Abstract
Objective. Children’s vulnerability to drug-related side effects has been highlighted in several studies. However, there is no consensus on the risk factors associated with these side effects. This study aimed to investigate risk factors associated with drug-related side effects in children. Methods. This scoping review was conducted across multiple databases. The search strategy was created with a focus on drug-related side effects, as they are more predictable based on the pre-determined risk factors. Data were collected, and reported narratively. Results. The demographic, health, hospital, and drug-related risk factors may cause drug-related side effects in children. Among them, low age, sex, polypharmacy, length of hospitalization, and medications used for comorbidities may increase the risk. Conclusion. While most of the risk factors might be similar in adults and children, their impact might be different in these 2 groups. Therefore, future studies should identify more details about the impact of the risk factors in children.
Graphical Abstract
Introduction
Drug use in children has always been an issue of concern for various reasons, such as their vulnerable and growing physiology and the differences in absorption, distribution, metabolism, and excretion of drugs compared to adults. 1 Indeed, as a vulnerable group, children are at increased risk of drug-related side effects, and the dosage and formulation of the drug should be considered at the time of administration. 2 As a vulnerable population, children are predisposed to encountering negative repercussions of medications. 1 These repercussions typically include adverse drug reactions (ADRs), adverse events (AEs), and side effects. 3 ADRs encompass unintended and detrimental reactions to a drug when administered at standard doses for preventive, diagnostic, or therapeutic purposes. These reactions span a spectrum from mild to severe, including fatal outcomes. 4 AEs, a broader term, encompass any unintentional consequences associated with medical intervention, medication errors, device malfunctions, and hospital-acquired infections. 3 Conversely, side effects represent unintended yet generally foreseeable outcomes of medication intake. Typically, drug–related side effects refer to unwanted outcomes of therapeutic usage that typically do not mandate treatment cessation, while toxic effects encompass ADRs. The latter occurs due to elevated drug concentrations, whether intentional or unintentional. 5
The safety of many drugs has not been adequately assessed in different age groups of children due to the limited data availability and ethical constraints of clinical trials. 6 In fact, despite differences between drug-related side effects in children and adults, relatively little attention has been paid to drug-related side effects in children, 7 and the limited knowledge of pediatricians about drug-related side effects may lead to potential complications and hospitalizations.7,8
Studies have shown that the incidence of drug side effects in children is between 0.6% and 16.8%,9,10 of which 3.9% can be serious and lead to death in children. 11 In addition, inpatient children are more susceptible to drug-related side effects due to the complexity of dosing and the difficulty in determining the safety and efficacy of medications. 7 It appears that children in intensive care units (ICUs) are at higher risk for drug-related side effects due to the administration of multiple drugs. 12 For example, the risk of a drug-related side effect in this ward increases by 1.7% with each additional drug. 13
It should be noted that there is no consensus on the risk factors associated with drug-related side effects in children. Previous studies have identified factors such as polypharmacy and low age as potential risk factors.7,8,12 However, there is still a need for more reliable data on the risk factors associated with drug-related side effects in the pediatric population. Therefore, this study aimed to investigate risk factors associated with drug-related side effects in children. The research question was as follows:
What are the risk factors (demographic, health-related, hospital-related, and drug-related risk factors) associated with drug-related side effects in children?
The results of the current study could be used in evidence-based drug therapy to ensure safer drug interventions and minimize potentially associated harms in the pediatric population.
Methods
This scoping review was conducted in 2023 and all studies related to the risk factors associated with drug-related side effects in children were reviewed. To conduct the scoping review, Arksey and O’Malley’s 14 framework was used. Before conducting this research, ethics approval (IR.IUMS.REC.1401.1007) was obtained from the Ethics Committee of Iran University of Medical Sciences.
Stage 1: Identifying the Research Question
A comprehensive understanding of the risk factors associated with drug-related side effects in children is essential for improving medication safety and minimizing potential harm. Therefore, the research question as noted in the introduction section was identified.
Stage 2: Identifying Relevant Studies
To identify relevant studies, 6 databases including Web of Science, PubMed, Ovid, Scopus, ProQuest, and the Cochrane Library were searched. The time frame was between 1st January 2013 to 1st November 2023. The search strategy consisted of 3 main terms: “risk factor,” “drug-related side effects,” and “children” along with their corresponding synonyms and MeSH terms. The main terms were combined using AND/OR logical operators to create search strategies for different databases (Supplemental Material 1). The reference lists of the included articles were also searched to identify additional studies that met our eligibility criteria. Moreover, key journals and conference proceedings were hand-searched to ensure that the relevant articles were included.
Stage 3: Study Selection
All relevant papers with different types of research methodologies including quantitative, qualitative, and mixed methods approaches which were published in English were included. Studies included in this scoping review investigated drug-related side effects in children as defined by the World Health Organization (WHO), encompassing individuals under 18 years old. 15 However, letters to the editor, protocols, and studies in which no risk factors associated with drug-related side effects in children were reported, were excluded from this study. Papers that were not published in English, their target group was adults, or their full texts were not available were also excluded.
The retrieved articles were organized using the Endnote software, and after removing duplicates, the remaining articles were screened in terms of the title and abstract relevancy to the aim of the study. Then, the full text of eligible studies was retrieved and reviewed. ET and HA contributed to screening the articles independently and any disagreements between them were resolved by discussing the issue with the third author (RA).
Stage 4: Charting the Data
The required data were extracted using a data extraction form which included the author (s’) name (s)/year of publication, country of the study, research objective, research methodology, and risk factors.
Stage 5: Collating, Summarizing, and Reporting the Results
The method of content analysis was used to extract data. Then, the extracted data were tabulated, summarized, and reported narratively.
Results
Initially, a total of 2412 articles were retrieved and after completing the screening process based on the PRISMA-ScR statement and applying the inclusion and exclusion criteria, 15 studies13,16-29 were identified for the final review. Figure 1 shows the process of selecting articles.
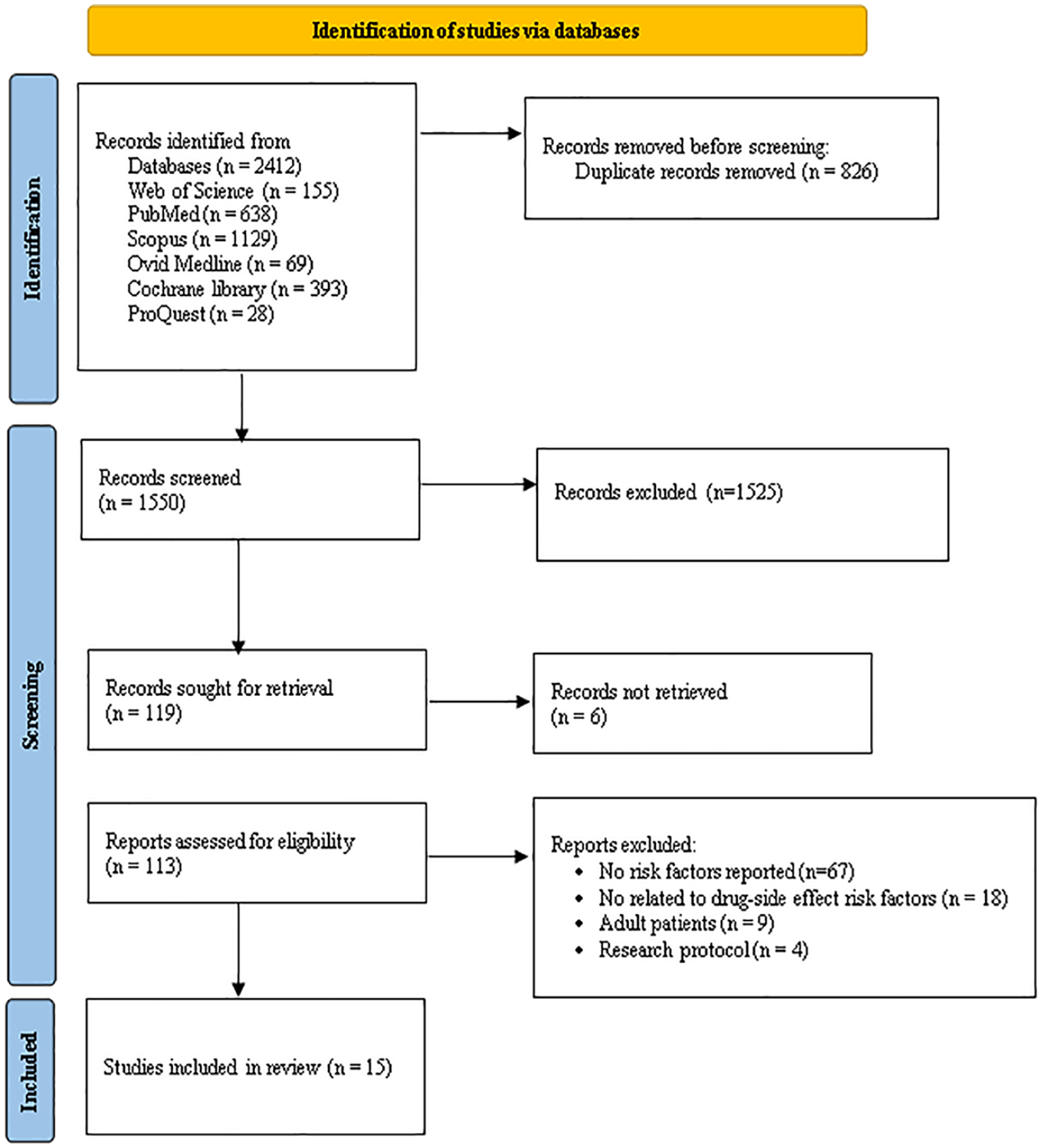
Selecting papers based on the PRISMA-ScR checklist. 30
Characteristics of the Selected Studies
The selected studies were conducted in China (n = 4),18,24,25,29 Brazil (n = 4),13,21,22,28 United Kingdom, 16 Japan, 27 India, 20 Italy, 26 Ethiopia, 19 Malaysia, 23 and Czech Republic. 17 The highest frequency of articles (n = 3) was published in 2020.25-27
About half of the studies were conducted as prospective observational studies (n = 8).16,19,20,23-25,27,28 Other studies were conducted using the retrospective cohort (n = 6)13,17,18,21,26,29 and systematic review approaches (n = 1). 22 Table 1 shows a summary of the articles included in the current research.
Summary of the Selected Articles.

Risk Factors Associated With Drug-Related Side Effects
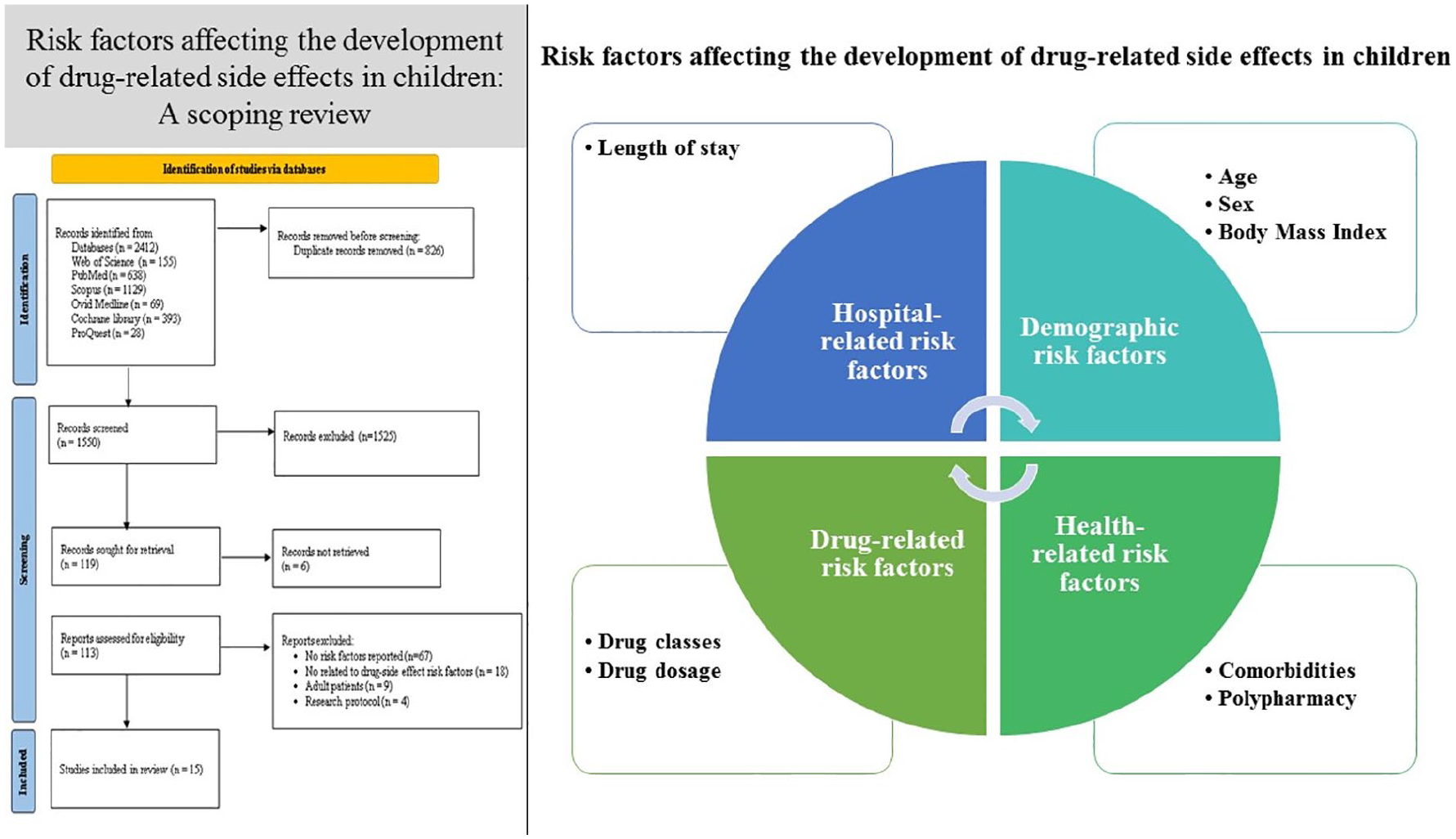
In general, the results showed that risk factors associated with drug-related side effects in children included demographic factors (age and sex), health-related factors (body mass index (BMI), comorbidities and polypharmacy), hospital-related factors (length of stay), and drug-related factors (including specific drug classes and drug dosage).
Demographic risk factors
The results of about half of the studies (n = 8) showed that the low age of the children is one of the risk factors for drug-related side effects.13,16,19,20,22,23,26,29 Silva et al 13 showed that children under 48 months of age were more likely to be at risk of drug-related side effects in the ICU. Similarly, other studies showed that the low age of patients admitted to the emergency department26,28 and inpatient units16,19,22,23,29 was significantly associated with drug-related side effects. Three studies also identified sex as a risk factor for drug-related side effects.17,20,22 Although the results of 2 studies indicated that male patients experienced drug-related side effects more frequently than females.20,22 Langerová et al 17 demonstrated that females were more significantly affected by drug-related side effects than males.
Health-related risk factors
The results of several studies showed that some diseases in children may have more significant impact on drug-related side effe-cts.16,17,19,22,25,27-29 Children with cancer,16,17,22,27 infectious diseases, 17 mental and behavioral diseases, 27 circulatory system disorders, 27 neurological disorders, 28 skin, 28 and respiratory diseases 28 may experience drug-related side effects more frequently due to the medications used for treating these conditions. The results of the study by Yu et al 29 also showed that an increase in the number of comorbidities is significantly related to the increase in the risk of drug-related side effects in children. Most studies also reported a significant association between polypharmacy and the risk of drug-related side effects due to the increase in the number of comorbidities in children and the subsequent use of multiple drugs.13,16,20,22-24,27,29 Furthermore, underweight children (low BMI) were found to be at a higher risk of drug-related side effects, as shown in the study by Yu et al. 29
Hospital-related risk factors
According to the results, an increase in the length of stay of children in hospital is significantly related to the increase in the frequency of drug-related side effects.19,24,25,29 Eshetie et al 19 showed that children who were hospitalized in the pediatric ward for more than 23 days were more likely to be exposed to drug-related side effects. Other studies showed that staying in the pediatric ward longer than 10 days may increase the likelihood of drug-related side effects.24,29 Liu et als’ 25 study indicated that risk of drug-related side effects in Chinese pediatric inpatients increased by 20.8% for each additional hospital stay day.
Drug-related risk factors
The results of some studies showed that the use of some commonly prescribed medications for children is more likely to result in drug-related side effects.16,19,21-24 There is a significant association between the use of antihistamines and antiallergic drugs, 19 central nervous system drugs, 19 endocrine drugs, 19 antiepileptic drugs, 23 antibacterial drugs 24 and general anesthetics,16,22 and the occurrence of drug-related side effects in children. In addition, the results of some studies showed that drug dosage effectively increases the risk of drug-related side effects in children.24,29 Andrade et al 21 showed that a history of drug-related side effects in children increases the likelihood of the side effect recurrence.
Synthesis
In general, it can be said that various risk factors may cause drug-related side effects in children. The most common risk factors associated with drug-related side effects were low age (n = 8, 20.5%), chronic comorbidities (n = 7, 17.9%), and polypharmacy (n = 8, 20.5%). It appears that the risk factors associated with drug-related side effects are interrelated and the presence of >1 risk factor may increase the likelihood of drug-related side effect. However, the weight of these factors might be different and dependent on the nature of the risk factors.
Discussion
This study aimed to investigate the risk factors associated with drug-related side effects in children. The results showed that demographic risk factors (low age and male sex), health-related risk factors (low BMI, chronic comorbidities, polypharmacy), hospital-related risk factors (length of hospitalization), and drug-related risk factors (type of drugs, dosage, history of drug-related side effects) were the most common risk factors associated with drug-related side effects in children.
As the results showed, there was a significant correlation between age and drug-related side effects in children. Indeed, the lower age of children in inpatient 31 and outpatient 32 departments can increase the likelihood of drug-related side effects. While Kunac et al 31 showed that the older age of hospitalized children favors the occurrence of drug-related side effects, Silva et al demonstrated that children <48 months old and hospitalized in the ICU were at a higher risk of drug-related side effects. Generally, some studies showed that children are at a lower risk of drug-related side effects compared to the adults and the elderly.32,33 Sex is another potential factor influencing the occurrence of drug-related side effects.34-36 Jung-Poppe et al 36 found that females were more likely at risk of drug-related side effects in inpatient, outpatient, and emergency departments.
The results of the present study showed that factors related to the health status of children, like using different drugs for treating diseases can significantly increase the likelihood of drug-related side effects. The results of other studies also showed that comorbidities can increase the frequency of drug-related side effects in adults.34,36,37 In these studies, neurological and mental health diseases, cardiovascular diseases, and type 2 diabetes were reported to be more prone to drug-related side effects,36,37 but the results of current research showed that children with cancer were more affected by drug-related side effects. Similarly, several studies highlighted polypharmacy as one of the most influential risk factors associated with drug-related side effects in adults33-35,37 and children.31,32 Low BMI was also found as a risk factor associated with drug-related side effects. 31
In addition to the health-related risk factors, most studies reported hospital-related factors, such as length of stay in hospitals as influential risk factors. The likelihood of drug-related side effects is higher for patients who require special care and long-term hospitalization.31,36,38,39 Kunac et al reported that drug-related side effects are more observed among the pediatric surgical cases rather than the pediatric medical cases. In their study, all pediatric surgical cases, 80% of pediatric medical cases, and 30% of neonatal intensive care unit (NICU) cases experienced drug-related side effects, 15 days or more after admission. 31
The findings also showed that pharmaceutical factors such as using a particular drug class, drug dosage, and the prior history of previous drug-related side effects in children may increase the likelihood of drug-related side effects. According to the results, the use of antihistamines and antiallergic drugs, central nervous system drugs, endocrine drugs, antiepileptic drugs, antibacterial drugs, and drugs for general anesthesia may lead to drug-related side effects in children. The role of the drug dosage as a risk factor was reported in another study. 31 The results are also in line with the findings of several studies in which the history of previous drug-related side effects was reported as a risk factor for increasing the likelihood of the side effect recurrence.40,41 However, the incidence of drug-related side effects in children may vary depending on the drug prescription periods, interactions with other drugs, and disorders in the organs that affect the pharmacodynamics and pharmacokinetics of drugs.1,11,31,41
It is critical to acknowledge that the underlying disease can have a significant impact on a child’s susceptibility to drug-related side effects. Different diseases may interact with medications in unique ways, exacerbating the effect of known risk factors. 42 For example, children with underlying liver or kidney dysfunction may have altered drug metabolism or excretion, increasing their risk of side effects from medications that are typically metabolized by these organs. 43 Furthermore, the medications used to treat specific diseases may have inherent risk profiles. Some chemotherapy drugs used to treat cancer, for example, are well known to cause specific side effects even when no other risk factors are present. 44 The interaction between disease and medication necessitates a more stratified approach to risk assessment in children. 45 By considering the specific disease being treated, along with established risk factors like age, comorbidities, and polypharmacy, healthcare professionals can develop more targeted medication regimens and implement closer monitoring strategies for potential side effects. Future research should delve deeper into the interaction between disease and medication-related side effects in children.
The topic of the current research is continually evolving along with emerging fields like pharmacogenetics holding immense promise for refining the related knowledge. 46 Pharmacogenetics delves into how individual variations in a person’s genes influence their response to medications, encompassing genes responsible for drug metabolism, transport, or target interaction. 47 While still in its early stages in children, pharmacogenetic research has the potential to revolutionize pediatric medication safety, 48 as demonstrated by its implications for adults that variations in the CYP2D6 gene can influence a patient’s response to certain pain medications. 49 Directly applying adult pharmacogenetic findings to children may not be suitable due to developmental differences; however, exploring this field in pediatrics holds immense promise. 50 Future studies tailored to investigate the role of pharmacogenetics in children’s drug response are imperative, potentially revealing how genetic variations interact with established risk factors like age, comorbidities, or polypharmacy to predict and prevent drug-related side effects. Integrating pharmacogenetics into the risk assessment process could usher in more personalized medication regimens and improved clinical outcomes for children, involving adjustments in medication dosages or selection based on a child’s unique genetic profile.46,48,50
Strengths and Limitations of the Study
This review presented an overview of the risk factors associated with drug-related side effects in children. The results can provide actionable insights for clinicians, researchers, and policymakers to improve pediatric medication safety practices. Moreover, by acknowledging the potential for variations in risk factors based on study design, population characteristics (age groups, specific diseases), and drug types, this review paves the way for future research to explore these nuances and refine our understanding of risk factors in specific contexts. By identifying knowledge gaps, such as the need for quantification and research in specific populations, this review guides future research efforts to improve medication safety for children.
However, this study had some limitations. First, only studies published in English were considered. There might be articles published in languages other than English that were not included in this study, mainly due to time and resource constraints. In addition, some studies were excluded, as access to their full texts was not possible, even after contacting the corresponding author. Moreover, the heterogeneity of the included studies, stemming from diverse methodologies and populations, may have influenced the synthesis and interpretation of the findings. Furthermore, due to the nature of the current study, we did not assess the quality of the included studies. In fact, the primary aim was to map the existing evidence rather than critically evaluate the individual studies. Future research could improve credibility by applying systematic review methodologies.
Conclusion
This study investigated the risk factors associated with drug-related side effects in children. This review provided insights into a variety of risk factors, including demographic, health, hospital, and drug-related factors. According to the results, most of the risk factors associated with drug-related side effects might be the same in adults and children; however, their impact might be different in these 2 groups. Overall, research about the impact of these factors, particularly in children, may lead to gaining a better understanding of patient safety. Future studies should focus on identifying other risk factors that influence drug-related side effects in children and include more details about the weight and impact of these factors by considering different characteristics of children.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X241273171 – Supplemental material for Risk Factors Associated With Drug-Related Side Effects in Children: A Scoping Review
Supplemental material, sj-docx-1-gph-10.1177_2333794X241273171 for Risk Factors Associated With Drug-Related Side Effects in Children: A Scoping Review by Esmaeel Toni, Haleh Ayatollahi, Reza Abbaszadeh and Alireza Fotuhi Siahpirani in Global Pediatric Health
Supplemental Material
sj-docx-2-gph-10.1177_2333794X241273171 – Supplemental material for Risk Factors Associated With Drug-Related Side Effects in Children: A Scoping Review
Supplemental material, sj-docx-2-gph-10.1177_2333794X241273171 for Risk Factors Associated With Drug-Related Side Effects in Children: A Scoping Review by Esmaeel Toni, Haleh Ayatollahi, Reza Abbaszadeh and Alireza Fotuhi Siahpirani in Global Pediatric Health
Footnotes
Author Contributions
ET and HA conceptualized the research. ET conducted the research and drafted the manuscript. HA supervised the study and commented on the manuscript. RA and AFS participated in the critical review of the manuscript. All authors reviewed the manuscript and declared their final approval for publication.
Data Availability
The data presented in this study are available upon request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded and supported by the Health Management and Economics Research Center, Health Management Research Institute, Iran University of Medical Sciences, Tehran, Iran (1402-2-113-26934).
Ethical Considerations
This study was performed in line with the principles of the declaration of Helsinki. Approval was granted by Iran University of Medical Sciences (IR.IUMS.REC.1401.1007).
Consent to Participate
Not applicable
Consent for Publication
All authors reviewed the manuscript and declared their final approval for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
