. All baseline characteristics are summarised in Table 1.
Baseline characteristics for patients with cT4bNxM0 or cTxN1-3M0 urothelial carcinoma treated with platinum-based induction chemotherapy or non-chemotherapy based induction therapy followed by radical surgery.
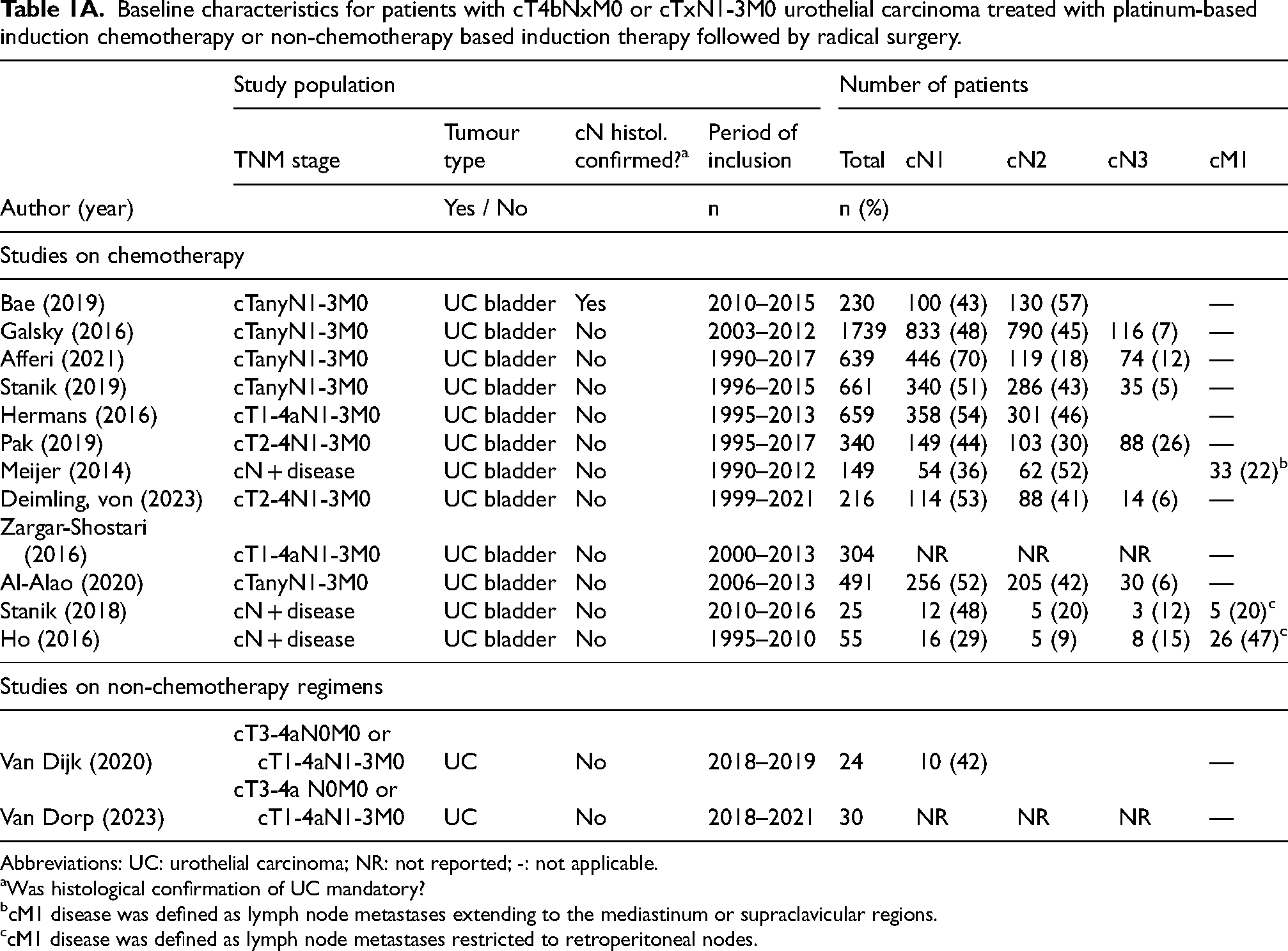
Abbreviations: UC: urothelial carcinoma; NR: not reported; -: not applicable.
Was histological confirmation of UC mandatory?
cM1 disease was defined as lymph node metastases extending to the mediastinum or supraclavicular regions.
cM1 disease was defined as lymph node metastases restricted to retroperitoneal nodes.
Baseline characteristics of treatment groups and regimens for patients with cT4bNxM0 or cTxN1-3M0 urothelial carcinoma treated with platinum-based induction chemotherapy or non-chemotherapy based induction therapy followed by radical surgery.
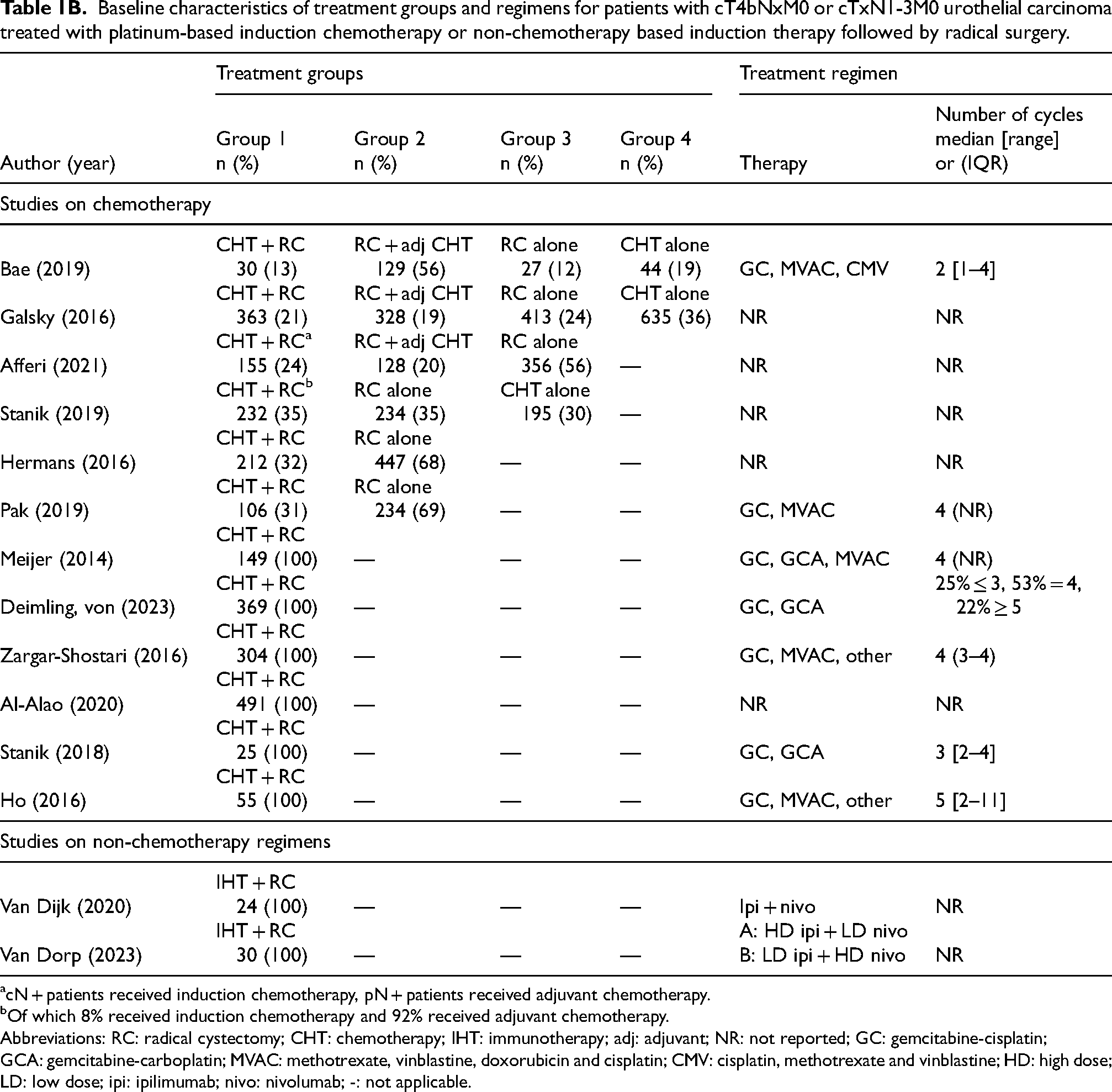
cN + patients received induction chemotherapy, pN + patients received adjuvant chemotherapy.
Of which 8% received induction chemotherapy and 92% received adjuvant chemotherapy.
Abbreviations: RC: radical cystectomy; CHT: chemotherapy; IHT: immunotherapy; adj: adjuvant; NR: not reported; GC: gemcitabine-cisplatin; GCA: gemcitabine-carboplatin; MVAC: methotrexate, vinblastine, doxorubicin and cisplatin; CMV: cisplatin, methotrexate and vinblastine; HD: high dose; LD: low dose; ipi: ipilimumab; nivo: nivolumab; -: not applicable.
Reported pathological complete response rate for patients with cT4bNxM0 or cTxN1-3M0 urothelial carcinoma treated with platinum-based induction chemotherapy or non-chemotherapy based induction therapy followed by radical surgery.
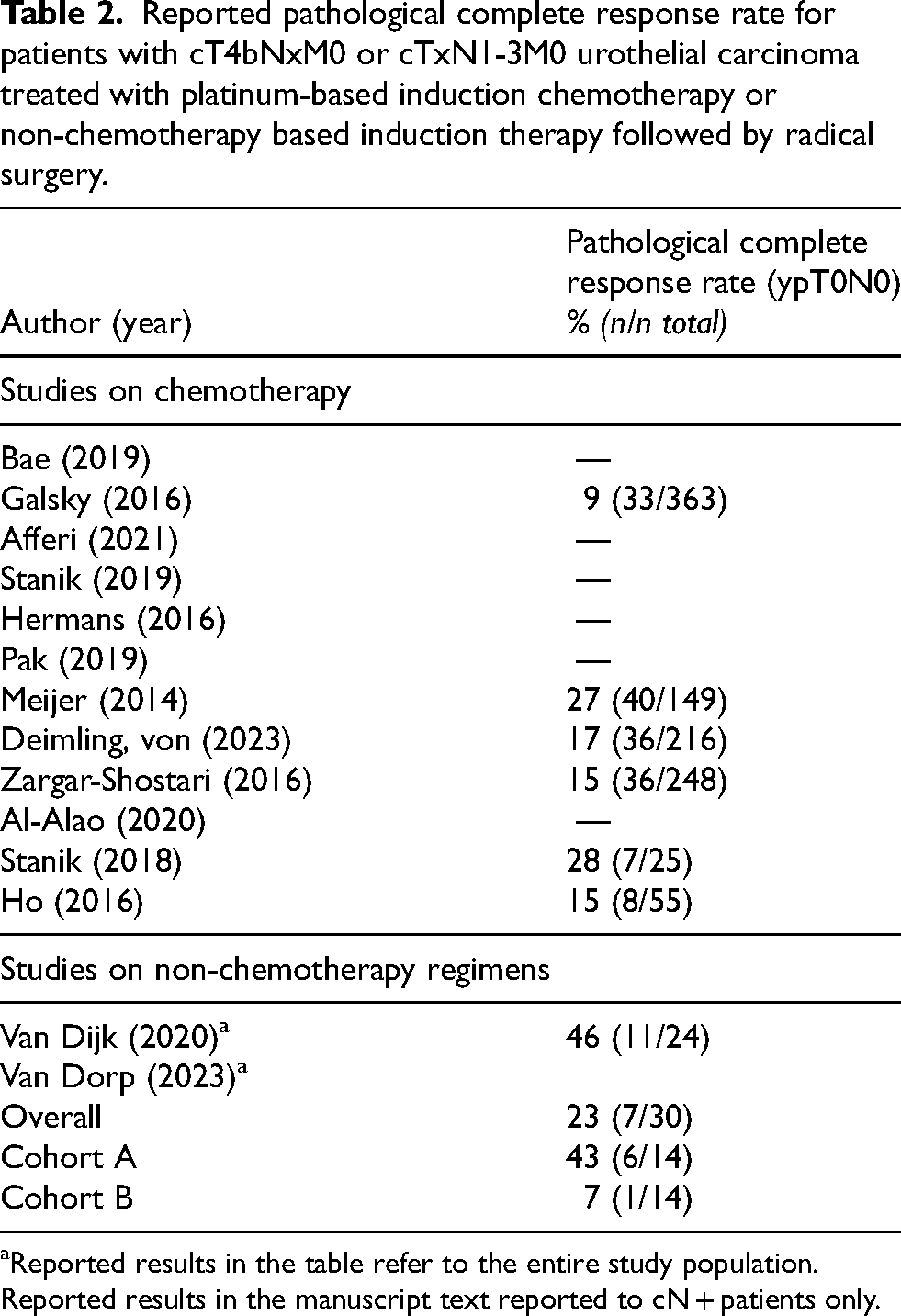
Reported results in the table refer to the entire study population. Reported results in the manuscript text reported to cN + patients only.
Induction treatment with chemotherapy regimens
Pathological outcomes
Six studies reported on pCR, defined as ypT0N0, after platinum-based chemotherapy.6,11,14,15,19,20 The observed pCR rate ranged from 9% up to 27%.11,15 Meijer et al. 11 reported the highest pCR rate of 27%, despite including 52% patients with stage cN2 and cN3. This study also included patients with cM1 disease, defined as lymph node metastases extending to the mediastinum or supraclavicular regions. However, since these patients did not undergo radical surgery, their inclusion did not affect the reported pCR rate. Reported pathological complete response rates are summarized in Table 2.
Two other studies included cM1 patients in their final analysis, defining cM + disease as metastases restricted to retroperitoneal nodes.19,20 Stanik et al. 19 reported that all five cM + patients exhibited partial or complete response after induction chemotherapy. Only one (13%) out of eight cN3-cM + patients achieved a pCR. Ho et al. 20 did not specify radiological response to chemotherapy but reported no differences between cM + (n = 26) and cN + patients (n = 29). The number of cM + patients achieving pCR was not reported. In both studies, cM + patients proceeded to radical surgery with dissection of the lymph nodes up to the renal hilum (retroperitoneal lymph node dissection). Histopathology showed that 12% to 15% had metastatic disease after surgery (ypM1), although it was unclear whether these patients were initially staged as cM + disease.19,20
The results of surgical resection specimens of the studies included are summarized in Supplementary Table 1. Eight studies reported the rate of ypT0 (regardless of ypN stage), ranging from 13% to 35%.13,23 The rates of ypN0 (regardless of ypT stage) varied from 35% to 57%.13,18 One study reported pathological complete downstaging, defined as yp ≤ T1, instead of pCR. This study did not provide separate information on ypT0. 17
Overall survival and cancer-specific survival
OS was not uniformly defined in the included studies. Five studies reported the median OS, which ranged from 23 to 49 months.6,12,13,16,18 Two studies reported 3-year OS rates, ranging from 34% to 58%,14,18 while four studies reported 5-year OS rates, varying from 25% to 34%.13,15,16,18
Several studies conducted a subgroup analysis of cN1 versus cN2-3 patients. Three studies reported cN1 patients to have a more favourable median OS than cN2-3 patients, with differences ranging from 7 to 26 months.6,12,18 Hermans et al. 17 reported similar results, with cN1 patients demonstrating better survival outcomes. The reported 3- and 5-year OS rates for cN1 patients were 66% and 58%, respectively, compared to 43% and 36% for cN2-3 patients. All OS outcomes are summarized in Table 3.
Reported overall survival outcomes and corresponding subgroup analyses for patients with cT4bNxM0 or cTxN1-3M0 urothelial carcinoma treated with platinum-based induction chemotherapy or non-chemotherapy based induction therapy followed by radical surgery.
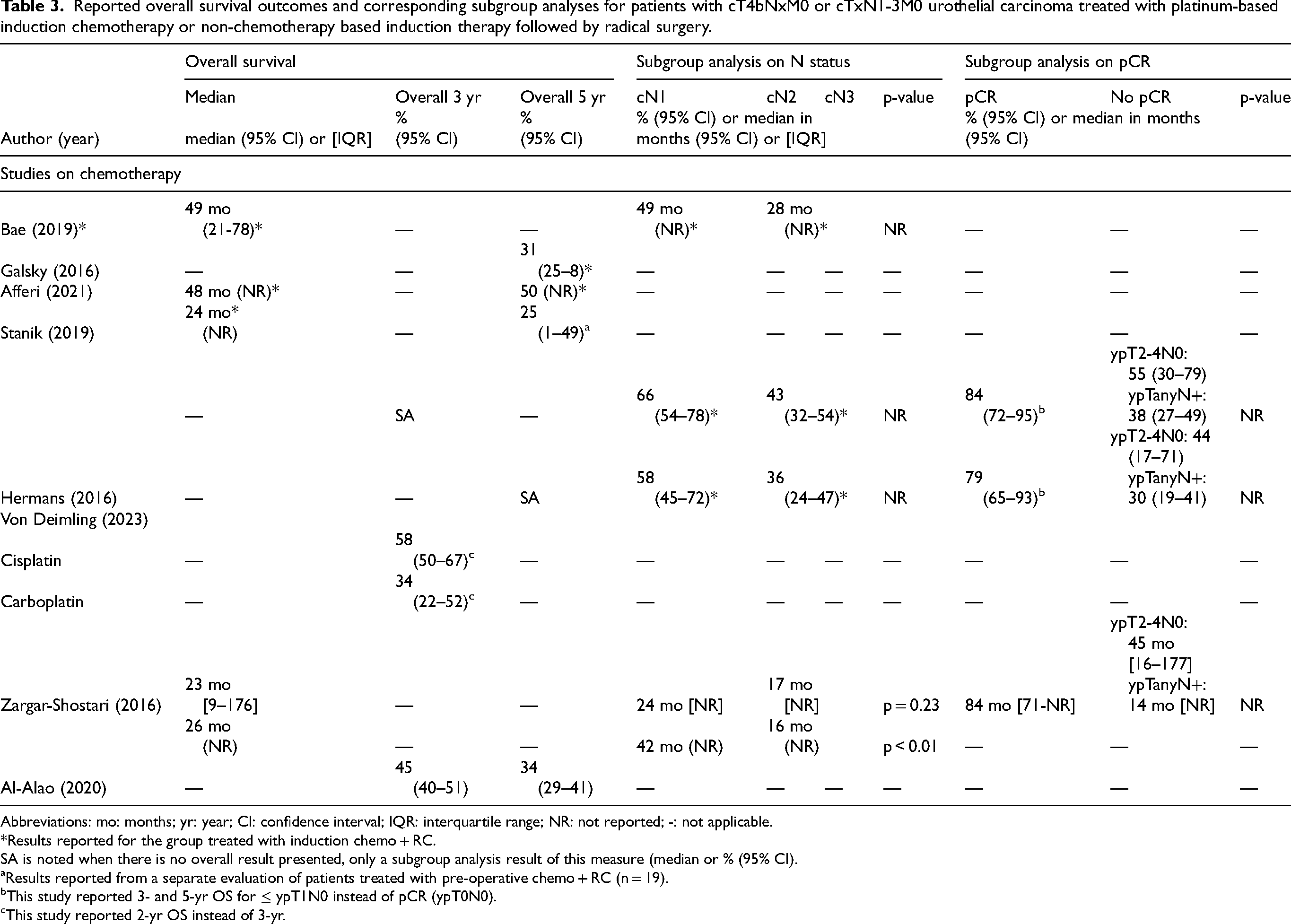
Abbreviations: mo: months; yr: year; CI: confidence interval; IQR: interquartile range; NR: not reported; -: not applicable.
*Results reported for the group treated with induction chemo + RC.
SA is noted when there is no overall result presented, only a subgroup analysis result of this measure (median or % (95% CI).
Results reported from a separate evaluation of patients treated with pre-operative chemo + RC (n = 19).
This study reported 3- and 5-yr OS for ≤ ypT1N0 instead of pCR (ypT0N0).
This study reported 2-yr OS instead of 3-yr.
Six studies reported on CSS.11,14,16,19,20,23 Two studies reported a median CSS of 20 months and 26 months.11,20 Three-year CSS rates ranged from 39% to 60%,14,19 while 5-year CSS rates varied from 30% to 49%.11,16,20,23 It should be noted that in one of the cohorts reporting a 5-year CSS, only 8% of patients received induction chemotherapy. 16 In one study, 3-year CSS rates did not differ significantly between cN1 and cN2-3 patients (cN1: 53% versus cN2-3: 56%). 19
Three studies reported 5-year CSS rates for cN1, cN2 and cN3 subgroups.11,19,23 Pak et al. 23 reported more favourable 5-year CSS for cN1-N2 compared to cN3 patients (68% versus 22%, respectively). Another study compared cN1 with cN2-3 patients, revealing longer median CSS (33 months versus 15 months) and a higher 5-year CSS rate (42% versus 23%) for the cN1 group. 11 Finally, one study did not observe a significant difference in 3-year CSS between cN1 and cN2-3 patients (53% vs 56%). 19 All CSS outcomes are summarized in Table 4.
Reported cancer-specific survival outcomes and corresponding subgroup analyses for patients with cT4bNxM0 or cTxN1-3M0 urothelial carcinoma treated with platinum-based induction chemotherapy or non-chemotherapy based induction therapy followed by radical surgery.

Abbreviations: mo: months; yr: year; CI: confidence interval; IQR: interquartile range; NR: not reported; NA: not applicable; pPR: pathological partial response (not further defined); pSD: pathologic stable disease (not further defined); pPD: pathological progressive disease (not further defined); -: not applicable.
Of which 8% received induction chemotherapy and 92% received adjuvant chemotherapy.
This study reported 2-yr CSS instead of 3 year
Induction treatment with non-chemotherapy regimens
Pathological outcomes
Results on non-chemotherapy regimens in patients with locally advanced UC were limited. Two studies were identified, both part of the NABUCCO trial, where patients with cT1-4aN1-3M0 or cT3-4aN0M0 UC received induction immunotherapy with ipilimumab and nivolumab.21,22 In the first cohort, 4 out of 10 patients with cN + disease achieved a pCR. 21 In cohort 2, treatment doses were adjusted: patients received either high-dose ipilimumab with low-dose nivolumab (cohort 2A) or low-dose ipilimumab with high-dose nivolumab (cohort 2B). In cohort 2A, two out of six patients with cN + disease achieved a pCR, compared to none out of eight in cohort 2B. Interestingly, four out of eight node-positive patients in cohort 2B had lymph node-negative disease (ypN0) at RC and three patients showed downstaging to non-muscle-invasive disease (ypTis/Ta/T1), which indicates a response to systemic induction immunotherapy. 22
Discussion
The objective of this review was to provide a literature overview on the clinical outcomes of patients with locally advanced irresectable or clinically node-positive UC treated with preoperative systemic induction therapy. Additionally, it aimed to compare the efficacy of induction chemotherapy with non-chemotherapy treatment alternatives. As literature review yielded insufficient and very heterogenous results, this review was conducted in a narrative manner. The included studies showed considerable heterogeneity with regards to eligible patient populations and reported outcomes, including pCR rates and survival. In summary, the reported pCR rates after treatment with induction chemotherapy ranged from 9% to 27%. Median OS varied from 23 to 49 months; whereas the 5-year OS and CSS rates ranged between 25% and 34% and 29% and 49%, respectively. These variations might be explained by several factors, including the heterogeneity of the included patient populations, inaccurate clinical staging and merging of subgroups.
Acknowledging that OS and CSS are the outcomes what it's all about for patients and for evaluating the real impact of induction therapy, available data and variability in reported outcomes hamper drawing firm conclusions on survival benefit. As pCR demonstrated to serve as a surrogate for the efficacy of chemotherapy including outcomes in muscle-invasive bladder cancer (MIBC) patients, 24 the pCR rate may primarily serve as a better outcome measure for the effect of induction therapy.
Several trials in the neoadjuvant setting demonstrated that patients with pCR survive longer than those with residual disease. A meta-analysis on 866 patients with cT2-4N0-xM0 MIBC, treated with neoadjuvant chemotherapy followed by RC, found that patients who experienced a pCR had a 55% lower risk of death (HR: 0.45; 95% CI, 0.36–0.56) and a 81% lower risk of recurrence (HR: 0.19; 95% CI, 0.09–0.39) than patients with any residual disease. Moreover, pCR was associated with a 60% lower risk of death compared to patients with residual muscle-invasive disease. 25 Additionally, patients with pCR had a median CSS of 127 months and 5-year CSS of 64% versus 20 months and 29% in all-comers. 26 These results demonstrate that achieving pCR is linked to better clinical outcomes and prolonged survival.
In locally advanced UC, the extent of the affected lymph nodes guides disease management towards the neoadjuvant (cN0-1) or induction setting (cN2-3). 2 Accurate clinical staging is therefore crucial in selecting an appropriate treatment strategy. Pre-operative induction chemotherapy is standard-of-care in both settings, although in the induction setting, adequate response to chemotherapy is a prerequisite to proceed to surgery. In literature, reported outcomes are often not stratified by clinical nodal status. 27 Patients with cN1, cN2, cN3 and even cM1 disease are frequently grouped together, which hampers the interpretation of outcomes for patients in the induction setting. Additionally, cross-sectional imaging has limited sensitivity to detect lymph node metastases, especially in non-enlarged lymph nodes, which contributes to discrepancies in clinical-pathological staging. Despite these acknowledged limitations, only one of the included studies required histological confirmation of the cN status. This suggests a potential for understaging in the present cohort. The presence of occult lymph node metastases is associated with inferior OS, illustrating the unfavourable impact of lymph node metastases on clinical outcome.3,4,28 Finally, throughout the inclusion period of several of the included studies, guidelines regarding N staging were changed. In 2010, the size of a pelvic lymph node metastasis was removed and cN staging became entirely reliant on the number and location of lymph nodes only.29–31 These factors may have contributed to heterogeneity of the included patient populations.
Several other observations arise regarding the patient populations encompassed in the included studies. While we intended to include data from patients with UC of the bladder, upper urinary tract and urethra, included studies reported almost exclusively on UC of the bladder. Moreover, uniform definitions of OS and CSS were lacking. Studies used either the initiation of systemic therapy or the date of surgery as baseline, which resulted in outcome discrepancies spanning several months, thereby complicating direct comparison between studies. Additionally, some studies lacked specific information on chemotherapy treatment regimens and the median number of cycles administered. This all resulted in limited comparability and generalizability of the results.
Furthermore, several studies did not differentiate between pre-operative and adjuvant chemotherapy, even though this is crucial when assessing pathological outcomes. Additionally, studies often failed to directly link individual clinical and pathological staging, which hampers the assessment of response to pre-operative systemic induction therapy. Patients were grouped together and individual clinical and corresponding pathological staging per patient was not provided. This data is important in clinical practice as response to systemic therapy may affect survival outcomes and future treatment perspectives. Finally, many retrospective studies reported on patients who proceeded to surgery after systemic treatment but no information was provided on patients starting induction therapy but who refrained from surgery due to various reasons, such as disease progression, toxicity or frailty. The current included studies are also subject to this selection bias.
Prognosis in patients with locally advanced UC may be affected by histological subtypes and surgical margin status. Different UC subtypes can affect outcomes, with some linked to increased probability of recurrence and progression and lower survival rates.32,33 Additionally, these subtypes might respond differently to chemotherapy. 34 Only two studies provided data on histological subtypes,14,23 and only three reported on surgical margin status,14–16 but did not stratify for this in the analysis. Future research should include these factors to better understand their impact on prognosis.
Literature on treatment modalities other than induction chemotherapy is scarce for patients with locally advanced irresectable or clinically node-positive UC. Two studies, which described cohort 1 and 2 of the NABUCCO trial, showed better pCR rates in the population treated with preoperative high dose ipilimumab and nivolumab when compared to pCR rates after induction chemotherapy (33% to 40% vs 9% to 27%). However, as the sample size of the NABUCCO cohorts was small and the study had a single-arm design, a fair comparison cannot be made. Therefore, the added value of double checkpoint inhibition in this setting needs further confirmation.
Although literature on immunotherapy in the induction setting is limited, it is commonly studied in metastatic UC. The Checkmate 901 study showed that in patients with treatment-naïve, unresectable or metastatic UC, adding nivolumab to gemcitabine-cisplatin (GC) resulted in a modest prolongation of median OS (OS: 21.7 versus 18.9 months; HR = 0.78, p = 0.02) and progression-free survival (PFS) (7.9 versus 7.6 months; HR = 0.72, p = 0.001) compared to chemotherapy alone. 35 Furthermore, the Keynote-A39 study showed that combining enfortumab vedotin (EV) with pembrolizumab nearly doubled median OS (31.5 vs 16.1 months; HR = 0.47, p < 0.001) and median PFS (12.5 vs 6.3 months; HR = 0.45, p < 0.001) in patients with treatment-naïve Iocally advanced or metastatic UC compared to chemotherapy alone. 36 These results are encouraging and as locoregionally advanced or irresectable cancer patients were eligible for these studies, a detailed subgroup analysis of their clinical outcomes would help further development of therapeutic options in this setting.
Current studies investigate the efficacy of EV combined with pembrolizumab in the neoadjuvant phase III trial (NCT04700124; Keynote B15), 37 and various PD-(L)1 inhibitors are examined in combination with or without chemotherapy in multiple phase III trials.37–39 A randomized phase II trial investigates the efficacy of ddMVAC chemotherapy with ddMVAC plus durvalumab in patients with cTanyN1-3M0 UC of the bladder (NCT05137262). 40 Additionally, the multicentre CHASIT study (NCT05600127) assesses the potential benefit of sequential chemo-immunotherapy in patients with cT4bNxM0 or cTxN1-3M0 UC of the bladder, ureter or urethra. 41 Rationale for this trial is based on the data from the Javelin-100 bladder trial, which showed survival benefit from maintenance avelumab in metastatic UC patients who have at least stable disease after chemotherapy. 42 The pCR rate is the primary outcome of both studies. Results of these trials may shed light on the efficacy of immunotherapy as induction treatment strategy and its possible implications in the clinic.
Overall, this review provided a detailed and comprehensive overview of the literature on clinical outcomes of systemic induction therapy for locally advanced UC. A major limitation was the disparity and heterogeneity of the reported data, which did not allow a direct comparison between chemotherapy and non-chemotherapy regimens and drawing definitive conclusions was therefore difficult. Other limitations included inconsistent definitions of OS and CSS, missing data on chemotherapy regimens administered, a lack of distinction between subgroup of patients who received induction versus adjuvant treatment and a lack of stratification of outcomes by clinical nodal status, i.e., cN1 versus cN2 versus cN3. Future studies should address these issues to improve the understanding of treatment efficacy in this disease setting.
Conclusion
In patients with locally advanced irresectable or clinically node-positive UC, the reported pCR rates following systemic induction chemotherapy were modest. Considerable heterogeneity between studies hampered direct comparison of the efficacy of induction therapy with chemo- versus non-chemotherapy regimens. Survival outcomes despite induction therapy and extensive surgery were poor with <50% 5-year overall survival. Future studies should consider patients with locally advanced irresectable and clinically node-positive UC as a separate clinical entity, having different prognosis and an aligned treatment strategy.
Supplemental Material
sj-docx-1-blc-10.1177_23523735241301646 - Supplemental material for Systemic induction therapy in patients with locally advanced or node-positive urothelial carcinoma: Evaluating treatment outcomes
Supplemental material, sj-docx-1-blc-10.1177_23523735241301646 for Systemic induction therapy in patients with locally advanced or node-positive urothelial carcinoma: Evaluating treatment outcomes by Vera C Rutten, Jan-Jaap Mellema, Tahlita CM Zuiverloon, Debbie GJ Robbrecht, Michiel S van der Heijden and Joost L Boormans in Bladder Cancer
Footnotes
Abbreviations
Acknowledgements
We would like to thank Wichor Bramer for his assistance in performing the search.
Author contributions
Concept and design: Rutten, Mellema, Boormans
Acquisition of data: Rutten, Mellema
Analysis and interpretation of data: Rutten, Mellema, Boormans
Drafting of manuscript: Rutten, Mellema
Critical revision of the paper for important intellectual content: Boormans, Zuiverloon, Robbrecht, van der Heijden
Supervision: Boormans, Zuiverloon, van der Heijden
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joost Boormans: JB performed consultancy work for BMS, Janssen, Astellas, Merck and Pfizer of which all financial rewards were attributed to the Erasmus Medical Center. In addition, JB was involved in research agreements for MSD and Janssen.
Tahlita Zuiverloon: TZ has been employed on project basis for Janssen Pharmaceuticals and Genecentric. TZ is an Editorial Board member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
Debbie Robbrecht: DR received other support consultation fees from Merck AG, MSD, Pfizer, Astellas, Astrazeneca and Janssen. Additionally, she received study grants from Merck AG. All fees and grants were paid to the institution.
Michiel van der Heijden: MvdH received research funding from BMS, Merck/MSD, AstraZeneca, Roche and 4SC, which were all attributed to the Netherlands Cancer Institute. Additionally, MvdH received consulting fees from BMS, Merck/MSD, AstraZeneca, Astellas, Pfizer, Seagen and Janssen, which were also fully paid to the institute.
Vera Rutten and Jan-Jaap Mellema have no conflicts of interest to disclose.
Supplemental material
Supplemental material for this article is available online.
Appendix A. List of search terms and databases
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
