Abstract
Objectives. To evaluate carotid artery intima-media thickness (CIMT) and lipid profile in children with epilepsy on long-term antiepileptic drug (AED) monotherapy. Methods. We included 60 children with epilepsy receiving valproate, carbamazepine, or levetiracetam monotherapy and 60 controls. A high-resolution B-mode ultrasound was used to measure (CIMT). Measurement of serum lipids was done. Results. Patients on valproate (0.44 ± 0.03, P ≤ .001), carbamazepine (0.43 ± 0.03with P ≤ .001), and levetiracetam (0.44 ± 0.02 with P ≤ .001) monotherapy showed significantly higher CIMT compared to controls. CIMT was correlated with age (P = .041, r = .112) AEDs{valproate (P = .005, r = .731), carbamazepine (P = .038, r = .365), and levetiracetam (P = .036, r = .155)}, duration of treatment (P = .001, r = .313), TC(P = .001, r = .192), TG (P = .014, r = .018), and LDL (P = .001, r = .219). HDL (P = .003, r = −.126). Seizure severity and Apo A1 were insignificantly involved. Conclusion. Long-term monotherapy with valproate, carbamazepine, and levetiracetam in epileptic children was associated with significant abnormalities in CIMT.
Introduction
Epilepsy is a common neurological disorder, and it affects over 50 million subjects all over the world. 1 The mainstay of treatment of children with epilepsy is the use of antiepileptic drugs (AEDs) as monotherapy or polytherapy. Older AEDs are Valproic acid (VPA), Carbamazepine (CBZ), Phenytoin, and, Phenobarbitone, and recent AEDs are Levetiracetam, Topiramate, Lamotrigine, and Oxcarbazepine. Carbamazepine, Valproate, Levetiracetam, and Lamotrigine are the most commonly used AED for monotherapy. 2
Most children with epilepsy require long-term antiepileptic drugs (AEDs). The prolonged use of AEDs may be associated with many adverse effects, including metabolic derangements, endocrinal disorders, organ toxicity, cognitive dysfunction, and psychiatric problems. 3 It was reported that chronic exposure to AEDs may play a role in the pathogenesis of atherosclerosis in patients with epilepsy by altering the lipid profile.4,5 The atherosclerosis process is characterized by remodeling of the arterial wall that may present as acute vascular events or pass unnoticed. The measurement of carotid artery intima-media thickness (CA-IMT) noninvasively by ultrasonography is considered a surrogate marker for the prediction of cardiovascular disease and atherosclerosis.6,7 Children on valproate therapy can develop adverse effects like an increase in body weight, elevated liver enzymes, abdominal pain, acute pancreatitis, and metabolic syndrome. Also, it affects the lipid profile by increasing the levels of serum leptin. Carbamazepine can affect lipid profile through enzyme induction.8-10 Levetiracetam can cause behavioral changes and psychotic disturbances.10-12 Changes in lipid profile and weight gain are the risk factors for the development of premature-onset cardiovascular diseases and atherosclerosis. The association between AEDs and CA-IMT remains controversial, with some authors reporting an increase in CA-IMT in epileptic patients on AEDs.5,13,14 However, other authors found no significant relationship between the use of AEDs and CA-IMT.15-17
We aimed to assess the association between CA-IMT and the use of long-term AEDs in epileptic children.
Patients and Methods
This prospective observational case-control study was conducted on 60 children diagnosed with epilepsy on AED monotherapy. The study was conducted in the pediatric department, at Benha University for over 1.5 years (from January 2021 to June 2022). Sixty healthy children of matched age and sex without underlying medical disorders served as a control group. Formal consent was obtained from the parents before enrollment into the study. Ethical clearance was obtained for the research study. Ethical guidelines of the 1964 declaration of Helsinki and its later amendments were followed.
Exclusion criteria:
- History of hepatic or renal disorder.
- Obese children, and those receiving drugs that affect lipid profile.
- Family history of obesity, atherosclerosis, cardiovascular or metabolic diseases.
- Poor compliance with medications or loss of follow up
The diagnosis of epilepsy was based on an abnormal electroencephalogram (EEG) at the time of diagnosis. All patients were receiving monotherapy, that is, Valproate, Carbamazepine, or Levetiracetam for at least 6 months duration. Children with epilepsy were classified according to the type of seizure and AED. 18
All Patients and controls were subjected to
(1) Detailed history taking with stress on age, sex, dietary history, type of convulsion (focal or generalized), type of treatment, duration of treatment, family history, response to treatment, last fits, frequency of seizure attacks, and the number of status epilepticus.
(2) Thorough Clinical examination and anthropometry: Body height and weight were measured in light clothes by a portable stadiometer. Body mass index (BMI) was calculated by dividing the weight by the square of the height (kg/m2) and plotted on the standard growth charts as per World Health Organization (WHO). BMI > 95th percentile is considered obese.
The Global Assessment of Severity of Epilepsy (GASE) Scale was also carried out for each epileptic child. 19
Laboratory investigations: Included measurement of serum lipids and liver function test.
They were measured by taking a 2 ml venous blood sample in the morning after overnight fasting (10-12 hours). Measurement of serum lipids included total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), apolipoprotein A1 (Apo A1), and triglyceride levels. They were measured using standard enzymatic methods by Boehringer Mannheim GmbH and a fully automatic analyzer. 20
Low-density lipoprotein cholesterol (LDL-C) and Very-low density lipoprotein cholesterol (VLDL-C) were calculated using the Friedewald et al formula (LDL-C = TC-(HDL-C+0.2TG and VLDL-C = TG/5). 21
TC (<170 mg/dL), TG (<90 mg/dL), HDL (>45 mg/dL), LDL (<110 mg/dL), VLDL (<30 mg/dL), and Apo A1 levels (>120 mg/dL) were taken as Standard reference values. 22
Measurement of CIMT
We used B-mode high-resolution ultrasonography to measure CIMT via a 5 to 7 MHz linear transducer. Both left and right carotid arteries were evaluated. Patients were in a supine position with the head extended and rotated to the other side during the examination. The common carotid artery, carotid bulb, and internal carotid artery were carefully scanned from the continuous angles to identify the thickest carotid IMT. The intima-media thickness (IMT) is defined as the distance between the intima-blood interface and the adventitia-media junction determined by the intima-media thickness (IMT). The mean value of the 3 determinations was calculated and the final values of IMT were averaged by the left and right mean IMT values (Figure 1).

Measurements of the intima-media thickness in the common carotid artery using 7 MHz linear probe.
Some of the CIMT literature, at least in the adult population, suggests elevated cardiovascular risk with absolute CIMT values greater than 1.0 or with values higher than the 75th percentile for age and gender. 23 In our study, a single cutoff point for CIMT between normal and abnormal children was established at 0.45 mm that defines the abnormality. 24
Statistical analyses: The analysis of data and variables was done using the software Statistical Package for Social Sciences (SPSS), version 16.0 (Chicago, IL, USA). Qualitative data are presented as numbers and percentages, whereas quantitative data with normal distribution are presented as means ± standard deviation or median with interquartile range. We used the student’s t-test to compare the data between cases and controls. However, a one-way analysis of variance (ANOVA) test was used to compare between and within the groups of children on AED monotherapy. This was followed by post hoc analysis between the studied groups. The relation between CIMT and different variables were analyzed using Pearson’s and Spearman’s correlation coefficients. Significance was considered at a P value of less than .05. Two stepwise models of multiple linear regression analysis were performed to assess the impact of independent variables on the mean CIMT as a dependent variable. Assessment of factors that are significantly correlated with mean CIMT was carried out initially by model 1 analysis, and significant factors thus identified were further subjected to model 2 analysis.
Ethical Approval and Informed Consent
The study was conducted after obtaining a written informed consent from concerned parents. Also, ethical clearance was obtained for the research study. The IRB of Faculty of medicine, Benha University, Egypt (Rc:7-4-2023) had approved the study. The study protocol conforms to the ethical guidelines of the 1964 Declaration of Helsinki and its later amendments.
Results
This study was conducted on 60 children with epilepsy on AED monotherapy. Their ages ranged from 2 to 14 years with a median age of 75 months. Thirty-six (60%) were males and 24 (40%) were females with an M:F ratio of 1.5:1. Sixty healthy children were selected as a control group. Their ages ranged from 3 to 14 years with a median age of 73 months. Thirty-two (53.3%) were males and 28 (46.7%) were females with an M:F ratio of 1.14:1.
Median age at the time of diagnosis of epilepsy was 6.2 years. None of our patients had overweight or obesity although the average weight and body mass index were higher in patients with epilepsy than controls. However, these changes were not significant.
Weight and height of patients and controls were within normal percentiles.
The average frequency of epileptic fits per month was 2.8 ± 5.3. However, 15(25%) patients showed infrequent seizures. The mean time elapsed from the last fit was 2.1 ± 3.4 months,
According to the type of AED monotherapy, 32 (54.3%) patients were on valproate, 11 (18.4%) patients were on carbamazepine, and 17 (28.3%) patients were on levetiracetam monotherapy. All AEDs were given with appropriate doses, good compliance, and were within normal therapeutic ranges. The most common type of seizure in our patients was a generalized tonic-clonic seizure and it was reported in 38 (63.3%) patients followed by a partial seizure in 14 cases (23.3%). The myoclonic seizure was reported in 5 cases (8.4%) and absence seizure was reported in 3 cases (5 %).
There difference between the studied groups regarding GASE scale (P value = .05) was significant (Table 1). Most cases have normal brain magnetic resonance imaging (MRI). However, 12 cases (20%) showed abnormal findings as brain atrophy in 5 cases (8.4%), gliosis in 4 cases (6.6%), cortical dysplasia in 2 cases (3.3%), and only 1 patient (1.7%) has a congenital arachnoid cyst. Clinical characteristics and demographic data of cases with epilepsy are shown in (Table 1). There was no statistically significant difference in the mean CIMT between males and females (0.43 mm ±0.o8 and 0.46 ± 0.11 respectively). We found a significant increase in the CIMT with the increase in age. The mean CIMT was significantly higher in patients with epilepsy compared to controls all over the age categories (P-value ≤ .001). The mean values of CIMT in different age categories in patients and controls are shown in (Table 2). The relationship between log-transformed CIMT and age of patients and controls after adjustment for AED use, duration of antiepileptic drug (AED) therapy, and blood levels of LDL and HDL is shown in (Figure 2).
Clinical Characteristics and Demographic Data of Cases of Epilepsy.

Values of the Mean cIMT in Different Age Groups Among Cases and Controls.


Relationship between log-transformed carotid intima media thickness (CIMT) and age of patients and controls after adjustment for AED use, duration of antiepileptic drug (AED) therapy, and blood levels of LDL and HDL.
The duration of AED therapy at the time of the study was between 0.5 and 1 year in 34 (56.7%), 1 and 2 years in 14 (23.3%), 2 and 3 years in 7 (11.7%), and more than 3 years in 5 (8.3%) children with epilepsy. The values of CIMT were significantly increased with the increase in the duration of AED monotherapy.
The relationship between log-transformed carotid intima media thickness (CIMT) and duration of antiepileptic drug (AED) therapy after adjustment for age, AED use, and blood levels of LDL and HDL is shown in (Figure 3).

Relationship between log-transformed carotid intima media thickness (CIMT) and duration of antiepileptic drug (AED) therapy after adjustment for age, AED use, and blood levels of LDL and HDL.
CIMT above normal reference value was reported in 43 (71.7%) patients, 26 (81.2%) patients in the valproate group, 5 (45.4%) patients in the carbamazepine group, and 12 (70.5%) patients in the levetiracetam group. None in the control group had CIMT above normal reference value. CIMT abnormalities were significantly higher in children on valproate than those on carbamazepine and levetiracetam (P-value .009 and .04) respectively. Also, patients on carbamazepine showed more significant abnormalities in CIMT than patients on levetiracetam (P-value .01) (Table 3).
Comparison of the Percentage of Patients With Above Normal Reference Value of CIMT and Abnormal Lipid Profile in the Group of Patients.

P1: Test the significance between valproate group and carbamazepine group. P2: Test the significance between valproate group and levetiracetam group. P3: Test the significance between carbamazepine group and levetiracetam group.
Abbreviations: CIMT, carotide intima media thickness.
Chi2 test with subsequent post-hoc analysis.
Among the 60 patients with epilepsy enrolled in the study, 44 (73.3%) patients showed abnormal lipid profiles. Twenty-nine (90.6%) patients in the valproate group, 7 (63.6%) patients in the carbamazepine group, and 8 (47.0%) patients in the levetiracetam group.
None in the control group showed abnormal lipid profiles.
Abnormalities in lipid profiles were significantly higher in patients on valproate than those on carbamazepine and levetiracetam (P-value .01 and .001) respectively. Also, patients on carbamazepine showed more significant abnormalities than patients on levetiracetam (P-value .03) (Table 3).
Our study showed BMI was higher among patients receiving valproate (15.3 ± 3.6 kg/m2), carbamazepine (15.1 ± 2.6 kg/m2), and levetiracetam (14.9 ± 1.9 kg/m2) compared with that of controls. However, the difference in BMI between cases and control was not statistically significant(P >.05).
None of the children had BMI more than the 95th centile. The comparison of BMI and CIMT between cases and controls is shown in (Table 4). Patients on valproate therapy showed significantly higher CIMT (0.44 ± 0.03, P ≤ .001 ) than controls. Also, Patients on carbamazepine and levetiracetam therapy showed significantly higher CIMT compared to controls (0.43 ± 0.03, with P ≤ .001 and 0.44 ± 0.02 with P ≤ .001 respectively). However, no significant difference in the CIMT was found between valproate, carbamazepine, and levetiracetam groups (P >.05) (Table 4).
Comparison of BMI and CIMT Between Cases With Epilepsy and Controls.
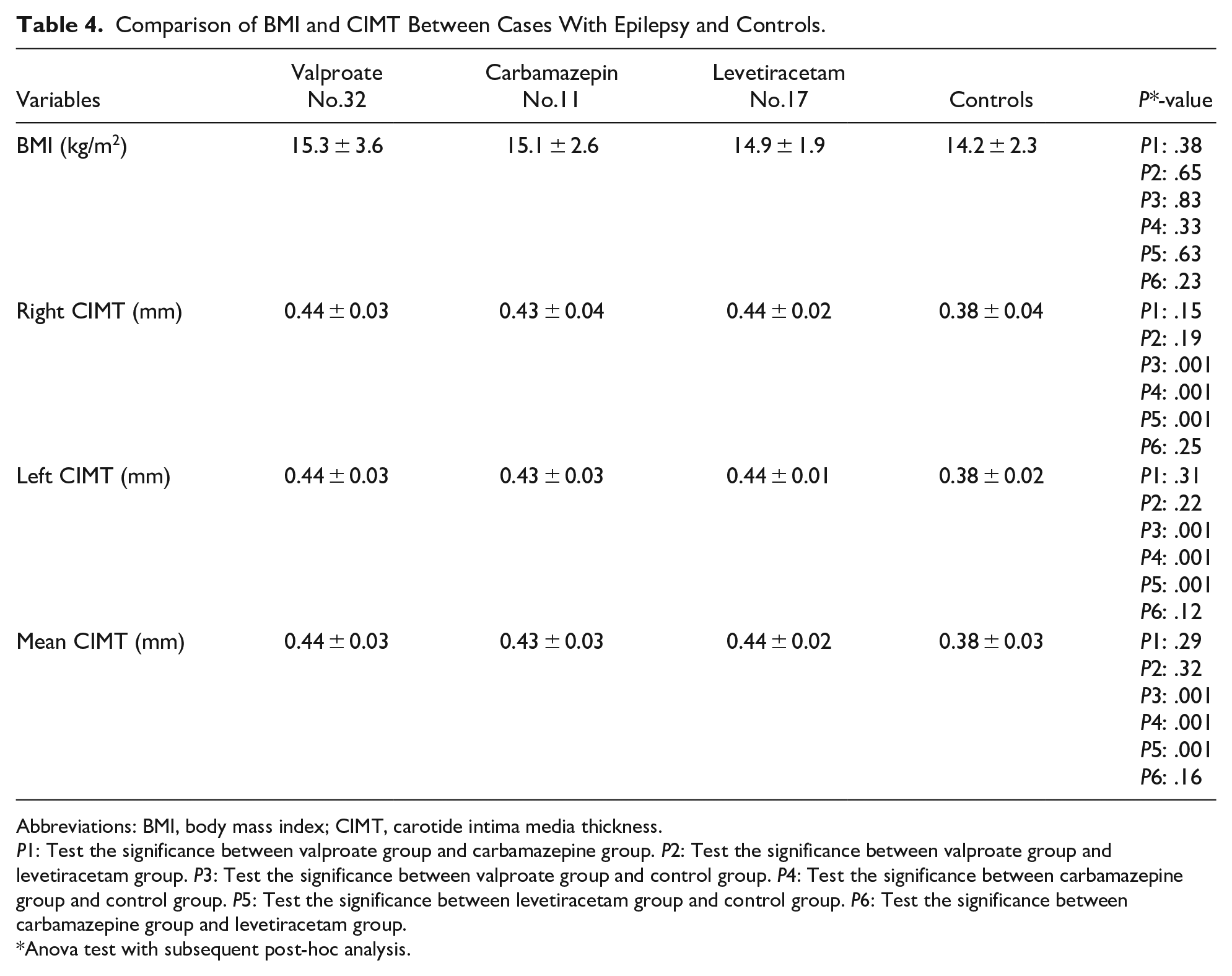
Abbreviations: BMI, body mass index; CIMT, carotide intima media thickness.
P1: Test the significance between valproate group and carbamazepine group. P2: Test the significance between valproate group and levetiracetam group. P3: Test the significance between valproate group and control group. P4: Test the significance between carbamazepine group and control group. P5: Test the significance between levetiracetam group and control group. P6: Test the significance between carbamazepine group and levetiracetam group.
Anova test with subsequent post-hoc analysis.
Serum levels of lipid profile including TC, TG, and LDL were significantly higher in children on both valproate and carbamazepine than that in children on levetiracetam and controls (P-value ≤ .001) with no significant difference between valproate and carbamazepine (P-value > .05). Also, no significant difference between levetiracetam and controls (P-value > .05). Regarding HDL, it was significantly lower in children on both valproate and carbamazepine than that in children on levetiracetam and controls (P-value < .001) with no significant difference between valproate and carbamazepine (P-value > .05). Also, no significant difference between levetiracetam and controls (P-value > .05). Regarding Apo A1, there was a significant difference between valproate, carbamazepine, and levetiracetam when compared to controls (P-value = .001). However, no significant differences were found between valproate, carbamazepine, and levetiracetam groups (P-value > .05). However, the differences for VLDL were not statistically significant (P > .05) (Table 5).
Comparison of Serum Lipid Profiles Among Cases With Epilepsy and Controls.

P1: Test the significance between valproate group and carbamazepine group. P2: Test the significance between valproate group and levetiracetam group. P3: Test the significance between valproate group and control group. P4: Test the significance between carbamazepine group and control group. P5: Test the significance between levetiracetam group and control group. P6: Test the significance between carbamazepine group and levetiracetam group.
Abbreviations: TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; VLDL-C, very low-density lipoprotein cholesterol; Apo A1, apolipoprotein A1; TG: triglyceride.
Reference value 23 *Anova test with subsequent post-hoc analysis.
CIMT was correlated with age (P = .041, r = .112) duration of treatment (P = .001, r = .313), TC(P = .001, r = .192), TG(P = .014, r = .018), and LDL(P = .001, r = .219).
CIMT was negatively correlated with HDL(P = .003, r = −.126). However, seizure severity and Apo A1 were insignificantly involved (P > .05). The correlations of CIMT, after adjustment for age, duration of (AED) therapy, and blood levels of LDL, and HDL with different variables are shown in (Table 6).
Correlations Between Adjusted CIMT and Studied Parameters in Patients With Epilepsy and Normal Controls.

CIMT was adjusted for age, duration of (AED) therapy and blood levels of LDL and HDL.
Abbreviations: CIMT, intima media thickness; LDL-C, low-density lipoprotein cholesterol. HDL-C, high-density lipoprotein cholesterol; Apo A1, apolipoprotein A1; AED, antiepileptic drug.
Epilepsy, use of AED, duration of AED therapy, and seizure frequency are defined as 0 in the control group.
Two series of statistical analyses were subsequently carried out to decipher the relationship between the augmented CIMT in patients with epilepsy and other identified risk variables. Based on the correlation analysis, our first series revealed that AED use(valproate or carbamazepine or levetiracetam), duration of AED therapy, age, blood levels of total cholesterol, TG, LDL-C, and HDL-C were significantly correlated with mean CIMT (Table 6). On the other hand, epilepsy, seizure severity and Apo A1 showed insignificant correlation. We further employed multiple linear regression analysis in our second series to identify from those variables the crucial determinant that underlies the augmented CIMT in patients with epilepsy.
Results from model 1 analysis revealed that whereas AED use (valproate or carbamazepine), duration of AED therapy, age, LDL, and HDL were significantly associated with log transformed CIMT. Epilepsy, TC, Apo A1, and TG were insignificantly involved. Model 2 stepwise regression analysis, which included only those 6 variables, provided further insights (Table 7).
Multivariate Regression Analysis of Association of Log-transformed CIMT With Candidate Risk Factors.

Log CIMT: dependent variable.
Abbreviations: CIMT, carotide intima media thickness; AED, antiepileptic drugs; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; VLDL-C, very low-density lipoprotein cholesterol; Apo A1, apolipoprotein A1; TC, total cholesterol; TG, triglyceride.
Discussion
The long-term use of AEDs can create adverse effects and complications by affecting several systems in children with epilepsy. The use of AEDs is associated with changes in the CIMT in children with epilepsy. CIMT has been linked to many vascular outcomes, including cardiovascular and cerebrovascular events. 25 There have been previous studies on the vascular side effects of AED therapy in children with epilepsy. However, the results are still controversial. 4
To our knowledge, the current study is one of the few studies conducted on children with epilepsy on AEDs to clarify their effects on CIMT and lipid profile. Most of the previous studies were about adults with epilepsy. This gives our study a powerful point of view. A major worry about the prescription of AEDs is whether or not these medications lead to the risk of atherosclerosis in children with epilepsy later on, and about their effects on CIMT, hence, provoking the incidence of cardiovascular and cerebrovascular complications.
In our study, we analyzed 60 children with epilepsy compared to 60 healthy controls. The gender distribution and clinical characteristics of our patients were comparable to other studies.4,25
Our study showed the difference in BMI between cases and controls was not statistically significant (P >.05). Contrary to our results, El-Farahaty et al., found a significantly higher BMI among cases receiving valproate and levetiracetam compared to controls (P = .003). 26 However, similar to our findings, Kolekar et al., did not report significant findings between BMI and AEDs monotherapy. 27 In a study of 114 children with epilepsy treated with valproate monotherapy over 24 months, obesity and metabolic syndrome were significantly reported in 46 and 20 children during the study period. 28
In present study, the mean values of CIMT in the controls were 0.38 ± 0.03, and most of our patients (71.7%) showed CIMT above normal reference value. These findings were comparable with the studies done by Mieczyslaw et al, Zhu et al and Dawson et al.29-31
Also, it was reported that the means of CIMT (in mm) were higher for valproate (0.64 ± 0.1 mm) and levetiracetam (0.563 ± 0.04 mm) groups with a P value of <.001. 26
In our study, the thickness of CIMT was significantly increased in patients receiving valproate, carbamazepine, and levetiracetam as compared to controls.
However, within the 3 studied groups of epilepsy, there was no significant difference in the CIMT. Our results were consistent with other studies.26,27,32
Also, we observed that with increasing age, the values of CIMT increased as well. However, no significant difference in the mean CIMT between males and females as reported in other studies.25,32
In a recent study conducted by Kolekar et al., they studied 42 epileptic children between 1 and 18 years of age over 18 months receiving AED monotherapy with valproate, phenytoin, or levetiracetam for at least 6 months. They reported significant abnormalities in CIMT with these AEDs. 27
We found that CIMT abnormalities were significantly prominent in children on valproate than those on carbamazepine and levetiracetam.
Our results are consistent with a study conducted by Erdemir et al., on 44 children with epilepsy on valproate monotherapy for more than 12 months. They found a significant increase in the CIMT among children receiving valproate (0.48 ± 0.06) compared to controls (0.43 ± 0.06), (P = .001). 32 Contrary to our findings, Ksoo et al. reported that CIMT was not significantly increased in patients receiving CBZ and VPA compared to controls. 25
In our study, we reported significant changes in lipid profile including TC, TG, LDL, HDL, and Apo A1 in children with epilepsy who received valproate and carbamazepine compared to levetiracetam and controls. Our results are consistent with previous studies that reported dyslipidemia with long-term AED therapy with elevation in the serum levels of TC and LDL.4,33,34
Chronic inflammatory reactions of epilepsy and lipid disturbances caused by long-term use of AEDs act as risk factors in the atherosclerotic changes in epileptic patients.
Some AEDs may increase lipids levels, whereas others may not. AEDs are either drugs such as carbamazepine, which enhance hepatic P450 cytochrome system activity, leading to increased cholesterol synthesis, or drugs inhibiting this enzyme activity, such as valproate, whereas levetiracetam is a non-enzyme inducer. 5
It was found that cytochrome P450 enzyme induction might reduce the feedback inhibition of 3-hydroxy-3- methylglutaryl coenzyme A synthesis, and subsequently resulted in increasing the level of cholesterol. 35
It was found that epileptic patients maintained on carbamazepine or valproate monotherapy—as in our study—have increased cholesterol and TG concentrations. 36
The role of AEDs in the pathogenesis of atherosclerosis is not fully understood. However, some explanations have been suggested. Long-term AED therapy can result in dyslipidemia, with elevated serum levels of TC, (LDL-C), and apolipoprotein A.4,33,34
LDL plays an important role in the atherosclerotic process by causing the retention of lipoproteins within the intima layer of blood vessels, increasing the permeability of the endothelial layer with the recruitment of inflammatory cells, and the formation of foam cells. 34 So, LDL contributes to increased CIMT.
Clinical changes in body weight are well-known adverse effects of AEDs. 37 Also, CIMT is associated with obesity and an increase in the BMI. 38 A fundamental mechanism of atherogenesis in obese patients or patients with increased BMI is low-grade systemic inflammation. 39 It follows the suggestion that the higher BMI exhibited by our patients under prolonged AED therapy may also contribute to the observed thickening of the CIMT via an inflammatory mechanism.
Hyperhomocysteinemia may partially explain atherosclerosis in epileptic children, which could increase tumor necrosis factor expression, enhance oxidative stress, and induce a proinflammatory state that might contribute to atherosclerosis. 36 AEDs with enzyme-inducing activity as carbamazepine may induce hyperhomocysteinemia by affecting the liver-enzyme induction of folate and vitamin B12.40,41
Carbamazepine is associated with elevated serum levels of total cholesterol and lowdensity lipoprotein due to their effects on cholesterol synthesis enzymes and C-reactive protein. 42 C-reactive protein (CRP), was found to be elevated in epileptic children receiving AEDs.4,43 C-reactive protein (CRP) can promote inflammation and atherosclerosis by affecting monocytes and endothelial cells and increasing the activity of plasminogen activator inhibitor. 44
Furthermore, it was found that older AEDs, such as carbamazepine cause a deficit in folate, which is a key cofactor in the breakdown of homocysteine. 45
It has been reported that there is completion effect between AEDs with enzyme inducing activity like carbamazepine and the cholesterol in the utilization of hepatic microsomal enzymes P450 system, leading to reduction in the cholesterol transformation in bile acids with subsequent increased serum cholesterol. 46
Also, it was reported that long-term AED therapy may result in low-grade systemic inflammation and increase in the oxidative stress. Oxidative mechanism is another independent factor associated with the acceleration of atherosclerosis in patients with epilepsy. There is now a consensus that atherosclerosis represents a state of heightened oxidative stress characterized by lipid and protein oxidation in the vascular wall. 47 Chronic production of reactive oxygen species may exceed the capacity of cellular antioxidants, resulting in oxidative modification of LDL-C, promotion of proinflammatory responses, recruitment of macrophage, and development of atherosclerotic lesion. 47
Recently, it was found that valproate may be associated with elevated serum homocysteine levels in epileptic patients either adults or children by impairing the absorption of folic acid and interfering with the metabolism of folic acid coenzymes.32,48,49
Among the 60 patients with epilepsy enrolled in our study, (73.3%) patients showed abnormal lipid profiles. Most patients were in the valproate group (90.6%).
El-Farahaty et al. studied the atherogenicity of the individual AED and they reported that children with epilepsy on valproate monotherapy had significant abnormalities in TC, LDL, HDL, TC/HDL, and LDL/HDL (P value of <.001). 26
We found significant changes in the lipid profile (TC, TG, LDL, and HDL) in children on both valproate and carbamazepine compared to those in levetiracetam and controls. However, no significant changes between valproate and carbamazepine were reported.
Ksoo et al. found that children with epilepsy who were receiving either carbamazepine or valproate had an increase in their total cholesterol, serum LDL, and serum HDL levels and these findings were comparable to other studies.5,25,28 Also, Ksoo et al. observed a decrease in the levels of triglycerides during the follow-up of their patients on valproate monotherapy which was similar to other studies.28,50
Consistent with our findings, Lima et al., observed that LDL values were greater in epileptic patients on AED therapy compared to controls, whereas HDL values were reduced. 51 Conversely, Büyükgöl and Güneş, concluded that valproic acid and carbamazepine had no effect on HDL that protect against coronary artery disease. 52 Also, Yamamoto et al., found patients who were receiving valproate showed a decrease in serum levels of HDL. 53
Regarding Apo A1, we reported a significant difference only between valproate, carbamazepine, and levetiracetam when compared to controls. Our findings are agreeing with Tomoum et al. who conducted a study on 22 children on either carbamazepine or valproate monotherapy, they found a significant decrease in Apo A1 levels in cases (114.7 ± 16.2 mg/dL) compared to controls (138.8 ± 10.4 mg/dL, t = 5.05). 54 In our study, we did not find significant changes in TC, TG, LDL, and HDL between levetiracetam and controls.
In our study, levetiracetam showed the lowest mean value of TG and cholesterol. This was in agreement with El-Farahaty et al. 26 who reported the lowest mean value of total cholesterol in patients treated with levetiracetam. Also, Kim et al. did not find changes in total cholesterol, low density lipoprotein cholesterol, and HDL-C levels in epileptic patients on levetiracetam monotherapy. 55
Our findings are supported by the study conducted by Attilakos et al. on 39 children with epilepsy who were treated with levetiracetam, they observed TG and TG/HDL ratios were significantly reduced at 6 and 12 months of levetiracetam treatment (P = .026 and P = .001 and P = .024, P = .003), respectively. Also, the ratio of LDL/HDL showed a significant reduction at 12 months of treatment (P = .025) with no significant changes in other lipid parameters. It was concluded that levetiracetam treatment has less atherogenic properties than other AEDs. 56 We observed that levels of VLDL did not show significant changes among studied groups which was comparable to other studies.57,58
Karatoprak et al., 2020, and Kolekar et al., 2021, concluded that non-obese children with epilepsy who receive monotherapy of valproic acid or levetiracetam may be at higher risk of developing subclinical atherosclerosis despite a normal lipid profile.27,59
In our study, we found significant positive correlations between CIMT and TC, triglyceride, and LDL. Also, we found a significant negative correlations between CIMT and HDL.These findings are comparable to Kolekar et al., who reported similar correlations between CIMT and individual lipid profiles. 27
Our results showed that AEDs especially valproate and carbamazepine, the duration of AED therapy and age were also correlated with CIMT. It is, therefore, reasonable to stipulate that AEDs notably valproate and carbamazepine, the duration of AED therapy and age are at least important and contributing risk factors for the increase in CIMT. From these correlations—highlighted in our study—, we can attribute changes in the CIMT to AEDs rather than epilepsy.
In concordance of our results, Luo et al., and Tan et al., found that the average CIMT of patients with epilepsy on valproate therapy was higher than that of healthy controls. In addition, the average CIMT of patients with valproate therapy with a duration of more than 1 year was higher than that of the patients with a valproate therapy duration of less than 1 year.4,60 Also, Ksoo et al. observed that the values of CIMT increased with increasing age as observed in our study. 25
The present study provides novel insights into these clinical observations by showing that the duration of prolonged use of AEDs is significantly associated with augmented CIMT as dependent variable in patients with epilepsy, alongside independent contributions of age, AEDs, LDL-C, and HDL- C.
A small sample of children with epilepsy was a limitation in our study. Also, we did not proceed to sample size calculation before conducting the study.
Future prospective studies on a larger sample of populations are recommended for stronger comments in this issue.
Conclusion
The present study has provided insights into AEDs by showing that long-term monotherapy with valproate, carbamazepine, and levetiracetam in children with epilepsy was associated with significant abnormalities in CIMT. These findings indicate that neurologists prescribing AEDs even levetiracetam should be aware of potentially unfavorable adverse effects, especially in patients at a high risk of vascular events as well as close follow-up of children with abnormal CIMT to prevent cardiovascular and cerebrovascular risks.
Also, choice of drug for patients with epilepsy under long-term AED therapy should be guided with their lipid profile.
Footnotes
Acknowledgements
All authors offer many thanks to all nurses and residents in Pediatric department, Benha University hospitals for their effort in this work.
Author Contribution
HHE conceived the study, MKE, REA, AHA, AOI, NMK, HAS, KAN, NSI, AMA, AAT, and WEA participated in its design and coordination. AMA, AAT, HAS, AHA, AOI, NMK, WEA, and KAN provided key technical guidance. MKE, REA, AHA, AOI, NMK, and HHE drafted the manuscript. HAS, KAN, NSI, AMA, AAT, and WEA critically revised the manuscript for important intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
