Abstract
Antimicrobial stewardship programs seek to improve patient outcomes, reduce cost, and hinder development of antimicrobial resistance. Collection of inpatient antimicrobial use data is foundational to these programs, and decisions to initiate and discontinue antibiotics are potentially amenable to improvement. In order to better understand our current practices and with a view toward improved antimicrobial stewardship, we reviewed charts of previously healthy children to age 16 years hospitalized with fever (without an evident localized source) and/or other findings suggestive of serious bacterial infection. Of 105 patients (18% 0-2 months of age, 42% 3-12 months, 25% 2-5 years), 100 (95%) received antibiotics, 72% for more than 2 days. Of 98 patients with negative body fluid cultures, 23 received antibiotics for 2 days or less, and 70 received antimicrobial therapy for more than 2 days. Focusing on selective initiation and earlier discontinuation of antimicrobial therapy in hospitalized children might reduce unnecessary antibiotic use.
Keywords
Introduction
In much of the world, antimicrobial stewardship programs seek to improve patient outcomes, reduce cost, and slow the spread of antimicrobial resistance by advocating for and/or enforcing judicious initiation, selection, and discontinuation of antimicrobial therapy. 1 A foundational component of effective medication stewardship is a monitoring program to identify and follow antimicrobial therapy. 1 Effective antimicrobial stewardship programs may be generalized throughout institutions or targeted toward specific patient groups. 2 In Europe, a children’s hospital’s quality improvement program led to significant reductions in overall antibiotic use. 3 Antimicrobial stewardship programs focused on children with acute respiratory infections have also led to improved care with decreased costs. 4
For children, empiric antimicrobial therapy is often considered and initiated before results of definitive diagnostic tests are available. Guidelines have been developed in North America to assist in management of febrile but otherwise well-appearing young infants. 5 However, with increasing resistance to commonly used antibiotics in newborns, several African and Asian countries report widespread resistance to what had been first line agents in standard antimicrobial recommendations for febrile infants. 6 Empiric antimicrobial therapy decisions should be based on the local antibiogram, as well as accessibility and affordability of antimicrobials. 6 A consensus group suggested that research should be prioritized that involves benchmarking and peer comparison of antibiotic use for common pediatric infections. 7 As an example, in a Jordanian study, 31% of pediatric inpatients received antibiotics, however the indications were not well-documented in medical records, and the timing of discontinuation was not consistent with recommended procedures. 8 At a US children’s hospital, key targets for improvement in antimicrobial use were found to relate specifically to ear, nose, and throat infections. 9 Thus, evaluation of local antibiotic prescribing habits can identify variations and non-standard practices; then, targeted interventions can potentially improve antimicrobial stewardship.
In our setting in the Arab Gulf region, children are frequently admitted to the hospital with concerns for serious bacterial infection yet without localizing signs or symptoms. In order to determine potential opportunities for improving antimicrobial stewardship, we undertook a retrospective chart review to determine our current antimicrobial use in children of various ages admitted with fever or other concerns for serious bacterial infection who did not have localizing signs or symptoms of infection
Methods
Sheikh Shakhbout Medical City is a growing local and regional hospital located in the suburbs of metropolitan Abu Dhabi, United Arab Emirates. The general pediatric hospital service cares for patients on a 30-bed unit. Staff physicians have been trained in various regions of the world, including North America, Europe, North Africa, and the Middle East. There are 25 pediatric residents, as well as students from 2 different universities. Patients are mostly from Abu Dhabi and include a mix of national backgrounds with about half being Emirati. This study took place during the COVID-19 pandemic, however our hospital was “COVID-free,” and patients who tested positive for SARS-CoV-2 were transferred to another facility for care.
Via retrospective chart review, previously healthy patients admitted with fever or other concerns for serious bacterial infection (shock, new seizure, oxygen desaturation) during 2021 were included if there was no sign of a localized infection (pneumonia, meningitis, urinary tract infection, osteomyelitis) at the time of admission. Patients with underlying medical conditions including pre-term birth were excluded from the study. Demographic information was noted, as were details about antibiotic use and laboratory testing.
Results
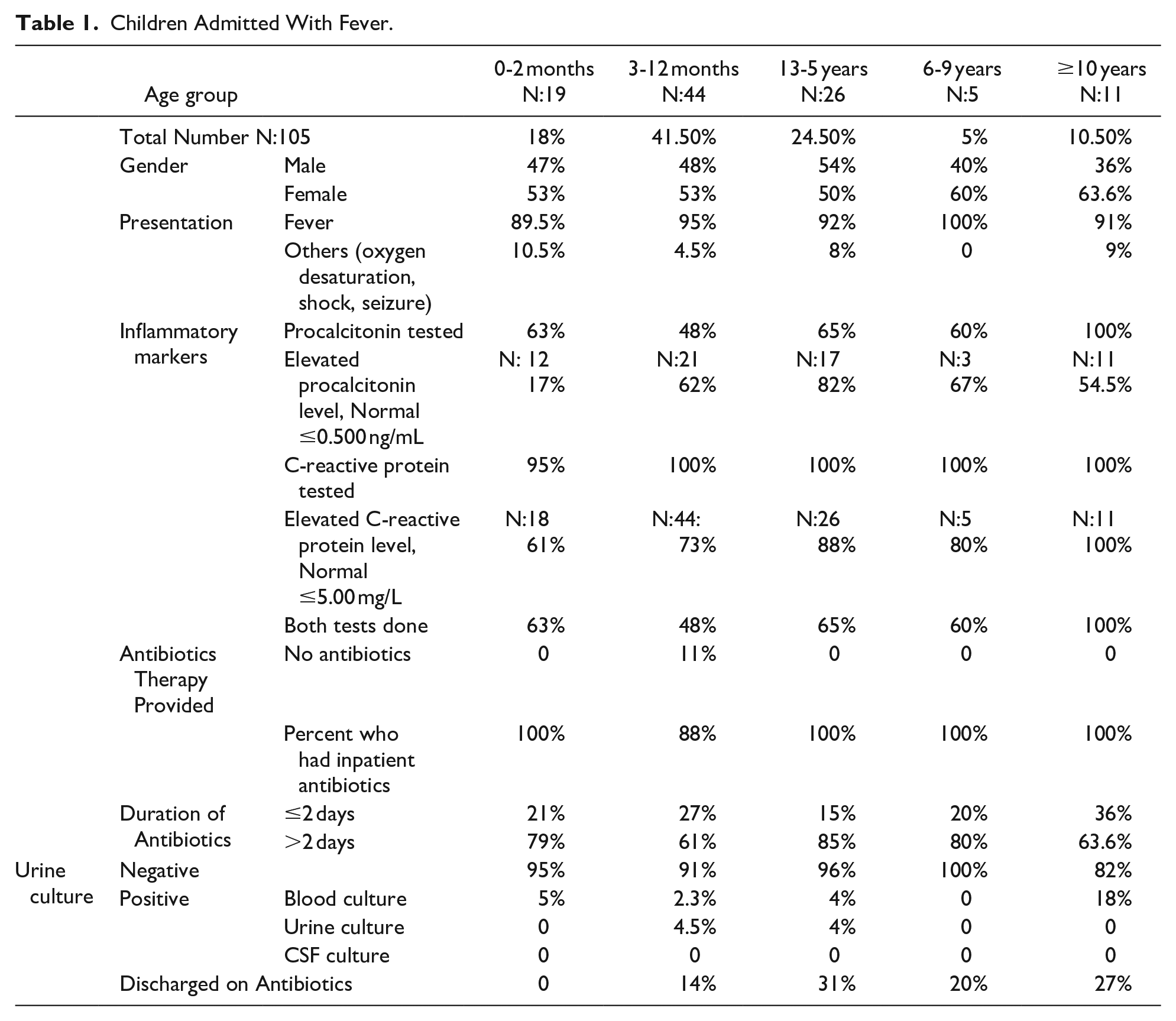
A total of 105 children were included in the study, with 85% less than or equal to 5 years of age; details are noted in Table 1. Procalcitonin was tested in 64 (61%) and was elevated in 37 (58%). C-reactive protein was tested in 104 (99%) and elevated in 81 (78%). Of 105 patients, 100 (96%) received antibiotic therapy. Cephalosporins were most commonly used, as noted in Table 2. Antibiotic treatment was continued for more than 2 days in 72% of patients, but only 18% were still on an antibiotic when discharged from the hospital.
Children Admitted With Fever.
Antibiotics Used During Hospitalization.

Seven patients had positive body fluid cultures. As noted in Table 3, 1 had positive blood and urine cultures, 4 had blood cultures positive for a pathogen, 2 had positive urine cultures (one for 2 different presumed pathogens), and 1 had a blood culture positive for a presumed contaminant.
Patients With Positive Culture Results, of Total n = 105.

Discussion
In our setting, antibiotic use was common in children presenting with findings concerning for serious bacterial infection. However, bacteremia and urinary tract infection were only identified in 7% of the children, despite the majority having elevated inflammatory marker test results. A significant number (1 of 5, 20%) of positive blood cultures included bacteria not considered to be pathogenic. Antibiotic therapy usually (72% of the time) continued for more than 2 days, despite negative culture results. These findings suggest that in our setting it might be possible to reduce unnecessary antimicrobial therapy by reducing initiation of antibiotic therapy and by more rapid discontinuation of antibiotic therapy when culture results are negative.
Most febrile children of more than 2 months of age do not have serious bacterial infections. Even during the first 2 months of life when clinical appearance is less reliable in predicting bacteremia, recent guidelines suggest the possibility of clinical observation without antimicrobial therapy. 5 In newborns, normal results of inflammatory marker tests such as C-reactive protein and procalcitonin, can support decisions not to give antibiotics. 5
In general, C-reactive protein and procalcitonin are most useful when not elevated given the high negative predictive value for serious bacterial infection. Elevated results are less specific for serious infection and have lower positive predictive values.10-12 Elevation of inflammatory markers, alone, should not be considered an indication to start antimicrobial therapy in febrile children, at least after the first 2 months of life.
Urinalysis, however, can often identify children who are most likely to benefit from antimicrobial therapy. In our study, 3 of the total of 6 children who eventually had body fluid cultures positive for pathogenic bacteria had abnormal urinalyses. One could postulate that the clinical appearance of many of the 97% of children with normal urinalysis results who did not end up having bacteremia might have been appropriately reassuring to allow careful observation without initiation of antibiotic therapy.
Molecular tests for single pathogens and multi-pathogen panels can also help identify possible etiologic agents in samples taken from febrile children. 13 Early identification of pathogens for which antimicrobial is not necessary (such as enterovirus and human herpesvirus 6) could allow treating clinicians to safely avoid antibiotic initiation or to discontinue unnecessary antimicrobial therapy early. 13 As for inflammatory marker testing, pathogen-specific testing is more useful to avoid antimicrobial use rather than to prompt the initiation of antimicrobial therapy. 14
The microbiologic etiology of bacteremia varies around the world. In much of Africa and Asia, even with lower rates of immunization against Haemophilus and pneumococcus than in Europe and North America, Gram-negative enteric bacteria and Staphylococcus aureus are more predominant than are other germs. 15 In addition, multi-resistant germs are commonly identified. 16 Thus, when antibiotics are deemed necessary, broader spectrum coverage might be necessary. Nonetheless, in our Middle East setting, only 1 of the 6 pathogen-positive isolates was resistant to cephalosporins.
When presumptive antibiotic therapy is appropriately started, de-escalation (or cessation) of treatment is warranted when negative culture results are available. With current laboratory technology, in settings with effective laboratory support, pathogen-positive blood cultures are almost always positive within 24 hours in newborns 17 and in older children. 18 In our setting, positive results are reported immediately, but negative results are only reported daily, prompting some clinicians to continue therapy until a “negative at 2 days” result is reported. Modification of systems so that clinicians are notified early about children with negative results might lead to shorter durations of antibiotic treatment. In addition, skepticism about the reliability of negative blood cultures leads some clinicians to continuing treatment for “clinical sepsis” based on a concerning initial patient presentation, even with negative culture results.
Contaminated body fluid culture samples pose another problem, with 20% of “positive” blood cultures in our study revealing a non-pathogen. Early notification of a “positive” culture result prompts prolongation of antibiotic therapy until the microbes are identified. As everywhere in the world, careful sampling and handling of culture specimens could reduce some unnecessary antibiotic treatment. In addition, non-elevated procalcitonin levels correlate with non-pathogen contaminants in blood cultures and could contribute to more rapid decisions to de-escalate antibiotic treatment. 19
As part of good antimicrobial stewardship, clinicians should monitor their own practices. While we identified potential over-initiation and over-prolongation of antibiotic treatment as possible targets for intervention, other centers in the Middle East have found their own specific targets to improve judicious use of antibiotics. Across the Arabian Gulf from us in Iran, excessive initiation of antibiotic treatment in both inpatient and outpatient settings was identified, and antimicrobial stewardship programs led to reductions in unnecessary antibiotic prescriptions. 20 In Jordan, it was the timing of discontinuation of antibiotic therapy that held promise as a good target for stewardship program intervention. 8 Similarly, in Europe, antimicrobial stewardship interventions in a children’s hospital led to less antibiotic use, less cost, and improved patient outcomes. 3 In a North American study, vancomycin use and the treatment of otorhinolaryngology infections were the targets identified as most amenable to helpful stewardship interventions. 9
There are a number of limitations to our study. First, it was retrospective, and we do not have consistent documentation of the thought processes and clinical assessments behind the decisions to initiate antibiotic treatment. Second, this is a relatively small study in one specific setting. As such, it is useful to us however might not be indicative of situations and needs in other settings. Nonetheless, this study demonstrates an approach to identifying potential targets for improved judicious use of antimicrobial therapy.
Antimicrobial stewardship is essential as we deal with severe infections, health care costs, and increasing antimicrobial resistance. Identification of potential sources of overuse of antibiotics, such as over-initiation of treatment for febrile children and excessively long durations of treatment after negative blood culture results, can serve as a means of targeting subsequent antimicrobial stewardship interventions.
Footnotes
Author Contributions
MKA conceived of this study and acquired the relevant data. MKA and PRF designed the study. Each of the four authors contributed to the analysis and interpretation of the data. MKA wrote the first draft of the paper, and PRF wrote subsequent drafts. Each of the four authors critically revised successive drafts of the paper for important intellectual content and approved of the final version to be published. Each author agrees to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
