Abstract
Hand, foot, and mouth disease (HFMD), with vesiculae on the hands, feet and mouth, is an infectious disease caused by many viral pathogens. However, the differences of immune response induced by these pathogens are unclear. We compared the clinical manifestations and the levels of immunologic indicators from 60 HFMD patients caused by different viral pathogens to analyze the differences in the immune response. It was shown that Th2 cytokines (IL-4 and IL-10) increased significantly in EV71-infected children; Th1 cytokines (IL-2 and IFN-γ) rose in CA16-infected children; both Th1 and Th2 cytokines elevated in non-EVG-infected children; only individual cytokines (such as IL-10) went up in EVG-infected children. Meanwhile, the antibodies induced by viral infection could not cross-interfere between the different pathogens. These differences might be due to variations in the immune response induced by the individual pathogens or to the pathogenesis of the infections by the individual pathogens.
Keywords
Introduction
In recent years, hand, foot, and mouth disease (HFMD) has been widely recognized as a highly infectious condition in children less than 5 years of age, especially in coastal areas of the Asian-Pacific region.1-3 On the Chinese mainland, outbreaks of HFMD have received substantial attention since 2008, following a large outbreak in Fuyang, Anhui Province. 4 Throughout the Chinese mainland, more than 12 million HFMD cases and 3200 deaths have been reported to date.5-11
The typical clinical manifestations of HFMD include vesicular eruptions and rashes on the hands, feet, and mouth and, in some cases, fever.12,13 Although these clinical manifestations are frequently mild and the prognosis is good, several children develop remarkable systemic neurological symptoms that lead to neurogenic pulmonary edema, cardiac and pulmonary failure, and other severe complications.13-16 Rapid disease progression can lead to death within 24 to 48 hours.17,18
Molecular epidemiologic data have demonstrated that the most common pathogens causing HFMD are enterovirus, coxsackievirus, and echovirus.19,20 Some characteristics of HFMD caused by these pathogens were similar, such as pathologic examinations show the presence of viral antigens in infected tissues,21,22 together with the increased expression of pro-inflammatory cytokines in the peripheral blood and, in severe infections, the cerebrospinal fluid.23-25 However, more than 80% of the severe cases and 90% of the deaths in HFMD are caused by enterovirus 71 (EV71) rather than by other viruses. 26 Importantly, repeated infections frequently develop in cases of coxsackievirus group A type 16 (CA16) infection, although CA16 infection usually leads to mild illness.27,28 These differences in the severity and appearance of HFMD suggest that the special immune responses were induced by the various pathogens.
In this study, we compared the clinical manifestations and associated immunologic indicators, including cytokines and chemokines, in the serum of HFMD patients to analyze the differences in the immune response and whether they were related to infection by a particular viral pathogen.
Materials and Methods
Ethics
HFMD patients were recruited from the placebo-controlled group of a phase III trial of an EV71 inactivated vaccine (prepared from human diploid cells). The clinical protocol and informed consent form were approved by the Ethics Committee of the Guangxi Center for Disease Control and Prevention (Guangxi CDC). The clinical protocol is registered in the ClinicalTrials website (NCT01569581).
HFMD Patients
Informed consent was obtained from the guardians of the 60 pediatric HFMD patients in the placebo-controlled group. These consisted of 16 patients positive for EV71 infection, 11 with positive CA16 infection, 15 positive for other enterovirus infections (EVG), and 18 who were negative for enterovirus infection (non-EVG). In parallel with the samples collected from these patients, samples from 5 healthy children without any clinical manifestations of HFMD who had also been included in the placebo-controlled group were also obtained.
Clinical Manifestations
The patients were observed for clinical manifestations, including vesicles, elevated body temperature, cough, and runny nose, according to the Guidelines for HFMD Diagnosis and Treatment, version 2010, published by the National Health and Family Planning Commission.29,30
Viral Etiology
Viral RNA was extracted from the stool samples or the throat swabs of the HFMD patients using the RNeasy mini kit (Qiagen, Hilden, Germany). 31 Etiological detection was carried out by real-time polymerase chain reaction, performed at the Guangxi CDC laboratories. The results were reconfirmed by the National Institutes for Food and Drug Control (NIFDC).
Cytokine and Chemokine Detection
The levels of cytokines and chemokines in the sera of the HFMD patients and controls (healthy children) were measured using Bio-Plex Pro human cytokine and chemokine kits (Bio-Rad, Hercules, CA) according to the manufacturer’s protocols. 32 This kit contains beads conjugated with monoclonal antibodies specific for IL-1β, IL-2, IL-4, IL-5, IL-6, IL-7, IL-8, and others. 32 Briefly, samples suspended in buffer were transferred to plates, incubated at room temperature for 1 hour with shaking at 850 ± 50 rpm, and then washed with the same buffer 3 times. Detection antibodies were added to the plates, which were then incubated as above for another 0.5 hours and then washed 3 times with the same buffer. SA-PE solution (Bio-Rad) was added to the plates in the dark, which were incubated for 10 minutes with shaking at 850 ± 50 rpm and washed 3 times with buffer. Finally, the beads were added and the samples were incubated with shaking as above for 30 seconds. The reactions were read using a Bio-Plex 200 system (Bio-Rad).
Anti-EV71 Neutralizing Antibody Detection
Specific anti-EV71 neutralizing antibody titers were detected using the classical micro-cytopathic effect neutralization assay according to the standard protocol.33,34 Briefly, the EV71 standard strain (NIFDC, China) was incubated with sera in different dilution at 37°C for 2 hours, transferred to 96-well plates containing a Vero cell suspension (104 per well), and mixed completely. After a 7-day incubation at 37°C, the samples were analyzed for the presence of cytopathic effects.
Statistical Analysis
Differences between groups were analyzed using SPSS software (Chicago, IL). A P value <.05 was considered to indicate a statistically significant difference.
Results
Pathogen Distribution of 60 HFMD Cases
HFMD is most often seen in children under 5 years of age. 35 In this study, HFMD developed in 33 children (33/60, 55%) younger than 2 years of age (Table 1). Among the 60 patients, 35 were male and 25 were female; 58 of the cases were mild and 2 were severe (Table 1). There was no significant difference between the age or sex of the patients and the degree of symptoms (except P = .018 between the CA16 and EVG groups).
Characteristics of the 60 Patients With HFMD Caused by Different Pathogens.

Abbreviation: HFMD, hand, foot, and mouth disease.
P < .05 of CA16-infected case versus non-EVG-infected case.
Clinical Manifestations of HFMD Cases Caused by Different Enteroviruses
The typical clinical character of HFMD is vesiculae or rashes on the hands, feet, and mouth.12,13 In this study, the typical rashes of the hands, feet, and mouth as well as the perianal region were seen in more than 60% of the patients (Table 2). Fever (body temperature >37.5°C) occurred in roughly 50% (Table 2). A few patients also had cough, runny nose, anorexia, and other symptoms (Table 2). In the 2 patients with severe HFMD, in addition to the typical rashes of the hands, feet, and mouth, high fever and oral vesicles developed in one patient, and a fever of 38.5°C accompanied by a mild neurological system disorder, manifested as a decreased response to stimulation, in the other (Table 2).
Clinical Characteristics of the 60 Patients With HFMD Caused by Different Pathogens.
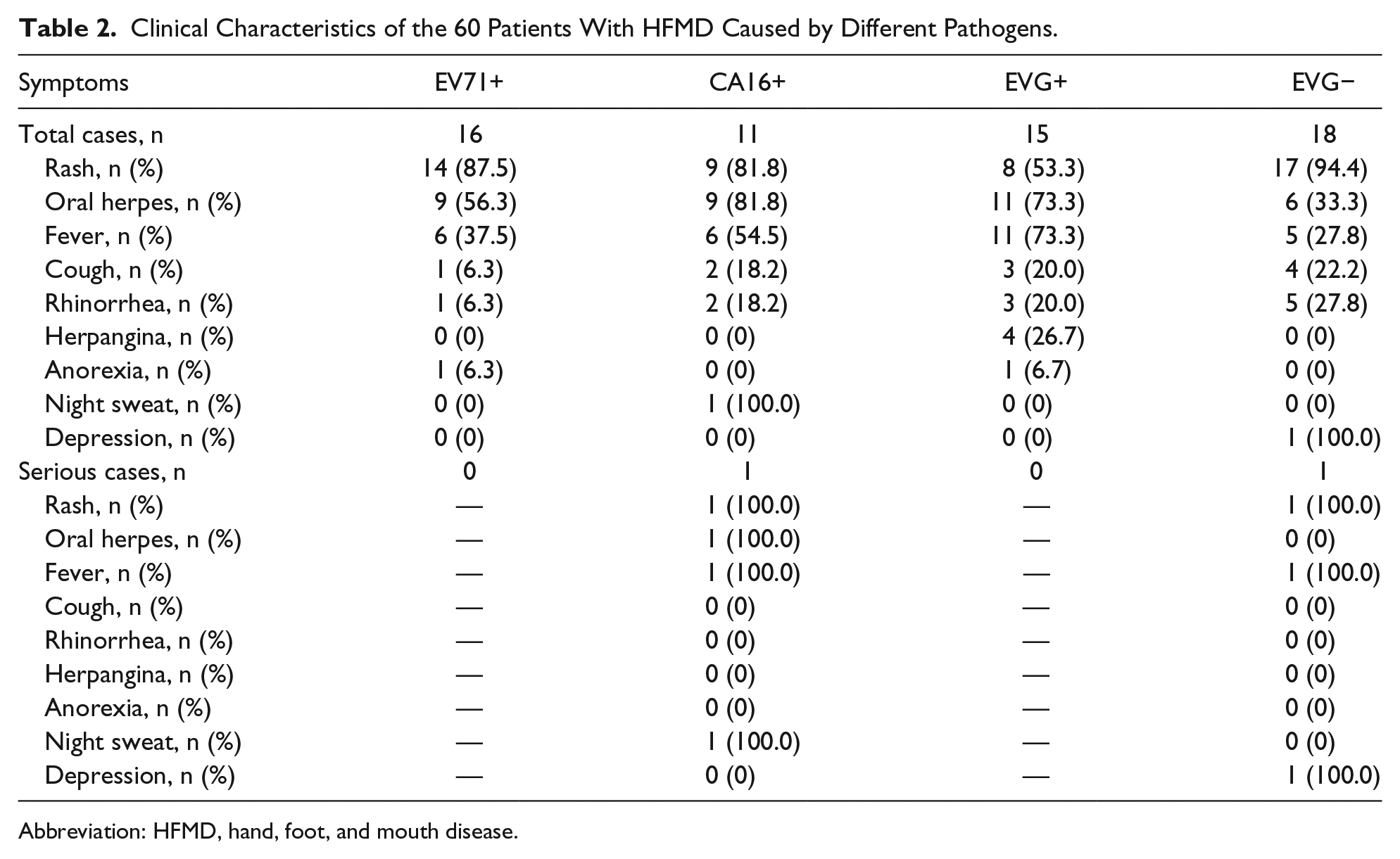
Abbreviation: HFMD, hand, foot, and mouth disease.
Cytokine Analysis of HFMD Cases Caused by Different Enteroviruses
Increased levels of pro-inflammatory cytokines have been clearly demonstrated in HFMD patients, 23 especially a remarkable increase in interleukin (IL)-6 in patients with severe EV71 infection.36,37 We therefore evaluated cytokine expression in our HFMD patients with respect to the different pathogens. Compared with the healthy children, EV71 infections was associated with high levels of IL-4 and IL-10, and CA16 infections associated with IL-2, IFN-γ, and IL-4, respectively (Table 3). By contrast, in the patients with HFMD caused by EVG, only an increase in the level of IL-10 was determined (Table 3). Nonenterovirus (non-EVG) infections triggered increases in IL-2, IL-16, IFN-γ, IL-4, and IL-10 (Table 3). These results suggested that the release of different pro-inflammatory cytokines was triggered by the different pathogens. The pro-inflammatory cytokines of the 2 patients with severe HFMD, caused by infection with CA16 and a non-EVG, respectively, showed similarities, including a clear elevation in IL-6 (Table 3). However, there are higher levels of IL-1 and IL-6 in the patient with the non-EVG infection than those of the patient with severe CA16 infection or the healthy children.
Cytokine Levels of the 60 Patients With HFMD Caused by Different Pathogens.

Abbreviation: HFMD, hand, foot, and mouth disease.
P < .05 of EV71-infected case versus non-EV-infected case.
P < .05 of EV71-infected case versus placebo.
P < .05 of CA16-infected case versus non-EV-infected case.
P < .05 of CA16-infected case versus placebo.
P < .05 of EV-infected case versus non-EV-infected case.
P < .05 of EV-infected case versus placebo.
P < .05 of non-EV-infected case versus placebo.
Chemokine Analysis of HFMD Cases Caused by Different Enteroviruses
Infections with exogenous pathogens trigger usually the release of a large variety of cytokines and chemokines.36,37 Our study showed that patients with different pathogen infections had high levels of the chemokines, including CCL1, CXCL6, CXCL12, CXCL13, and CX3CL1 (Table 4). Of these, the levels of CXCL6, CXCL12, and CXCL13 were significantly higher in HFMD patients with infections attributed to all of EV71, CA16, EVG, and non-EVG (Table 4). The level of CXCL5 was lower in HFMD patients with EV71, CA16, and EVG infections than in the healthy children (Table 4). In the 2 patients with severe HFMD, CCL25 levels were significantly higher than in the healthy controls (Table 4). However, in the patient with severe non-EVG HFMD, the levels of CXCL1, CXCL2, CCL3, CCL2, and CCL20 were much higher than in either the patient with severe CA16 infection or the healthy controls (Table 4).
Chemokine Levels of the 60 Patients With HFMD Caused by Different Pathogens.

Abbreviation: HFMD, hand, foot, and mouth disease; OV, over the highest level.
P < .05 of EV71-infected case versus non-EV-infected case.
P < .05 of EV71-infected case versus placebo.
P < .05 of CA16-infected case versus non-EV-infected case.
P < .05 of CA16-infected case versus placebo.
P < .05 of EV-infected case versus non-EV-infected case.
P < .05 of EV-infected case versus placebo.
P < .05 of non-EV-infected case versus placebo.
Anti-EV71 Immune Response in HFMD Caused by Different Enteroviruses
Cross-neutralization capacities between different enteroviruses have been demonstrated.38,39 Because EV71 infections can cause severe disease and even death,13,14 in this study we explored the interference of sera taken from HFMD patients infected with different pathogens against EV71 infection. The results showed that the sera of the patients caused by CA16, EVG, and non-EVG were failed to interfere EV71 infection, namely, being negative for anti-EV71 neutralizing antibody, whereas, as expected, serum from the patient with EV71 infection was positive (Figure 1).

Neutralizing antibodies against EV71 from the 60 patients with HFMD caused by different pathogens.
Discussion
HFMD is a highly infectious disease caused by different pathogens and is primarily seen in children.20,35 The basic clinical manifestations of HFMD in the majority of cases consist of rashes and vesicles involving the hands, feet, and mouth.12,13 However, there is epidemiological and immunological evidence of different manifestations of HFMD depending on the infecting strain of virus, in terms of both the pathology and the immune response.40,41 In this study, in accordance with the Good Clinical Practice guidelines and the Ethics Committee of our institution, we evaluated the clinical records and serum samples of 60 pediatric HFMD patients infected by different viral pathogens. These patients were drawn from the placebo group of a phase III trial of an EV71-inactivated vaccine. They did not show significant difference between the age or sex of the patients and the degree of symptoms. All 60 patients exhibited the typical, aforementioned symptoms of HFMD. In the 33 patients under 2 years of age (55%), fever was also present. In our 2 patients with severe HFMD, the infections were caused by CA16 and a non-EVG infection. However, our sample of 60 patients was too small to confirm viral specificity, such as which strain was easy to cause a severe case.
Furthermore, as functional responses of the host to the infecting pathogens, some special pro-inflammatory cytokines and chemokines were increased in our HFMD patients. These cytokines were associated with the immune system activation, such as CXCL6, which is closely associated with neutrophil migration recruit, 42 CXCL12 and CXCL13, which are associated with B cell migration and recruitment and antibody production.43-45
Although the previous study reported that the levels of IL-6, tumor necrosis factor-α (TNF-α), IP-10, IL-10, MCP-1, or CXCL9 were significantly higher in HFMD patients with neurological symptoms than in either those without or in healthy children,36,37 the expression of many chemokines associated with inflammation, except IL-4 and IL-10, did not show any remarkable increase, likely a storm, in most HFMD cases, not even in the 2 severe cases. It is not that the inflammatory responses were milder in the patients caused by non-EV71 virus than those induced by EV71, by which severe cases with neurological symptoms are often caused.13,14
Interestingly, along with the common clinical manifestations of vesicular eruptions on the hands, feet, and mouth, different profiles of cytokine and chemokine responses were noted in our HFMD patients infected by different viruses. These can be summarized as follows: EV71 infections were characterized by significantly increased levels of Th2 cytokines (IL-4 and IL-10) and CA16 infections by significantly increased levels of Th1 cytokines (IL-2 and IFN-γ). By contrast, non-EVG infections seemed to directly trigger parallel increases in both Th1 and Th2 cytokines, whereas in EVG infections variations were detected only in individual cytokines, such as IL-10. Similarly large, virus-specific variations occurred in the levels of multiple chemokines. For example, the levels of CXCL1 were significantly increased in EVG-infected patients, CCL21 in CA16-infected patients, and CCL17 in non-EVG-infected patients. The differences in these immune effectors might be due to variations in the immune response induced by the individual pathogens or to the pathogenesis of the infections by the individual pathogens. 46
Finally, the neutralizing assay using sera from the HFMD patients showed no cross-interference between the different pathogens that cause HFMD. Our previous data and phase III trial of an EV71 inactivated vaccine had demonstrated effective immune protection induced in the individuals infected by EV71 or immunized by the vaccine. Thus, repeat HFMD infection might reflect an infection by the different pathogen or that the abnormal immune response might be related to CA16 or EVG infection.
Taken together, our results suggest the importance of HFMD in determining the exact pathogen when considering the common clinical manifestations induced by the infection of different viruses.
Author Contributions
QL contributed to conception and design, contributed to acquisition, contributed to acquisition, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring itegrity and accuracy; YZ contributed to conception and design, contributed to analysis and interpretation of data, drafted manuscript, critically revised manuscript, gave final approval and agrees to be accountable for all aspects of work ensuring itegrity and accuracy, JW contributed to aquisation and analysis od data, critically revised manuscript, gave final approval, JP contributed to acquisition, critically revised manuscript, gave final approval and agrees to be accountable for all aspects of work ensuring itegrity and accuracy, LL contributed to acquisition, gave final approval and agrees to be accountable for all aspects of work ensuring itegrity and accuracy, CY contributed to acquisition, critically revised manuscript and gave final approval; YL , WL, GL, MF, YL, SF, LC, contributed to acquisition and analysis of data and gave final approval
Footnotes
Acknowledgements
Many thanks all the investigators from Guangxi Provincial Center for Disease Control and Prevention, Guilin City Center for Disease Control and Prevention, Yangshuo County Center for Disease Control and Prevention, Lipu County Center for Disease Control and Prevention, Yongfu County Center for Disease Control and Prevention, Lingui County Center for Disease Control and Prevention, Lingchuan County Center for Disease Control and Prevention, Xin’an County Center for Disease Control and Prevention and Quanzhou County Center for Disease Control and Prevention who worked on the trial.
Authors’ Note
Authors Jingjing Wang and Jing Pu contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Basic Research Program (2011CB504903), the National High-Tech R&D Program (2012AA02A404), the Technology Development Research Institutes (2013EG150137), the State Project for Essential Drug Research and Development (2012ZX09101319 and 2014ZX09102042) and Yunnan Province Project (2013FA024, 2014FB191 and 2014HB066).
