Abstract
Hepatitis B is a major global health concern and can be prevented in the era of vaccination. Impaired immunological memory to primary immunization is a common chemotherapy-related complication among cancer survivors. The study aimed to determine protective immunity against hepatitis B virus (HBV) and anamnestic response to booster vaccination. In all, 107 pediatric cancer survivors previously immunized with primary hepatitis B vaccination were enrolled. A hepatitis B booster dose was administered to those with suboptimal seroprotection (anti-HBs < 10 mIU/mL) and 2 additional doses were subsequently administered at 1 and 6 months to those whose anti-HBs remained low. Clinical and serologic parameters were analyzed. Sero-protective rate against HBV (anti-HBs ≥ 10 mIU/mL) among survivors was 20.6% with geometric mean titer (GMT) of 95.7 ± 265.6 mIU/mL. Anamnestic response was 61% after a booster vaccine among those with suboptimal seroprotection and 100% after 2 additional booster doses among those whose anti-HBs remained low. GMTs among those survivors after the First and third booster vaccines were 320.0 ± 412.4 mIU/mL and 826.5 ± 343.8 mIU/mL, respectively. Age at diagnosis was a significant independent risk factor for adequate seroprotection (adjusted OR = 0.84, 95%CI: 0.71-0.99) with a P-value of .034. No associated risk factors to predict optimal anamnestic response to booster vaccination were identified. Loss of immunological memory to primary hepatitis B immunization is an inevitable complication among most pediatric cancer survivors; therefore, assessing adequate seroprotection is essentially required. For those with limited accessibility to serologic tests, completion of full 3-booster-dose series is alternative and highly recommended.
Keywords
Background
Cancer is one of the leading causes of death worldwide; however, the survival rate of childhood cancer has significantly increased over the past decades.1,2 One of the key elements unquestionably contributing to this achievement is improved supportive care not only during cancer therapy but continuously after achieving treatment goals. Cytotoxic chemotherapy is considered an important element of pediatric cancer treatment and has been implemented in most treatment regimens. However, 1 inevitable chemotherapy-related side effect is immune-suppression.3,4 Innate system and adaptive system immunity are profoundly affected from cytotoxicity of the drugs. For this concept principle, many pediatric cancer survivors experience loss of immunological memory and protective acquired immunity from their previous immunization contributing to a higher risk of serious infection.
Hepatitis B virus (HBV) transmission is one of the major public health challenges worldwide in which more than 350 million infected cases were have been reported. 5 The transmission is caused by an enveloped DNA acid virus that infects the liver resulting in hepatocellular inflammation and necrosis. The virus is commonly transmitted from mother to child perinatally as well as through contact with blood and other body fluids from an infected HBV positive person. 6 Chronic HBV infection defined as persistence of hepatitis B surface antigen (HBsAg) for 6 months or more, is another global public health concern. More explicitly, chronically infected individuals could experience lifelong serious and fatal complications including cirrhosis and hepatocellular carcinoma in which nearly 1 million fatal cases were estimated annually.5,7 Unfortunately, the majority of newly infected pediatric cases are asymptomatic with only few of those experiencing mild symptoms that last several weeks; therefore, most cases are unaware of their HBV positive status and often present when they have already developed advanced liver disease.
Comprehensive hepatitis B immunization programs targeting infants with the first dose administered at birth adequately provide protective immunity in up to 90% to 95% of the cases and successfully prevent HBV transmission during the perinatal period and early childhood. 8 The immunization strategy has been globally implemented across health care systems and proven to be highly effective in reducing the incidence of HBV and its transmission among endemic areas specifically in low and middle income countries. The standard hepatitis B immunization regimen includes 3 routine dose vaccinations and birth dose vaccinations. According to this effective immunization regimen, the global proportion of chronically HBV positive children under 5 years of age decreased to under 1% in 2019 compared with 5% during the prevaccination era. 9
In general, clinical and basic laboratory parameters are insufficient to differentiate acute HBV infection from hepatitis caused by other viral pathogens; therefore, more advanced laboratory confirmation is essentially required including detecting HBsAg and immunoglobulin M antibody to the hepatitis B core antigen (HBcAg) or anti-HBc. Hepatitis B e antigen (HBeAg) is a laboratory indicator of high levels of active viral replication and usually found among cases with highly HBV infectivity. 9 For children who are primarily immunized against HBV, protective immunity can be determined by measuring antibodies to HBsAg (anti-HBs) at 1 to 3 months after administering the last dose of the primary immunization series. The anti-HBs concentration of 10mIU/ml or higher is considered a reliable marker of adequate immune protection against HBV transmission. However, significant numbers of cancer survivors who were previously immune-suppressed from prior intensive systemic chemotherapy experience loss of humoral immunity against HBV.10,11 Those particular individuals require hepatitis B booster vaccination with the ultimate goal to restore long lasting protective immunity against the virus.
Changes of immunological memory to hepatitis B vaccination after chemotherapy among pediatric cancer survivors have been reported in several studies12-15; however, anamnestic response to hepatitis B booster vaccination among those specifically selected survivor populations is still not well-characterized. Herein, we conducted a prospective observational study to determine protective immunity against HBV among pediatric cancer survivors as well as anamnestic response to hepatitis B booster vaccination among those survivors who had suboptimal protective titers of anti-HBs. This study was the first study to explore anamnestic response to hepatitis B booster vaccination in this unique and specific survivor population.
Methods
Subject Selection
One hundred seven pediatric cancer survivors followed at the Division of Hematology and Oncology, Department of Pediatrics, Phramongkutklao Hospital from April 30, 2018 to May 1, 2019 were enrolled in this study. The study was also registered and approved by the Thai Clinical Trials Registry (TCTR20201021003). The study’s inclusion criteria included pediatric cancer survivors who were in complete remission status, had complete cessation of chemotherapy or immunosuppressive therapy for 6 months or more and were previously immunized with primary hepatitis B immunization series prior to diagnosis of cancer. Survivors receiving hepatitis B booster vaccination after completion of treatment, experiencing clinical or serologic evidence of hepatitis B infection including positive anti-HBc and/or positive HBsAg or having a history of blood transfusion within 3 months were excluded from the study. The study’s withdrawal criteria included those lost to follow up or any situation in which the survivors or their parents declined to participate in the study.
Ethical Approval and Informed Consent
Written informed consent and assent forms to take part in the study were obtained from all participants including the children themselves as well as their parents or legal guardians before engaging in the study. This prospective cross sectional study was approved by the Institutional Review Board, Royal Thai Army Medical Department according to the ethics principles of the Declaration of Helsinki (1975) and its revision (reference number: IRBRTA 395/2560).
National Hepatitis B Immunization Policy and Strategy
The national immunization strategy against HBV has been implemented across the health care systems in Thailand since 1992. The standard hepatitis B immunization regimen including 3 routine dose vaccinations during infancy and birth dose vaccination is mandatory and has been successfully introduced to all Thai children.
Outcome Measurement
The primary outcome of this study was to determine immunological memory to primary hepatitis B immunization among pediatric cancer survivors previously immunized before diagnosis of cancer. The secondary outcome was to evaluate anamnestic response to hepatitis B booster vaccination among those survivors exhibiting suboptimal protective titers of anti-HBs.
Study Design and Method
The study adhered to the Preferred Reporting items for OBservational studies in Endodontics (PROBE) guidelines. Study schema is shown in the flow diagram in Figure 1. After informed consent and assent were obtained from participating survivors, clinical information including survivor’s age, sex, body weight, nutrition status, history of previous cancer and its treatment and hepatitis B immunization status before diagnosis of cancer were collected. Serum anti-HBs were obtained among all participating survivors on the first visit (day 0). The survivors presenting anti-HBs titer reaching the sero-protective level (anti-HBs titer of 10 mIU/mL or more) were considered having adequate seroprotection against HBV and no longer required further follow-up in the study. On the other hand, those who had presenting anti-HBs titer of <10 mIU/mL were considered having suboptimal seroprotection against HBV and did require a dose of hepatitis B booster vaccination with the repeated anti-HBs titer scheduled on the following visit. On the second visit (month 1), serum anti-HBs was repeated among survivors with low titer levels. The survivors who had increased anti-HBs titer up to 10 mIU/mL or more were considered having sufficient protective anamnestic response against HBV and no longer required further follow-up in the study. Again, those who still had anti-HBs titer of <10 mIU/mL were considered having suboptimal seroprotection against HBV and required two additional doses of hepatitis B booster vaccination administered on that day (second visit or month 1) with the subsequent dose given in the next 5 months (third visit or month 6). In addition, those particular survivors were also required to have anti-HBs titer repeated at 1 month after the last dose of booster vaccination (fourth visit or month 7). The survivors who had increased anti-HBs titer up to 10 mIU/mL or more were considered having sufficient protective anamnestic response against HBV; however, those still presenting anti-HBs titer level of less than 10 mIU/mL were considered nonresponders.

Study flow diagram.
Statistical Analysis
Baseline values of selected variables were analyzed and presented as mean with standard deviation (SD) or median (range) for continuous variables and calculated using frequency and percentage for categorical variables. Comparison between 2 independent data sets were analyzed using Fisher’s exact test for categorical data and independent sample t-test for normally distributed data or Mann–Whitney U test for data not normally distributed. Univariate and multivariate analyses were performed using multiple logistic regression to analyze the prognostic factors to predict adequate protective immunity against HBV defined as anti-HBs titer of 10 mIU/mL or more as well as anamnestic response to hepatitis B booster vaccination. STATA/MP Software, Version 12 (STATA Corp., TX, USA) was used and a P-value <.05 was considered statistically significant.
Results
Survivor Characteristics
Clinical characteristics among 107 participating pediatric cancer survivors including age, sex, body weight, nutritional status, history of previous cancer and its treatment and hepatitis B immunization status before diagnosis of cancer are summarized in Table 1. Most participating survivors received an initially diagnosis of cancer at pre-school ages and their status at enrollment ranging from childhood to adulthood with a median duration from diagnosis to enrollment of 7.1 years. Males were more predominant than females at a ratio of 1.6:1. The most common cancer type was leukemia resembling a typical cancer distribution among children followed by solid tumors and lymphoma, in rank. Most survivors were from urban areas, presented normal nutritional status and already completed 3 doses of hepatitis B vaccination before initiating chemotherapy. Two survivors were lost to follow-up after their first visit and 1 was lost to follow-up after the second visit.
Survivor Demographic Data.

Data are presented as mean ± SD and median (range) for continuous variables and number (%) for categorical variables.
Geometric Mean Antibody to Hepatitis B Surface Antigen (anti-HBs) Titer and Sero-Protective Rate
Protective immunity against HBV was measured and reported as a geometric mean titer (GMT) of anti-HBs. Interestingly, the overall GMT among pediatric cancer survivors enrolled in this study was 95.7 ± 265.6 mIU/mL with sero-protective rate against HBV (anti-HBs titer of 10 mIU/mL or more) of 20.6% Table 2. A hepatitis booster vaccination was administered to the survivors who had suboptimal seroprotection to HBV (n = 83), and GMT measured at 1 month after the booster dose was 320.0 ± 412.4 mIU/mL. Furthermore, three fifths (61.4%) of those successfully showed anamnestic response to the booster vaccine. According to the study’s protocol, described in Figure 1, those remaining survivors who still had suboptimal seroprotection to HBV (n = 31) despite a dose of booster vaccine were subsequently given 2 additional hepatitis B booster vaccinations in which their GMT measured at 1 month after the last dose of booster vaccination was 826.5 ± 343.8 mIU/mL. Surprisingly, all of those achieved optimal immune protection against HBV.
Geometric Mean Antibody to Hepatitis B Surface Antigen (Anti-HBs) Titer and Sero-Protective Rate.

Data are presented as mean ± SD and median (range) for continuous variables and number (%) for categorical variables.
Two survivors were lost to follow-up after first visit.
One survivor was lost to follow-up after second visit.
Association between Different Cancer Types and Optimal Protective Immunity against HBV
Among 107 participating pediatric cancer survivors with various cancer diagnoses and treatments, association between their previous underlying cancer type and adequate protective immunity against HBV posttreatment was explored. However, no statistically significant difference of sero-protective anti-HBs was found among different types of cancer including leukemia, lymphoma, solid tumors and histiocytosis as well as among the cancer subtypes as shown in Figure 2A to D. Although insignificant differences were observed, more than one half of the survivors previously receiving a diagnosis of leukemia including acute lymphoblastic leukemia (ALL) and acute myeloid leukemia and all enrolled survivors receiving a previous diagnosis of germ cell tumor, Hodgkin lymphoma and osteosarcoma were found to have suboptimal immune protection against HBV as shown in Figure 2B to D.
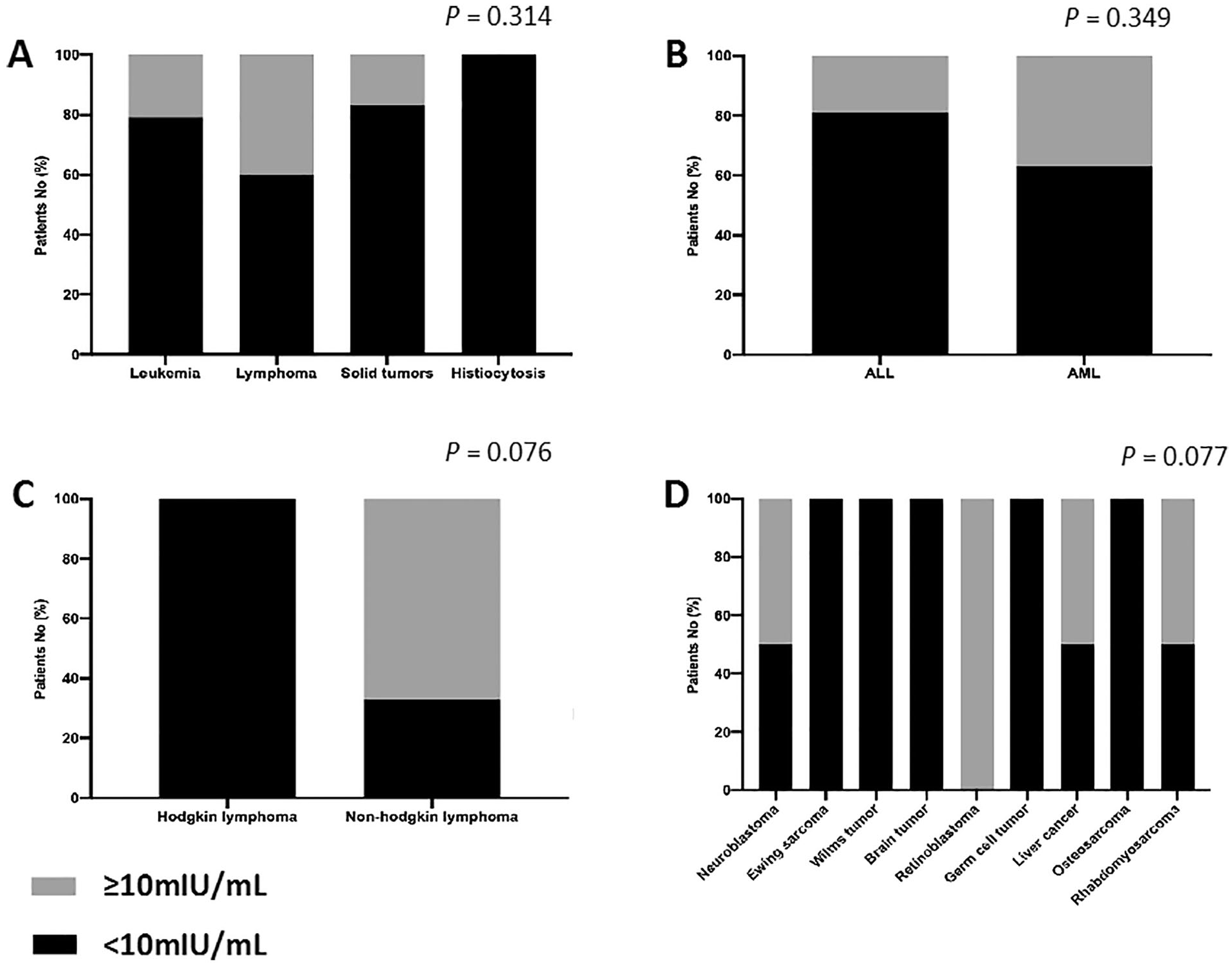
Protective immunity against HBV among different cancer types. (A) All cancer types (B) Leukemia types. (C) Lymphoma types. (D) Solid tumor types.
Associated Risk Factors to Predict Adequate Seroprotection against HBV
Associated risk factors, potentially contributing to sufficient protective immunity against HBV defined as an anti-HBs titer of 10 mIU/mL or more among enrolled pediatric cancer survivors, were evaluated using multiple logistic regression analysis as shown in Table 3. Interestingly, age at diagnosis was found to be a significant independent risk factor for adequate seroprotection (adjusted OR = 0.84, 95%CI: 0.71-0.99) with a P-value of .034. Neither survivors’ sex, nutritional status, underlying disease nor time intervals during or after cancer treatment were found to be predictive factors for optimal protective immunity against HBV. Moreover, additional potential predictive factors for an adequate anamnestic response after a dose of booster vaccination among survivors with suboptimal anti-HBs titers were further analyzed as shown in Table 4. Neither survivors’ age at diagnosis, sex, nutritional status, underlying disease, time intervals during or after cancer treatment nor anti-HBs titers at first visit were found to be influential factors in predicting optimal protective immunity against HBV.
Potential Associated Risk Factors for Adequate Seroprotection Against HBV.
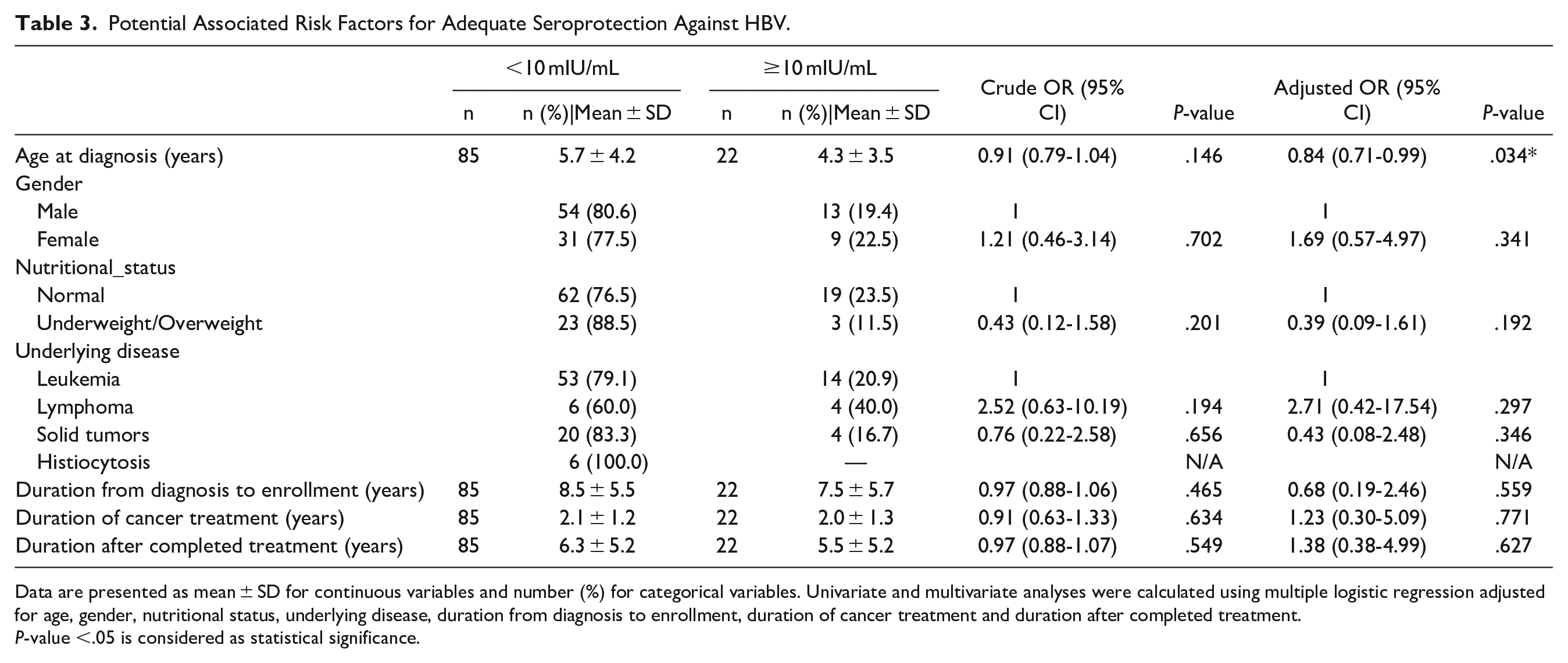
Data are presented as mean ± SD for continuous variables and number (%) for categorical variables. Univariate and multivariate analyses were calculated using multiple logistic regression adjusted for age, gender, nutritional status, underlying disease, duration from diagnosis to enrollment, duration of cancer treatment and duration after completed treatment.
P-value <.05 is considered as statistical significance.
Potential Associated Risk Factors for Adequate Anamnestic Response to a Dose of Hepatitis B Booster Vaccination.
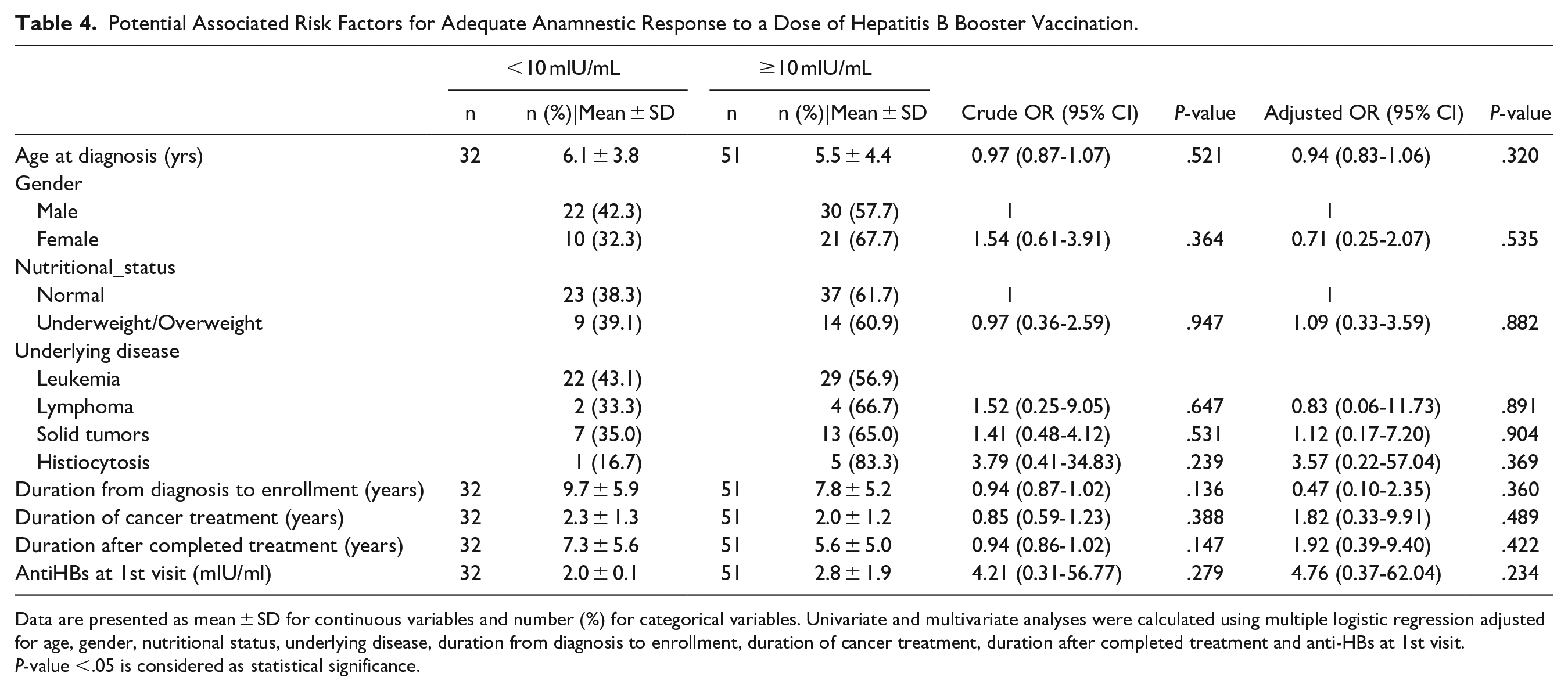
Data are presented as mean ± SD for continuous variables and number (%) for categorical variables. Univariate and multivariate analyses were calculated using multiple logistic regression adjusted for age, gender, nutritional status, underlying disease, duration from diagnosis to enrollment, duration of cancer treatment, duration after completed treatment and anti-HBs at 1st visit.
P-value <.05 is considered as statistical significance.
Discussion
Immune suppression is a common unavoidable side effect from systemic chemotherapy among patients with cancer. Defective innate and adaptive immune mechanisms could adversely impair immune protection against foreign pathogens subsequently resulting in increased susceptibility to severe infection. In addition to the diminished defensive immunological property, remote sero-protective memory, passively developed from preceding primary immunization series, administered during infancy before initiating chemotherapy could also be affected, in which this defectivity has been noticeable in a certain number of patients. Although loss of protective immunity to primary immunization is a common foreseen complication among patients with cancer as well as survivors, the condition has been unfortunately overlooked by many healthcare professionals given no apparent clinical findings specifically related to impaired immune protection and the complexity of unique and specific laboratory tests required to confirm diagnosis. Hepatitis B status is considered one of the most common global health concerns especially in developing countries; however, a gradual decrease of disease incidence has been observed after introducing hepatitis B immunization administered during early infancy.16,17 Despite high, protective anti-HBs antibody levels of more than 95% after completing primary immunization, persistent protective immunity followed up at 20 years was significantly decreased measuring as low as 37%. However, high anamnestic response to a booster hepatitis B vaccine among those immunocompetent subjects was observed, in which the restoration of protective anti-HBs antibody levels up to 84% to 95% could be achieved.18-22 Unfortunately, the anamnestic response to a booster hepatitis B vaccine among pediatric cancer survivors remains poorly understood. Regarding our knowledge, this constitutes the first study to characterize anamnestic response to hepatitis B booster vaccination in this specific cancer survivor population.
One hundred and seven pediatric cancer survivors were enrolled in this study and anti-HBs titer level was measured. Although newly developed transitional B cells and naïve B and T cells among cancer survivors are rapidly reconstituted within 1 month after completing chemotherapy, the recovery of different memory B and T cell subpopulations is delayed, in which those specific immune cells might still not be completely reconstituted despite continuous follow-up to 5 years. 23 Loss of immunological memory to primary hepatitis B immunization has previously been studied and reported among pediatric cancer survivors with the sero-protective rate against HBV ranging from 54% to 80%.13-15 However, our study and the recent study from Fayea et al 12 found a lower proportion of survivors having seroprotection as low as 21% and 14%, respectively. This discrepancy might be related to the differences in the intensity of chemotherapeutic regimen between different time intervals. In addition, survivors with a previous diagnosis of ALL was reported to have significantly decreased protective immunity against HBV with the sero-protective rate of approximately 40% 13 ; however, the lower sero-protective rate of 20% among pediatric ALL survivors was observed in this study. Again, this inferiority of immune protection might represent higher intensive steroids containing a chemotherapeutic regimen for childhood ALL in this current era. This information addresses the need to assess adequate seroprotection against HBV in all pediatric cancer survivors.
As previously mentioned, a dose of booster vaccination could effectively restore protective immunity in immunocompetent subjects who had loss of immunological memory to primary immunization.18-22 Interestingly, only 61% of pediatric cancer survivors could achieve adequate anamnestic response after receiving a dose of hepatitis B booster vaccination. However, 2 additional doses of hepatitis B booster vaccination subsequently administered to those whose anti-HBs remained low successfully restored optimal protective immunity against HBV in all remaining survivors. Regarding the above findings, anti-HBs titer should be rechecked among all pediatric cancer survivors with suboptimal protective immunity (anti-HBs < 10 mIU/mL) receiving a dose of booster vaccination, and 2 additional booster vaccines should absolutely be administered to all survivors whose anti-HBs remained low. Concerning certain difficult circumstances such as survivors having financial limitation and unable to pay for the serologic test or some institutions where accessibility to the test is limited, completing the full 3-booster-dose series pro-actively is an alternative and highly recommended to completely restore protective immunity against HBV.
Unlike the studies from Karaman et al 13 and Zignol et al, 15 age at diagnosis was found to be a significant independent risk factor for adequate seroprotection in this study. Explicitly, the older the age at diagnosis of cancer, the lower would be the likelihood for adequate seroprotection against HBV after completing treatment. This finding could be explained from physiologic decline of vaccine-induced immunity, which has been waning overtime. 24
The limitation of this study included the small sample size of participating pediatric cancer survivors, which might have contributed insignificant results. In addition, data obtained from the unique and specific populations in this study might not be generally applicable among cancer survivors at different age ranges.
Conclusion
Loss of immunological memory to primary hepatitis B vaccination among pediatric cancer survivors is an important chemotherapy-related complication, and may be more common than previously estimated. Therefore, adequate seroprotection against HBV should be assessed in all survivors. Although only just over one half of the survivors developed anamnestic response to a dose of booster vaccination, 2 additional booster doses given at 1 and 6 months after the first booster dose were found to be highly effective and should be administered to all remaining survivors with suboptimal protective anti-HBs titers. For certain circumstances in which accessibility to the serologic test is limited, completing the full 3-booster-dose series pro-actively would provide a high chance of completely restoring immune protection against HBV.
Footnotes
Acknowledgements
The funding from the Phramongkutklao College of Medicine, Royal Thai Army was used to conduct the study, analyze and interpret the study results and submit the study for publication. The authors would like to thank the patients and families for participating in the study.
Authors’ Contributions
DS involved in patients’ care, collected, analyzed, interpreted the patient’s data and wrote the manuscript; WT involved in patients’ care, collected, analyzed and interpreted the patient’s data; CT involved in patients’ care, analyzed and interpreted the patient’s data; PR involved in patients’ care, collected, analyzed, interpreted the patient’s data and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
