Abstract
Objective: Our aim is to (1) ascertain the proportion of pediatric patients at a tertiary hospital in Western Massachusetts over a 10-year period with hospital-acquired venous thromboembolism (VTE) of particular characteristics and (2) determine whether ACCP or Cincinnati Children’s guidelines would have recommended VTE prophylaxis in these patients. Setting: Urban teaching hospital in the United States. Participants: Data from 98 477 pediatric hospital admissions (roughly 10 000 admission per year) from 2008 to 2017 were reviewed. There were a total of 177 VTE cases identified. Outcome measures: Hospital-acquired venous thromboembolism (including deep venous thrombosis and pulmonary embolism). Result: 177 charts were extracted that carried the diagnosis of VTE based on ICD-9 and ICD-10 codes over a 10-year-period. Among these patients, 34 (19%) met the inclusion criteria for HA-VTE; 5 (16%) would qualify for prophylaxis according to ACCP and 7 (21%) according to Cincinnati Children’s guideline. The most common age group to have a VTE was infants under 1 year of age (41%), and the most common characteristic was the presence of a central line (82%). Age outside of the recommended range was the sole reason that excluded patients from prophylaxis qualification per Cincinnati Children’s. Conclusion: HA-VTE carries increased morbidity and mortality. Although recognition and prevention of HA-VTE in adult populations are routine, prophylaxis for pediatric HA-VTE is not commonly practiced. This may be due to paucity of strong evidence supporting prophylaxis and the challenge of identifying risk factors for HA-VTE. Our results suggest that published guidelines recommend prophylaxis in only a minority of pediatric patients who would have subsequently developed HA-VTE. Further modification and validation of current guidelines are needed to effectively prevent pediatric HA-VTE.
Strengths and Limitations
This study was a retrospective analysis at a single tertiary care academic center that serves Western Massachusetts over a 10-year period.
Analyzing data from a single academic center limits generalizability to other pediatric hospitalizations.
The hospitalizations were identified using ICD-9 and ICD-10 data, and as such may miss some HA-VTE hospitalizations.
Data was collected over a 10-year span in a tertiary hospital center with about 10 000 pediatric hospital admissions per year. However, the incidence of HA-VTE might be higher if the outpatient population within 90 days of hospital discharge was included.
Introduction
Pediatric venous thromboembolism (VTE), while significantly less common than in adults, is increasing in incidence.1-4 It is estimated that there are 34 to 58 VTE cases per 10 000 pediatric hospital admissions, 3 and annual incidence is estimated at 0.07 to 0.49 per 10 000 children.5,6 Causes of rising VTE incidence include better diagnostic modalities, 1 increased use of central venous catheters (CVC) in children, 7 and increasing supportive care for medically complex children. 8 Additionally, VTE is being recognized as an important preventable cause of morbidity and mortality.5,9 Documented complications include prolonged hospitalizations, need for potentially invasive treatments and long-term anticoagulation, and chronic disability in cases of post-thrombotic syndrome (PTS), pulmonary hypertension and pulmonary insufficiency.10,11 Canadian VTE Registry identified mortality attributable to pediatric VTE in 2.2% of patients, recurrent thrombosis in 8.1% and PTS in 12.4% with an average follow-up period of 2.86 years.12,13 Although VTE prophylaxis may aid in the prevention of these devastating complications, concerns have been raised for the risks of pharmacological thromboprophylaxis (pTP) related adverse events in a population with immature hemostatic systems, different underlying disease processes and generally shorter hospitalizations.2,14
VTE is a well-recognized complication of hospitalization in adults, which has led to the widespread implementation of universal thromboprophylaxis policies including assessment, early prevention, and reporting guidelines.8,15,16 Children’s Hospitals’ Solutions for Patient Safety collaborative conducted a 3-year cohort study to reduce hospital-acquired harms in pediatric patients, including VTE. 17 Evidence supporting the use of pTP in children is lacking and mostly extrapolated from adult practice.1,8,18 Unlike adults, in children, clots are infrequently spontaneous or idiopathic (5-10% in children as compared to 40% in adults). 1 Through observational studies, several modifiable and non-modifiable risk factors have been identified.6,15 The current available evidence-based clinical practice guidelines pertaining to antithrombotic therapy in children recommended by the American College of Chest Physicians (ACCP) limit the use of pTP to a subset of children with cavopulmonary anastomosis, myocardial dysfunction, pulmonary hypertension, cyanotic congenital heart disease and children receiving prolonged total parenteral nutrition (TPN).4,8 Other identifiable risk factors include intensive care unit stay, CVC, mechanical ventilation, and length of hospital stay. 19 Additional risk factors identified include sepsis or acute infection, oncological disorders, preexisting coagulation disorders, surgery or trauma, congenital heart disease, systemic lupus, nephrotic syndrome, inflammatory bowel disease, hyperosmolar states, obesity, immobility (which is challenging to define in children20,21), and specific medication use such as L-asparaginase and estrogen.1,2,10,15,22
In order to address concerns regarding VTE morbidity, mortality, and healthcare-related costs based on the known risk factors, several authors have developed institutional risk assessment models and thromboprophylaxis guidelines based on known risk factors.11,15,16,20,22,23 These tools suggest that certain pediatric patients warrant further consideration for primary or secondary thromboprophylaxis, in particular if multiple risk factors coexist.1,5,10,15,24 For example, PEDS-CLOT is a risk assessment tool developed at the Riley Hospital for Children based on retrospective chart review which seeks to stratify children into low, moderate, and high risk, followed by different prophylactic recommendations. 23 Examples of other institutions with such tools include Johns Hopkins, 16 Cincinnati Children’s, 22 Miami Children’s, 15 and the Princess Margaret Hospital of Western Australia. 25
Not surprisingly, randomized controlled trials on the prevention of pediatric VTE are difficult to achieve in pediatric patients. It is primarily related to the low incidence of pediatric VTE; as a result, prospective validation of these tools is similarly challenging. However, retrospective analysis of the demographic data of patients with pediatric VTE is easily attainable. The goal of this study is to analyze the demographic data of pediatric patients diagnosed with VTE at Baystate Children’s Hospital, establish whether based on existing guidelines from other institutions whether our patients would have qualified for VTE prophylaxis, and obtain the data as a baseline for future quality improvement. Collection of this data will not only inform the practice at Baystate Children’s Hospital but also adds to the existing body of research on prevention of pediatric VTE.
Methods
Study Population and Data Sources
This was a retrospective chart review. As part of a quality improvement project, this did not require full review by the Institutional Review Board based on Human Subjects Research Determination assessment. Our aim was to identify patients with hospital acquired VTE and their associated demographic data in hospitalized pediatric patients aged 21 and under at Baystate Children’s Hospital. Additionally, we aimed to determine if these patients would qualify for existing pediatric guidelines.
Baystate Children’s hospital is a 110-bed and 57-bassinette hospital as part of the Baystate Medical Center in Springfield, Massachusetts. It is the only accredited full-service children’s hospital in Western Mass and serves as a regional referral center. Annual pediatric hospital admissions are estimated to be 10,000 at Baystate Children’s Hospital.
HA-VTE is defined as deep vein thrombosis (DVT) and pulmonary embolism (PE) diagnosed >48 hours from admission. Exclusion criteria included patients aged 22 or older, or signs or symptoms of possible VTE on admission including dyspnea, chest pain, calf pain, asymmetric calf swelling, or asymmetric calf redness.
Outcomes
Patient charts were extracted using ICD-9 and ICD-10 billing codes (Table 1) with corresponding radiographic evidence of clot. A manual retrospective chart review was conducted seeking to identify HA-VTE in hospitalized pediatric patients. Patients ≤21 years old from 1/1/2008 to 12/31/2017 with VTE were identified using ICD-9 and ICD-10 codes. VTE was defined as deep vein thrombosis (DVT) including clots in deep veins in the upper and lower extremities, cerebral sinuses, right atrium, abdominal veins, and pulmonary embolism (PE). Patients were excluded if they were admitted with VTE or developed VTE within 48 hours of admission. Once the presence of VTE was established, demographic data including age, sex, ethnicity, unit of admission, and presence of VTE prophylaxis was gathered.
ICD-9 and ICD-10 Codes for VTE.

Abbreviations: VTE, venous thromboembolism.
In addition to establishing the overall incidence and identifying characteristics of hospital-acquired VTE (HA-VTE) at our institution, we sought to determine the proportion that would qualify for mechanical and/or pharmacologic prophylaxis using established clinical guidelines from ACCP 12 and Cincinnati Children’s Hospital. 22 These 2 guidelines were chosen as they were considered to be more widely used based on our discussions with pediatric hematologists at our institution. ACCP recommended children with certain conditions such as cavopulmonary anastomosis, primary pulmonary hypertension, prolonged TPN to receive prophylactic anticoagulation therapy without regard to age range. In contrast, Cincinnati children’s hospital has an algorithm that stratified risk categories in children between 10 and 18 years of age based on impaired mobility and other known risk factors of VTE, such as the presence of a CVC, history of VTE, history of thrombophilia, trauma, infection, and inflammation, etc. These risk factors along with contraindications to prophylaxis were collected (Table 2). Cases that would qualify for ACCP or Cincinnati’s prophylaxis guidelines after assessing contraindications to mechanical or pharmacologic prophylaxis were identified and listed in Tables 3 and 4. Mechanical prophylaxis includes sequential compression devices and graduated compression stockings, and pharmacologic prophylaxis is via low molecular weight heparin injection. The definition of altered mobility is subjective in this case, and it was identified as a risk factor from patients with fractures, osteomyelitis, sedation, and neurologic conditions that would impair mobility. This definition of altered mobility should not be confused with subjects who may have long-term reduced mobility compared to their peers.
Risk Factors Included in 2 Pediatric VTE Guidelines.
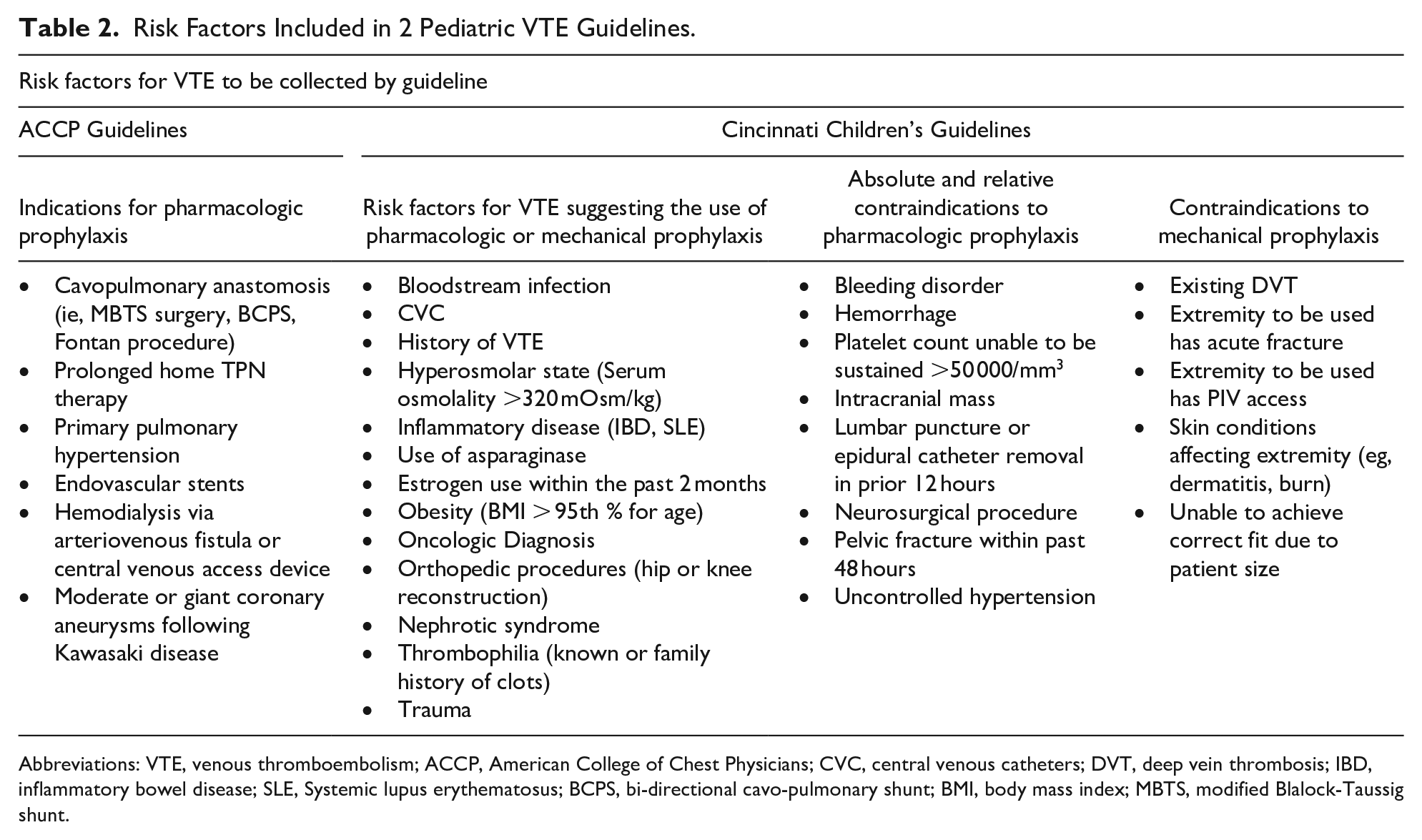
Abbreviations: VTE, venous thromboembolism; ACCP, American College of Chest Physicians; CVC, central venous catheters; DVT, deep vein thrombosis; IBD, inflammatory bowel disease; SLE, Systemic lupus erythematosus; BCPS, bi-directional cavo-pulmonary shunt; BMI, body mass index; MBTS, modified Blalock-Taussig shunt.
Cases Qualifying for VTE Prophylaxis per ACCP Guidelines.

Abbreviations: VTE, venous thromboembolism; ACCP, American College of Chest Physicians; DIC, disseminated intravascular coagulation; TPN, total parenteral nutrition; NEC, necrotizing enterocolitis.
Cases Qualifying for VTE prophylaxis per CCHMC Guidelines.

Abbreviations: VTE, venous thromboembolism; CCHMC, Cincinnati Children’s Hospital Medical Center; CVC, central venous catheters.
Ethical Approval and Informed Consent
This was the first phase of a quality improvement project designed to determine appropriate venous thromboembolism prophylaxis for hospitalized pediatric patients at Baystate Children’s Hospital. As such it was determined that the activity does not constitute human subjects research as defined by federal regulations, and IRB review was not required (IRB reference # BH-19-002, Baystate Medical Center Institutional Review Board). The Standard for Quality Improvement Reporting Excellence (SQUIRE) guidelines were consulted by the authors to guide the publication of these results.
Results
Out of 98 477 hospital admissions (roughly 10 000 admission per year) from 2008 to 2017, there were a total of 178 VTE cases identified, or approximately 18 cases per 10 000 admissions. Of patients with a VTE diagnosis code, roughly 1 in 5 cases were hospital-acquired (19% of the total VTE cases), or about 3.4 cases per 10 000 admissions (Figure 1). 41% of the HA-VTE patients were under 1 year of age, and another 41% were age above 10 (Figure 2). The most common characteristic of HA-VTE (82%) was the presence of a CVC. 16% (5/34) of the HA-VTE cases would qualify for ACCP guideline prophylaxis, with all 5 cases being associated with TPN requirement. 21% (7/34) of the HA-VTE cases would qualify for Cincinnati’s guideline prophylaxis, with all 7 cases being associated with the presence of a CVC. Only one patient (subject 142) would have qualified for prophylaxis under both guidelines. Brief case presentations that would qualify for ACCP and Cincinnati Children’s guideline are described below (Tables 3 and 4). All of the cases that did not qualify for Cincinnati’s prophylaxis were due to patient’s age outside of the guideline range. All patients that are ≥18 years would qualify for prophylaxis per Baystate’s adult VTE prophylaxis guidelines.
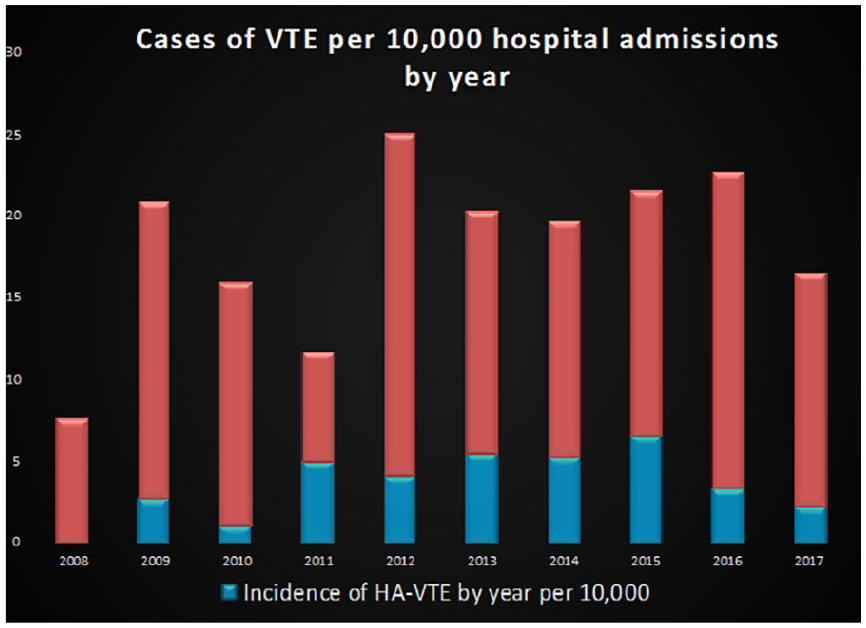
Incidence of HA-VTE per 10 000 hospital admissions per year.

Age distribution of HA-VTE.
Discussion
Our study demonstrated the majority of HA-VTE were infants under age 1, with an additional one-fifth of children between the ages of 10 to 17, and one-fifth between the ages of 18 to 21. This is similar to the current literature, with a bimodal age distribution of VTE seen in infants and teenagers/young adults. 11
The incidence of VTE at our institution is about 18 cases per 10 000 hospital admissions from the 10-year retrospective chart review. The rate is lower in comparison to the results of a large multicenter 7-year retrospective cohort study, which showed 34 to 58 VTE cases per 10 000 pediatric hospital admissions. 3 This study also found a yearly increase of the incidence of VTE in pediatric hospitals, which was not observed in our study. At Baystate Children’s Hospital, 3.4 cases were identified per 10 000 hospital admissions, whereas 30 per 10 000 hospital admissions were identified in Johns Hopkin’s, 24 and 12 to 37 HA-VTE cases per 10 000 admissions were seen in a large multi-institutional study via the CHAT registry (The Children’s Hospital-Acquired Thrombosis). 26 The significant lower HA-VTE incidence is likely due to their higher percentage of patients with complex medical conditions requiring tertiary and quaternary care, which may have predisposed patients to HA-VTE conditions. In addition, the Hopkins study included patients with a VTE diagnosis within 90 days of previous hospitalizations, and the CHAT project included patients who developed VTE within 30 days of hospital discharge. We mainly studied the inpatient population and did not include the outpatient population with new VTE diagnosis after recent hospital discharge, which may have contributed to the observed lower HA-VTE incidence.
Prevalent characteristics of HA-VTE identified in our study were the presence of a CVC which was seen in 82% of our HA-VTE cases; orthopedic surgery, complex medical comorbidities, trauma, and obesity. Not surprisingly, this was similar to the risk factors in the general population, including adults.19,21,26,27
Of note, Baystate Children’s Hospital also admits young adults up to age 21. In our study, all patients ages range from 18 to 21 would qualify for our Baystate adult prophylaxis guideline with either mechanical prophylaxis or pharmacologic prophylaxis. A small portion (16-21%) of the HA-VTE patients <18 years old would qualify for either ACCP or Cincinnati Children’s guideline. No contraindications to mechanical or pharmacologic prophylaxis were identified in patients who would qualify for Cincinnati’s prophylaxis guideline. All cases that qualified for pharmacologic prophylaxis by ACCP guideline were due to prolonged TPN requirement. Multiple risk factors identified in patients who would qualify for Cincinnati Children’s were the presence of a CVC, altered mobility, as well as a few other comorbidities. The Hopkins study similarly has found that patients with 4 or more complex chronic conditions are 4 times more likely to develop HA-VTE. 24
Despite the presence of a CVC being the most common risk factor for VTE, ACCP does not recommend routine thromboprophylaxis. 12 The EINSTEIN-Jr study also showed a small portion of young children with VTE received no or only short duration of anticoagulation. 27 This is likely due to concerns that the risk of hemorrhage outweighs the benefit of prophylactically treating all children with a CVC with antithrombotic agents. According to Nowak-Gottl, the major bleeding risk with low molecular weight heparin prophylaxis is 0.3% whereas minor bleeding risk is about 3%. 28
Strengths and Limitations
This study was a retrospective analysis at a single tertiary care academic center that serves Western Massachusetts over a 10-year period. Analyzing data from a single academic center limits generalizability to other pediatric hospitalizations. The hospitalizations were identified using ICD-9 and ICD-10 data, and as such may miss some HA-VTE hospitalizations. Data was collected over a 10-year span in a tertiary hospital center with about 10 000 pediatric hospital admissions per year. However, the incidence of HA-VTE might be higher if the outpatient population within 90 days of hospital discharge was included. In addition, as we studied only patients with known VTE, it is unknown what proportion of patients without VTE would have qualified for prophylaxis based on the ACCP or Cincinnati Children’s VTEP guidelines.
Conclusion
HA-VTE carries increased morbidity and mortality. Although recognition and prevention of HA-VTE in adult populations are routine, prophylaxis for pediatric HA-VTE is not commonly practiced. This may be due to the rarity of the disease and the challenge of identifying risk factors for HA-VTE. Our results suggest that published guidelines recommend prophylaxis in only a minority of pediatric patients in our institution who would have subsequently developed HA-VTE. Further modification and validation of current guidelines are needed to effectively prevent pediatric HA-VTE.
Footnotes
Author Contributions
Elaine M. Fan: collected and analyzed the data, drafted the abstract and paper.
Deirdre Lewis: collected and analyzed the data, revised the paper.
Thomas Presti: collected and analyzed the data, revised the paper.
Nura El-Haj: drafted the initial proposal.
Weijen Chang: research mentor, helped guide the IRB approval and research direction, revised and submitted the paper.
Authors’ Note
Elaine M. Fan is now affiliated with Intermountain Primary Children’s Hospital in Salt Lake City, Utah. Deirdre Lewis is now affiliated with Cohen Children’s Medical Center in New Hyde Park, New York. Nura El-Haj is now affiliated with UCSF Benioff Children’s Hospital Oakland in Oakland, CA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
