Abstract

Case
A 7-month-old girl presented with daily fever for 1 month before presentation. Mother reported daily fevers, temperatures ranging between 101 and 104°F. Her fevers resolved for a week in between the course of 1 month. However, she again started developing fevers ranging between 102 and 104°F along with arching of her back for the past 1 week before presentation. Her mother also noted increased irritability, especially when she would try to lay her flat on the back, increased sleepiness and decreased interaction. She had 3 episodes of emesis which were non-bloody and non-bilious and contained undigested formula milk. Hence, her mother decided to bring her to the emergency department.
She was born full-term and her mother had poorly controlled gestational diabetes. There were no complications during her delivery or in the perinatal period. The patient had a swelling over her lower back since birth. No imaging of the back swelling was previously performed.
Her vital signs consisted of a temperature of 39.2°C, heart rate 150 beats/minute, blood pressure 80/48 mmHg, respiratory rate 38 breaths/minute. On examination, she was irritable with hyperextension of her neck and back resembling opisthotonus posturing. She had severe neck stiffness with increased fussiness while trying to move her neck. She had a midline circular swelling in the lower back measuring 1.5 × 1.5 cm, soft to firm in consistency which was 3 cm away from the anal verge with a slit overlying the swelling. The base of the slit was not visualized. There was no hypertrichosis or abnormal pigmentation noticed over the back or anywhere else over the skin. She was spontaneously moving all her extremities with no other neurological deficits. Complete blood count showed leukocytosis (white count: 21.2 K/fl) with neutrophilic predominance (absolute neutrophilic count: 15.5 K/fl). C-reactive protein was elevated to 24 mg/L. Blood was sent for culture. Ultrasound of the back swelling was performed which showed a well circumscribed hypoechoic mass in the subcutaneous tissue without internal vascularity (Figure 1). Lumbar puncture was attempted 3 times, however, it was unsuccessful to draw cerebrospinal fluid (CSF) for analysis. Further imaging and history revealed the diagnosis.
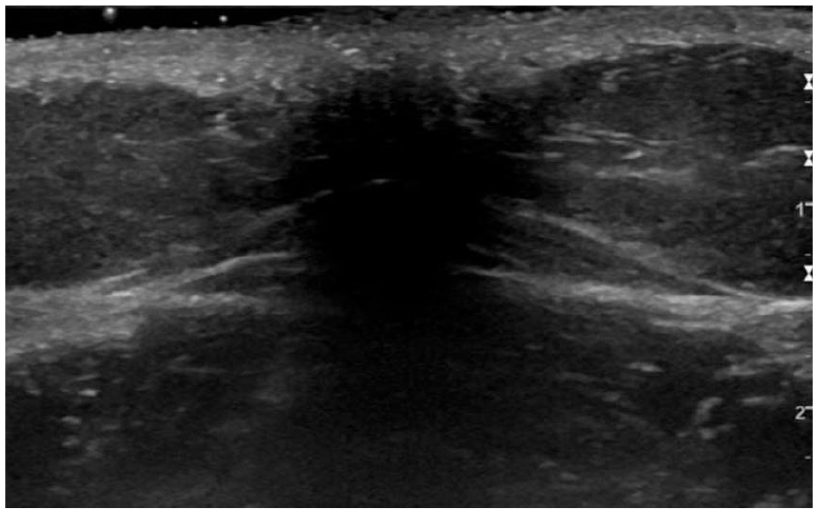
Ultrasound of the lower back swelling shows a well circumscribed hypoechoic mass in the subcutaneous tissue.
The patient was empirically started on intravenous vancomycin and ceftriaxone for suspected meningitis. Multiple attempts including fluoroscopic guided lumbar puncture performed at L2-L3, L3-L4, and L4-L5 level failed to draw CSF. On further inquiring, the mother reported occasional fluid drainage from that back swelling. Therefore, magnetic resonance imaging (MRI) of the spine was performed which showed a dorsal dermal sinus entering the spinal canal at the L5-S1 level with associated significant heterogeneous enhancing lesions filling the entire spinal canal between L1 and S2 concerning for subdural empyema. There were punctate areas of diffusion restriction in the distal thecal sac concerning for dermoid/epidermoid cyst (Figures 2A, B and 3A, B).

(A) Sagittal T1W series showed dorsal dermal sinus cord (arrow) with extensive spinal abscess (dashed arrow). (B) Sagittal T1W post gadolinium series showed dorsal dermal sinus (arrow), cord abscess with extensive spinal intradural and epidural abscess (dashed arrow).

(A) Sagittal diffusion weighted imaging (DWI) series showed diffusion restriction of the spinal abscess (arrow). (B) Axial T1W post gadolinium series demonstrates thick wall spinal intradural abscess (dashed arrow).
Soon after MRI, her right lower extremity became limp. On examination, the entire right lower extremity was flaccid with no deep tendon reflexes and no response to painful stimuli. Antibiotics were escalated to meropenem and she emergently underwent L1-L5 laminectomy, dermal sinus tract excision, subdural abscess drainage and washout with tethered cord release. Anaerobic culture grew Anaerococcus species and Actinomyces neuii. Blood cultures remained negative. The dermal sinus tract measured 2.2 cm in length and 0.1 cm in diameter. Immunohistochemistry stained positive for S100 and glial fibrillary acidic protein confirming neural tissue. Postoperatively, her right lower extremity continued to remain limp with no spontaneous movements. She initially had occasional urinary retention, showing up to 380 ml on a bladder scan, concerning for neurogenic bladder which resolved later on. The patient continued to be afebrile after surgery and was stable. She was started on early physical and medical rehabilitation with continued IV antibiotics for a total duration of 5 weeks. Repeat MRI was performed at the end of IV antibiotics therapy which showed near-complete resolution of the subdural abscess.
Discussion
Dermal sinus tract (DST) is a rare form of spinal dysraphism, with an incidence ranging between 1 in 2000 and 3000 live births.1,2 It is comprised of an ostium at the midline of neuroaxis with an epithelial tract extending from the skin surface into deeper structures of the spinal cord. 3 It is thought to develop during the third and eighth weeks of gestation due to incomplete disjunction or abnormal adhesion between the neuroectoderm and surface ectoderm. 4 DST can occur anywhere in the midline from occiput to coccyx, rarest being the cervical region (<1%) and more commonly in thoracic (10%), lumbar (40%), lumbosacral (12%), sacral (23%), and sacrococcygeal junction (13%). 5 They are usually associated with overlying cutaneous manifestations such as hair tuft, abnormal pigmentation, skin tags, and raised plaques, vascular malformations such as hemangiomas, lipomas or signs of localized infection such as redness, discharge, or induration. 3 DSTs should be distinguished from the benign and more commonly seen sacral dimples. Lesions that are >2.5 cm from anal opening, >5 mm in size and those associated with cutaneous stigmata mentioned above should raise suspicion for an underlying spinal dysraphism. 6 Other developmental anomalies associated with DST are inclusion tumors (dermoid/epidermoid cyst, teratoma), tethered cord and split cord.3,7
DST serves as a portal of entry for bacterial pathogens into different intraspinal compartments (epidural, subdural, and intramedullary), causing meningitis or abscesses. The frequency of these infections in the presence of DST has been ranging from 10% to as high as 75% in the past.3,8,9 Presenting symptoms are usually non-specific and include fever, meningismus, acute neurological deficit, and bladder incontinence.10,11 Some children can have purulent drainage of fluid from the ostium of the DST which can suggest an underlying infection. Causative organisms isolated from spinal abscesses have been Staphylococcus spp., Streptococcus spp., E. coli, Actinomyces spp., Pneumococcus spp, and Proteus. Our patient’s abscess drainage grew Anaerococcus and Actinomyces neuii which are a part of human microbiota that was introduced following a breakdown in her protective mucocutaneous protective barriers resulting in infection. Microbiological pathogens may not be isolated in all cases and approximately 30% have sterile cultures which could be due to prior antibiotic treatment or chemical meningitis from the overlying dermoid cyst fluid.9-12 Ultrasound has low sensitivity in detecting dermal sinus tracts and it mainly depends on the expertise of the radiologist performing it. Unfortunately, in our patient, ultrasound was unable to detect the dermal sinus tract and thus lumbar puncture was attempted. Typically, in known DST cases, LP is contraindicated. Hence, in the current era, MRI is considered to be the modality of choice as it provides detailed soft-tissue differentiation, reconstruction across different planes, ability to delineate spinous structures accurately, and no radiation risks. 10
The mainstay of treatment is usually a combination of intravenous antibiotics along with surgery. Intravenous antibiotics were mostly used for at least 4 to 6 weeks. Surgery involves resecting the DST from its surface, extending all the way deeper until complete exploration of the tract is accomplished, along with laminectomy and drainage of the underlying abscess. Timing of the surgery has been equivocal as some authors recommended immediate surgical drainage at diagnosis whereas others recommended initial control of infection with antibiotics followed by surgical drainage and repair of DST later.9,10,12 However, recent review articles have consistently shown better neurological outcomes with early surgical intervention before the development of infection.9-11 Neurological deficits in these children could either be due to underlying neurological malformation or spinal abscess causing vascular compromise and local mechanical pressure.
Conclusion
Patients with midline back cutaneous manifestations such as hair tuft, abnormal pigmentation, skin tags, and raised plaques, vascular malformations such as hemangiomas, subcutaneous lipomas or swelling such as dermoid/epidermoid cyst should raise suspicion for underlying spinal dysraphism. It is important to differentiate dermal sinus tracts from benign sacral dimple as the former is >2.5 cm from the anal verge, >0.5 cm in size and may be associated with an overlying neurocutaneous marker as mentioned above. Patients with DST are at risk for central nervous system infections such as meningitis, epidural, subdural, or intramedullary abscess depending on the extension of the tract. Early identification and prompt surgical intervention is essential to prevent complications and improve the outcome in these patients.
Footnotes
Acknowledgements
We thank Deniz Altinok, MD a neuro-radiologist at Children’s Hospital of Michigan for his imaging review and guidance with revision of this manuscript.
Author Contributions
All authors contributed equally in drafting the case report, reviewing pertinent literature and revising the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Informed consent was obtained verbally from the mother of the child and all personal information will be kept confidential.
