Abstract
The vaccine effectiveness (VE) of childhood measles-mumps-rubella (MMR) vaccine to reduce childhood rubella infections in the US during the 1990s/2000s was undertaken in a retrospective longitudinal cohort study. SAS and StatsDirect software were utilized to examine non-identifiable linked eligibility and claim healthcare records prospectively generated from the Florida Medicaid system in the Independent Healthcare Research Database (IHRD). A total of 33 839 children received a single MMR vaccination (vaccinated) and 44 154 children never received a rubella-containing vaccine (unvaccinated) were continuously eligible from 1990 to 2009 for Florida Medicaid within the first 10 years following birth. Cox proportional hazards models determined VE against diagnosed rubella (ICD-9 code: 056.xx). Children receiving MMR were at significantly reduced risk of rubella in unadjusted (VE = 80.7%, 95% confidence interval = 73.7%-85.8%) and adjusted (VE = 78.6%, 95% confidence interval = 70.8%-84.3%) models as compared to unvaccinated children. Between 1991 and 2009, in the combined vaccinated-unvaccinated cohort examined on a yearly basis, a significant inverse correlation between increasing MMR vaccine population coverage and a decreasing incidence rate of diagnosed rubella was observed. This first large-scale population epidemiological study supports the routine use childhood MMR vaccination to significantly reduce childhood rubella infections and also supports its ability to induce “herd immunity.” This study, coupled with a recently published epidemiological study showing childhood MMR vaccination significantly reduced measles infections, provide powerful epidemiological evidence strongly supporting MMR vaccination as an effective tool to improve public health.
Introduction
Rubella “German measles” is a contagious viral infectious disease that mostly infects school-aged children and young adults. 1 In the pre-vaccine era, the global incidence and prevalence of rubella was high. 1 Symptoms of rubella infection are usually characterized by a low-grade fever, sore throat, and a rash. The rash starts on the face and spreads to the rest of the body. 1 In the United States (US), the last major rubella epidemic occurred from 1964 to 1965 with an estimated 12.5 million persons infected with rubella. 1 Among those infected with rubella, there were more than 2000 encephalitis cases, 11 250 miscarriage cases, and 20 000 congenital rubella syndrome (CRS) cases. 1 The introduction of rubella-containing vaccines in the US in 1969 led to a dramatic decrease in rubella cases. 1
M-M-R® II vaccine (Merck & Co, Inc, Whitehouse Station, NJ, USA) was introduced into routine childhood vaccine programs in the US during the 1980s. Initial MMR vaccination is recommended for children between 12 through 15 months of age by the American Academy of Pediatrics (AAP), the American Academy of Family Practitioners, and the Advisory Committee on Immunization Practices (ACIP). 2 Routine childhood MMR vaccine administration has eliminated continuous rubella transmission in the US, since 2004. It is not medically ethical to undertake placebo-controlled trials to evaluate rubella vaccine effectiveness (VE) among American children. Instead, rubella VE studies must focus on retrospective cohort studies. 3 The purpose of this study was to examine during the 1990s/2000s in the US the impact of childhood MMR VE to reduce the incidence of rubella infections among children in the Florida Medicaid system.
Methods
Independent Healthcare Research Database (IHRD)
As described in previous studies,4-7 non-identifiable healthcare records generated from the Florida Medicaid system compose the Independent Healthcare Research Database (IHRD). The Agency for Health Care Administration (AHCA) of the state of Florida provided the Florida Medicaid eligibility and claim files that were utilized to create the IHRD. The IHRD allows for a person’s eligibility and claim records to be linked by a unique recipient identifier code. The IHRD was examined using the SAS system for Windows, version 9.4 (Cary, NC, USA).
Ethics Approval and Informed Consent
IHRD data were assembled and accessed under approval (No. 11.10.0011) by the Liberty Institutional Review Board (IRB) (Deland, FL). The Liberty IRB determined that the research undertaken in the IHRD did not involve human subjects, and, as such, no informed consent was necessary.
Study Participants
A schematic flowchart of the IHRD data examined is shown in Figure 1. A cohort of 8 440 941 persons with eligibility in Florida Medicaid at specific times from July 1990 to June 2009 was evaluated. These persons were of all ages with specified genders and dates of birth. A total of 1 871 728 children were eligible for Florida Medicaid from their date of birth and a total of 193 453 children were continuously eligible for Florida Medicaid for the initial 10 year period following birth. Among those children with ≥10 outpatient office visits during the initial 10 year period following birth, a sub-cohort of 101 736 children was identified. The identification and selection methods of the cohort children examined were described in previous studies.4-7

A schematic flowchart of the data examined in the present study.
Vaccination Status
The status of vaccination was determined for each cohort member examined from current procedural terminology (CPT) codes filed on claims, as described in previous studies.4,5 Rubella vaccination (codes: 90706, 9947, W1942), MMR vaccine (codes: 90707, W1943), measles and rubella vaccine (codes: 90708, W1944), rubella and mumps vaccine (codes: 90709, W1945), measles, mumps, rubella, and varicella (MMRV) vaccine (code: 90710) were the CPT codes examined in this study. The vaccinated cohort examined in this study included only children receiving a single dose of MMR vaccine (codes: 90707, W1943) during the study period. The unvaccinated cohort in this study included children not receive any rubella-containing vaccine during the study period. Children receiving more than 1 dose of MMR vaccination or other rubella-containing vaccines were excluded from this study. The date of MMR vaccination was determined from the date of service for the first claim in chronological order with a CPT code specifying MMR vaccine.
Outcomes
Outcomes, as described in previous studies, were identified from International Code for Disease, 9th revision (ICD-9) codes for rubella-related diagnoses (code: 056xxx) documented on claims for the children examined in this study.4-7 Laboratory data confirming rubella diagnoses was not available in the IHRD. The onset of rubella infection was assumed to be the date of service for the first claim in chronological order with a rubella diagnosis.
Statistical analyses
SAS and StatsDirect, version 3.3.3 (Merseyside, UK) were utilized, and a 2-sided
Person-years of follow-up began on the date of birth and continued until the end of eligibility (a maximum of 10 years after birth) or until the date of the first rubella diagnosis in the unvaccinated cohort. Person-years of follow-up began on the date of MMR vaccine administration and continued until the end of eligibility (a maximum of 10 years after birth) or until the date of the first rubella diagnosis in the vaccinated cohort.
Cox proportional hazards models in SAS were used to evaluate overtime in years the potential relationship between MMR vaccination and the outcome of a rubella diagnosis. The exact method was utilized for handling ties in failure times. In Model I, no adjustments were made for any covariates. In Model II, adjustment for the covariates of gender (categorical variable), date of birth (continuous variable), and county of residence (continuous variable) were made. Overall, MMR VE against rubella infections was determined as ((1–hazard ratio) × 100).
A regression analysis in StatsDirect was utilized to evaluate in a combined cohort (MMR vaccinated and unvaccinated children) the relationship between the yearly rubella incidence rate and the coverage of MMR vaccine.
Results
The demographic characteristics of the 33 839 MMR vaccinated children and 44 154 unvaccinated children are shown in Table 1. The gender distributions among MMR vaccinated children (male/female ratio = 1.13) and unvaccinated children (male/female ratio = 1.14) were similar. The mean dates of birth were similar among MMR vaccinated children as compared to unvaccinated children. The demographic characteristics of the 375 children diagnosed with rubella examined in this study are shown in Table 2.
Demographic Characteristics of the Children Examined in This Study. a

ICD-9 = International Code of Disease, ninth revision; MMR = measles, mumps, rubella; std = standard deviation.
All children examined in this study were enrolled from their date of birth for 120 consecutive months. All children had non-changing dates of birth and gender status. All children had ≥10 outpatient office visits.
Children received only 1 dose of MMR vaccine and includes only children diagnosed with rubella post-MMR vaccine administration (272 children were excluded with rubella diagnoses pre-MMR vaccine administration).
Children received no doses of any rubella-containing vaccine.
Demographic Summary of the Children Diagnosed With Rubella Examined in This Study. a
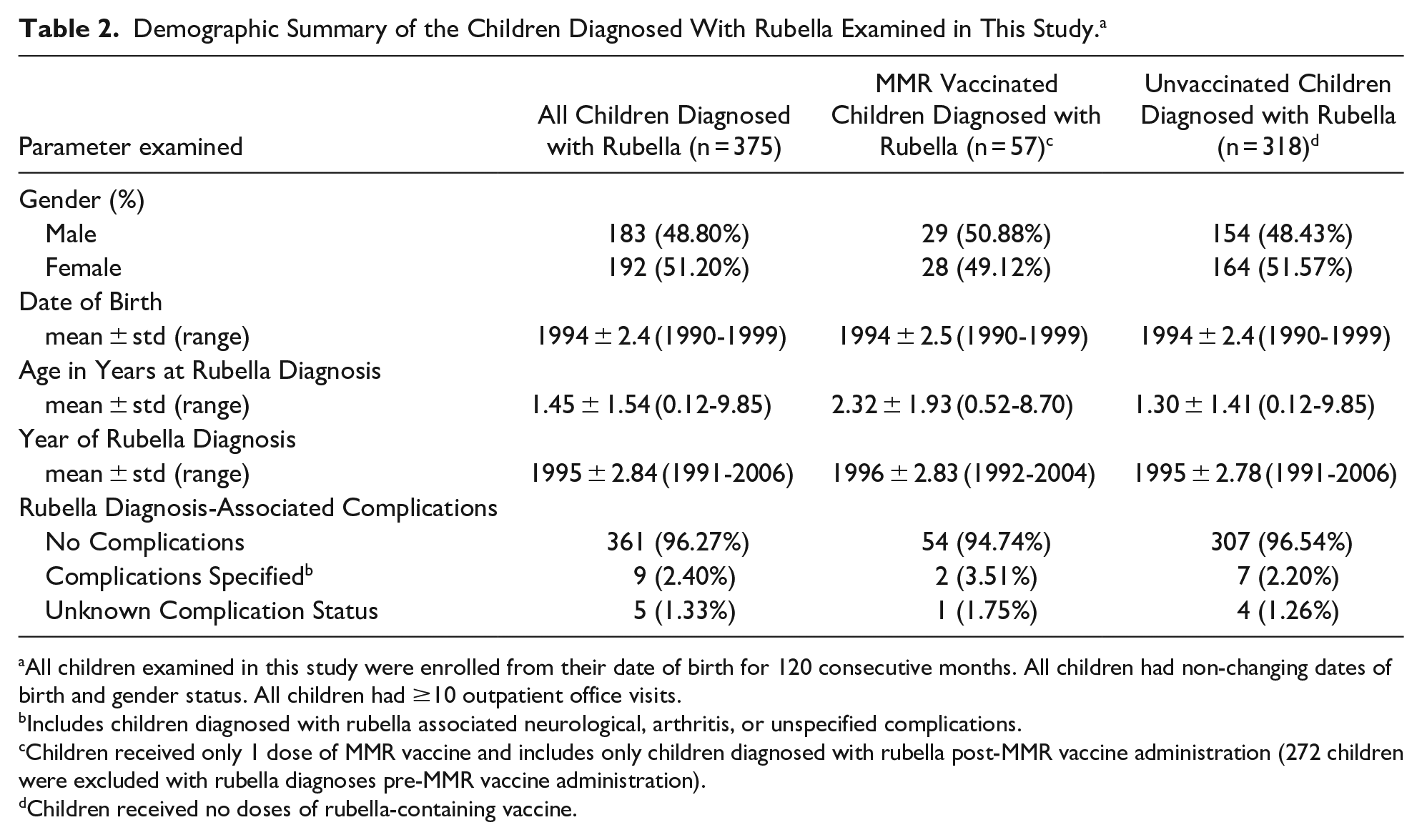
All children examined in this study were enrolled from their date of birth for 120 consecutive months. All children had non-changing dates of birth and gender status. All children had ≥10 outpatient office visits.
Includes children diagnosed with rubella associated neurological, arthritis, or unspecified complications.
Children received only 1 dose of MMR vaccine and includes only children diagnosed with rubella post-MMR vaccine administration (272 children were excluded with rubella diagnoses pre-MMR vaccine administration).
Children received no doses of rubella-containing vaccine.
Table 3 shows the results of the Cox proportional hazards models constructed to determine childhood MMR VE. Childhood MMR VE against rubella infections was 76.1% (95% confidence interval [CI] = 68.3%-82.0%) in the unadjusted and 73.5% (95% CI = 64.9%-80.1%) in the adjusted models. Figure 2 reveals the Cox proportional hazards survival plot generated when examining the incidence rate of rubella over the period of years children were followed in the MMR vaccinated cohort (from age of vaccination) and the unvaccinated cohort (from birth). Figure 2 shows that there was a clustering of diagnosed rubella infections in the initial period post-MMR vaccination (<6 months) as compared to the later period post-MMR vaccination (>6 months) among MMR vaccinated children. In addition, Figure 2 reveals that among unvaccinated children, most rubella infections were diagnosed within the first 2 years of follow-up. This initial period was then followed by a second follow-up period (from the 2nd to 6th year) of more modest increases in diagnosed rubella infections. A final follow-up period (from the 6th to 10 year) with relatively few rubella infections as compared to the first 2 follow-up periods was observed.
Cox Proportional Hazards Model Results Examining the Relationship Between MMR Vaccination and Diagnosed Rubella.

CI = confidence interval, VE = vaccine effectiveness
Children received only 1 dose of MMR vaccine.
Children received no doses of rubella-containing vaccine.
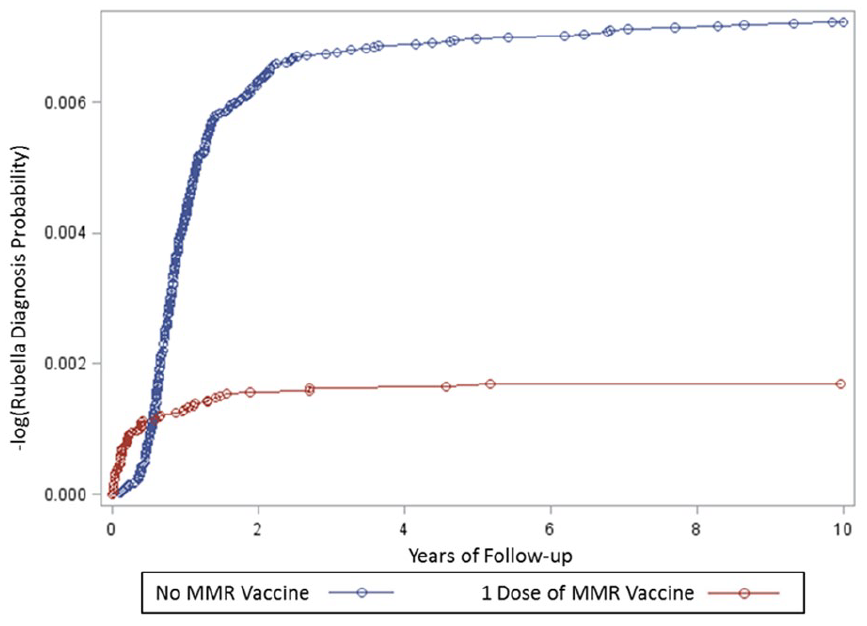
A Cox proportional hazards survival plot evaluating cases of rubella diagnosed over the period of follow-up in the MMR vaccinated cohorta in comparison to the unvaccinated cohortb
Table 4 and Figure 3 summarizes in the yearly incidence rate of diagnosed rubella cases in the combined cohort of children examined (children vaccinated with MMR and unvaccinated children) as compared to the overall coverage of MMR vaccination. Between 1991 and 2009 in the combined cohort examined on a yearly basis, there was a significant increase in MMR vaccine coverage from 0.13% to 43.4% and a significant decrease in diagnosed rubella cases from 28.24 per 10 000 children to 0.0 per 10 000 children. The linear regression statistic revealed that the equation of the trend line was MMR vaccine coverage (%) = −1.69 (diagnosed rubella incidence per 10 000 children) + 38.4 (
A Summary of Diagnosed Rubella Cases by Year of Diagnosis and MMR Vaccine Coverage Among a Combined Cohort of Children (MMR Vaccinated and Unvaccinated Children) Examined in This Study.

Children received only 1 dose of MMR vaccine.
Children received no doses of rubella-containing vaccine.

A summary of the yearly incidence of rubella diagnoses in comparison to the yearly childhood MMR vaccination coverage for the combined vaccinated-unvaccinated cohort examined in the present study.
It was also observed in this study that there were 11 diagnosed rubella cases clustered between 8 and 14 days after MMR vaccine administration. Table 5 summarizes the number of diagnosed rubella cases by day post-MMR vaccination.
A Summary of Diagnosed Rubella Cases by Day post-MMR Vaccination. a

Children received only 1 dose of MMR vaccine (n = 33 839). During the 7 day period from 8 through 14 days post-MMR vaccination as compared to the 76 day period from 15 through 90 days post-MMR vaccination, there was a significant clustering of diagnosed rubella cases (rate ratio = 6.29, 95% confidence interval = 2.70-13.9,
During the 7 day period from 8 through 14 days after MMR vaccine administration as compared to the 76 day period from 15 through 90 days after MMR vaccine administration, there was a significant clustering of diagnosed rubella cases (rate ratio = 6.29, 95% CI = 2.70-13.9,
Cox Proportional Hazards Model Results Examining the Relationship Between MMR Vaccination and Diagnosed Rubella (diagnosed > 14 days post-MMR vaccination).

CI = confidence interval, VE = vaccine effectiveness
Children received only 1 dose of MMR vaccine.
Children received no doses of rubella-containing vaccine.
Discussion
Important and compelling new epidemiological quantitative data regarding childhood MMR VE routinely administered to American children to prevent rubella infections were observed in this retrospective longitudinal cohort study of prospectively collected healthcare data. It was also observed among the combined cohort of children (vaccinated and unvaccinated) examined that as MMR vaccine coverage increased, a corresponding significant decrease in the incidence rate of infections occurred.
MMR VE was examined in a Cochrane Collaboration recently published study. 9 They identified a single cohort study that measured the effectiveness of childhood MMR vaccination against rubella infections during a rubella outbreak in a Chinese school system. 10 The study revealed the risk ratio for rubella infections was significantly reduced among those children receiving childhood MMR vaccination (with different strains of the rubella component) as compared to unvaccinated children (VE = 89%, 95% CI = 56%-95%). The study was rated by the Cochrane Collaborative investigators as offering low certainty evidence regarding the effectiveness of rubella vaccination against rubella infection, but the certainty of the evidence was upgraded 1 level to moderate certainty based upon the large effect size observed in the study. Other investigators reported that increasing population MMR vaccine coverage was associated with significant reductions in the incidence rate of rubella infections. For example, investigators examined the rubella infection rate in Vojvodina, Serbia over a 24 year period. 11 The investigators described in the pre-vaccination period from 1978 to 1993 that there were sporadic rubella cases combined with outbreak cases of rubella. A rubella vaccination program was initiated from 1993 onwards. As the program expanded, it was observed that there was initially a period of localized rubella epidemics from 1993 to 2006, and from 2006 to 2017 there were only sporadic rubella cases. The aforementioned study results are consistent with the present study results.
Childhood MMR VE observed in this study are biologically plausible. Investigators undertook a 10 year follow-up study of anti-rubella antibody titers following a dose of childhood MMR vaccination administered at between 1 and 2 years of age. 12 They observed antibodies against rubella declined moderately in the period post-MMR vaccination, but seropositivity well above the threshold remained after 10 years. It was even suggested that rubella vaccination may induce lifelong antibodies. 13
VE of the rubella to measles component of the MMR vaccine is another interesting finding of this study. The results of this study revealed that the rubella component of MMR vaccine was about 75% effective in the 10 years post-immunization. In a previous study of the IHRD, using similar epidemiological methods, the measles component of MMR vaccine was about 80% effective in the 10 years post-immunization. 4 These observations from the IHRD are consistent with previous hypothetical projections from a large meta-analysis of seroconversion rates among children receiving MMR vaccine and not exposed to wild-type viruses post-immunization. 14 It was estimated in the 35 years post-MMR immunization, the portion of persons that would become seronegative was about 30% for measles and 35% for rubella. Therefore, it was observed in both studies that the measles component relative to the rubella component of MMR vaccination was 5% more effective. The consistency of observations between both studies helps to validate the IHRD and our methodology, while also significantly supporting the effectiveness of MMR vaccination.
Strengths/Limitations
Eligibility and claim records from the Florida Medicaid system prospectively generated on a completely independent basis from the study design employed in this study is an important strength of the current study. The children studied in the IHRD were continuously enrolled within the Florida Medicaid system from birth until 10 years-old (120 consecutive months of enrollment from birth). Children examined in this study were selected for their active utilization of the Florida Medicaid system. The period of enrollment and the requirement of active utilization of the Florida Medicaid system by the children in the cohorts examined are important because they help to minimize factors associated with differences in enrollment and healthcare-seeking behaviors.
Large cohorts of vaccinated and unvaccinated American children were examined in this assessment of the IHRD. It was previously described that there are a substantial number of hard-to-reach under-immunized low-income infants and preschool children that participate in the Medicaid system, and hence, the number of unvaccinated children available for examination in this study. In response to the large unvaccinated populations in the Medicaid system, the US Centers for Disease Control and Prevention (CDC) during the 1990s undertook an aggressive campaign to increase immunization coverage among this population. 15 The result is that the IHRD is relatively unique in its ability to examine VE among American children.
Minimization of statistical chance or unknown confounders/biases is another important strength of this study. A limited number of statistical tests were performed, and most of the results were highly statistically significant, so it was deemed unlikely that statistical chance could explain the results observed. It is also unlikely that unknown confounders/biases could account for the results observed because significant associations remained in the statistical models constructed even when adjusting for several potential covariates. Finally, in this study, the results observed were consistent with previous epidemiological observations on different populations and are biologically plausible.
A limitation of this study is that some children examined in the IHRD had diagnostic or immunization status misclassifications. It is believed that such phenomena should apply equally to both the vaccinated and unvaccinated cohorts examined. Further, any such misclassifications would most likely result in biasing the study findings toward the null hypothesis. In addition, exposure to wild-type rubella may be influenced by potential geographical differences over multiple years. It was assumed that the chances of exposure to wild-type rubella would be similar, but future studies should further investigate this phenomenon in more detail.
Rubella diagnostic status was based upon ICD-9 diagnoses reported by healthcare providers in administrative claim records to the Florida Medicaid system and no information was available regarding whether rubella cases were laboratory confirmed or not. This limitation is important because, as described by the CDC, many rash illnesses can mimic rubella infection, so a clinical diagnosis in the absence of confirmatory lab testing may be unreliable. 16 The CDC reports that in order to confirm acute or recent rubella infection, there are a variety of testing options available, including: the presence of rubella-specific immune globulin (Ig)M antibody, polymerase chain reaction (PCR) detection of rubella virus, or an increase in IgG antibody specific to rubella in acute- and convalescent-phase sera. Despite the lack of access to specific lab test results in the IHRD, it was presumed that diagnosing healthcare providers would have undertaken the appropriate clinical and laboratory testing to confirm a rubella diagnosis for each child. Furthermore, whatever diagnostic ambiguities may exist for a rubella diagnosis among the children examined in the IHRD, it would be expected to occur with equal frequency regardless of vaccination status, and, therefore, not bias the results observed.
The apparent occurrence of the induction of “herd immunity” in this study may have reduced VE. 17 Uniformity in diagnosed rubella cases during the study period was not observed. A potential explanation for this phenomenon is that increasing “herd immunity” as a consequence of increasing MMR vaccine coverage in the overall population examined occurred throughout the study period examined. The data suggest that unvaccinated children were deriving a benefit of protection against rubella infection from vaccinated children. Future studies should further evaluate the impact of increasing “herd immunity” on rubella disease patterns.
A final limitation of this study was that the vaccinated children examined received only 1 dose of MMR vaccine. A second childhood dose of MMR vaccination is currently recommended by the ACIP. 2 It is hypothesized that additional MMR vaccine doses would offer even more significant protection against rubella infections. Future studies should further explore the impact of additional doses of childhood MMR vaccination on VE against rubella infections.
Conclusion
This longitudinal cohort study revealed a significant decline in the incidence rate of rubella infections following a single dose of childhood MMR vaccination among American children over a 20 year period. It should be noted that the current ACIP recommendations call for the routine administration of 2 doses of childhood MMR vaccination. 2 Similarly, another recently published longitudinal cohort study showed the incidence rate of measles infections significantly declined following routine childhood MMR vaccination in American children over a 20 year period. 4 In both studies, it was also observed that increasing childhood MMR vaccine uptake among the cohorts examined was able to induce “herd immunity” with the consequence that ongoing rubella and measles infections were eliminated without the need for 100% of the children receiving a childhood MMR vaccination. Taken together, these 2 new longitudinal cohort studies provide powerful epidemiological evidence strongly supporting MMR vaccination in childhood to control the public health impacts of childhood measles and rubella infections among American children. Future studies should build upon the current results by continuously monitoring children to determine how long MMR vaccine induced immunity persists among vaccine recipients.
Footnotes
Authors’ Contributions
Mark Geier and David Geier undertook study design, conducted the research, and drafted the paper. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported the nonprofit Institute of Chronic Illnesses, Inc. Dr. Mark Geier and Mr. David Geier are directors of the Institute of Chronic Illnesses, Inc.
Ethics Approval and Informed Consent
The data in IHRD were assembled and accessed under approval (No. 11.10.0011) by the Liberty Institutional Review Board (IRB) (Deland, FL). The research undertaken was determined by the Liberty IRB to not involve human subjects, and, as such, no informed consent was necessary.
