Abstract
Introduction
Inflammatory mediators produced by macrophages play a key role in the immune response to sepsis. Agonism of the cannabinoid receptor two represents a promising anti-inflammatory strategy that can modulate macrophage activation in this context. In this article, we highlight the use of the synthetic cannabinoid HU-308 as an anti-inflammatory therapeutic agent in an in vitro model of macrophage inflammation.
Methods
To this end, RAW 264.7 murine macrophages were stimulated with lipopolysaccharide and their production of inflammatory mediators was evaluated using enzyme-linked immunosorbant assays. Further, we evaluated macrophage viability with HU-308 treatment and confirmed the expression of the cannabinoid receptor 2.
Results
We identified a dose-related reduction in the secretion of IL-6, TNF, and CXCL8.
Conclusion
HU-308 is a promising candidate for reducing inflammation secondary to macrophage activation, indicating the value of additional research on this topic.
Introduction
Macrophages play an integral role in the immune response during sepsis by recognizing pathogen-associated molecular patterns (PAMPs) and activating neighbouring cells via secretion of pro-inflammatory cytokines and chemokines. 1 Upon sensing Gram-negative bacterial lipopolysaccharide (LPS) via intracellular and extracellular recognition systems, tissue-resident macrophages polarize to a pro-inflammatory (M1) phenotype characterized by the secretion of pro-inflammatory cytokines (e.g., IL-6, TNF) and chemokines (e.g., CXCL2). 2 These inflammatory mediators activate a suite of host defense mechanisms including leukocyte recruitment and production of reactive oxygen species (ROS), mechanisms which aid in pathogen killing but also cause collateral tissue damage, thus perpetuating organ dysfunction and risk of death. 3 Modulating activation of macrophages during sepsis thus represents a potentially important therapeutic target.
The endocannabinoid system (ECS) is an endogenous signaling network comprising cannabinoid type 1 and 2 receptors (CB1 and CB2), endocannabinoid ligands (endocannabinoids [eCBs]), and their cognate enzymes. 4 CB2 is highly expressed in immune cells and is capable of modulating inflammation in vivo and in vitro. 5 Many macrophages and macrophage-derived cells in humans have been demonstrated to express CB1 and CB2 and respond accordingly to pharmacological agonists/antagonists of both receptor subtypes. 6 This raises the question as to whether CB2 activation in macrophages may be a potential therapeutic target.
In this brief report, we investigate the impact of CB2 activation on LPS-stimulated RAW 264.7 macrophages. We employ the CB2-selective agonist HU-308, which possesses minimal off-target activity and low signaling bias. 7 Herein, we demonstrate that HU-308 reduces cytokine secretion in CB2-expressing RAW 264.7 cells.
Methods
RAW 264.7 (TIB-71, American Type Culture Collection, via Cedarlane Labs, Burlington, ON, Canada) cells were cultured in 60 mm culture dishes with Dulbecco’s Modified Eagle’s Medium (DMEM; Wisent Bioproducts, Saint-Jean-Baptiste, QC, Canada) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Avantor, Radnor, PA, USA). Cultures were incubated at 37°C in a 5% CO2 humidified atmosphere and sub-cultured every 2–3 days to a maximum of 12 passages. After overnight incubation to allow adherence, cells were washed once with warm Dulbecco’s phosphate-buffered saline (DPBS; Gibco, Carlsbad, CA, USA), and experimental reagents were added simultaneously (co-treatment model) in warm DMEM containing 10% FBS prior to 24 h incubation. LPS (from Pseudomonas aeruginosa; Sigma-Aldrich, Oakville, ON, Canada) was administered in DPBS constituting 0.04% of the final media volume to a final concentration of 1 μg/mL. HU-308 (Tocris Bioscience, Bristol, UK) was administered in DMSO (Sigma-Aldrich, Oakville, Canada) constituting 0.5% of the final media volume to final concentrations of 5 μM or 10 μM. Dexamethasone (Sandoz, Basel, Switzerland), a glucocorticoid receptor agonist and immunosuppressant, was used as a positive control and administered in media to a final concentration of 100 nM. All vehicle concentrations were normalized for each experimental condition.
Cell viability was assayed in 96-well plates via the Cell Counting Kit-8 (CCK-8; Dojindo, Gaithersburg, MD, USA) colorimetric assay according to the manufacturer’s recommendations. Upon expiration of the 24 h timepoint, 10 μL/well of CCK-8 solution was added per 100 μL/well culture media and plates were incubated for 1–2 h (depending on the rate of color development) in standard cell culture conditions (37°C, 5% CO2). Absorbance was quantified using a Cytation 5 Cell Imaging Multi-Mode Reader (Agilent, Santa Clara, CA, USA). Experimental assays in this study were performed in 3–8 technical replicates (i.e., 3–8 wells) per experimental condition. Cell viability for each experimental condition was calculated as percent of absorbance relative to non-stimulated conditions.
Cytokine production was assessed in cell culture supernatants by ELISA. Supernatant was collected from the 24-well plates at 24 h culture, centrifuged (2000×g, 4°C, 10 min) and stored immediately at −20°C (short-term) or −80°C (long-term). Prior to assay, supernatant was thawed, vortexed briefly, and centrifuged at 2000 g at 4°C for 5 min. Samples were assayed in duplicate according to manufacturer’s recommendations using the following ELISA kits: mouse IL-6 (Invitrogen, Carlsbad, CA, USA), TNF alpha (Invitrogen, Carlsbad, CA, USA), and MIP-2/CXCL2 (Peprotech, Cranberry, NJ, USA). Absorbance was quantified via plate reader (Cytation 5 Cell Imaging Multi-Mode Reader; Agilent BioTek, Santa Clara, USA) at 450 nm with wavelength correction at 570 nm for TMB substrate (IL-6, TNF), and 405 nm with wavelength correction at 650 nm for ABTS substrate (CXCL2). Data are reported in ρg/mL.
To examine intracellular CB2 expression at 24 h of cell culture, cells in 24-well plates were washed three times with cold PBS, fixed in ice-cold methanol for 30 min, washed three times with cold PBS again, and stored at 4°C. Cells were then permeabilized with 0.2% Triton X-100 in PBS for 10 min, washed three times in PBS, and blocked for 30 min in PBS with 0.1% Tween-20 (PBST) with 1% bovine serum albumin (BSA). After washing once in PBST, cells were incubated overnight at 4°C with primary antibody (CNR2 Recombinant Rabbit Monoclonal Antibody, Invitrogen, Carlsbad, CA, USA) with a 1:200 dilution in PBST with 1% BSA. Then, cells were washed three times in PBST, and incubated for 30 min with fluorescent secondary antibody (Goat anti-Rabbit IgG Cross-Adsorbed Secondary Antibody, Alexa FluorTM 488, Invitrogen, Carlsbad, CA, USA). Finally, cells were washed three times in PBST. Imaging was performed in cell culture plates using the ZEISS Axio Observer at 40X magnification.
Results
To evaluate the inflammatory response of RAW 264.7 macrophages stimulated with LPS, we evaluated cytokine and chemokine expression using an ELISA. The RAW 264.7 macrophages demonstrated robust secretion of IL-6, TNF, and CXCL2 relative to unstimulated conditions (Figure 1(a)). Incubation with HU-308 significantly reduced this cytokine secretion: IL-6 and CXCL2 secretion were strongly attenuated at both of the tested doses of HU-308, while TNF secretion was modestly reduced at 10 μM HU-308. Enhanced suppression was observed at the higher dose of HU-308, suggesting dose-dependent effects. A similar suppressive effect was observed through incubation with dexamethasone (positive control), which exerted similar, although slightly more potent, suppression of cytokine release. Impact of HU-308 in LPS-stimulated RAW macrophages. (a) Cytokine levels in supernatant as determined by ELISA. (b) Viability was determined in 96-well plates via CCK-8 assay. (c) Representative immunofluorescence images depicting bright field images and CB2 immunoreactivity; scale bars = 100 μm. HU (5) and HU (10) indicate 5 and 10 μM of HU. All drug concentrations are in μM. Data are expressed as mean ± SD. Statistically significant differences are depicted as *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001.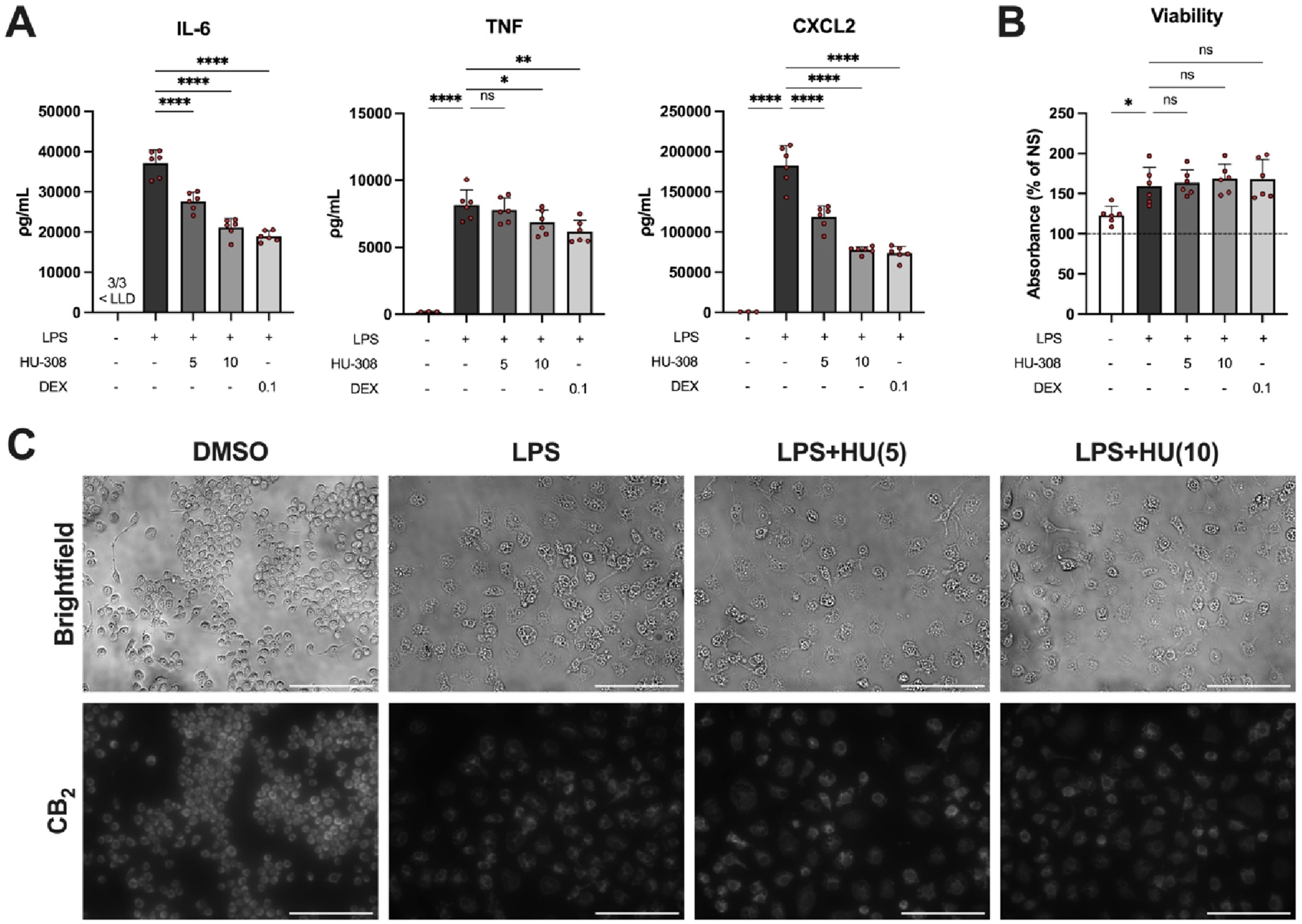
We also examined RAW 264.7 viability to evaluate the suitability of HU-308 for in vitro studies with the RAW 264.7 cells. While LPS stimulation induced a proliferative response, no significant alteration of cell viability was observed in response to HU-308 or dexamethasone treatment (Figure 1(b)).
Finally, we employed fluorescent microscopy to visualize CB2 immunoreactivity in the RAW 264.7 cells. All conditions demonstrated CB2 immunoreactivity, indicating the expression of the CB2 receptor by RAW 264.7 cells. No marked differences were observed among the HU-308 treated conditions (Figure 1(c)).
Discussion
The present report demonstrates that the CB2 agonist HU-308 diminished LPS-induced secretion of IL-6, TNF, and CXCL2 in RAW 264.7 cells in a dose-related manner. Secretion of these inflammatory mediators from macrophages plays a critical role in the inflammatory response during sepsis, and may initiate or exacerbate organ dysfunction. Additionally, the RAW 264.7 cell line was shown to express the CB2 receptor, confirming a previous report by Mukhopadhyay et al. 8 Taken together with the lack of cell death observed when treated with HU-308, this indicates that this cell line represents a valuable tool for evaluating the anti-inflammatory effects of CB2 agonists in vitro.
HU-308 was previously reported to reduce NLRP3 inflammasome expression through an autophagy-dependent mechanism in LPS and dextran sodium sulfate-stimulated RAW 264.7 macrophages. 9 Similarly, a later study found that reductions in inflammatory mediator production by HU-308 were driven by inhibition of NLRP3-mediated pyroptosis. 10 Collectively, these studies may implicate pro-autophagic and anti-pyroptotic effects in the observed anti-inflammatory effects of HU-308 in RAW macrophages.
These findings suggest that CB2 agonists such as HU-308 could be useful in reducing LPS-induced pro-inflammatory mediator release by macrophages during sepsis. An important limitation of this study is the utilization of an immortalized macrophage cell line, which does not fully recapitulate the diverse phenotypes of tissue-resident macrophages in vivo. Thus, future studies ought to investigate the relevance of these results in specific primary macrophage populations involved in sepsis, such as peritoneal macrophages, alveolar macrophages, and Kupffer cells.
Footnotes
Acknowledgments
The authors would like to acknowledge significant contributions in the form of technical expertise by Dr Karla Valenzuela, Dalhousie University, and in the form of provision of research space by Dr Melanie Kelly, Dalhousie University.
Author contributions
Conceptualization, S.H. and C.L.; methodology, S.H.; formal analysis, S.H.; investigation, S.H.; resources, Z.C.; data curation, S.H.; writing—original draft preparation, S.H.; writing—review and editing, S.H, K.D., Z.C., J.Z, and C.L.; visualization, S.H.; supervision, C.L.; project administration, J.Z. and C.L. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was supported by the Anesthesia Research Fund provided by the Department of Anesthesia, Pain Management, and Perioperative Medicine, Dalhousie University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data and details of resources can be found within the article.
