Abstract
Objectives. Antimicrobial resistance (AMR) poses a significant threat to global public health, with substantial mortality rates attributed to AMR-related infections. Pediatric populations face heightened vulnerability due to prevalent antimicrobial misuse. This study aimed at addressing the significant threat of antimicrobial resistance (AMR) and its associated mortality rates. Methods. This retrospective cross-sectional multicentric study investigated antibiotic prescribing patterns in pediatric wards of 4 secondary care hospitals affiliated with Aga Khan University Hospital. The study utilized the WHO Access, Watch, and Reserve (AWaRe) classification framework. Data from 6934 encounters were analyzed. Results. Antibiotics were prescribed in 78.1% of encounters, with intravenous administration being predominant (98.6%). Ceftriaxone was the most prescribed antibiotic agent (45.8%), and third-generation cephalosporins constituted the most prevalent antibiotic class (54.4%). Pneumonia exhibited the highest prescription rate (99.9%), with Watch group antibiotics being predominantly prescribed (>80%) across hospitals. Conclusion. These findings underscore the urgency for targeted interventions to optimize prescribing practices and mitigate resistance.
Keywords
Introduction
Antimicrobial resistance (AMR) stands as a formidable threat to global public health, with implications ranging from increased mortality rates to compromised treatment efficacy in infectious diseases. In 2019 alone, approximately 4.95 million deaths were associated with drug-resistant infections, and 1.27 million of these deaths were directly caused by AMR, underscoring its urgent and pervasive nature. 1 Particularly vulnerable are young children, with 1 in 5 deaths due to AMR occurring in children under 5 years old. 2 Notably, regions such as sub-Saharan Africa and South Asia bear a disproportionate burden of AMR-related fatalities, highlighting the critical need for comprehensive interventions in low- and middle-income countries (LMICs). 1 In South Asia, countries like India, Pakistan, and Bangladesh grapple with alarming statistics regarding AMR. For instance, India faces an estimated annual burden of over 2 million deaths attributed to AMR-related infections by 2050. 3 Pakistan is also witnessing a rise in AMR rates, complicating the treatment of common infections. 4 Similarly, Bangladesh struggles with the spread of resistant pathogens, compounded by resource constraints. 5
The vulnerability of infants and young children to bacterial infections further accentuates the urgency of addressing AMR, particularly in settings like Pakistan where antimicrobial misuse is prevalent. 6 Children, constituting significant consumers of antibiotics globally, face heightened risks due to factors such as variable body weights and immature immune systems. 7 In Pakistan, the rate of premature births is notable, with approximately 15% of births occurring prematurely. 8 Additionally, a considerable percentage of children in Pakistan are underweight, with around 32% of children under the age of 5 classified as underweight. 9
To mitigate the escalating threat of AMR, the World Health Organization (WHO) has championed initiatives such as the Global Action Plan on AMR, advocating for enhanced surveillance, and optimization of antimicrobial use. 10 In this vein, the WHO introduced the Access, Watch, and Reserve (AWaRe) classification system to guide antibiotic prescribing and monitoring efforts. 11 The AWaRe classification system, categorizes antibiotics not only based on their therapeutic value but also considering their impact on antibiotic resistance. This framework divides antibiotics into 3 categories: Access, Watch, and Reserve, to guide their appropriate use and minimize resistance development. The Access group includes antibiotics with a lower resistance potential, while the Watch and Reserve groups contain those with higher resistance risks and should be used sparingly. By promoting judicious antibiotic use, AWaRe aims to balance effective treatment and the mitigation of resistance spread. 12
The significance of the AWaRe classification lies in its potential to foster tailored antimicrobial stewardship strategies, particularly in high-burden settings where surveillance data are scant. 13 By emphasizing the prioritization of narrow-spectrum antibiotics (Access group) over broad-spectrum agents (Watch group), the AWaRe classification aligns with global efforts to curb AMR. 14 Notably, the WHO targets to ensure that at least 60% of prescribed antibiotics belong to the Access group, thereby restraining the emergence of resistance. 15
Pakistan has developed a National Action Plan for Antimicrobial Resistance (AMR), aligning with the Global Action Plan on AMR by the World Health Organization. This comprehensive plan outlines strategic objectives to combat AMR through improving awareness, strengthening surveillance, reducing the incidence of infection, optimizing the use of antimicrobial agents, and ensuring sustainable investment. The present study addresses one of the key objectives of Pakistan’s National Action Plan by evaluating the current patterns of antibiotic use and resistance, thereby providing critical data to inform targeted interventions and policies aimed at mitigating AMR. 16
Assessing the implementation of the AWaRe classification in pediatric settings is paramount, given the unique prescribing considerations for children and neonates. 17 However, existing literature predominantly focuses on adult populations, leaving a critical gap in understanding pediatric antibiotic practices, particularly in LMICs like Pakistan. 18 Against this backdrop, this study endeavors to investigate the prescribing patterns of antibiotics in the pediatric wards of secondary care centers, utilizing the AWaRe classification as a framework.
In 2021, the WHO Essential Medicines List (EML) Working Group classified antibiotics in the EML for Children (EMLc) into 3 groups: Access, Watch, and Reserve. (The AWaRe Classification). Traffic light color codes have been suggested to indicate different categories. Access antibiotics (green), Watch antibiotics (amber), and Reserve antibiotics (red). 19
The Access group: It contains generally narrow spectrum antibiotics recommended as first and second choice for most common clinical infection syndromes. The WHO 13th General Program of Work 2019-2023 includes a country-level target of at least 60% of total antibiotic consumption being Access group antibiotics.
The Watch group: It contains generally broader spectrum antibiotic classes corresponding to the highest priority agents with toxicity or resistance concerns on the list of critically important antimicrobial drugs for human medicine.
The Reserve group: It consists of last resort antibiotics for targeted use in multidrug resistant infections.
Specifically, this multicentric study conducted at secondary care centers within Aga Khan University Hospital aims to elucidate the AWaRe patterns in pediatric inpatients. By examining antibiotic utilization and adherence to AWaRe guidelines, this research seeks to shed light on the appropriateness of antibiotic prescribing practices in this population. Moreover, insights gleaned from this study can inform the development of tailored antibiotic prescription policies and stewardship programs, tailored to the unique needs of pediatric patients.
Methods
Study Design
It is a retrospective, descriptive, cross sectional, multi centric study.
Setting
The hospitals were selected through convenience sampling. Retrospective data was obtained from the pediatric wards of all 4 secondary care urban teaching hospitals of Aga Khan University Hospital for the year 2022. These community hospitals are in Karachi and Hyderabad, each of which although lacking intensive or special care units for neonates and pediatrics, have well-equipped nurseries, well baby units, and pediatric wards along with urgent care units. The study population comprised children from low- and middle-income countries (LMICs), characterized by limited access to advanced healthcare facilities and a higher burden of infectious diseases.
Inclusion Criteria
All admitted patients (from 1 month until 12 years) from 1st January 2022 till 31st December 2022, in the pediatric wards who have been prescribed at least one systemic antibiotic were included in the study.
Exclusion Criteria
Exclusion criteria includes single day admissions for diagnostic tests. Patients who have been prescribed with topical antimicrobial, antifungal, antiviral, anthelmintic and antiprotozoal and antituberculosis were excluded as these are not incorporated in the list of WHO AWaRe classification of antibiotics. Patients who have acquired nosocomial/hospital acquired infections were excluded.
Outcomes
The frequency of the prescribed Access, Watch and Reserved antibiotic groups used in the pediatric wards of secondary care centers of Aga Khan University Hospital according to WHO (AWaRe) classification over the past 1 year were determined.
The most common prescribed antibiotic group according to WHO Access, Watch, and Reserve (AWaRe) classification in the pediatric wards of secondary care centers over the last 1 year were determined.
Access to Watch ratio for the pediatric wards of each secondary care hospital were determined individually.
The frequency of the prescribed Access, Watch and Reserved antibiotic groups used, by age and clinical indications was determined.
Sample Size
All children between the ages of 1 month until 12 years were included in the study, who fulfill the inclusion and exclusion criteria; from 1st January 2022 to 31st December 2022. Retrospective data was collected over a period of 3 months. A total of 6934 patients were admitted in the hospital; from these 5414 patients received antibiotics, and were included in the study
Data Collection and Variables
Hospital admission data and electronic discharge summaries were used to extract data on the total number of patients admitted in pediatric wards, patients’ age, gender, and diagnosis.
Hospital pharmacy database with EMAR (electronic medication administration record) was used to extract data on the total number of patients receiving systemic antibiotics, names and number of antibiotics prescribed per admission and their route of administration; IV (intravenous) or oral. The administration routes were mostly intravenous, no intramuscular and subcutaneous injections were given.
Antibiotic drugs were coded on the basis of the WHO Anatomical Therapeutic Chemical (ATC) classification system. 20 Antibiotics which have been classified as J01 category (antibiotics for systemic use) under the ATC classification system were included in this study. The antibiotics prescribed was then categorized into Access, Watch and Reserve groups of antibiotics according to WHO AWaRe classification.
Combination antibiotic was also prescribed, in which more than 1 antibiotic was used for treating infection. Combinations of beta-lactam and aminoglycoside antibiotics are frequently used in the treatment of pediatric infections.
Statistical Analysis
We utilized the Global-PPS pediatric data for our analysis, focusing on variables consistently collected across all datasets. These variables include patient antibiotic use (type, route of administration, frequency of use), type of treatment (empirical or targeted), and diagnosis. All data were entered into a Microsoft Excel file. The statistical analyses were done using SPSS version 22.0. Categorical variables were presented as frequencies and percentages. Measures of relative consumption, expressed as a percentage of total consumption of groups of antibiotics, were derived for each antibiotic. The frequency and percentages of the respective Access, Watch and Reserved antibiotic groups was calculated.
Measures to Minimize Bias
All pediatric patients full filling the inclusion and exclusion criteria, admitted in the pediatric wards from 1st January 2022 to till 31st December 2022 was included to minimize the selection bias. Data was entered anonymously.
Ethical Approval
This study was reviewed and approved by the ERC—ethical review committee of Aga Khan University Hospital on 24-March-2023. Reference number: 2023-8507-24288.
As there was no contact with the patients, all data was collected from EMAR (Electronic medication administration record), discharge summaries and patient files, we did not need patient consent.
Result
The study examined antibiotic prescribing patterns in pediatric wards of 4 secondary care centers of Aga Khan University Hospital, focusing on WHO’s Access, Watch, and Reserve (AWaRe) classification. As shown in Table 1. A total of 6934 encounters were analyzed across 4 hospitals: A, B, C, and D. Antibiotics were prescribed in 78.1% of encounters, with the highest prescription rate observed in C (88.1%, n = 1550), followed by B (84.3%, n = 1190), A (71.3%, n = 2104), and D (70.0%, n = 570) hospitals. Intravenous antibiotics were commonly prescribed, with C hospital showing the highest rate (99.9%, n = 1548). Combination antibiotic therapy was utilized in 25.9% of encounters, with B hospital having the highest rate (40.6%, n = 483).
Antibiotic Prescribing Patterns by Hospital Setting.

Data based on the antibiotic of the first choice.
Distribution of Antibiotic Agents and Classes Across the 4 Hospitals.

The high rate of intravenous antibiotic administration can be attributed to the fact that children admitted to these hospitals often have difficulty tolerating oral medications when they are acutely ill.
Ceftriaxone is the most commonly prescribed antibiotic across the four hospitals, accounting for 45.8% of antibiotic therapies, while the third-generation cephalosporin class dominates overall prescriptions at 54.4% as mentioned in Table 2.
As shown in Figure 1, analyzing antibiotic therapy distribution across diagnostic conditions, pneumonia had the highest prescription rate (99.9%, n = 1398), followed by diarrheal illness (75.3%, n = 1558) and enteric fever (99.8%, n = 809). Combination antibiotic therapy rates varied across diagnoses, with sepsis showing the highest rate (83.9%, n = 213), as mentioned in Table 3, followed by bronchiolitis (39.7%, n = 142), Pneumonia (31.6%, n = 442) and UTI (22.5%, n = 41).

Antibiotic therapy distribution according to diagnosis.
Antibiotic Prescribing Patterns by Diagnostic Conditions.
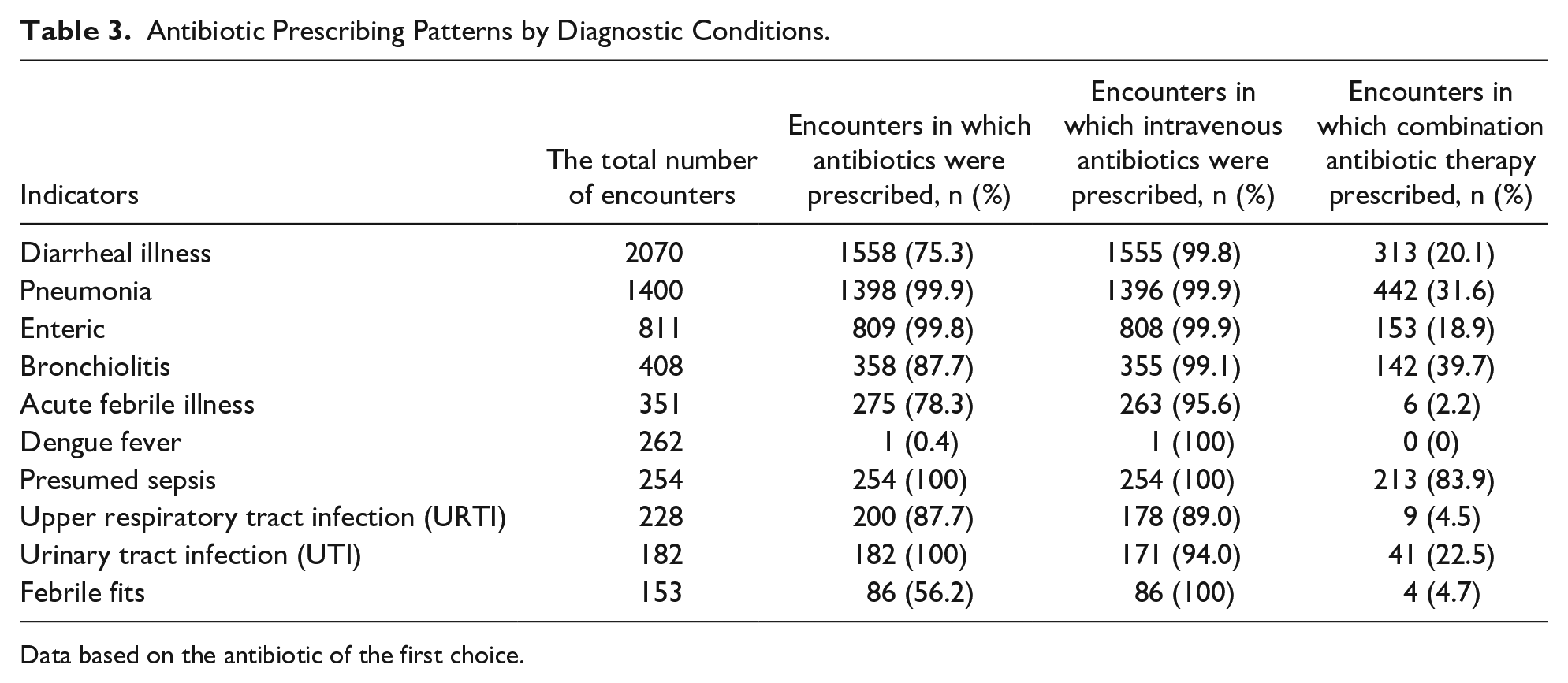
Data based on the antibiotic of the first choice.
Additionally, the distribution of antibiotic therapy according to AWaRe categories was examined across hospitals Figure 2. In A, B, C, and D hospitals, Watch group antibiotics constituted the majority of prescriptions (>80%), with third-generation cephalosporins being the most commonly prescribed.

Antibiotic therapy distribution according to AWaRe categories across hospitals.
Similarly, antibiotic therapy distribution according to AWaRe categories across various diagnoses was assessed (Figure 3) Third-generation cephalosporins were frequently prescribed for both respiratory tract conditions and other common diagnoses, such as presumed sepsis and urinary tract infections, while macrolides were common for cough and bronchiolitis.

Antibiotic therapy distribution according to AWaRe categories across various diagnoses.
Discussion
The study identified a high prevalence of antibiotic prescriptions among pediatric inpatients, with 78.1% of encounters involving antibiotic use. This observation aligns with global trends emphasizing the substantial reliance on antibiotics in pediatric healthcare settings. 21 Intravenous administration was predominantly favored, indicating the severity of conditions or the preference for rapid therapeutic action. Furthermore, the considerable utilization of combination antibiotic therapy (25.9%) underscores potential issues with the misuse or overuse of antibiotics, where combination therapy is being employed even for the treatment of single diseases. 22
Ceftriaxone emerged as the most frequently prescribed antibiotic agent, consistent with its broad spectrum and versatility in treating various infections. Third-generation cephalosporins, as a class, dominated the antibiotic prescriptions, followed by fluoroquinolones and carbapenems. 23
The distribution of antibiotic therapy varied across diagnostic conditions, with pneumonia exhibiting the highest prescription rate. This finding reflects the commonality of bacterial pneumonia in pediatric populations and emphasizes the need for appropriate empirical antibiotic coverage. 24 However, the high prescription rates across various diagnoses, including diarrheal illness, bronchiolitis and acute febrile illness warrant further scrutiny to ensure judicious antibiotic use and mitigate the risk of unnecessary antimicrobial exposure. 25
Comparisons with previous studies underscore the consistency of findings regarding the high prevalence of antibiotic use in pediatric settings. For instance, a study conducted in 2020 by Miao et al in different types of hospital in China, reported a similar high rate of antibiotic prescriptions among pediatric inpatients, with intravenous administration being the preferred route, mirroring our study’s findings. 26 However, while our study focused on secondary care centers in Pakistan, Smith et al’s study provides insights from a different healthcare setting, highlighting the universality of the issue. 27 Furthermore, a study by Labi et al among 716 pediatric inpatients in Ghana revealed comparable trends in antibiotic prescribing patterns, with third-generation cephalosporins being the most commonly prescribed class, akin to our findings. 28 This consistency across different regions underscores the global nature of antibiotic misuse in pediatric healthcare.
Variations in prescribing patterns and antibiotic utilization across different regions emphasize the influence of local factors. For example, a study in 2022 by Fabre et al in Latin America found regional disparities in antibiotic prescribing practices, attributed to differences in healthcare infrastructure and access to antimicrobial stewardship programs. 29 Similarly, Islam et al’s study in Bangladesh highlighted the impact of socioeconomic factors on antibiotic use in pediatric populations, indicating the need for context-specific interventions. 30 These findings suggest that while overarching strategies are crucial, tailored approaches addressing local determinants are essential to combat antimicrobial resistance effectively.
While our study provides valuable insights into antibiotic prescribing practices in secondary care centers, further research incorporating primary care settings and community-based data is warranted. For instance, a study by Ardillon et al conducted a multicentric community-based cohort study in Madagascar, Senegal, and Cambodia, revealing widespread inappropriate antibiotic prescribing among pediatric outpatients. Despite varying intercountry practices, common risk factors for inappropriate prescription were identified, emphasizing the necessity of targeted interventions to optimize antibiotic use in LMICs at the primary care level. 31 Additionally, Ding et al’s study in rural area of China demonstrated unique challenges in antibiotic prescribing, highlighting the importance of rural-specific strategies to promote prudent antibiotic use. 32 Incorporating data from diverse healthcare settings can enrich our understanding of pediatric antibiotic usage and inform comprehensive antimicrobial stewardship initiatives.
In summary, while our study contributes valuable insights into antibiotic prescribing patterns in pediatric wards of secondary care centers, it is essential to consider regional variations and diverse healthcare settings when designing antimicrobial stewardship programs. By leveraging findings from multiple studies and adopting a multifaceted approach, stakeholders can develop context-specific interventions to optimize antibiotic use and mitigate the threat of antimicrobial resistance in pediatric populations.
Implications of the study findings extend to the development and implementation of antimicrobial stewardship programs tailored to pediatric populations. Targeted interventions focusing on optimizing antibiotic prescribing practices, promoting adherence to evidence-based guidelines, and enhancing surveillance of antimicrobial resistance can mitigate the emergence and spread of resistant pathogens. 33 Additionally, educational initiatives targeting healthcare professionals and caregivers can raise awareness about the appropriate use of antibiotics and the importance of antimicrobial stewardship in safeguarding pediatric health.
Limitations
Limitations of the study include its retrospective design, it inherently relies on existing medical records and electronic databases, which may introduce selection bias due to incomplete documentation or variability in record-keeping practices across different hospitals. This could impact the accuracy and completeness of data extraction, potentially influencing the study’s findings and conclusions.
Secondly, the study’s focus on secondary care centers with specific capabilities—such as nurseries, well-baby units, and pediatric wards—limits its generalizability to other healthcare settings within Pakistan, including primary care facilities or tertiary hospitals with intensive care units. Variations in patient demographics, disease severity, and healthcare practices between different hospital types may not be fully captured, thereby limiting the broader applicability of the study’s findings.
Thirdly, the exclusion criteria, which omit patients with single-day admissions for diagnostic purposes and those prescribed non-systemic antibiotics (eg, topical agents), may overlook a subset of patients whose antibiotic prescribing patterns could differ significantly from those included in the study. Additionally, the exclusion of patients with nosocomial infections might underestimate the prevalence and impact of antibiotic resistance within hospital settings.
Furthermore, while the study categorizes antibiotics according to the WHO AWaRe classification, it primarily focuses on prescribing patterns and does not evaluate clinical outcomes such as treatment efficacy, adverse events, or the development of antimicrobial resistance. These outcomes are crucial for assessing the appropriateness and effectiveness of antibiotic use, providing a more comprehensive understanding of the implications of prescribing practices.
Addressing these limitations through prospective studies that incorporate diverse healthcare settings, comprehensive data collection methods, and robust outcome measures would enhance the validity and applicability of findings, supporting evidence-based strategies for optimizing antibiotic use and combating antimicrobial resistance in pediatric populations.
Conclusion
In conclusion, this study offers valuable insights into the antibiotic prescribing patterns observed in pediatric wards of secondary care centers. The findings reveal a notable prevalence of antibiotic use, with a predominant preference for broad-spectrum agents. Additionally, the considerable utilization of combination antibiotic therapy highlights potential issues with the misuse or overuse of antibiotics, where combination therapy is being employed even for the treatment of single diseases. These observations emphasize the necessity for targeted interventions aimed at optimizing antibiotic prescribing practices and fostering antimicrobial stewardship within pediatric healthcare settings. Such initiatives are essential for safeguarding the efficacy of antimicrobial therapy and mitigating the emergence and spread of antimicrobial resistance.
Footnotes
Acknowledgements
None
Author Contributions
All authors contributed significantly to the work reported in the manuscript. Afia Tariq Butt and Sobia Ramzan, conceptualized the study and develop the methodology. Dania Shabbir and Amna Sajjad conducted the data collection and analysis. Murk Shaikh provided critical revision and interpretation of results. Maryam Iqbal Syed and Mohammad Aadil Qamar oversaw the project administration and manuscript preparation. All authors contributed to drafting the manuscript, reviewed, and approved the final version of submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The primary study was approved by the ethical review committees of Aga Khan University Hospital (2023-8507-24288).
