Abstract

Introduction
Persons with untreated atrophic thyroiditis develop a permanent height deficit. Adequate thyroxine replacement therapy can improve growth and the probability of attaining normal adult height. However, height deficit is related to duration of thyroxine deficiency, regardless of the adequacy of thyroxine replacement therapy after diagnosis. 1 Several studies reported that use of adjunctive therapy with growth hormone (GH) and gonadotropin-releasing hormone agonist (GnRHa) to improve the final height of patients.2-5 The rationale for this therapy is to prolong the growth period by inhibiting pubertal progression and delaying epiphyseal fusion, 6 although GH deficiency in atrophic thyroiditis patients usually improves after L-thyroxine treatment.
We report a case of autoimmune atrophic thyroiditis in a boy who presented with GH deficiency even after his enlarged pituitary gland had decreased in size. Combined treatment with GH, GnRHa, and L-thyroxine was successful in allowing the patient to attain normal adult height.
Case
An 8-year-old boy presented with short stature at our hospital. Growth data were plotted on Japanese growth charts for males aged 0 to 18 years (V-Link Co, Ltd, Tokyo, Japan). The heights of his father and mother were 173 cm (+0.4 SD) and 154 cm (−0.8 SD), respectively. Mid-parental target height ([paternal + maternal heights +13]/2) was 170 cm. His height at the first visit was 110 cm (−2.9 SD; see Figure 1). Magnetic resonance imaging showed pituitary enlargement. Evaluation of serum hormone levels showed primary hypothyroidism and GH deficiency. Serum free T3 was 2.20 pg/mL (2.2-4.1 pg/mL), free T4 was 0.12 ng/dL (0.8-1.9 ng/dL), thyroid-stimulating hormone (TSH) was >1230 µIU/mL (normal range = 0.4-4.0 µIU/mL), and the peak value of GH in response to insulin was 1.4 ng/mL. Anti-TSH receptor antibody was elevated, at 25.5% (<10%), while antithyroglobulin antibody and antithyroid peroxidase antibody levels were normal.
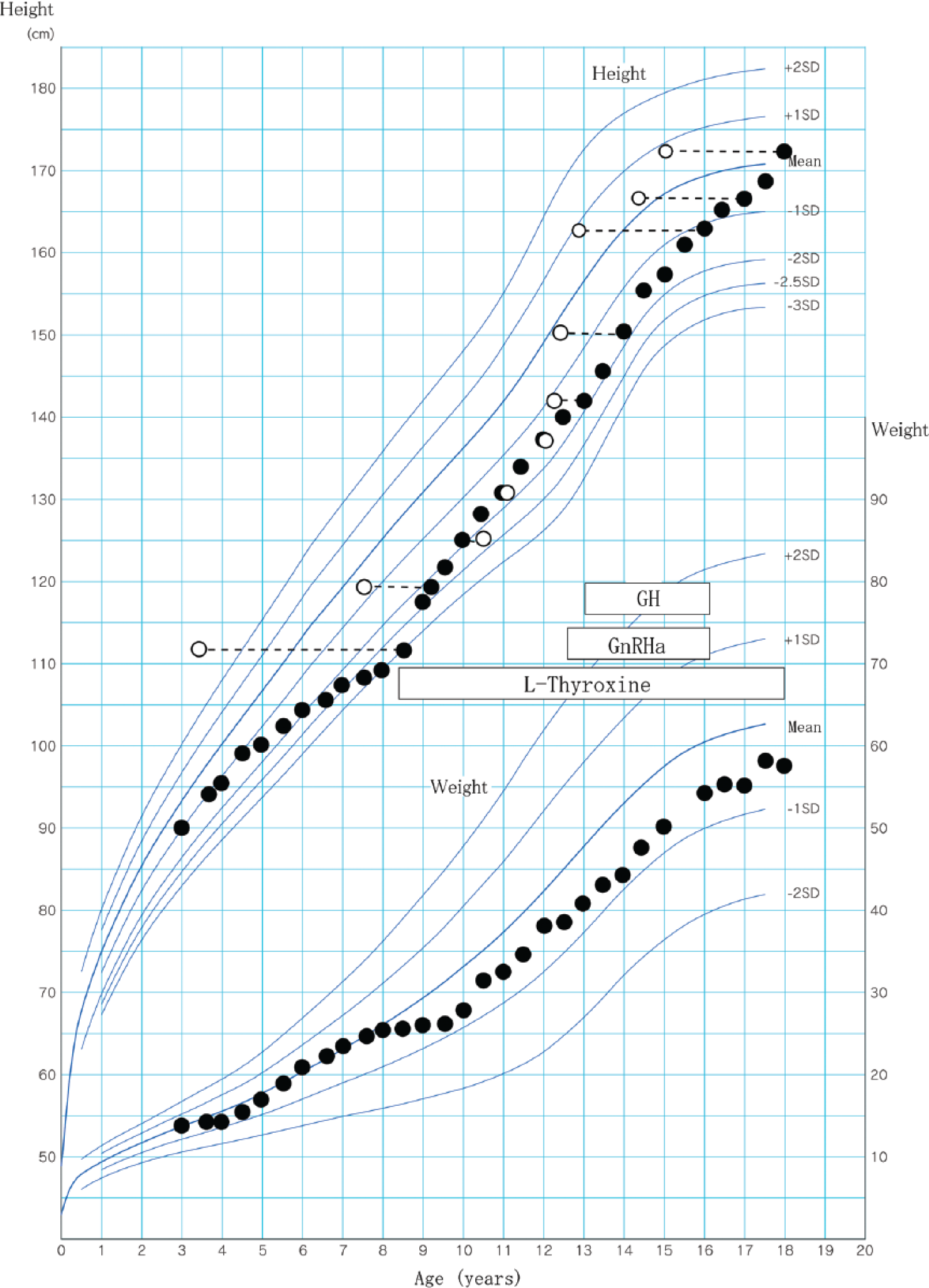
Cross-sectional growth chart for the present patient (Case 6).
Bone age was assessed by the Tanner and Whitehouse (TW2) method and was markedly retarded (3 years 5 months). We started thyroxine replacement therapy, which was adjusted to normalize thyroxine and TSH concentrations. Soon after beginning treatment with L-thyroxine, pituitary enlargement resolved, and his growth rate improved from 1.6 cm/year to 8.5 cm/year.
Five years after treatment, although his height had reached 136.7 cm (−1.6 SD) at age 12 years, his bone age was 12 years 0 months, an increase of more than 8 years. His pubertal stage was Tanner II of penis (absence of pubic hair), and peak luteinizing hormone and follicle-stimulating hormone values in response to a LHRH provocation test were 6.5 mIU/mL and 2.6 mIU/mL, respectively. Monthly GnRHa injections (leuprorelin 30 µg/kg) were started at age 12 years 7 months. Because he showed low growth rate (<−1.5 SD) for 2 years, we reconfirmed his GH deficiency twice (peak GH values in response to insulin, 0.8 ng/mL at age 11 years 10 months and 1.7 ng/mL at age 12 years 10 months). Insulin-like growth factor 1 was 146 ng/mL (125-557 ng/mL, −1.7 SD). The Japanese Foundation for Growth Science and the Japanese Health Insurance System for Pediatric Chronic Diseases approved our case as GH deficiency affected by pituitary gland enlargement. At age 13 years 0 months, daily GH replacement injections (0.2 mg/kg/week) were begun. Combined GnRHa/GH treatment was discontinued at age 16 years 1 month. At age 18 years his height was 172.8 cm (+0.3 SD).
Written informed consent was obtained from the patient and his legal guardian for publication of this case report.
Discussion
Most patients with atrophic thyroiditis exhibit rapid catch-up growth during the first 3 years after the start of thyroxine replacement treatment. In addition, transient GH deficiency usually improves after L-thyroxine treatment. However, they also experience a marked advance in bone age. 1 To avoid progression of puberty and improve final height, several patients were treated with combination therapy comprising L-thyroxine, GH, and GnRHa (Table 1).2-5 Persistent GH deficiency after L-thyroxine treatment was confirmed in Cases 1, 2, and 6 (our case) but was not described in the other cases. A recent study reported 2 patients with persistent GH deficiency after L-thyroxine treatment. 7 Such GH deficiency is likely attributable to pituitary enlargement resulting in compression of the pituitary stalk. We believe that pituitary gland enlargement caused irreversible damage to the somatotroph in our patient.
Effect of Combined Treatment With L-Thyroxine, Growth Hormone (GH), and Gonadotropin-Releasing Hormone Agonist (GnRHa).
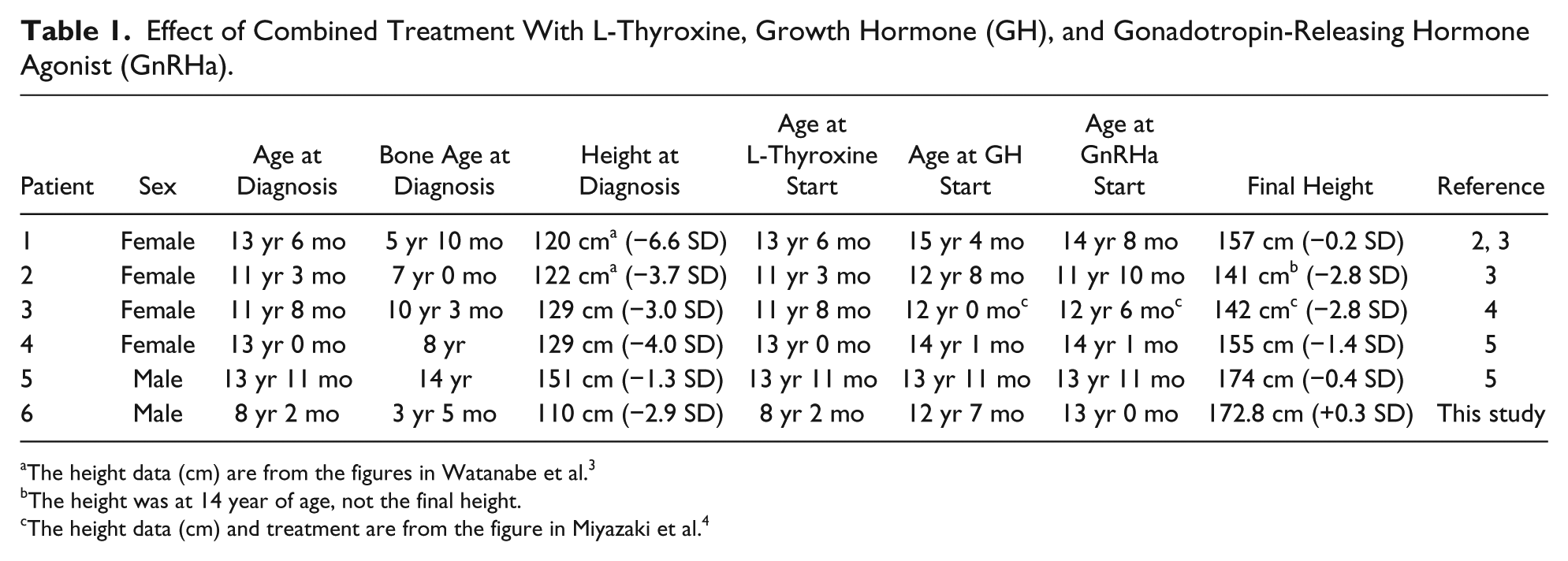
The height data (cm) are from the figures in Watanabe et al. 3
The height was at 14 year of age, not the final height.
The height data (cm) and treatment are from the figure in Miyazaki et al. 4
After combination therapy, 5 of the 6 cases, including our case, had improved SD scores for final height. Only one patient (Case 3) failed to respond to therapy, because she had reached puberty (menarche at age 10 years 3 months) before the start of treatment. 4 Our patient had a satisfactory height outcome because the diagnosis was made at a younger age, as compared with the other cases, and because combination therapy was started before puberty. We conclude that potential GH deficiency should be evaluated before puberty, to improve final height, even if catch-up growth is achieved with L-thyroxine therapy.
Japan has 2 highly effective systems for identifying and treating children with poor growth. First, all preschool/school children are seen by pediatricians who measure their height and weight at biannual or annual health check-ups. Thus, at-risk children are identified quickly, and pediatricians can accurately evaluate height velocity before diagnosis. Second, the Japanese health insurance system provides coverage to all Japanese. When a medical condition is diagnosed, the insurance system reimburses patients for most medical expenses, even when the treatment cost is extremely high, as is the case for hormone therapies. Our patient benefited from these systems and was thus able to attain normal adult height.
Author Contributions
TO: contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
NT: contributed to conception; contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
KN: contributed to design; contributed to interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MO: contributed to conception; contributed to acquisition; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
