Abstract
There is a global concern about children presenting with inflammatory syndrome with variable clinical features during the ongoing COVID-19 pandemic. This paper reports the first pediatric case of bilateral serous retinal detachment and conjunctival hemorrhage as a revealing pattern of the pediatric inflammatory multisystem syndrome temporally associated with SARS-CoV-2 (PIMS-TS). Despite the severity of multisystemic involvement, the management with steroids was very successful. Complete remission was obtained within 3 months of acute onset.
Keywords
Introduction
The pediatric inflammatory multisystem syndrome temporally associated with coronavirus disease 19 (PIMS-TS), also called multisystem inflammatory syndrome in children (MIS-C), features a toxic shock-like syndrome or Kawasaki-like syndrome in the setting of SARS-CoV-2 positive diagnostic testing. 1 The leading cause is a severe cytokine storm resulting in a broad spectrum of clinical phenotypes, ranging from mild self-resolving to severe life-threatening presentations. 2 Renal and ophthalmological involvement are some of the rarest manifestations reported in the literature.3,4
Case Description
A 12-year-old non-consanguineous girl with no medical history presented to the emergency department for a bilateral acute vision loss with conjunctival hemorrhage evolving for 2 days. The family reported that the child had poor compliance with the preventive measures against coronavirus disease 2019 (COVID-19) during the previous month while attending public places like school. Also, several cases of COVID-19 were confirmed within her schoolmates. The girl suffered from flu-like symptoms of a fever of 39°C associated with a runny nose, sneezing, a dry cough, and diarrhea 4 weeks before admission. At that time, the parents did not seek medical advice. The child received self-medication with antipyretics at home. Her flu symptoms resolved spontaneously within 7 days. After her hospital visit, the girl was admitted to the pediatric ward. She was hemodynamically stable. Vital signs were as follows: a body temperature of 37.1°C, a blood pressure of 120/60 mmHg, a pulse rate of 68 beats per minute, a respiration rate of 20 breaths per minute, and oxygen saturation level of 98%, and a capillary refilling time of 2 seconds. The urine dipstick found microscopic hematuria of 4× and proteinuria of 1×. The diuresis was 2.5 cc/kg/h. The child had no edema or signs of volume overload. The ophthalmological examination found a periorbital ecchymosis associated with a significant, diffuse, and bilateral subconjunctival hemorrhage (Figure 1a). The best-corrected distance visual acuity was 0.3 logMAR in the right eye and 0.4 LogMAR in the left eye. The anterior segment examination showed no inflammation. The fundoscopy revealed a bilateral serous retinal detachment (SRD) affecting predominantly the posterior pole. Retinal angiography showed a dye leakage of the juxta macular area with diffusion into the SRD bulla on both sides. Meanwhile, the spectral-domain optical coherence tomography (SD-OCT) found a bilateral SRD more extensive on the left side (Figure 1b and c). Evaluation for cells in the posterior vitreous was negative in both eyes. The remaining physical examination was unremarkable. Cerebral and facial computed tomography angiography was normal. Laboratory tests revealed an acute kidney injury, with urea nitrogen of 3.29 g/l, a creatinine of 68.4 mg/l, and an eGFR of 9 ml/min/1.73 m2. The girl had hyponatremia of 133 mmol/l, hypochloremia of 92 mmol/l, and hypocalcemia of 77 mg/l. Also, she had a metabolic acidosis, with a bicarbonate level of 14 mmol/l. The remaining plasma electrolytes were within the normal range, with potassium of 5 mmol/l and a phosphor of 50 mg/l. The 24 hours proteinuria and microalbuminuria levels were high (respectively 760 mg and 142.6 mg). On the other hand, albumin and protein levels were within the normal range. Urinalysis found leukocytic and hematic casts. Complete blood count found a neutrophilic leukocytosis of 13 294/mm3 and normal hemoglobin level and platelets count (11.8 g/dl and 233 000/mm3, respectively). Acute phase reactants were high, with a CRP of 118 mg/l, an ESR of 77 mm the first hour, ferritin of 272 ng/ml, and fibrinogen of 3.7 g/l. D-dimers and amylase were high (with 1.17 ng/ml and 56 ui/l, respectively). Meanwhile, PT was prolonged at 59%. Concurrently, the child had a normal aPTT level of 22.2 seconds with no hepatic cytolysis or cholestasis. Likewise, complement, CPK, and troponin levels were within the normal range. The echocardiography and the abdominal echography were normal. A kidney biopsy was performed and showed diffuse endocapillary glomerulonephritis. Direct immunofluorescence found deposits of IgG and C3 in the capillary walls. The epidemiological context (the COVID-19 pandemic), the telltale symptoms (the flu signs), as well as the results of the paraclinical examinations (showing features of inflammation with multisystemic involvement) made the diagnosis of PIMS-TS very likely. From this perspective, we performed SARS-CoV-2 laboratory tests. The RT-PCR on the nasopharyngeal swab was negative, while the SARS-CoV-2 serology on the blood sample found negative IgM antibody and positive IgG antibody. Negative serologies of CMV, EBV, HSV, HBV, HCV, and HIV eliminated another viral cause of this condition. Meantime, repeated blood cultures to look for a bacterial or fungal infection remained sterile. Furthermore, the ASLO and angiotensin-converting enzyme II levels were within the normal range, and the QuantiFERON TB-gold plus test was negative. No additional clinical signs suggested an autoimmune disease (eg, systemic lupus erythematous, Gougerot-Sjögren syndrome, juvenile systemic arthritis, Wegener granulomatosis, or polyarteritis nodosa). In this context, anti-nuclear antibodies, anti-DNA antibodies, rheumatoid factor, anti-SSA/SSB, anti-Rnp, and anti-nuclear anti-cytoplasmic antibodies were negative. Management consisted of starting pulses of methylprednisolone at 1 g/1.73 m2/day for 3 consecutive days, followed by oral prednisone at 2 mg/kg/day for the first 4 weeks. Evolution was marked by normalization of renal function, plasma electrolytes, acute phase reactants, neutrophils polynuclear count, amylase, and PT levels within 1 week. After 1 month of acute onset, the ophthalmological examination showed improved visual acuity with 0.05 logMAR in the right eye and 0.1 logMAR in the left eye. Also, there was a significant recovery of the subconjunctival hemorrhage on both sides (Figure 2a). As for the fundoscopy, there was a persistent SRD of the left eye, while the right eye was normal. The SD-OCT revealed a rapid remission and fast reduction of the SRD of the right eye, whose macular architecture was restored to normal (Figure 2b). Meanwhile, the SRD of the left eye had partially resolved (Figure 2c). That is when we started tapering oral steroids at a dose of 1 mg/kg/day. By the end of the second month, remission was complete. The best-corrected distance visual acuity was 20/20 in both eyes. There was a total regression of the subconjunctival hemorrhage (Figure 3a), the SRD on both sides, and a normal macular profile of both eyes on SD-OCT (Figure 3b and c). Withdrawing oral steroids was completed by the end of the third month of treatment. The child remains healthy and asymptomatic after a follow-up period of 5 months.
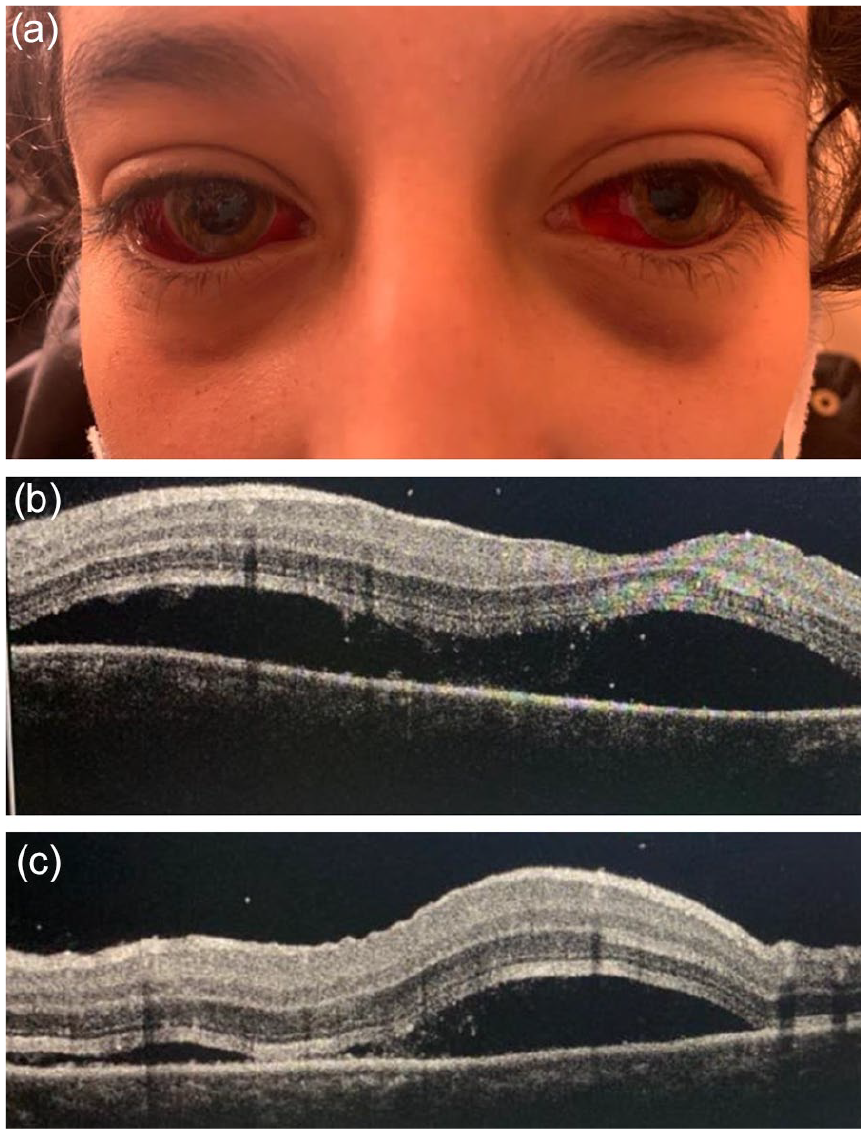
(a) Periorbital ecchymosis associated with a significant, diffuse, and bilateral subconjunctival hemorrhage. (b) SRD of the left eye. (c) SRD of the right side.
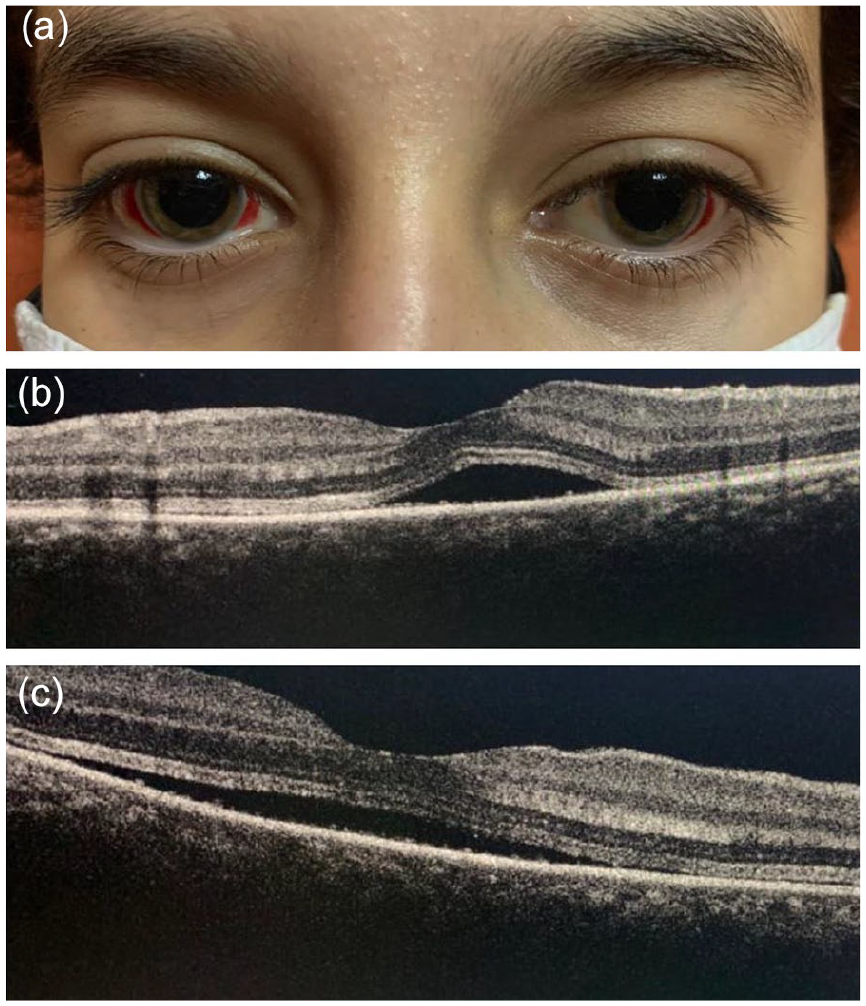
(a) Significant recovery of the subconjunctival hemorrhage on both sides. (b) Reduction of the SRD of the right eye. (c) Persistent SRD of the left eye.

(a) Total regression of the subconjunctival hemorrhage. (b and c) Normal macular profile of both eyes on SD-OCT.
Conclusions
SARS-CoV-2 can drive an exaggerated immune response. 5 In Adults, there is growing evidence to suggest an association between prior SARS-CoV-2 infection and subsequent development of autoimmune disorders, such as Systemic Lupus Erythematosus, Guillain–Barré syndrome, Kawasaki disease, and Rheumatoid Arthritis. 6 The most-reported autoimmune phenomenon associated with COVID-19 in children is Pediatric Inflammatory Multisystem Syndrome temporally associated with SARS-CoV-2 (PIMS-TS). 7 Several cases of PIMS-TS have been reported worldwide.7,8 The pathophysiology of PIMS-TS is currently not well understood. 9 Classically, PIM-TS patients have no symptoms suggestive of acute COVID-19 but somewhat positive serum test results for IgG antibodies, suggesting that this syndrome results from an abnormal immune response to the virus. 10 The existence of molecular mimicry might be involved. 11
The symptoms resemble both Kawasaki disease and toxic shock syndrome. 12 PIMS-TS is typically characterized by single or multiorgan dysfunction. 2 Several reports suggest many myocardial dysfunctions and coronary artery involvement in addition to systemic symptoms. 13 Less severe presentations, such as abdominal pain, vomiting, and diarrhea, have also been reported. 14 However, this is -to our knowledge- the first case to present with SRD and conjunctival hemorrhage complicating acute glomerulonephritis and hepatic failure. Exudative retinal detachment occurs when fluid accumulates between the neurosensory retina and the retinal pigment epithelium. Ocular diseases or multisystem conditions may lead to exudative retinal detachment. 15 Gass has suggested that the uremic state in patients with renal failure can lead to serous retinal detachment. 16 Acute kidney injury (AKI) is a common finding in PIMS-TS. 17 Estimates from single centers and systemic reviews vary between 10%and 73%. 18 Older age increased markers of inflammation and left ventricular systolic dysfunction were associated with AKI in PIMS-TS. 19 Data of kidney histopathology findings are limited. 20 Renal biopsies from adults with COVID-19 found diverse histological lesions: (1) acute tubular necrosis, (2) tubular interstitial nephritis, (3) thrombotic microangiopathy, (4) immune complex glomerulonephritis, (5) minimal changes disease, or (6) collapsing glomerulopathy. 21 It is not clear if all these observations are the direct effect of SARs-Cov2 infection, the multisystemic inflammatory response, or a preexisting condition. 22
In summary, acute immune complex glomerulonephritis leading to SRD can be part of the multisystemic involvement in PIMS-TS. Steroids are effective in inducing complete remission. More prospective controlled studies and systemic reviews are needed to shed light on the pathophysiology of ocular and renal involvement in PIMS-TS.
Footnotes
Author Contributions
HN: writing the paper. RQ, AB, IA: revision and final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
All data analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.
Statement of Ethics
The parents have given their written informed consent to publish their child’s case.
Ethics Approval
Ethics approval was not required from the ethics committee of Mohammed VI university hospital center of Marrakesh.
