Abstract
Objectives. This study aimed to find the association between clinical characteristics, cytogenetics, and post-induction outcomes of childhood acute lymphoblastic leukemia. Methods. The study was conducted at the Indus Hospital in Karachi. Initial total leukocyte count (TLC), cytogenetics, CNS status, and post-induction remission status were recorded. Results. Out of 108 children diagnosed with ALL, 66 (61.1%) were male and 42 (38.9%) were female. The majority 90 (83.3%) had B-ALL. CNS1 status was observed in 76 (84.4%) B-ALL and 18 (88.9%) T-ALL. All T-ALL and 89 (98.8%) B-ALL achieved remission post-induction. In B-ALL, 50 (55.5%) had a normal diploid karyotype, and 22 (24.4%) had numerical abnormalities. No typical gene rearrangement was observed in 66 (73.3%), 11 (12.2%) had BCR::ABL1, 10 (11.1%) had ETV6::RUNX1 and 3 (3.3%) KMT2A on FISH. No significant difference was observed between cytogenetics and clinical characteristics (P > .05). Conclusion. The study provides valuable data on childhood acute lymphoblastic leukemia in the Pakistani population.
Introduction
Childhood Acute Lymphoblastic Leukemia (cALL) is an aggressive malignancy of lymphoid cells most prevalent in children, with 75% of cases occurring under the age of 15 years. B-ALL is the most common, whereas T-ALL is infrequent with a dismal prognosis. In 2016, the World Health Organization (WHO) further classified B-ALL with recurrent genetic abnormalities into nine categories and those without recurrent genetic abnormalities as not otherwise specified (NOS). There is no subclassification of T-ALL based on genetic abnormalities. 1
Three varieties of recurrent genetic aberration in ALL are aneuploidy, balanced chromosomal translocations, and molecular genetic abnormalities. 2 The first two are part of the diagnostic workup, directly impacting therapeutic decisions and clinical outcomes. The last one aims to discover new molecular targets for a precise understanding of biology and eventually translate into clinical practice for the best management. 3 The risk stratification of cALL into high-risk (HR) and standard-risk (SR) depends on precise prognostic factors. It is essential for the choice of induction regimen. 4 It has been reported that 50% of patients of B-ALL have normal findings on cytogenetic analyses and are classified under standard risk (SR). 5 T-ALL falls under HR. 6 Patients with less than 0.01% leukemic cells at the end of induction have an excellent treatment outcome. In contrast, high levels (ie, ≥1%) of minimal residual disease (MRD) are at higher risk of relapse. 7 They are considered for intensive chemotherapy, stem cell transplantation, or targeted therapy. 8
In the developed world, survival rates of cALL have improved dramatically. However, in underdeveloped nations, it differs due to delays in diagnosis, financial constraints, and access to treatment. 9 In Pakistan, cALL is the second most common cancer and contributes to 17.9% of all cancers. 10 Only a few centers have complete diagnostic facilities. Hence, data is scarce and inconsistent. We hypothesize the clinical features and cytogenetics of cALL in our setting are similar to other low/middle-income countries. The study aims to investigate the association between clinical characteristics including gender, age, TLC at diagnosis, immunophenotype, CNS status, post-induction outcome, risk groups, and cytogenetic abnormalities of cALL
Methods
Indus Hospital is a privately run tertiary care hospital in Karachi, Pakistan with an established and fully equipped pediatric oncology ward providing free-of-cost services. It receives patients from all over Pakistan.
Study Participants
OpenEpi was used for sample size calculation. The sample size was calculated on a prevalence of 17.9% with a confidence interval of 95% and a margin of error of 7.5%. 10
Children diagnosed with ALL were included in the study. The inclusion criteria were: (a) B-ALL or T-ALL, (b) age 1 to 16 years, and (c) received induction therapy at the study site. Exclusion criteria were (a) infantile leukemia, (b) acute leukemia other than ALL, (c) children receiving treatment for ALL, (d) relapsed ALL, (e) non-consenting patients, and (f) incomplete workup/loss to follow-up.
Study Design Tools and Collection of Data
A single-center, cross-sectional study, was conducted at the pediatric hematology-oncology unit of The Indus Hospital, Karachi, from Jan 2019 to Dec 2022. Total leukocyte count (TLC) at presentation, Cytogenetics, CNS status, FISH, and post-induction remission status were recorded. Cytogenetics of T-ALL was not done due to institutional policy. TLC was analyzed on the hematological analyzer Coulter DxH 900 (Beckman Coulter, USA). Initial risk stratification was determined with the patient’s age and TLC following the National Cancer Institute (NCI) risk stratification criteria; HR constitutes ≥10 years of age, or TLC ≥ 50 × 109/L and SR includes patients ≤10 years and with a TLC count ≤50 × 109/L. Chemotherapy was given according to the modified Berlin-Frankfurt-Munster (BFM) protocol. For B-ALL, Fluorescence in situ Hybridisation (FISH) was carried out to identify recurrent genetic abnormalities BCR::ABL1, KM2TA, and ETV6::RUNX1. Chromosome analysis was done using the conventional G-banding technique. Their results were interpreted according to the international system of cytogenetic nomenclature (ISCN).11,12 CSF cytology and D/R were used for the assessment of CNS status. Slides were prepared by cytospin and examined by a hematopathologist for detailed reporting of morphology. CNS status was classified as CNS1: absence of blasts on cytospin preparation, regardless of the TLC, CNS2: the presence of <5/µl TLC count with the presence of blasts and CNS3: >5/µl TLC with the presence of blasts, traumatic tap with >10 red blood cells/µl and presence of blasts and/or signs of CNS leukemia. On day 35, bone marrow aspirate was assessed morphologically for induction response. Complete hematological remission is defined as absolute neutrophil count (ANC) 1.0 × 109/L; platelet count 100,000/µl, and bone marrow blast <5% after induction therapy. Based on all the above findings, patients were stratified into SR and HR according to the criteria in Table 1.
Criteria for Standard Risk and High Risk.
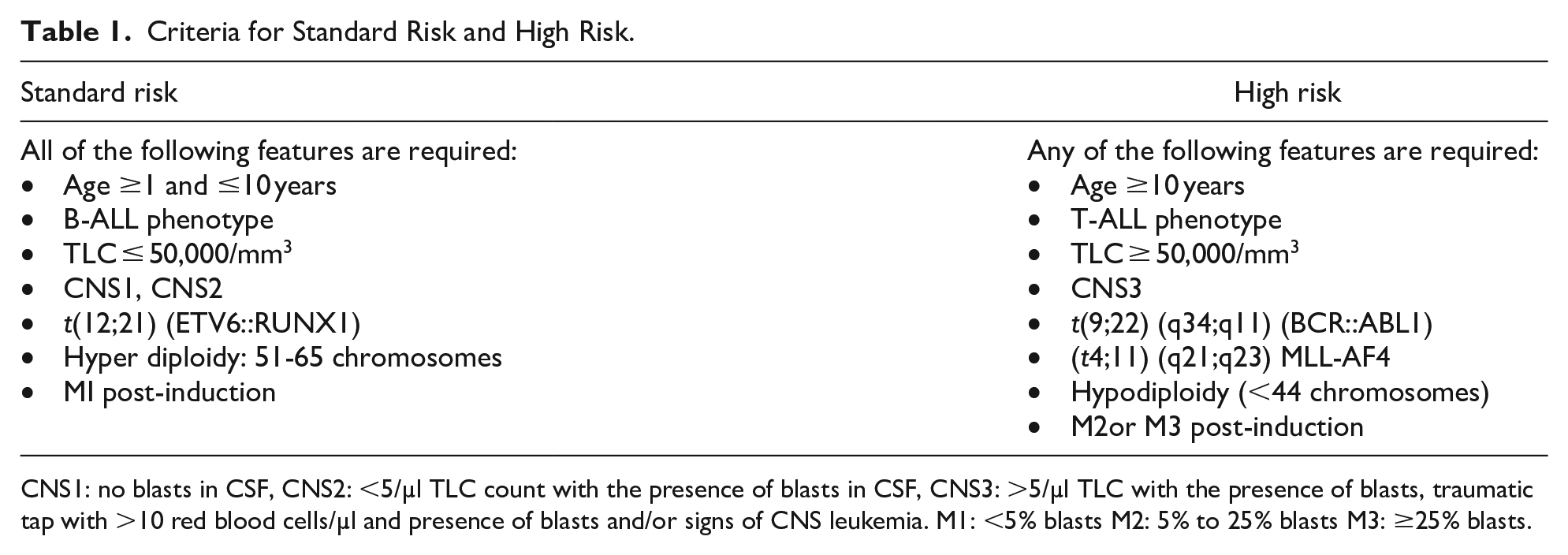
CNS1: no blasts in CSF, CNS2: <5/µl TLC count with the presence of blasts in CSF, CNS3: >5/µl TLC with the presence of blasts, traumatic tap with >10 red blood cells/µl and presence of blasts and/or signs of CNS leukemia. M1: <5% blasts M2: 5% to 25% blasts M3: ≥25% blasts.
Ethical Approval and Informed Consent
The study was approved by the Institutional Review Board of Indus Hospital (IRD_IRB_2018_09_001). Parents/or a guardian of all participants provided written informed consent prior to enrolment in the study. Assent was taken from older children whose parents/guardians gave written informed consent.
Data Analysis Procedure
Data was analyzed on SPSS Version 22. Quantitative data were presented as mean ± standard deviation (SD). Frequencies and percentages were reported for the qualitative variables and analyzed by chi-square or Fisher exact test. P < .05 was considered statistically significant.
Results
Patients’ Characteristics
A total of 108 children diagnosed with ALL met the inclusion criteria. The mean age of the children was 6.55 ± 4.06 years. Eighty-five (79%) of the patients were between 1 and 9 years, and 23 (21%) were between 10 and 16 years. Sixty-six (61.1%) were males and 42 (38.9%) were female. The male to female ratio was 1.6:1. On immunophenotype, 90 (83.3%) had B-ALL, and 18 (16.7%) had T-ALL. The mean TLC was 64.7 ± 114.7 × 109/L.
Association Between Age Groups and Clinical Characteristics of Children with ALL
In both age groups, there were more males than females [1-9 years: 51 (60%) and 15 (65.2%)] (Table 2). B-ALL was the most common immunophenotype in both age groups and was statistically significant [1-9 years: 75 (88.2%) and 10-16 years: 15 (65.2%) P < .0001]. The mean TLC of 10 to 16 years was higher than 1 to 9 years (89.67 ± 151.6 × 109/L vs 58 ± 102.5 × 109/L]. Most 1 to 9 years and 10 to 16 years had TLC ≤ 50 × 109/L [63 (74.1%) and 15 (65.2%) respectively]. CNS1 was higher in both age groups [1-9 years: 73 (85.9%) vs 19 (82.6%) respectively]. CNS3 was higher in 10 to 6 years 3 (13%). Majority of 1 to 9 years had SR 53 (62.4%) and 10 to 16 years had HR 15 (65.2%).
Association Between Age Groups of Children with ALL and Clinical Characteristics.
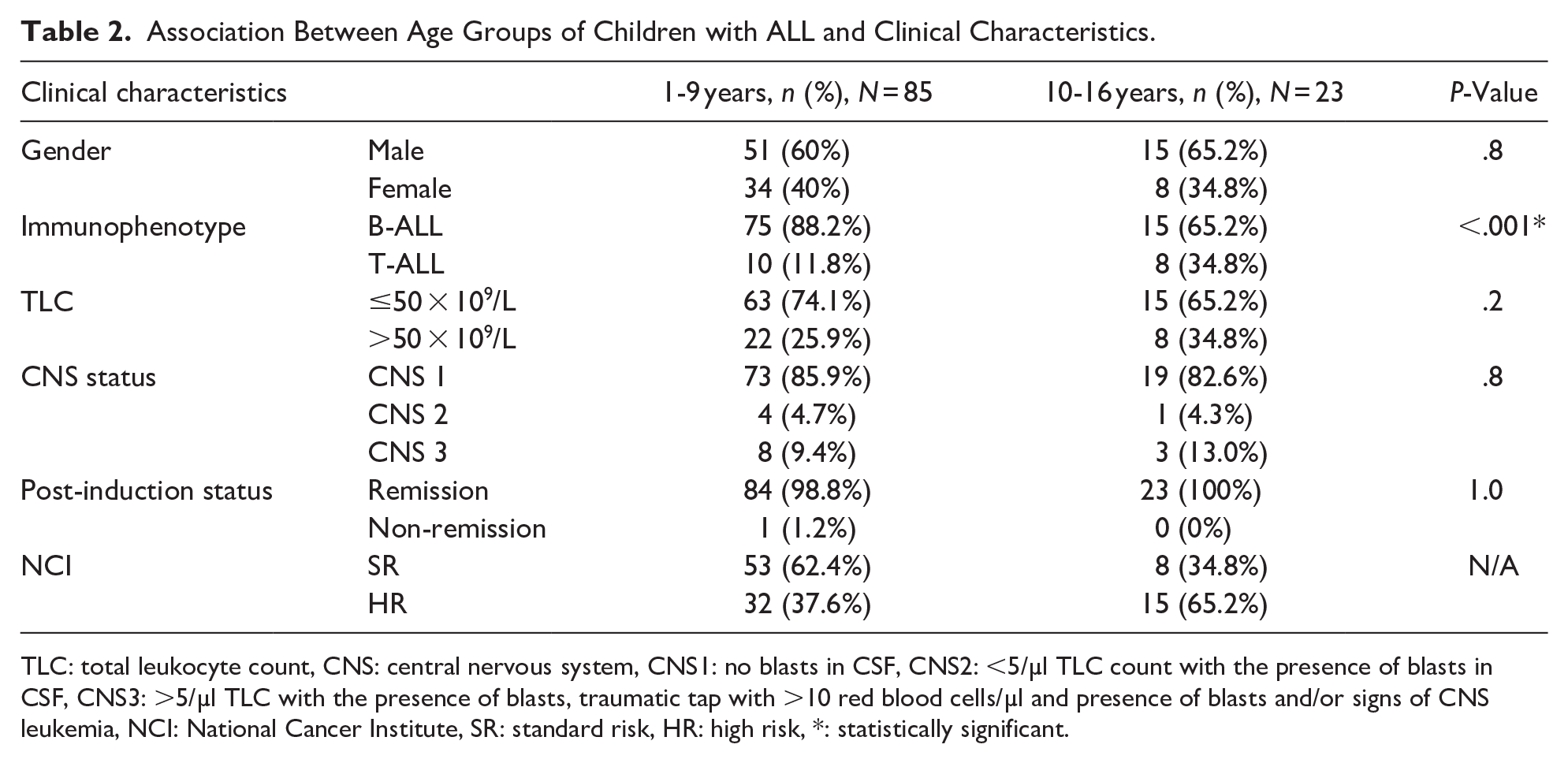
TLC: total leukocyte count, CNS: central nervous system, CNS1: no blasts in CSF, CNS2: <5/µl TLC count with the presence of blasts in CSF, CNS3: >5/µl TLC with the presence of blasts, traumatic tap with >10 red blood cells/µl and presence of blasts and/or signs of CNS leukemia, NCI: National Cancer Institute, SR: standard risk, HR: high risk, *: statistically significant.
Association Between Immunophenotype and Clinical Characteristics of Children with ALL
The mean age of T-ALL was higher than B-ALL [9.67 ± 2.95 years vs 5.92 ± 3.97 years] (Table 3). B-ALL and was common in males, 56 (62.2%). The mean TLC of T-ALL was higher than B-ALL [194.8 ± 178.7 × 109/L vs 38.76 ± 74.9 × 109/L]. Most B-ALL 76 (84.4%) had TLC ≤ 50 × 109/L and T-ALL 16 (88.9%) had TLC ≥ 50 × 109/L. The association between TLC and immunophenotype was significant(P < .001). CNS 1 was common in both B-ALL 76 (84.4%) and T-ALL 18 (88.9%). CNS3 was higher in T-ALL 2 (11.1%). In B-ALL, 61 (67.8%) were in SR was and 29 (32.2%) in HR. All children with T-ALL 18 (100%) were in HR, according to Table 1 criteria.
Association Between Immunophenotype and Clinical Characteristics of Children with ALL.

WBC: white blood count, CNS: central nervous system, CNS1: no blasts in CSF, CNS2: <5/µl TLC count with the presence of blasts in CSF, CNS3: >5/µl TLC with the presence of blasts, traumatic tap with >10 red blood cells/µl and presence of blasts and/or signs of CNS leukemia, NCI: National cancer institute, SR: standard risk, HR: high risk, *: statistically significant, N/A: statistics not applicable.
Cytogenetics of Children with B-ALL
Fifty (56%) had a normal diploid karyotype and 22 (24%) had numerical abnormalities (Figures 1 and 2). Among numerical abnormalities, one case of trisomy 8 was found in a 10-year-old female with TLC of 17 × 109/L, CNS 1, no abnormalities on FISH, and in remission.
Sixty-six (73.3%) had no typical gene rearrangement on FISH for tested probes, 3 (3.3%) had KM2TA, 10 (11.1%) had ETV6::RUNX1, and 11 (12.2%) had BCR::ABL1.
Association Between Karyotype and Clinical Characteristics of Children with B-ALL
Normal diploid karyotype was found in most males 32 (64%) 1 to 9 years 41 (82%) TLC ≤ 50 × 109/L 43 (86%) and CNS1 43 (86%) (Table 4). No association was observed between karyotype and clinical characteristics.
Association Between Karyotype and Clinical Characteristics in Children with B-ALL.

TLC: total leukocyte count, CNS: central nervous system, CNS1: no blasts in CSF, CNS2: <5/µl TLC count with the presence of blasts in CSF, CNS3: >5/µl TLC with the presence of blasts, traumatic tap with >10 red blood cells/µl and presence of blasts and/or signs of CNS leukemia.
Association Between FISH and Clinical Characteristics of Children with B-ALL
None of the analyzed fusion genes were present in most males 42 (63.6%), 1 to 9 years 56 (84.2%) TLC ≤ 50 × 109/L 58 (87.9%) and CNS1 55 (83.3%) (Table 5). No association was observed between FISH and clinical characteristics.
Association Between FISH and Clinical Characteristics in Children with B-ALL.

TLC: total leukocyte count, CNS: central nervous system, CNS1: no blasts in CSF, CNS2: <5/µl TLC count with the presence of blasts in CSF, CNS3: >5/µl TLC with the presence of blasts, traumatic tap with >10 red blood cells/µl and presence of blasts and/or signs of CNS leukemia.
Discussion
In this study, most T-ALL 16 (88.9%) had hyperleukocytosis (Table 3). Furthermore, the mean age and initial TLC of T-ALL were higher than B-ALL. These findings are consistent with local and international literature.13-15 Age and initial TLC an independent risk factors in B-ALL but they have less impact on the prognosis of T-ALL due to its aggressive nature.16,17 10.2 to 19.2% cALL present with hyperleukocytosis (TLC ≥ 100 × 109/L) which can lead to early neurological, respiratory and metabolic complications and has worse long-term survival.18,19 Chainansamit found that the patients with hyperleukocytosis had a higher relapse rate than those without it. 20 Other studies by Kittivisuit et al and Dongyan et al found that hyperleukocytosis in cALL is associated with older age, T-cell immunophenotype, mediastinal mass, hepatomegaly, and splenomegaly.19,21
The male-to-female ratio was 1.6:1. The higher proportion of males compared to females has been reported in local and other Asian countries.22-24 Social reasons such as gender bias in seeking healthcare and preferential neglect for the female child due to resource constraints must be considered. However, boys still have inferior event-free survival (EFS) and overall survival (OS) as compared to girls according to a recent report by Children’s Oncology Group (COG). 25
A higher frequency of CNS leukemia was observed in the study (Tables 2 and 3) compared to other local literature by Fadoo et al 13 and Meeraj et al. 26 This is most likely due to delayed identification and referral. Another explanation is the delay in the first dose of intrathecal chemotherapy as it is administered on day 8 of induction as in our current protocol. 27
CNS3 was more common in the T-ALL and 10 to 16 years age group (Tables 2 and 3). Sirvent et al concluded that CNS3 is an adverse prognosis factor in cALL without cranial irradiation. 28 Additionally, T-ALL predisposes to CNS involvement. The proposed mechanism for CNS tropism is the enhanced expression of chemokines and adhesion molecules by T-cells in the cerebrospinal fluid (CSF), which facilitates their interaction with the choroid plexus and subarachnoid vessels.29,30 Different cytogenetic subtypes in ALL, such as E2A-PBX1, BCR::ABL1 and KMT2A are associated with a higher incidence of CNS leukemia. 31 CNS1 was the most frequent among all cytogenetic abnormalities (Tables 4 and 5) which is similar to studies from India by Chadha et al and Shawana et al from Palestine.32,33 New techniques for the detection of leukemic blasts in the CSF such as multicolor flow cytometry, PCR on cell-free DNA, and circulating microRNA are currently undertrial to further enhance detection. 33
Both FISH and karyotyping were done to analyze cytogenetic abnormalities in B-ALL. 55.5% had normal diploid karyotype and 73.3% had no findings on FISH. The most common chromosomal aberrations were hyperdiploidy 19 (21%) on karyotype and BCR::ABL1 11 (12.2%) on FISH (Figures 1 and 2). No statistical significance was observed between cytogenetics and clinical characteristics of B-ALL (Tables 4 and 5). These findings are similar to a study from Iran. 34

Frequency of karytype findings and breakup of numerical abnormalities.

Results of interphase FISH for ALL panels.
There is limited local research on the clinical implications of ALL’s cytogenetics. In a multi-institutional report on cALL by Fadoo et al, karyotype and FISH findings were consistent with our study although they analyzed selected samples with FISH due to cost constraints. 13 Khan et al found that 40% of patients with ALL had chromosomal abnormalities, including 17.7% hyperdiploidy, 8% near-tetraploidy and 13.1% t(9;22). However, they included both adults and children with ALL. 35 Sheikh et al reported 48.8% with cALL had an abnormal karyotype, with hyperdiploidy being the most common (13.4%) followed by t(9;22) (7.08%). 36 Although the sample size of these studies was larger than ours, only karyotyping was done and associated clinical characteristics and induction outcomes were not reported. Korejo and Rana et al found 30.6% and 19% hyperdiploidy respectively in cALL. Additionally, post-induction remission was significantly higher with favourable cytogenetics than the poor cytogenetics group.37,38 Niaz et al 39 also had similar findings as Korejo et al but they included adults with ALL. In the present study, 107/108 (99.1%) patients went into remission. This higher finding is attributed to the small sample size and inclusion of patients with a complete workup.
Out of 11 cases of BCR::ABL1 detected by FISH, four were concordant with karyotype. Discordance was 7/11 (63%). This signifies the simultaneous use of both techniques in the risk assessment of cALL. This marked discordance is due to the limited sample size. Chromosomal microarray (CMA) detects genomic gains and losses at a much higher resolution than G-banded karyotyping. In studies by Mitrakos et al and Chen et al, CMA detected chromosomal abnormalities in 81% and 77.8% of the cALL patients and adult ALL with normal karyotypes, respectively.40,41 Hence, CMA can detect significant cryptic submicroscopic variants. Another study by Brown et al found that RNA-sequencing identified rearrangements that were missed by FISH and karyotyping. 42
There was one case of isolated Trisomy 8 in B-ALL. So far, only a few patients with isolated trisomy 8 have been reported.43,44 In acute myeloid leukemia, trisomy 8 is an intermediate risk. Liz et al. found that trisomy 8 alters microRNA expression, fostering leukemogenesis, treatment response, and prognosis; this is yet to be explored in cALL.
The study provides valuable data on cALL in the Pakistani population. Complete diagnostic profiles including special testing like flow cytometry, FISH and cytogenetics are available in only a few centers therefore these data are very limited for our patients. Cytogenetic analysis was done with karyotyping and FISH to present the distribution and characteristics of chromosomal abnormalities of B-ALL. This is essential for assessing the risk group and determining the appropriate therapeutic strategy.
We acknowledge that there are several limitations to the study. The sample size was small. Therefore, some of the categories were merged like all numerical cytogenetic abnormalities presented as one group. Moreover, cytogenetic testing is not performed in T-ALL cases as it is not required for risk stratification and does not impact the protocol. Post-induction remission status was included based on the morphological assessment as flow cytometry data for the minimal residual disease was not included in the study. Lastly, the study was limited to the assessment of post-induction remission instead of long-term follow-up to study disease-free survival and overall survival.
Conclusion
Our study is based on the experience of a single institute with cALL. The majority of the patients were male, and B-ALL was the most common immunophenotype. In B-ALL, 61 (67.8%) were in SR was and 29 (32.2%) in HR. We observed a significant association between immunophenotype, age groups, and TLC. Hyperdiploidy was the most common cytogenetic abnormality, followed by BCR::ABL1. There were no statistically significant differences between clinical characteristics and cytogenetics of cALL. The findings reported are similar to local and international data from low-income countries.
cALL will continue to bank upon diagnostic testing to identify known and currently unknown genetic alterations indispensable for the management and clinical development of “more tailored, less toxic” precision medicine therapies.
Footnotes
Acknowledgements
We would like to thank all the children and their parents or guardians for participating in this study. Additionally, we express our gratitude to the staff of the Pediatric Oncology ward at Indus Hospital for their dedication, and to Ms. Fariha Anum for her statistical expertise.
Authors’ Note
Talat Mirza is also affiliated to Ziauddin College of Molecular Medicine.
Author Contribution
NT contributed to the acquisition, analysis, interpretation and drafting of the manuscript. SM and TM contributed to the conception and design and critically revised the manuscript. ZB and NM contributed to the acquisition, analysis, and interpretation and critically revised the manuscript. All authors approved the work to be published. They are responsible for the integrity and originality of the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
