Abstract
Introduction
Lead has been identified as a public health concern in Africa as its widespread use has resulted in extensive environmental pollution and significant health problems. 1 Childhood exposure to lead from recycling of used lead-acid batteries (ULAB) in unregulated environments is an important cause of lead toxicity especially in low and middle-income countries. 2 Children are most vulnerable to the effects of environmental lead pollution due to immature organs resulting in health effects including impaired cognitive function and anemia.3,4 The Centres for Disease Control and Prevention previously considered a blood lead level (BLL) > 10 μg/dL in children as the threshold for intervention. 5 However, current guidelines recommend that for children with BLLs ≥ 5 μg/dL the source(s) of lead exposure should be identified and appropriate action taken to reduce and terminate exposure. 6
In Kenya, a study carried out to determine environmental lead contamination due to recycling of ULABs in informal settlements in Nairobi (including Kariobangi where the current study was conducted) found widespread soil and house dust lead contamination with values as high as 58,194 µg/ft 2 compared to the US Environmental Protection Agency (EPA) standard of 40 µg/ft 2 . 7 This study did not measure BLLs but extrapolated what the levels would be in children residing in the study area using an Integrated Exposure Uptake Biokinetic (IEUBK) model and estimated that 94.0% to 99.7% of the children would have BLLs above 10 µg/dL assuming that they consumed 500 mg of lead contaminated soil daily. 7 A study carried out in an informal settlement called Uhuru Owino located close to a lead smelting factory in Mombasa, Kenya found that 48% of children had BLLs > 5 µg/dL. 8 Uhuru Owino has a history of environmental pollution related to release of untreated waste from the lead smelting factory. However, the health impact of this pollution on the population residing in the area is yet to be published.
Several studies have revealed an association between elevated BLLs and iron deficiency. Lead inhibits 3 enzymes necessary for heme synthesis while low levels of serum iron stimulate intestinal absorption of lead.9,10 Increasing BLLs have been associated with reduction in both blood hemoglobin and iron stores hence the association between increasing BLLs and iron deficiency anemia (IDA).9,11 Children are not only vulnerable to the health effects of environmental lead contamination but are also at an increased risk of IDA due to physiological demands associated with rapid growth. 12 It is therefore necessary, when evaluating BLLs in children, to assess for IDA to better mitigate the impact of lead toxicity in this vulnerable population. 11
The diagnosis of IDA is often wrought with challenges due to inherent limitations of existing diagnostic tests. Bone marrow examination is invasive and not practical for routine evaluation of iron status while serum ferritin, the preferred alternative according to WHO, is spuriously elevated during inflammation. 13 Red blood cell indices such as mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH) and mean corpuscular hemoglobin concentration (MCHC) can be used to screen for iron deficiency.14,15 As these parameters are readily available as part of a complete blood count, they may be useful in the evaluation of iron deficiency especially in settings where direct assessment of iron stores isn’t feasible.
We set out to determine BLLs in children residing in an informal settlement in Nairobi where a previous study had shown high levels of environmental contamination due to recycling of ULABs. 7 We further sought to determine the association between BLLs, serum ferritin and red blood cell (RBC) indices in children with and without anemia.
Methodology
This was a descriptive study conducted in an informal settlement in Kariobangi, Nairobi County.
Study Population
Children aged 6 months to 7 years were recruited into the study from a church located in the vicinity of the ULABs recycling shacks. This age group was selected as it comprises the children most vulnerable to the neurological effects of lead. Recruitment was restricted to those living within a 2 km radius from the recycling area. This distance was determined subjectively as the distance covered in 30 minutes of a brisk walk. Initial recruitment of the first 23 participants started in March 2020 but had to be suspended for 2 years due to government restrictions on movement and gatherings related to the SARS-COV-2 pandemic. The study resumed in October 2022 and was completed in December 2022.
Informed Consent
Written informed consent was obtained from the children’s legal guardian after which a questionnaire (Supplemental File) was filled to collect information on potential lead exposure.
Sample Collection and Analysis
Venous blood samples were collected in evacuation tubes (BD Vacutainer® Blood Collection Tube, US) for complete blood count (CBC), ferritin and lead analysis. Lead analysis was done on ethylene diamine tetra acetic acid (EDTA) whole blood samples collected in special tubes for trace element analysis. CBC samples were transported at 4°C to 8°C to the AKUHN main laboratory for analysis within 12 hours of sample collection. All other samples were transported at ambient temperature. CBC analysis was done on a Sysmex XN analyzer, ferritin on a Siemens Atellica® IM analyzer and lead testing was outsourced to a referral laboratory (Metropolis Labs, India) where analysis was done using an inductively coupled plasma mass spectroscopy method. We provided the results for all the laboratory tests done to the parents and/or guardians of the children with elaborate explanations given by a pediatric hematologist on what the results meant. Further, appropriate interventions for those with elevated BLLs were given with the primary intervention being advice on moving away from the ULAB recycling areas. For those identified as having IDA, iron supplementation was provided for 3 months at an appropriate dose with the guardians advised to visit their preferred healthcare provider for follow up. Children found to have BLLs above 10 μg/dL and with neurological symptoms were referred to a pediatric neurologist for further evaluation and management. Participant confidentiality was maintained by using a unique study identification number and limiting access to study documents to the study investigators. All questionnaires and consent forms were kept in a secure locker that could only be accessed by the principal investigator.
Ethical Consideration
Ethical approval for the study was obtained from the Aga Khan University Health Research Ethics committee, Nairobi, Kenya (2017/REC-101) and from the National Commission of Science, Technology and Innovation based in Nairobi, Kenya (845832). Written informed consent was obtained from the parents and/or guardians of all participants prior to enrollment in the study. This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Data Analysis
A sample size of 195 was determined to be sufficient using the single proportion formula,
16
assuming that 90% of children would have BLLs above 45 μg/dL as extrapolated by an environmental study carried out in the study area,
7
a confidence level of 98% and a margin of error of 5%. Descriptive statistics were used to summarize participant characteristics and laboratory data. The proportion of children with BLLs above 5 and 10 µg/dL was presented as a percentage with corresponding 95% confidence intervals (CIs). Exploratory analysis to determine the relationship between BLLs and ferritin was done using Spearman’s correlation coefficient. The relationship between log transformed BLLs and relevant CBC parameters while controlling for ferritin was determined using partial correlation analysis. Independent-samples median test was used to compare medians for various CBC parameters and BLLs between those with and without anemia. The specific parameters of interest were RBC count, red cell distribution width (RDW), mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH) and mean corpuscular hemoglobin concentration (MCHC). Children with ferritin levels < 12 µg/L were considered iron deficient and those with a hemoglobin < 11 g/dL were considered anemic as per WHO recommendations.13,17 IDA was thus defined as a hemoglobin < 11 g/dL and a ferritin < 12 µg/L. The same analysis was carried out using a cut-off of 30 µg/L as recommended by WHO for children with infection or inflammation.
13
A
Results
A total of 215 children were recruited and had blood samples collected for analysis. Of these, 2 were subsequently excluded from the analysis due to incomplete laboratory data. Subsequent analysis was done using data from 213 children among whom were 114 (53.5%) females. The study participant characteristics are summarized in Table 1.
Characteristics of Study Participants.

Only 2 (0.9%, 95% CI: 0.1-3.4) children had BLLs greater than 10 μg/dL with values of 10.96 and 24.38 μg/dL. Seventeen (8.0%, 95% CI: 4.7-12.5) had BLLs ≥ 5 μg/dL. The 97.5th percentile value was 7.39 µg/dL. Figure 1 shows the distribution of BLLs according to sex.
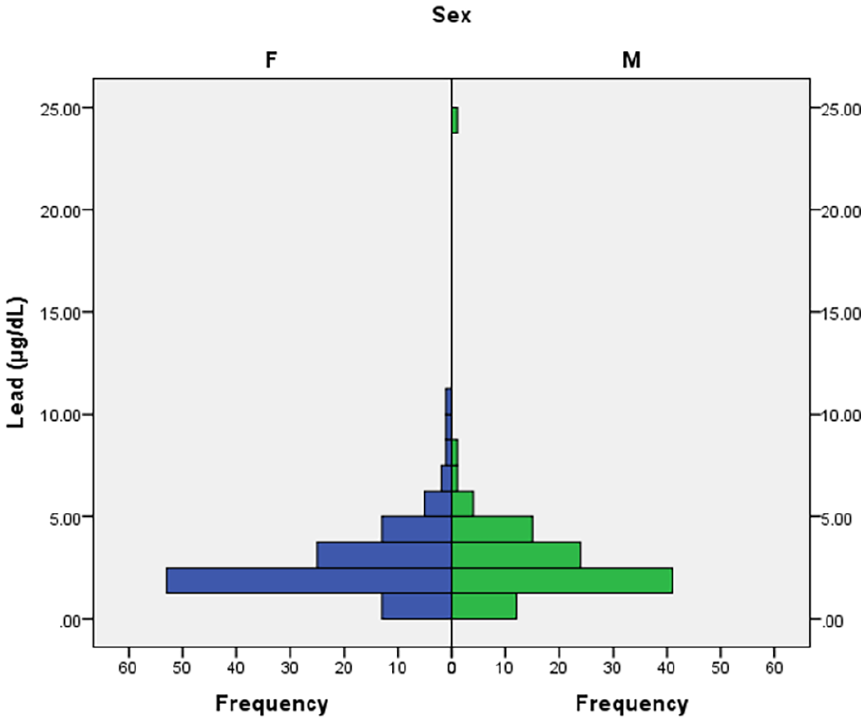
Blood lead levels distribution by sex.
There was a weak negative correlation between BLLs and ferritin (
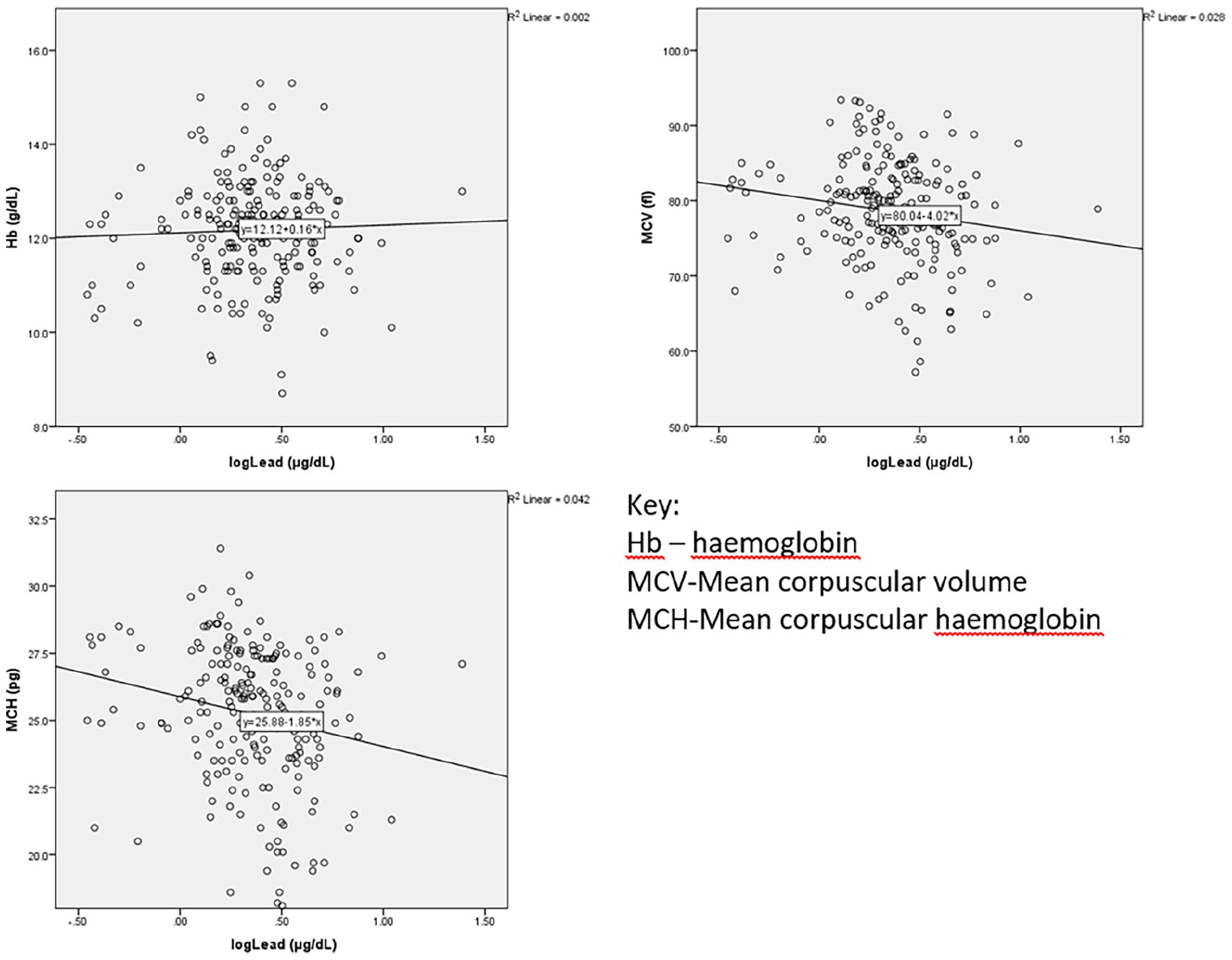
Association between log blood lead level and hemoglobin, MCV, MCH.
The difference in median BLLs between those with and without anemia was not statistically significant regardless of whether one was iron deficient or not as shown in Figure 3. In children with ferritin levels ≥ 12 µg/L, there was a significant difference in RBC parameters between those with and without anemia. Those with anemia had lower MCV, MCH, MCHC and higher RDW as shown in Figure 3. For those with ferritin < 12 µg/L, the only significant difference was in MCH where those with anemia had a lower median value.

Association between ferritin, anemia, lead, and RBC indices.
Nineteen (8.9%) children had ferritin levels below 12 µg/L and 10 (4.7%) children had IDA as shown in Table 2. Using a ferritin cut-off of 30 µg/L, IDA was 10.3% as shown in Table 2.
Ferritin and Hemoglobin Levels.

Discussion
Children in Africa continue to be exposed to lead from several sources including mining, cottage industries, petrol, paint, battery manufacture and recycling as highlighted in a 2015 WHO publication on lead exposure in African children. 18 As of 2011, it was estimated that Africa had lost up to US$ 134.7 billion (4.05% of Gross Domestic Product) as a result of early childhood lead exposure which impacts on lifetime economic productivity. 19 We determined the prevalence of BLLs ≥ 10 μg/dL in an environment known to have lead contamination to be 0.9% which was much lower than the 99.9% that had been predicted using the IEUBK model in a previous study. 7 However, biological equivalents and reference values for lead in blood are lacking and neurodevelopmental health effects have been reported even at BLLs below 5 μg/dL.20-22 Nigg et al 23 found reduced cognitive function and increased incidence of attention-related and problem behaviors in children aged 6 to 17 years of age with BLLs < 5 μg/dL.
The current study’s 97.5th percentile BLL of 7.4 μg/dL is higher than what was obtained from a similar population in the US. 24 The difference is possibly a consequence of the unregulated recycling of ULABs in the study setting given the previous finding of high levels of lead in the environment. 7 However, the median BLL of 2.3 μg/dL is lower than the mean BLL of 7.4 μg/dL found in children living near a battery recycling and smelting factory in an informal settlement in Mombasa, Kenya. 8 Seventeen (8%) children had levels above 5 μg/dL with 1 having neurological symptoms requiring further evaluation. With progressive exposure, these children are at risk of lead associated neurodevelopmental impairment, which could result in significant economic impact to them and the community. Increase in BLLs has been associated with decline in intelligence quotient scores at 3 and 5 years of age, with this decline being greater for those with BLLs below 10 μg/dL. 4 There is therefore a need for concerted efforts to regulate recycling of ULABs to prevent unnecessary exposure.
There was a weak negative correlation between BLLs and ferritin. Lead interferes with heme biosynthesis by inhibiting δ-aminolaevulinic acid dehydratase (δ-ALAD) enzyme which eventually results in a microcytic hypochromic picture. 25 We found a weak negative correlation between BLLs and both MCV and MCH even after controlling for ferritin which is in keeping with what has been found in other studies.9,26 The positive correlation between BLLs and RBC count has also been reported from a similar study carried out in China where increasing BLLs was associated with an increase in RBC count in children with BLLs < 10 μg/dL. 26 The mechanism underlying this observation might be similar to the relative erythrocytosis seen in patients with thalassemia trait which also causes microcytosis and hypochromia. 27
The association between BLLs and iron status has been shown to be dependent on the stage of iron depletion with higher BLLs seen in the stage of IDA. 28 Decline in hemoglobin in the setting of iron deficiency is the last stage and is often preceded by a decline in markers of iron status such as ferritin as well as changes in RBC indices such as MCV and MCH. 29 The absence of a correlation between BLLs and hemoglobin in this study might be due to the low prevalence of IDA given that adequacy of iron stores can modify this effect. 28 Hegazy et al 30 found a negative correlation between BLLs and ferritin, as well as between BLLs and hemoglobin in children attending pediatric clinics in Egypt but this was in a setting where the prevalence of IDA was 50%. Iron supplementation is therefore important when managing cases of elevated BLLs especially in those with IDA. 28
The prevalence of IDA was lower than what we expected given that the study participants were children largely drawn from an informal settlement where it was assumed that malnutrition would be prevalent. Increasing the ferritin cut-off to 30 µg/L only increased the prevalence of IDA to 10.3% which is still much lower compared to what has been published from other African countries. Muriuki et al 31 found the prevalence of IDA using a ferritin cut-off of 12 µg/L to be 24.3% in a cohort of children aged less than 8 years recruited from a rural setting in Kilifi. Of note is that Kilifi is an area that is endemic to infectious diseases like malaria which could compromise the diagnostic accuracy of the WHO recommended ferritin cut-off of 12 µg/L to diagnose iron deficiency in the setting of acute inflammation. Based on the WHO definition of IDA, the prevalence in Africa has been shown to vary from as low as 21.7% in The Gambia to as high as 42.0% in South Africa. 31 The high altitude of Nairobi (>1700 m above sea level) may have masked true anemia given that increase in altitude is accompanied by a physiological increase in hemoglobin levels in order to compensate for relative hypoxia. 32
The determination of iron status in children being evaluated for possible lead toxicity is important as it guides clinical decision making on whether to give iron supplementation. We compared medians of various RBC indices between children with and without anemia and found that those with anemia and seemingly adequate iron stores (ferritin ≥ 12 µg/L) had lower MCV, MCH, MCHC, and higher RDW. This pattern of RBC indices derangement is suggestive of IDA and may be used to determine the need for iron supplementation in otherwise healthy children presenting with anemia but normal ferritin levels.
Limitations
Our study has several limitations. We collected data over a period of more than 2-years due to the COVID-19 pandemic which necessitated a 2-year suspension of the study in line with government and institutional recommendations. The extent to which this impacted on the demographics of the study population and ULAB recycling practices is unknown. Another limitation is that we waited for study participants to avail themselves for recruitment. A better strategy would have been to randomize households and carry out recruitment at a household level to ensure that every child in the study area had an equal chance of being recruited. However, this was not done due to safety concerns for the research staff which informed the decision to centralize recruitment in a church compound that was considered safe. By targeting participants that presented at a particular location, it is possible they were not representative of the entire population. We did not recruit a reference pediatric population to enable us compare BLLs and determine whether children residing in the study area have relatively higher BLLs compared to those living in areas not known to have environmental lead contamination. Our sample size calculation assumed that 90% of study participants would have BLLs > 45 μg/dL based on a previous study carried out in the same area that had extrapolated BLLs using environmental lead levels. Based on our findings, our assumption was wrong as the highest BLL was 24.38 μg/dL with only 8% of children having BLLs > 5 μg/dL. Lastly, we used a subjective measure to determine whether the recruits resided within 2 km of the ULAB recycling sites. It is possible that some of them resided in areas that were outside the intended study area and would invariably have low BLLs.
Conclusion
Our study confirms the presence of above-recommended lead levels in children residing in an informal settlement that may be an indicator of existence of other toxic waste materials. Relevant authorities should consider legislating laws on safe disposal of used motor vehicle batteries to protect the population from toxic effects. There was, however, no correlation between lead exposure and anemia.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X241263162 – Supplemental material for Assessment of Ferritin and Lead Levels in Children Living in an Informal Urban Settlement in Kenya
Supplemental material, sj-docx-1-gph-10.1177_2333794X241263162 for Assessment of Ferritin and Lead Levels in Children Living in an Informal Urban Settlement in Kenya by Geoffrey Omuse, Daniel Maina, Maureene Ondayo, Syama Sinuff, Stanley Onyango, Alex Mugo, Pauline Samia and William Macharia in Global Pediatric Health
Footnotes
Acknowledgements
None.
Author Contributions
GO: contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. DM: contributed to conception and design; contributed to acquisition; critically revised manuscript. MO: contributed to design; critically revised manuscript. SS: contributed to conception and design; contributed to acquisition; critically revised manuscript. SO: contributed to analysis and interpretation; critically revised manuscript. AM: contributed to analysis and interpretation; critically revised manuscript. PS: contributed to design; critically revised manuscript. WM: contributed to design; critically revised manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
