Abstract
Early recognition and management are the key elements to prevent febrile neutropenia associated mortality. The prospective observational study aimed to investigate prognostic accuracy of serum lactate to predict septic shock within 48 hours among hemodynamically stable children with febrile neutropenia. In all, 99 pediatric oncology patients who developed febrile neutropenia were enrolled in the study. Clinical information during 48 hours and serum lactate at the time of enrollment were analyzed. Among 99 participating patients, 10 developed septic shock and 4 of those expired. No significant difference was found of patients’ baseline characteristics and basic laboratory parameters between patients with and without septic shock. Serum lactate was significantly elevated among patients developing septic shock (P-value < .001) and those who expired (P-value .002). Receiver operating characteristic (ROC) curve was created to identify the best cutoff value for initial serum lactate associated with the development of septic shock within 48 hours. Baseline serum lactate more than 2.5 mmol/L showed the largest area under the ROC curve to predict the septic shock development within 48 hours (ROC area, 0.90; 95% confidence interval [CI], 0.81-0.98), with sensitivity, specificity, negative predictive value, and accuracy of 80.0%, 92.1%, 97.6%, and 90.9%, respectively. Serum lactate level determined early at the time of febrile neutropenia was an effective surrogate marker for developing septic shock within 48 hours among hemodynamically stable, pediatric oncology patients. The level more than 2.5 mmol/L was the best threshold to start preemptive aggressive hemodynamic monitoring and prompt treatment to ensure adequate tissue perfusion.
Background
The survival rate of childhood cancer has dramatically increased over the past decades. 1 Improved supportive care throughout the entire duration of cancer treatment is one of the key elements significantly contributing to this success. Systemic chemotherapy is considered the backbone of treatment and has been incorporated in most pediatric cancer treatment regimens. However, the chemotherapy’s toxicity to hematopoietic cells could result in decreased numbers of immune cells contributing to immune-suppression and a higher risk of serious infections.2,3
Infection is the leading cause of mortality among pediatric patients with cancer undergoing systemic chemotherapy accounting for 60% to 80% in leukemia, 50% to 60% in lymphoma and 40% to 50% in solid tumor cases. 4 Severe sepsis and septic shock are life-threatening complications caused by immune and coagulation responses to an infection in which pro-inflammatory and anti-inflammatory cytokines produced in response to inflammation could contribute to systemic vasodilation (hypotension), impaired myocardial contractility, endothelial damage, and activation of the coagulation system.5,6 The most common etiology for sepsis is occult bacteremia from gram-positive and gram-negative bacterial pathogens. 7 Risk factors for severe sepsis associated with death are presence of comorbidity especially uncontrolled blood sugar, vital signs instability, severity and site of infection, delayed empiric antibiotic administration and inadequate intravenous fluid resuscitation.8,9 In addition, patient’s immune status is another determining factor for dismal outcomes for those patients developing severe sepsis. Neutrophil is a type of white blood cell that plays a major role in innate immune response. 10 The cell is considered as the first line defense against infection and its functions are mainly mediated by phagocytosis and release of cytotoxic granules. 11 Low numbers of circulating absolute neutrophil count (ANC) especially among patients with cancer undergoing chemotherapy is significantly correlated with the increased risk of infection-related mortality.12,13 Therefore, early recognition and management of this urgent circumstance is critical to prevent worsening clinical course contributing to increased morbidity and mortality as well as delayed next chemotherapy cycles and necessary dose reductions of chemotherapy, which could compromise efficacy of treatment and increase risk of disease relapse. Although investigation and management guidelines for patients with febrile neutropenia have been implemented in most cancer treatment centers, the practical algorithms especially in pediatric populations differ and vary among institutions.14 -17
Serum lactate is a laboratory parameter that plays an important role in determining the status of acid-base homeostasis in the body and adequacy of tissue oxygenation and perfusion. This laboratory marker is increasingly used to assess and monitor clinical severity among patients with suspected sepsis developing tissue hypoperfusion, multiple organ dysfunction or hypotension. 18 Under normal physiologic conditions, lactate is rapidly eliminated by the liver with normal serum lactate level measured less than 1 mmol/L. High serum lactate level also correlated with increased mortality when incorporated with clinical hypotension. 19 Furthermore, elevated serum lactate level above 2 to 3 mmol/L was found to be an excellent predictor for septic shock to occur in adult populations.20,21 Therefore, serum lactate level >2 mmol/L has recently been used as one of clinical criteria to define septic shock and the level is now routinely obtained at the time when septic shock is suspected. 22 However, only few studies have explored the additional role of serum lactate as a surrogate marker to predict developing septic shock specifically among children with cancer developing febrile neutropenia. 23
Herein, we conducted a prospective observational study to determine clinical usefulness of serum lactate level in predicting septic shock within 48 hours specifically among hemodynamically stable, pediatric oncology patients developing febrile neutropenia, and which optimal cut off level of serum lactate should be established.
Methods
Patient Selection
Ninety-nine pediatric oncology patients undergoing chemotherapy at the Division of Hematology and Oncology, Department of Pediatrics, Phramongkutklao Hospital from January 1, 2019 to January 31, 2020 were enrolled in this study. The study was registered and approved by the Thai Clinical Trials Registry (TCTR20200716002). The study’s inclusion criteria included patients aged less than 18 years undergoing chemotherapy for cancer treatment developing febrile neutropenia defined as a single oral temperature measurement of greater than or equal to 38.3°C, a temperature of greater than or equal to 38.0°C sustained over 1 hour along with ANC of lower than 500 cells/mm3 or lower than 1000 cells/mm3 with a trend to be lower than 500 cells/mm3 in 48 hours. The study’s exclusion criteria consisted of patients hemodynamically unstable at the beginning of a febrile neutropenic episode or experiencing prolonged fever more than 72 hours.
Ethical Approval and Informed Consent
Written informed consent and assent forms to take part in the study were obtained from all participants including the children themselves as well as their parents or legal guardians before engaging in the study. This prospective study was approved by the Institutional Review Board, Royal Thai Army Medical Department according to the ethics principles of the Declaration of Helsinki (1975) and its revision (reference number: IRBRTA 152/2561).
Outcome Measurement
The primary outcome of this study was to investigate the prognostic accuracy of serum lactate level to predict the occurrence of septic shock within 48 hours after developing febrile neutropenia and which optimal cut off level of serum lactate should be established. The secondary outcomes were to determine the association between serum lactate level and mortality risk and to identify additional determinant factors associated with septic shock. Sepsis is defined according to the International Pediatric Consensus Conference, and septic shock was characterized as sepsis with the presence of hypotension (<5th percentile for age or systolic blood pressure, <2 SD below normal for age) with need for vasoactive agents. 24
Study Design and Method
The study adhered to the Preferred Reporting items for Observational studies in Endodontics (PROBE) guidelines. Study schema is shown in the flow diagram in Figure 1. After informed consent and assent were obtained from participating patients, clinical information including patient’s demographic data and the 4 main vital sign measurements (body temperature, respiratory rate, heart rate, and blood pressure) were collected at the time of developing febrile neutropenia. Serum lactate level was also drawn in addition to our usual septic workup investigational studies for children experiencing a febrile neutropenic episode consisting of complete blood count (CBC), electrolytes, blood urea nitrogen, creatinine, liver function test as well as blood and urine cultures. According to our institutional standard procedures, all patients with febrile neutropenia were hospitalized and empiric antibiotics were administered intravenously as soon as specimen cultures were obtained; however, antibiotic regimens varied among each individual patient depending on his/her clinical information and the judgment from each infectious disease specialist. Participating patients were then closely monitored clinically and their clinical and laboratory data were collected for the next 48 hours after enrolling in this study.

Study flow diagram.
Statistical Analysis
Baseline values of serum lactate and other selected variables were analyzed and presented as mean with standard deviation (SD) or median (range) for continuous variables and calculated using frequency and percentage for categorical variables. Comparisons between two independent data sets were analyzed using Fisher’s exact test for categorical data and Mann-Whitney U test for continuous data. Receiver operating characteristic (ROC) curve to predict septic shock development was generated based on the serum lactate level. An ROC curve is a graphical plot of sensitivity (true positive rate) versus false positive rate (1—specificity; or 1—true negative rate) to identify the optimal cut-off point. In this study, the curve represents values of sensitivity and specificity for each measurement of serum lactate level at the time of developing febrile neutropenia, and the optimal cut off point for developing septic shock was explored based on the level. The area under the ROC curve was calculated using the extended trapezoidal rule. 25 STATA/MP, Version 12 Software (STATA Corp., TX, USA) was used and a P-value < .05 was considered statistically significant.
Results
Patient Characteristics
Patient characteristics including age, sex, weight, BMI, vital signs, cancer type, types of chemotherapy, and empiric antibiotics administered, associated symptoms and initial investigational results are summarized in Table 1. Most participating patients were older children at pre-adolescent age. Females were slightly more predominant than males at a ratio of 1.1:1. Patients with various types of cancer were enrolled in this study. The most common cancer type was acute lymphoblastic leukemia (ALL) resembling a typical cancer distribution among pediatric patients. Neuroblastoma and osteosarcoma were the second and third most common cancer types, respectively, due to their requirement for highly intensive treatment associated with more potent immunosuppressive effects resulting in a higher risk of febrile neutropenic complications. All patients underwent various types of chemotherapeutic regimens. Upon developing a febrile neutropenic episode, one half of the participating patients were noted to have localizing symptoms including upper respiratory symptoms, gastro-intestinal symptoms and mucositis, respectively. Empiric antibiotic regimens administered were broad-spectrum covering both gram-positive and gram-negative bacterial organisms. Most patients experienced pancytopenia although some, with a diagnosis of acute lymphoblastic leukemia and disease, relapsed and were noted to have leukocytosis with prominent peripheral leukemia blasts. In addition, serum lactate level is described in Table 1.
Patient Demographic Data.
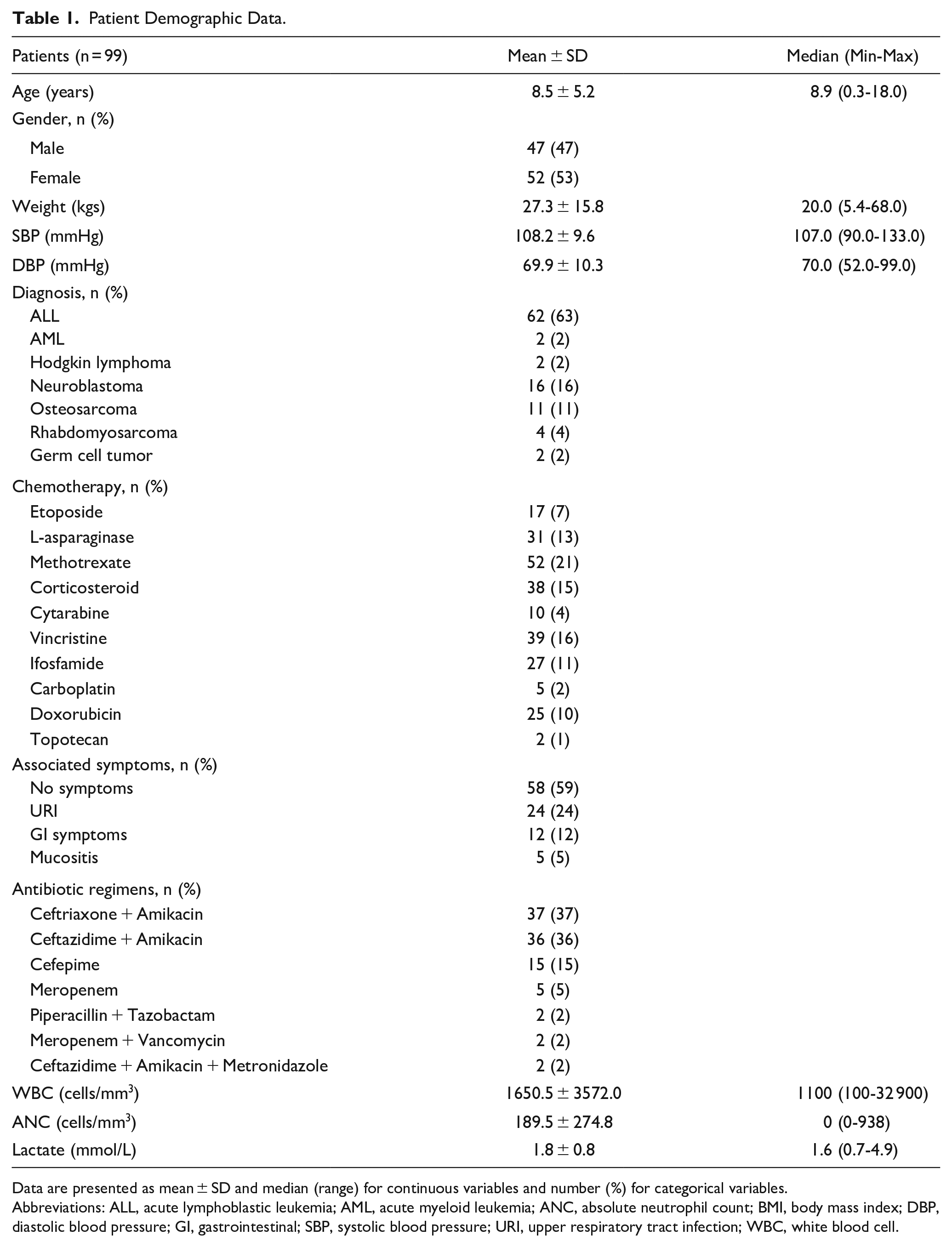
Data are presented as mean ± SD and median (range) for continuous variables and number (%) for categorical variables.
Abbreviations: ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ANC, absolute neutrophil count; BMI, body mass index; DBP, diastolic blood pressure; GI, gastrointestinal; SBP, systolic blood pressure; URI, upper respiratory tract infection; WBC, white blood cell.
Associated Factors among Febrile Neutropenic Patients with and without Shock
Among 99 hemodynamically stable patients with febrile neutropenia enrolled in this study, 10 developed septic shock within 48 hours after febrile neutropenic episodes. Despite aggressive resuscitation and treatment, 4 of those patients with septic shock clinically deteriorated and expired. As shown in Table 2, serum lactate level and various associated factors were explored between participating patients experiencing septic shock and those who did not. Interestingly, serum lactate level obtained at the febrile neutropenic episodes was significantly elevated among patients developing septic shock within 48 hours (mean serum lactate level 3.1 ± 1.0 mmol/L) compared with those not experiencing septic shock (mean serum lactate level 1.8 ± 1.7 mmol/L) with a P-value of <.001, as shown in Figure 2a. In addition, serum lactate level obtained at the febrile neutropenic episodes was also significantly elevated among patients who expired within 48 hours (mean serum lactate level 3.5 ± 1.3 mmol/L) compared with those still alive (mean serum lactate level 1.9 ± 1.7 mmol/L) with a P-value of .002, as shown in Figure 2b. Isolated pathologic organisms from blood cultures were also found significantly more than those among patients developing septic shock (70%) compared with those who did not (4.5%) with a P-value of <.001 in which pathologic organisms isolated for both blood and urine cultures are described in Table 3.
Associated Factors among Patients with Febrile Neutropenia With and Without Shock.

Data are presented as mean ± SD and median (range) for continuous variables and number (%) for categorical variables. Comparison between two independent data sets was analyzed using Fisher’s exact test for categorical data and Mann-Whitney U test for continuous data; *P-value < .05 was considered as statistically significant.
Abbreviations: ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ANC, absolute neutrophil count; BMI, body mass index; DBP, diastolic blood pressure; GI, gastrointestinal; SBP, systolic blood pressure; URI, upper respiratory tract infection; WBC, white blood cell.
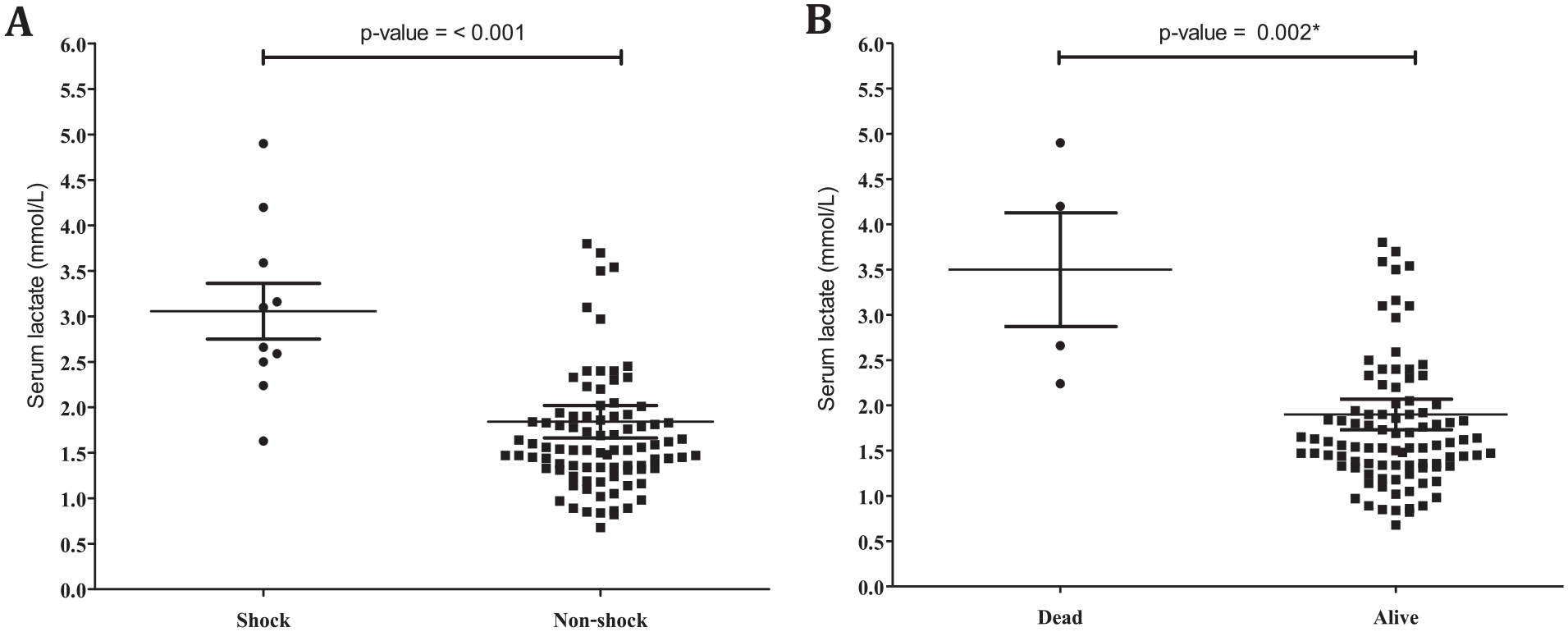
Serum lactate levels (mmol/L) obtained at the time of febrile neutropenic episodes among hemodynamically stable patients. (A) Patients developing septic shock within 48 hours versus those who did not. (B) Patients expiring within 48 hours versus those who were alive.
Isolated Organisms from Blood and Urine Cultures.
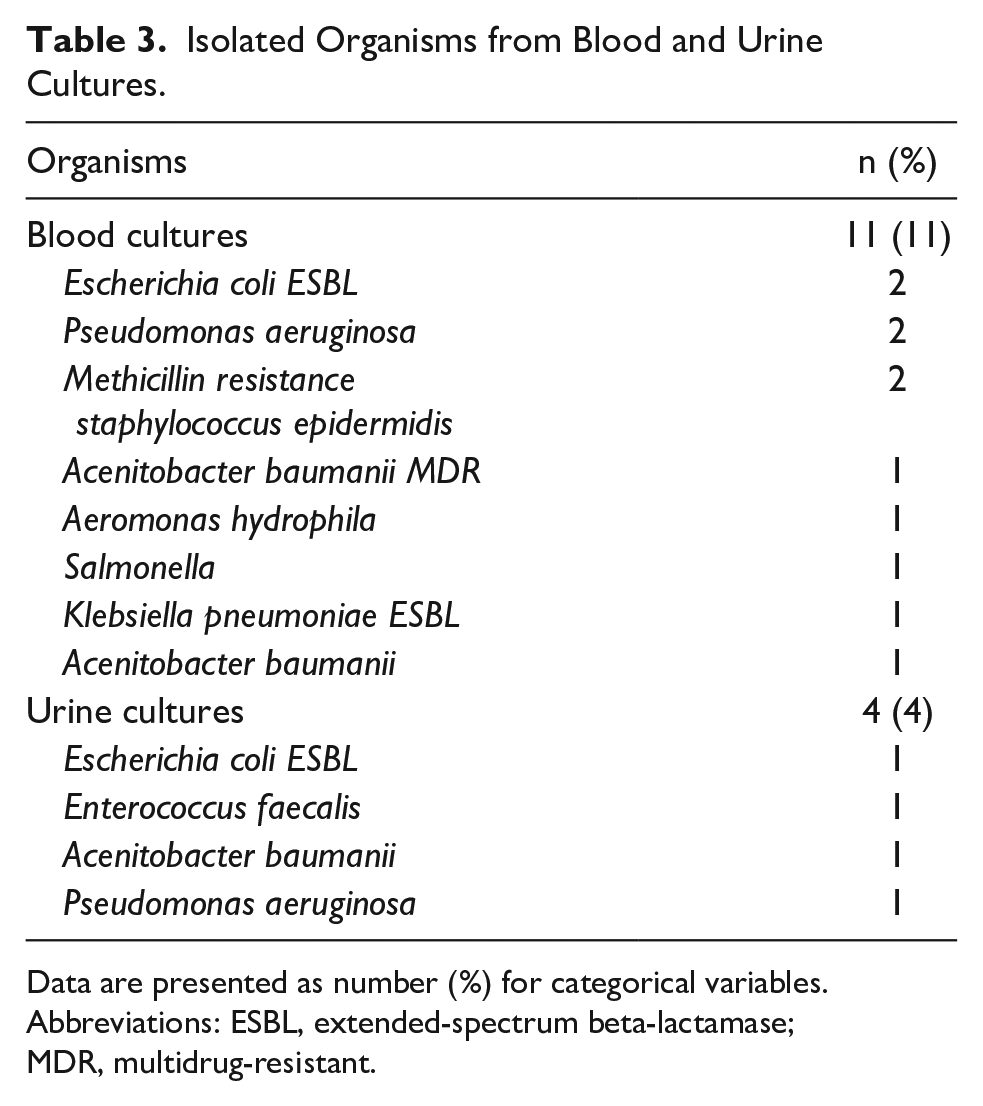
Data are presented as number (%) for categorical variables.
Abbreviations: ESBL, extended-spectrum beta-lactamase; MDR, multidrug-resistant.
Optimal Cut-Off Calue of Serum Lactate Level for Developing Septic Shock
The ROC curve was used to define the optimal cut-off value of serum lactate level that may predict developing septic shock within 48 hours after febrile neutropenic episodes among hemodynamically stable patients. The AUC of the resultant ROC curve was 0.90 [standard error = 0.044, 95% confidence interval (CI) = 0.81-0.98] as shown in Figure 3, which confirmed that serum lactate level had a good ability to correctly classify septic shock development within 48 hours after febrile neutropenic episodes. Patients were dichotomized according to the chosen serum lactate cut off, and sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) were then calculated. Statistically, the serum lactate level of 2.5 mmol/L was found to be the optimal predictor for developing septic shock with a sensitivity of 80.0% (95% CI = 44.4-97.5), specificity of 92.1% (95% CI = 84.5-96.8) and accuracy of 90.9%. On the other hand, a traditional cut-off 2.0 mmol/L on serum lactate level for developing sepsis had a sensitivity of 90.0% (95% CI = 55.5-97.0), specificity of 78.7% (95% CI = 68.7-86.6), PPV of 53.3% (95% CI = 26.6-78.8), NPV of 97.6% (95% CI = 91.7-99.7) and accuracy of 79.8%.
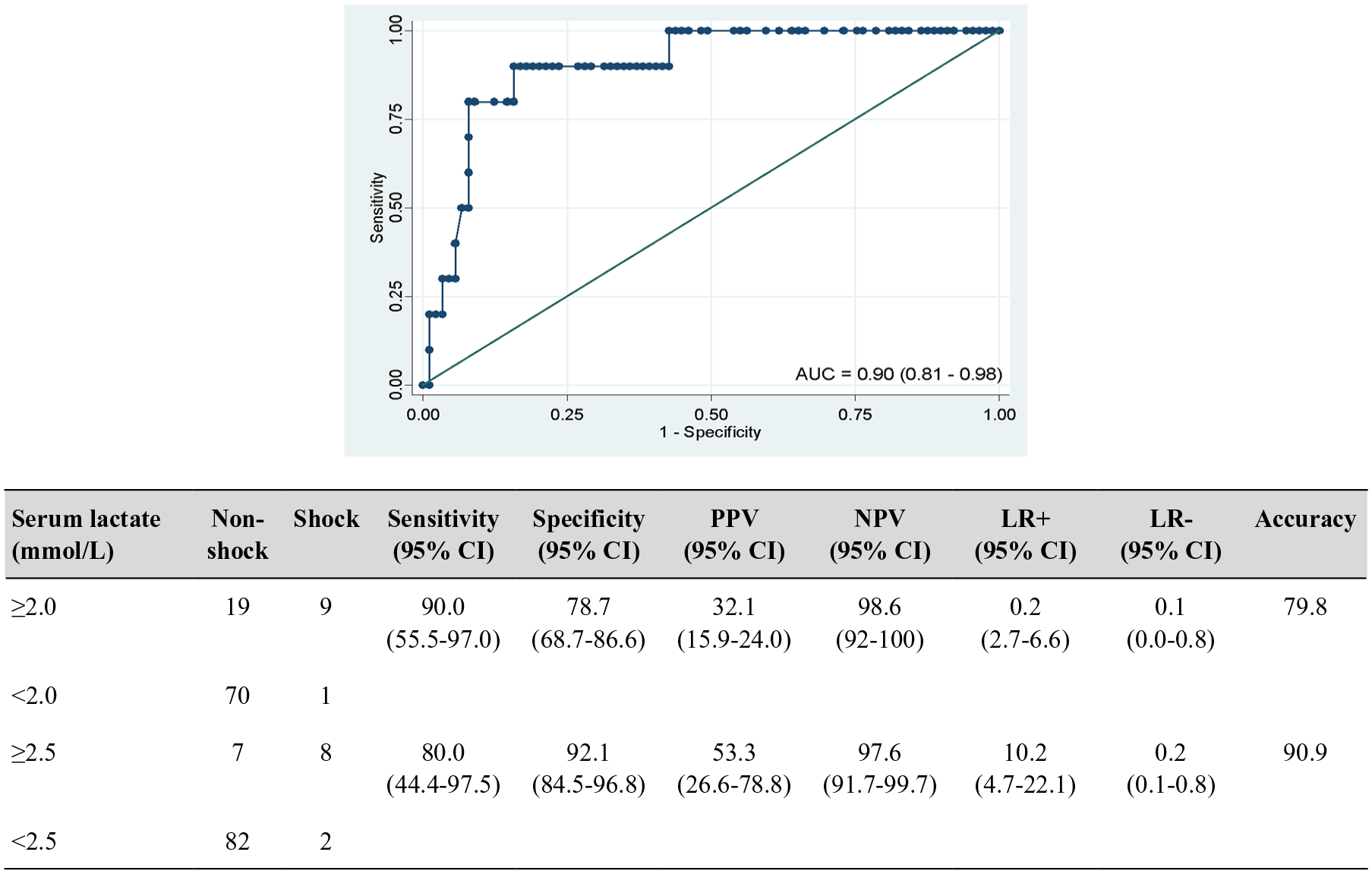
ROC curves to predict developing septic shock within 48 hours based on the serum lactate level. The curve represents values of sensitivity and specificity for each measurement of serum lactate level at the time of developing febrile neutropenia. The AUC was 0.90.
Discussion
Febrile neutropenia is a life-threatening complication in cancer treatment, 26 which could contribute to neutropenic sepsis and septic shock. 27 Several potential risk factors associated with high fatality among patients with febrile neutropenia include patient’s underlying physical conditions, high virulent and multidrug resistant organisms, delayed recognition of the complication and inappropriate antimicrobial treatment. 28 Despite the advancement in empiric antibiotic regimen, many patients are delayed in recognizing this condition and eventually progress to septic shock and death. Therefore, identifying the determinant factors to predict developing septic shock upfront among hemodynamically stable patients with febrile neutropenia is essentially required to prevent this serious condition from happening. Serum lactate is a sensitive marker typically elevated at the time of developing septic shock.18,19,21 However, the additional role of this biomarker in predicting developing septic shock among hemodynamically stable, pediatric oncology patients developing febrile neutropenia remains poorly defined as well as for which optimal cut off levels should be established.
Herein, we conducted a prospective observational study to determine clinical usefulness and prognostic accuracy of serum lactate level as an effective predictor of septic shock within 48 hours among hemodynamically stable, pediatric oncology patients developing febrile neutropenia. Ninety-nine children with cancer experiencing febrile neutropenic episodes were enrolled and involved to the end of the study with no lost follow-up cases. Most patients with neutropenia were classified at severe stage (ANC less than 500 cells/mm3) and had other cell lines (red blood cells and platelets) suppressed. Patients’ demographic data and characteristics including initial vital sign measurements, underlying diagnosis, associated symptoms beside fever and CBC did not significantly differ between patients developing septic shock within 48 hours and those who did not. However, serum lactate level was significantly higher among patients developing septic shock within 48 hours than the levels among those who did not. In addition, serum lactate level was also significantly elevated among patients who deceased compared with those who were alive. Serum lactate level has been included as a new emerging vital sign in recent clinical criteria for septic shock and routinely obtained at the time when septic shock is suspected. 22 The findings from our study provided the additional role of serum lactate level as an effective surrogate marker for developing septic shock within 48 hours after febrile neutropenic episodes among hemodynamically stable, febrile neutropenic, pediatric oncology patients. Therefore, measuring serum lactate level upfront upon developing febrile neutropenic episodes among these specific patient populations despite their stable hemodynamic statuses is highly recommended. The clinical benefit of serum lactate level from our study correlates well with data from adult oncology patients reported by Mato et al 20 Moreover, an ROC curve was created to identify which optimal cut off serum lactate level should be established specifically in pediatric oncology populations. According to new clinical criteria for septic shock, a serum lactate level more than 2 mmol/L has been included as one of the definitions of septic shock. 22 However, the result from our ROC curve also revealed the highest true positive rate of 2.0 mmol/L cut off value of serum lactate level for developing septic shock within 48 hours. This finding also provided the additional role of 2.0 mmol/L cut off value of serum lactate level to predict septic shock given its high sensitivity. Interestingly, the highest true negative rate of 2.5 mmol/L cut off value of serum lactate level for developing septic shock within 48 hours was identified with a specificity of 92.1%. Therefore, serum lactate level more than 2.5 mmol/L should constitute the best threshold to initiate preemptive aggressive hemodynamic monitoring such as transferring patients to a semi-intensive care unit, establishing appropriate vascular access and consider starting prompt treatment such as adjustment of intravenous fluid hydration to ensure adequate tissue perfusion.
The limitations of this study included the small sample size of participating patients, which might have contributed to insignificant differences of some associated factors between patients who developed septic shock and those who did not. In addition, data obtained from the unique and specific populations in this study might not be generally applicable to all patients at different age ranges and with all cancer types. Moreover, this study mainly focused on developing septic shock within 48 hours after febrile neutropenic episodes, so the role of serum lactate level on long term morbidity and mortality among hemodynamically stable children with febrile neutropenia should be further explored.
Conclusions
In addition to the clinical usefulness of serum lactate level to define septic shock, the level determined early at the time of febrile neutropenia among hemodynamically stable, pediatric oncology patients is also an effective surrogate marker to predict developing septic shock within 48 hours. The serum lactate level more than 2.0 mmol/L should raise awareness for developing septic shock within 48 hours; however, the level more than 2.5 mmol/L could be considered as the threshold to start preemptive aggressive hemodynamic monitoring and prompt treatment to ensure adequate tissue perfusion.
Footnotes
Acknowledgements
The funding from the Phramongkutklao College of Medicine and Hospital, Royal Thai Army was used to conduct the study, analyze and interpret the study results and submit the study for publication. The authors would like to thank the patients and families for participating in the study.
Authors’ Contributions
DS involved in patients’ care, collected, analyzed, interpreted the patient’s data and wrote the manuscript; WP involved in patients’ care, collected, analyzed and interpreted the patient’s data; CT involved in patients’ care, analyzed and interpreted the patient’s data; PR involved in patients’ care, collected, analyzed, interpreted the patient’s data and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Data Availability Statement
The data that support the findings of this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding from the Phramongkutklao College of Medicine and Hospital, Royal Thai Army was used to conduct the study, analyze and interpret the study results and submit the study for publication.
