Abstract

Introduction
Congenital hypothyroidism, if left untreated, can lead to severe neurodevelopmental consequences in the newborn. In addition to congenital causes of hypothyroidism, maternal autoimmune thyroid disease can lead to transfer of thyrotropin receptor antibodies across the placenta and may cause a transient hyperthyrotropinemia or hypothyroidism in the infant or more rarely neonatal thyrotoxicosis.1-7 The newborn screen is designed to detect congenital hypothyroidism, but testing modality varies state by state. In New York State, a 2-tiered screen with primary free thyroxine and a reflex thyroid-stimulating hormone is used. There are currently no guidelines for obtaining additional testing in infants with maternal hypothyroidism apart from the newborn screen, and current literature has yielded mixed results on the topic.1,3,8-14 Therefore, the question of whether additional screening apart from the newborn screen is warranted in these cases and the optimal timing to do so is controversial.
In contrast, neonatal thyrotoxicosis is a rare but life-threatening condition that can be observed most frequently in poorly controlled maternal Graves’ disease. The newborn screen is not designed to identify this disorder, and there are recommendations to obtain routine screening of infants born to mothers with hyperthyroidism. 4
Through a quality improvement project at our institution, we found that nursery providers were obtaining serum thyroid function testing (TFT) in well infants in the setting of maternal hypothyroidism in the newborn nursery an estimated 60% to 80% of the time over the past 5 years. We seek to assess how both maternal and provider characteristics influence the decision of newborn nursery providers to obtain TFT in healthy newborns across New York State.
Methods
This was a cross-sectional survey study with institutional review board approval from Albert Einstein College of Medicine. A list of newborn nurseries in New York State was obtained from the New York State Newborn Screening Program, and surveys were sent electronically to nursery administrators to distribute to providers at 138 hospitals in New York State using SurveyMonkey. Three surveys were physically collected at local hospitals. We excluded providers from our own institution. Providers were surveyed on situations in which they would obtain TFT in a healthy newborn in the newborn nursery. We collected data including provider type, practice setting, years of medical experience, and perceived access to pediatric endocrinology. Study personnel were blinded to both provider and institution.
We received 38 responses from nurse practitioners, general pediatricians, and neonatologists from a number of community hospitals, private hospitals, and academic centers. We expected at least 90% of providers to order TFT in the setting of maternal hyperthyroidism and 60% or less of providers to order testing for maternal hypothyroidism. Our study was powered to detect this difference with 80% power and a P value of .05. Responses were analyzed using descriptive statistics and Fisher’s exact testing with SPSS statistical software to detect differences in testing based on provider characteristics.
Results
Eighty-one percent of providers considered a maternal thyroid disease to be an indication to obtain TFTs in the well newborn. Sixty-five percent of surveyed providers ordered testing in the setting of maternal hyperthyroidism compared with 38% in the setting of maternal hypothyroidism. This difference was found to be significant at 5.786 with P value of .016 using McNemar’s test. Providers were surveyed on other situations they would obtain TFT based on both maternal history and infant characteristics (Figure 1).
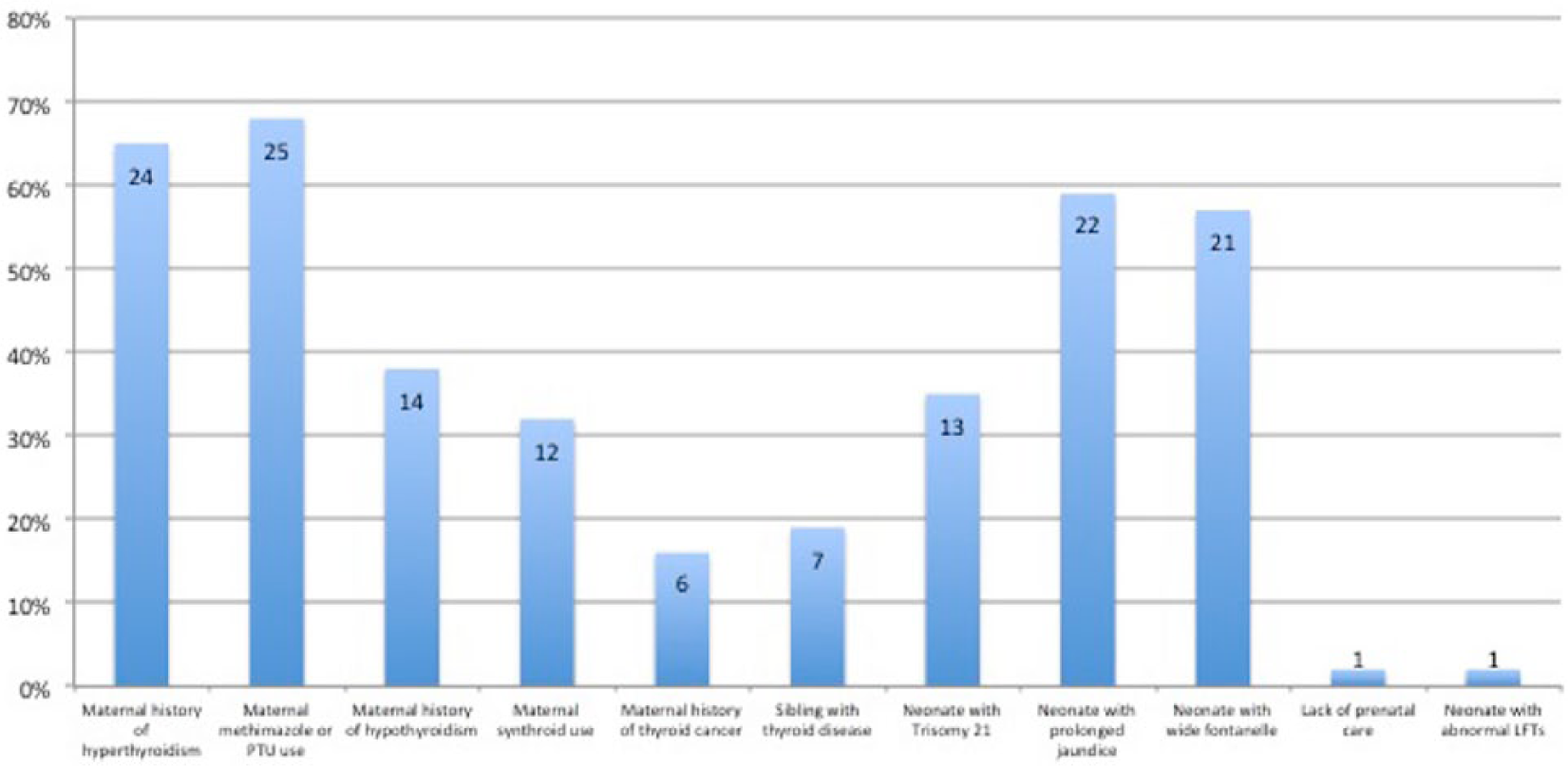
Situations in which providers obtain thyroid function testing (TFT) in the newborn nursery.
Two-sided Fisher’s exact testing was performed to detect differences in practice based on provider characteristic. Fifty-six percent of neonatologists performed serum thyroid testing in the setting of maternal hypothyroidism versus 14% of general pediatricians, with P value of .027. Fifty-six percent of providers with 0 to 10 years of experience performed serum thyroid testing in infants with trisomy 21 versus 19% in providers with 11+ years of experience, with P value of .036 (Figure 2). There were differences noted in provider characteristics for other situations but none of these reached statistical significance.
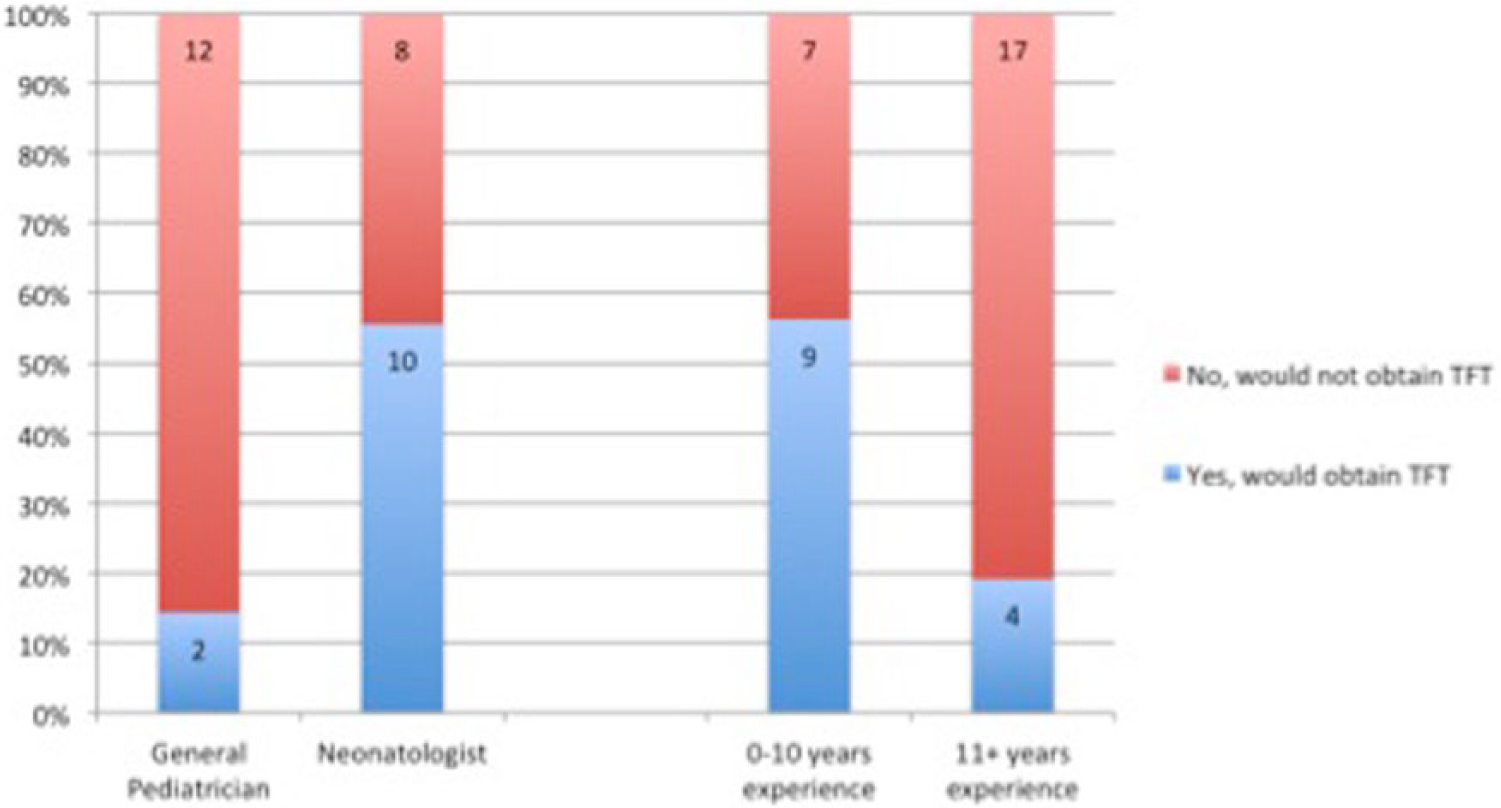
Characteristics of providers ordering thyroid function testing (TFT) in newborns with (A) maternal hypothyroidism and (B) trisomy 21. Both subsets reached statistical significance.
Discussion
We found that TFTs are commonly being obtained in well newborns with maternal hypothyroidism in the nursery period in New York State, despite the lack of formal recommendations. A few recent studies have demonstrated that additional testing did not identify any additional cases of hypothyroidism apart from the newborn screen, leading to their recommendation that additional testing is unnecessary.9-13 Other recent studies have demonstrated utility in additional testing in the newborn period, although optimal timing of testing has not been determined.1,3,6,8,14 Additional testing obtained unnecessarily has a significant impact on resource allocation and referrals; moreover, TFTs obtained too early after birth may be affected by the newborn’s physiologic thyroid-stimulating hormone surge and therefore yield false positive results.8,14 This topic is controversial, as some amount of additional testing may be considered acceptable to ensure a diagnosis with preventable developmental implications is not missed.
In contrast to maternal hypothyroidism, we found that TFTs in the setting of maternal hyperthyroidism are not being obtained as universally as we had predicted. There are guidelines for obtaining additional testing in these cases depending on presence of maternal thyrotropin receptor antibodies, whether diagnosis occurred during the pregnancy, and if the mother is taking an antithyroid medication. 3 We hypothesize this discrepancy between recommendations and clinical practice may be the result of providers not being as comfortable evaluating neonatal hyperthyroidism and referring to an endocrinologist instead of ordering testing themselves, providers not being as familiar with the consequences of neonatal hyperthyroidism as compared with hypothyroidism, providers expecting affected infants to have apparent signs and symptoms and deferring testing, or providers unfamiliarity with current guidelines. It will be important to obtain further studies regarding barriers to improve adherence to guidelines given the morbidity associated with this condition.
We found that neonatologists are obtaining TFT in the nursery more frequently than general pediatricians for maternal hypothyroidism. This may due to the nature of the training of neonatologists and the need to obtain serum sampling for various reasons more frequently in their patient population. We also found that providers with fewer years experience are obtaining TFT more frequently than providers with more experience in infants with trisomy 21. Current guidelines suggest that infants with trisomy 21 have thyroid function monitored at 6 months and 12 months; however, there are no guidelines for obtaining testing in the neonatal period apart from the newborn screen. 15
Despite the risk to infants of mothers with hypo- and hyperthyroidism, we found that maternal thyroid disease was not a consideration in 19% of surveyed providers. Additional guidelines would be helpful in standardizing practice; however, even without such guidelines, providers must consider maternal thyroid disease when evaluating the newborn infant. Further studies are needed to determine how widespread these practices are and to support recommendations for situations and optimal timing of obtaining additional testing on infants. Such studies may be difficult to conduct, however, given the differences in newborn screening methodology between states.
The primary limitation of our study is the small sample size. Although we found a correlation between provider characteristics and decision to send TFTs in 2 situations, our study may not have been powered to detect other differences. The study was also limited to New York State and may not be an accurate representation of provider practices across the country. New York State relies on a 2-tiered screening system, which may influence provider’s attitudes regarding the need for additional serum sampling to determine newborn thyroid status. Last, our survey questions did not further characterize maternal thyroid disease to assess provider decision-making based on presence of antibodies or control of disease.
Conclusion
Serum TFT in healthy term newborns in the setting of maternal hypothyroidism is a prevalent practice among New York State nursery providers despite the introduction of newborn screening tests. In contrast, TFT in the setting of maternal hyperthyroidism is not universal. Maternal thyroid history is an important consideration in the evaluation of the newborn and should be taken into account when deciding to perform neonatal serum TFT, especially in cases with maternal history of hyperthyroidism. There is a need to ensure primary providers are aware of current recommendations regarding screening infants with maternal hyperthyroidism and a need for additional studies to explore potential barriers to adherence to current guidelines. Last, there is a need for clearer guidelines on recommendations for obtaining TFT apart from the newborn screen in well newborns in the setting of maternal thyroid disease.
Footnotes
Author Contributions
DD: Contributed to acquisition and analysis; drafted the manuscript; gave the final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MS: Contributed to acquisition of data; drafted the manuscript; gave the final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
LU: Contributed to conception and design; critically revised the manuscript; gave the final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
LL: Contributed to design; critically revised the manuscript; gave the final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
