Abstract
Esophagogastroduodenoscopy (EGD) is commonly performed for abdominal pain in children, and in the presence of alarm symptoms, the diagnostic yield of this procedure is high. The aim of this study was to investigate the utility of EGD when performed for uncomplicated abdominal pain in children in the absence of alarm symptoms. Charts of all children less than 18 years of age who underwent EGD at the University of Florida between January 2016 and October 2018 were reviewed. Of 1478 children who underwent EGD for any indication, 287 patients (male-to-female = 123:164) were discovered to have undergone EGD, in the absence of colonoscopy, for uncomplicated acute or chronic abdominal pain, nausea, vomiting, gastroesophageal reflux, and/or dyspepsia, with no alarm symptoms. A significant change in clinical management was noted in 20 (7.0%) children. The findings of this procedure changed clinical management in a minority of our study participants.
Introduction
Abdominal pain is common in children, affecting quality of life, causing angst for caregivers, and, in some cases, leading to more serious complications. 1 In the absence of alarm symptoms, initial evaluation relies on the clinical history and physical examination, and prior experience reveals that extensive testing is not necessary when there is high suspicion for a functional gastrointestinal disorder. 2 In cases where alarm symptoms are present (ie, dysphagia, weight loss, persistent diarrhea or vomiting, fever, slow growth, gastrointestinal bleeding, anemia, high erythrocyte sedimentation rate (ESR) or C-reactive protein (CRP), or daily nonsteroidal anti-inflammatory drug/corticosteroid therapy), further diagnostic testing may be indicated. 3 Esophagogastroduodenoscopy (EGD) is the definitive test for many diseases of the esophagus, stomach, and duodenum and has demonstrated high diagnostic yield in the evaluation of abdominal pain, especially in the presence of alarm symptoms.4 -6 What has not been well-established, however, is to what degree EGD leads to a significant change in the management of this common childhood complaint when alarm symptoms are absent.
EGD is often performed in children with refractory abdominal pain, dyspepsia, gastroesophageal reflux, or when there is suspicion of gastrointestinal inflammation.7,8 Histamine-2 (H2) blocker or proton pump inhibitor (PPI) therapy is effective for the treatment of gastritis and esophagitis; however, persistent symptoms or apparent lack of response to this therapy often results in further evaluation.
6
EGD remains the gold standard for diagnosing
The purpose of this study was to determine if EGD is useful in the diagnosis and management of uncomplicated abdominal pain in children and how often the findings of this procedure yield a significant change in clinical management.
Methods
Sample
This retrospective review selected children (<18 years of age) who underwent initial EGD at the University of Florida, a large academic, research, and tertiary referral center, between January 2016 and October 2018. Our study was approved by the hospital institutional review board.
Subjects with previous known organic diseases (such as inflammatory bowel disease, celiac disease, EoE, cystic fibrosis, cerebral palsy, epilepsy, autoimmune disease, or cancer), feeding difficulties, those with psychiatric disorders, and those who underwent concomitant procedures (ie, colonoscopy, capsule endoscopy, and bronchoscopy) were excluded from the study. Patients undergoing EGD for the evaluation of abnormal celiac serology, caustic ingestion, or foreign bodies were excluded. Patients discovered to be deceased at the time of the review were excluded. The history of prior EGD was not considered exclusionary unless the procedure was performed more than once within the study timeframe, in which case only the first of these procedures was included.
Alarm Symptoms
The presence of alarm symptoms was documented in each case. Alarm symptoms were considered to be dysphagia, weight loss (>5% in the past 2 months), persistent diarrhea or vomiting (daily for >2 weeks), fever (temperature >100.4°F), unexplained retardation of growth (height at less than the third percentile for age), gastrointestinal bleeding (by history or occult blood in stool), anemia (hemoglobin less than 11 mg/dL), elevation of ESR (ESR > 20) or high-sensitivity CRP (CRP > 5), or a recent history of daily corticosteroid or nonsteroidal anti-inflammatory drug use for >7 days within the past month. Those children with one or more alarm symptom were excluded.
Procedure
EGD was performed by a member of a team of pediatric gastroenterologists in an endoscopy suite using a fiber-optic gastroscope. The patient was placed under general anesthesia by a certified anesthesia provider. Gross endoscopic findings were recorded, and in most cases, biopsies were taken from the esophagus, stomach, and duodenum. All biopsies were reviewed by a histopathologist with subspecialty expertise in pediatric and/or gastrointestinal pathology and again by a multidisciplinary team of pathologists and gastroenterologists at a weekly quality assurance conference.
Gross Findings
The presence of erythema, ulcers, erosions, nodularity, or other clinically significant anomaly in the esophagus, stomach, or duodenum was noted. The presence of furrowing, rings, or white patches in the esophagus was noted. Minor structural anomalies (ie, hiatal hernia, capacious duodenal bulb) or luminal contents (ie, retained food, bilious secretions) were not recorded unless significant enough to induce an immediate change in clinical management.
Histology
In most cases, a biopsy was taken from the distal esophagus, gastric antrum, and third portion of the duodenum. One or more additional biopsies were taken from the mid or proximal esophagus in cases where the gross findings suggested the possibility of EoE. Additional biopsy specimens were taken if the endoscopic findings raised suspicion of other anomalies.
Normal histopathology was defined as the absence inflammation and, for the esophagus, squamous epithelium without basal cell hyperplasia or intraepithelial eosinophils; for the stomach, normal site-specific mucosa; and for the duodenum, normal villous architecture. Mild reactive mucosal changes were not considered to be clinically significant. The degree of eosinophilic infiltration of the esophagus was divided into 2 categories: less than 15 per high-powered field (HPF) and 15 or more per HPF.
Postprocedure Management
Change in management was defined as the immediate addition of a new class of medication (other than an H2-blocker, PPI, or pharmaceutical treatment for functional gastrointestinal problems such as a pro-motility or antispasmodic agent), the discontinuance of a medication, referral to a non-gastrointestinal specialist, and/or recommendation for further testing.
Statistical Analysis
Data were collected and managed using REDCap version 8.8.2. Basic intake cohort descriptives were recorded, including age, gender, date of procedure, preoperative symptoms, concomitant procedures, preprocedural use of an H2-blocker or PPI,
Data were analyzed using SAS 9.4 (Cary, NC). Demographics and key EGD characteristics of pre- and postoperative rates were computed overall and among those patients with and without change in postoperative diagnosis or treatment. To test for differences between those with and without postoperative changes, Fisher’s exact tests were used (2-sided α = 0.05). Preoperative diagnoses, alarm symptoms, as well as gross procedure and histology results were compared between those with and without postoperative changes.
Ethical Approval and Informed Consent
The institutional review board of the University of Florida waived the need for ethics approval and the need to obtain consent for the collection, analysis, and publication of the retrospectively obtained and anonymized data for this noninterventional study.
Results
From January 2016 to October 2018, EGD was performed on 1478 children for numerous indications. Exclusion criteria were applied and 287 qualified for this study. The patients in our study were between 1 and 17 years (mean = 11.2 ± 4.0 years), and 57.1% were female (164/287). Preoperative symptoms were generalized abdominal pain (71, 24.7%), right lower quadrant abdominal pain (1, 0.4%), left upper quadrant abdominal pain (15, 5.2%), right upper quadrant abdominal pain (8, 2.8%), epigastric abdominal pain (91, 31.7%), periumbilical abdominal pain (28, 9.8%), lower abdominal pain (2, 0.7%), nausea/vomiting (81, 28.2%), dyspepsia (10, 3.5%), heartburn (36, 12.5%), and gastroesophageal reflux (39, 13.6%). See Table 1.
Basic Intake Cohort Descriptives.
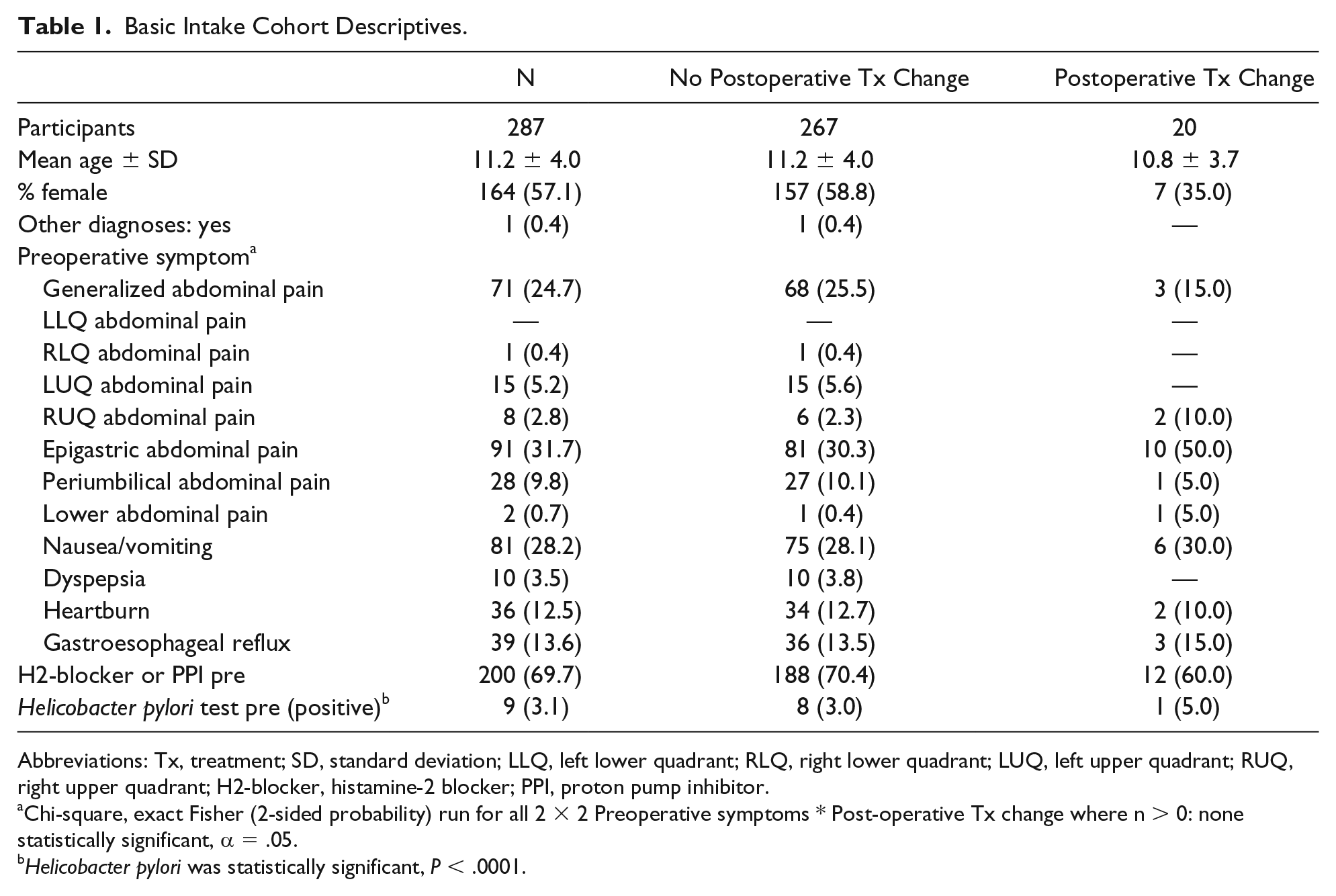
Abbreviations: Tx, treatment; SD, standard deviation; LLQ, left lower quadrant; RLQ, right lower quadrant; LUQ, left upper quadrant; RUQ, right upper quadrant; H2-blocker, histamine-2 blocker; PPI, proton pump inhibitor.
Chi-square, exact Fisher (2-sided probability) run for all 2 × 2 Preoperative symptoms * Post-operative Tx change where n > 0: none statistically significant, α = .05.
EGD was grossly normal in 203 (70.7%), and other findings were as follows: esophagus erythema (19, 6.6%), esophagus erosions (3, 1.1%), esophagus nodularity (8, 2.8%), esophagus furrows (14, 4.9%), esophagus rings (3, 1.1%), esophagus white patches (4, 1.4%), stomach erythema (24, 8.4%), stomach nodularity (17, 5.9%), stomach ulcers/erosions (5, 1.7%), duodenum erythema (9, 3.1%), duodenum erosions (9, 3.1%), duodenum nodularity (2, 0.7%), and other (1, 0.4%). See Table 2.
Gross Procedure and Histology Results.
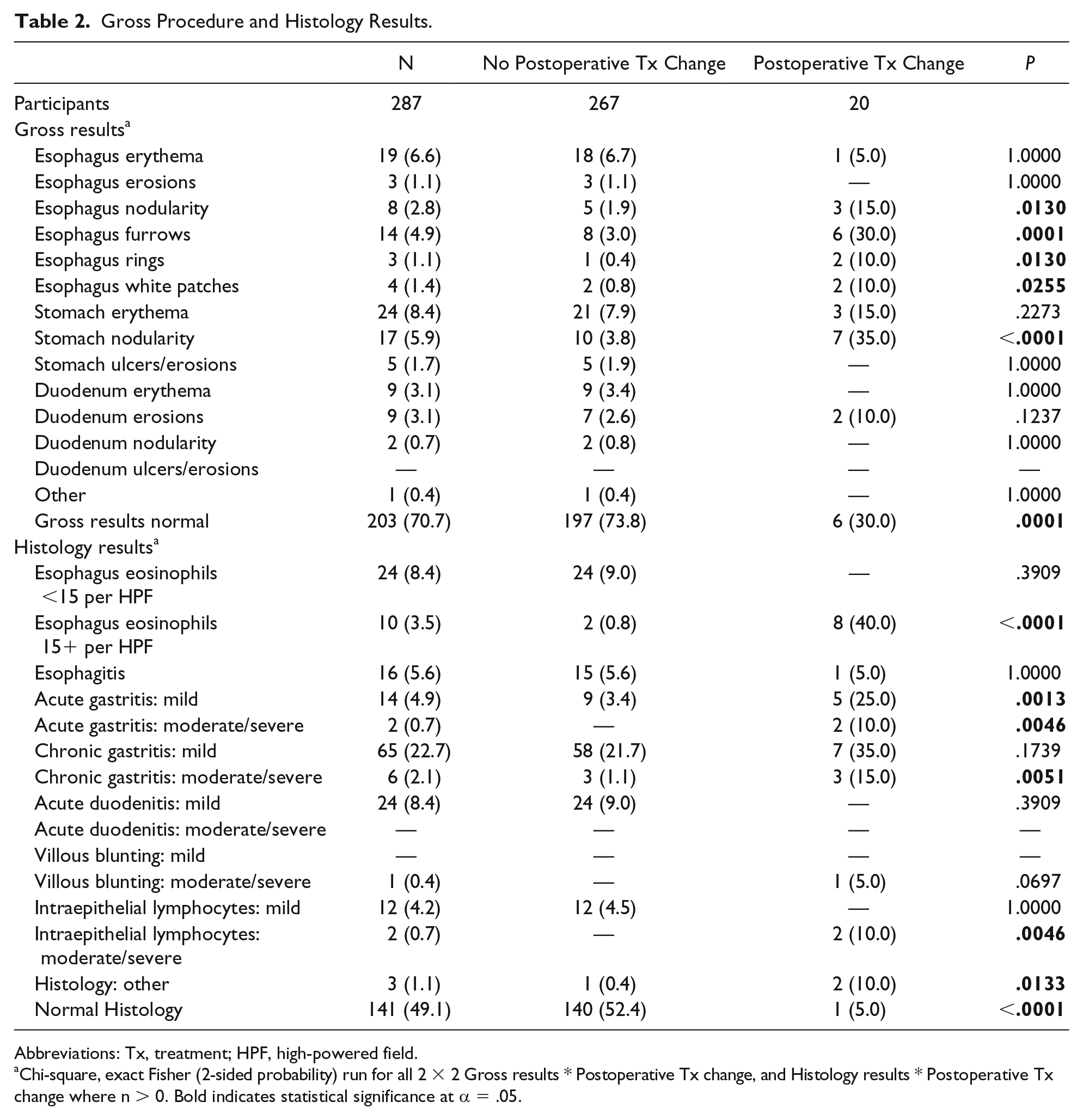
Abbreviations: Tx, treatment; HPF, high-powered field.
Chi-square, exact Fisher (2-sided probability) run for all 2 × 2 Gross results * Postoperative Tx change, and Histology results * Postoperative Tx change where n > 0. Bold indicates statistical significance at α = .05.
Histological evaluation was normal in 141 (49.1%), and other findings were as follows: esophageal eosinophils (<15 per HPF; 24, 8.4%), esophageal eosinophils (15+ per HPF; 10, 3.5%), acute esophagitis (16, 5.6%), mild acute gastritis (14, 4.9%), moderate/severe acute gastritis (2, 0.7%), mild chronic gastritis (65, 22.7%), moderate/severe chronic gastritis (6, 2.1%), mild acute duodenitis (24, 8.4%), moderate/severe duodenal villous blunting (1, 0.4%), mild intraepithelial duodenal lymphocytes (12, 4.2%), moderate/severe duodenal intraepithelial lymphocytes (2, 0.7%), and other (3, 1.1%).
H2-blocker or PPI therapy was attempted in 200 (69.7%) prior to the procedure and 12 (6.0%) of these patients required a change in clinical management as a result of EGD findings. Of the 87 patients who did not undergo a trial of H2-blocker or PPI therapy prior to the procedure, 8 (9.2%) required a change in management. Stool
Patients with positive celiac serology were excluded from this study while 82 (28.6%) of the included patients had negative serology. No celiac testing was performed in 205 (71.4%).
A substantial change in clinical management was noted in 20 (7.7%). Of these, treatment was initiated for EoE in 8,
Discussion
The objective of this study was to determine if EGD for children with uncomplicated acute or chronic abdominal pain, nausea, gastroesophageal reflux, and/or dyspepsia produced a significant change in clinical management. Other investigators have demonstrated high diagnostic yield when EGD was performed for these indications, especially in the presence of alarm symptoms; however, these studies considered mild nonspecific inflammation in the esophagus, stomach, and duodenum to be a positive result.5,13 These data were consistent with our findings; however, we discovered that, in the absence of
PPI therapy was initiated in our patients who were not already taking an H2-blocker or PPI when acid-related gastrointestinal inflammation was discovered endoscopically. Nonetheless, since a trial of PPI therapy is a relatively safe and inexpensive intervention and is recommended as first-line treatment for suspected or known gastric acid–related disorders (heartburn, retrosternal, or epigastric pain) in children, 14 we did not consider the initiation of a H2-blocker or PPI as a change in management for our patients. Also, our results did not demonstrate that prior acid blockade correlated with the need for a change in clinical management. We believe that these data, in addition to the paucity of serious findings for our patients with uncomplicated abdominal pain, support the North American Society for Gastroenterology, Hepatology, and Nutrition (NASPGHAN) and European Society for Gastroenterology, Hepatology, and Nutrition (ESPGHAN) consensus guidelines for a 4- to 8-week trial of PPI therapy in children with suspected acid-related gastrointestinal disorders prior to considering EGD. 14
A clinical report by the American Academy of Pediatrics and NASPGHAN on chronic abdominal pain found that functional abdominal pain can be reliably differentiated from organic disease without diagnostic testing when alarm symptoms are absent. 7 The statement recommended against performing EGD in children with chronic or recurrent abdominal pain; however, the indications for EGD in the setting of acute or severe abdominal pain are less clear. While efforts were made to define long-lasting intermittent or constant abdominal pain in children in this report, it is not known to what degree health care providers are following consensus recommendations for evaluation, diagnosis, and treatment. Many children with chronic or recurrent abdominal pain are referred to a pediatric gastroenterologist with an expectation for further workup. Despite evidence that suggests EGD is not necessary in the majority of children with uncomplicated abdominal pain, this procedure remains a common diagnostic choice.
Patients with known EoE, dysphagia, or positive celiac serology prior to EGD were excluded from this study. Nonetheless, EoE was discovered in 7 of our patients with a variety of complaints (3 with abdominal pain, 2 with gastroesophageal reflux or vomiting, and 2 with both abdominal pain and vomiting). It is not known if EoE was the cause of our patients’ symptoms or an incidental finding. Screening for celiac disease should be performed with serologic testing, followed by small bowel biopsy for positive values, when there is low clinical suspicion of this disorder, 11 and therefore, the diagnosis of celiac disease by small bowel histology, in the absence of a positive serologic test, was an unexpected finding in our patients. Two children with no prior celiac testing were discovered to have celiac disease by histology in our study. Both of these children were later found to have positive celiac serology and both clinical improved on a gluten-free diet.
Although noninvasive tests for
It is recognized that negative endoscopy findings may provide reassurance that supportive care is appropriate. The knowledge of normal EGD findings can be therapeutic for patients with functional symptoms exacerbated by anxiety due to uncertainty of the diagnosis. 17 In this retrospective review, it is not possible to determine if negative findings prevented further unnecessary medical visits, testing, or treatment. In our patients, negative endoscopy results were sometimes used to support treatment for functional symptoms such as the initiation of a promotility (ie, azithromycin) or antispasmodic agent (ie, hyoscyamine, dicyclomine); however, the utility of these strategies was not studied. A study by Bonilla et al 17 demonstrated that negative EGD results did not affect long-term outcomes for functional abdominal pain in children, and we agree that EGD should not be performed to reassure families and patients on the benign nature of this condition.
The authors acknowledge limitations of the study, including its retrospective nature. This study did not include children with dysphagia, feeding difficulties, suspected foreign body, gastrointestinal bleeding, or abnormal celiac serology as an indication for the procedure. It also did not include children undergoing concomitant colonoscopy or other procedure (ie, capsule endoscopy, pH probe, feeding tube, or gastric stimulator placement, bronchoscopy, or other surgical procedure). It is suspected that the likelihood of EGD yielding a change in management would be significantly higher in this unstudied group. Further prospective, larger, and multicenter studies are needed to validate our findings and to determine the diagnostic utility of EGD in children with indications outside our study population.
Procedural complications are rare for children undergoing EGD 18 and the potential benefit of direct visualization of gastrointestinal disease cannot be overstated. 19 Histopathology can further clarify the diagnosis in some cases, but there may be discrepancy between macroscopic abnormalities and histological findings in others. 20 The decision to perform EGD is often complex, requiring the joint decision making of patients, caregivers, and providers, all of whom must balance the potential risk and benefit with the cost and efficacy of the procedure. Our study results indicate that EGD has limited utility in the evaluation of abdominal pain in healthy children who do not have alarm symptoms and the findings of this procedure changed clinical management in a minority of our study participants whether or not they were already taking an H2-blocker or PPI.
Footnotes
Author Contribution
RAR: Contributed to conception and design; contributed to analysis; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
SLF: Contributed to analysis; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MJG: Contributed to analysis; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
AS: Contributed to analysis; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MKD: Contributed to conception and design; contributed to analysis; drafted the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by a Children’s Miracle Network Research Discovery Award.
