Abstract
Introduction
Cerebral microbleeds (CMBs) signify subclinical brain parenchymal damage resulting from cerebral microvascular pathology. Histological analyses demonstrate the pathological accumulation of hemosiderin-laden macrophages surrounding the blood vessels. The relationship between CMBs and cognitive impairment has garnered increasing attention. Existing research indicates that CMBs are associated with cognitive impairment in patients with Alzheimer’s disease, lacunar infarcts, and Parkinson’s disease (Fazekas et al., 1999; Nakamori et al., 2020; Zhu et al., 2022). CMBs are key indicators of increased risk for cognitive impairment, stroke, and mortality, substantially affecting patients’ physical and mental health as well as their quality of life (Vázquez-Justes et al., 2022; Zand et al., 2018). Early diagnosis and management of cerebral microbleeds are clinically significant for improving the prognosis of dementia patients, including those with Alzheimer’s disease, vascular dementia, and mixed dementia (Charidimou et al., 2018; Hussein et al., 2023). The detection of CMBs may be more critical in the management of vascular dementia because they directly indicate underlying vascular pathology. This, in turn, guides clinicians in taking targeted measures to prevent further vascular damage. In neurodegenerative dementias such as Alzheimer’s disease, the presence of CMBs may reflect vascular comorbidity. In these cases, CMBs may serve more as an auxiliary diagnostic marker rather than a key factor directly influencing treatment decisions. The association between cerebral microbleeds and cognitive impairment is garnering considerable attention in the medical community. Furthermore, the early intervention of CMBs has emerged as a focal point in neuroscientific research.
Susceptibility Weighted Imaging (SWI), a cutting-edge Magnetic Resonance Imaging (MRI) technique, has the advantage of being able to discriminate calcium from blood, which is one of the key benefits of SWI sequences over conventional sequences such as T2*-weighted gradient echo (2D-GRE) (Haller et al., 2021; Martínez Camblor et al., 2023). The sensitivity of SWI in detecting CMBs depends on its magnetic field strength, higher spatial resolution, and the use of longer echo times. These factors enhance magnetic susceptibility contrast and prominence (Haller et al., 2021). This technique excels in identifying minuscule cerebral hemorrhagic lesions, often undetectable in standard MRI sequences, rendering it the most sensitive MRI method for detecting CMBs (Martínez Camblor et al., 2023; Puy et al., 2021). On Susceptibility Weighted Imaging, cerebral microbleeds manifest as distinct, oval or round lesions, typically measuring 2 to 10 mm in diameter. These lesions, encircled by brain parenchyma, present as hypointense foci. Their paramagnetic properties result in signal loss attributable to susceptibility effects (Greenberg et al., 2009; Haller et al., 2018). The number and location of CMBs detected by SWI can aid in assessing the risk of ischemic and hemorrhagic strokes. This information can influence clinical decision-making, treatment planning, and prognosis (Wach-Klink et al., 2021).
Presently, there is a lack of consensus regarding the risk factors for CMBs. Population-based studies find that CMBs are associated with traditional cardiovascular and cerebrovascular risk factors, including age, male gender, hypertension, smoking, and excessive alcohol consumption (Haller et al., 2018; Wach-Klink et al., 2021). CMBs are also associated with a history of stroke, atrial fibrillation, the use of antiplatelet drugs, the use of anticoagulants, and white matter hyperintensities (WMH) (Horstmann et al., 2015; Lau et al., 2017; Liu et al., 2020). However, some studies have found inconsistent correlations between CMBs and diabetes, dyslipidemia, and white matter hyperintensities (WMH) (Lv & Li, 2023; Wach-Klink et al., 2021).
As one of the markers of cerebral small vessel disease, CMBs may serve as potential indicators for detecting, quantifying, and predicting cognitive impairment. The relationship between CMBs and cognitive dysfunction has garnered increasing attention in recent years. The connection between the quantity of CMBs, their spatial distribution within the brain, and cognitive function is still not well-defined (Hussein et al., 2023; Lv & Li, 2023). Cognitive impairment includes executive dysfunction and a decline in information processing speed. It also involves varying degrees of impairment in language, memory, attention, and visuospatial abilities (R. Yang et al., 2023). Brain networks are complex, large-scale networks mapping the brain’s structure and function, characterized by “small-world” properties. There is a strong spatial correspondence between cognitive functions and brain network modules, which may offer advantages over traditional structural lesions in explaining cognitive dysfunction (Du & Xu, 2019). Although numerous studies indicate that the number of CMBs is associated with the severity of cognitive impairment, different locations of CMBs represent different underlying pathologies. Multiple spatially distributed CMBs may affect cognitive function by disrupting brain network connectivity (Mitaki et al., 2023; R. Yang et al., 2023). Nannoni et al. demonstrated that the number of CMBs is independently associated with cognitive impairment, with significant detriments observed in executive function and information processing speed (Nannoni et al., 2022). A bibliometric analysis revealed that the relationship between CMBs and cognitive impairment is an active research area. Most studies support a close association between the two, though some research has not found a significant correlation between CMBs and cognitive dysfunction (R. Yang et al., 2023). The relationship between different quantities and locations of CMBs and various cognitive domains of cognitive impairment requires further research and exploration.
Therefore, this study utilizes SWI to diagnose cerebral microbleeds, with the goal of elucidating the risk factors linked to CMBs and examining the relationship between CMBs and cognitive decline. It aims to lay a groundwork for the etiological research of cognitive disorders and aspires to open new avenues for the prevention and treatment of cognitive impairment.
Materials and Methods
Subjects
A cohort of 236 patients diagnosed with cerebral small vessel disease was selected from Department of Neurology, Second Affiliated Hospital of Shandong First Medical University, spanning from September 2022 to September 2023. Based on susceptibility-weighted imaging (SWI), 124 cases were categorized into the cerebral microbleeds group (CMBs group) and 112 cases into the non-cerebral microbleeds group (Non-CMBs group). Both groups signed informed consent forms. This study obtained approval from the hospital’s ethics committee.
Inclusion Criteria: 1. Age range: 50 to 80 years. 2. Diagnostic cranial MRI indicators of cerebral small vessel disease, including one or more of the following: lacunar infarct, white matter lesions, cerebral microbleeds, enlarged perivascular spaces, or new small subcortical infarcts (axial diameter <20 mm). 3. Ability to participate in neuropsychological assessments. 4. Absence of medications affecting cognition. 5. Provision of informed consent.
Exclusion Criteria: 1. Large vessel infarcts evident on cranial MRI. 2. Cranial vascular Doppler ultrasound, carotid ultrasound, MRA, CTA, or other tests confirming cerebral or carotid artery stenosis ≥50%. 3. Conditions known to impair cognition, such as Alzheimer’s disease, Cerebral amyloid angiopathy, Parkinson’s disease, epilepsy, encephalitis, brain tumors, history of brain trauma, preexisting diagnosis of mild cognitive impairment (MCI), etc. 4. Severe cardiac, hepatic, renal, or other vital organ diseases. 5. Significant visual, auditory, or speech disabilities impacting participation. 6. Exclusion of acute, large-area cerebral infarction and cerebrovascular disease sequelae.
Clinical Data and Cognitive Function Assessment
Clinical data and laboratory test results were systematically recorded for all participants, encompassing demographic and health-related variables (gender, age, education level, body mass index), medical history (hypertension, diabetes, hyperlipidemia, coronary heart disease, cerebral infarction, cerebral hemorrhage, smoking, alcohol consumption), and laboratory values (fibrinogen, uric acid, glycosylated hemoglobin, cholesterol, triglycerides, high-density lipoprotein, low-density lipoprotein, homocysteine).
The collection and measurement methods are as follows: Body Mass Index (BMI) was calculated by dividing weight (in kilograms) by the square of height (in meters). Smoking history was defined as smoking more than one cigarette per day on average for a continuous period exceeding 6 months. Alcohol consumption history was defined for men as a daily intake of more than 40 g of pure alcohol for a continuous period exceeding 6 months, and for women as a daily intake of more than 20 g of pure alcohol for a continuous period exceeding 6 months. Information on the presence of hypertension, diabetes, dyslipidemia, coronary artery disease, and history of stroke was obtained from medical records or clinical diagnosis. Blood samples were taken after at least 8 hr of fasting upon the patient’s admission to measure blood markers such as uric acid, blood glucose, and lipids.
Cognitive function was assessed by seasoned neurologists using the MoCA scale, ensuring consistent language, tools, and settings for the evaluation. The MoCA encompasses various cognitive domains, including visuospatial and executive functions, memory, attention, language, abstraction, delayed recall, and orientation. Scores were education-adjusted, with ≥26 indicating normal cognition, and an additional point given for those with ≤12 years of education.
A GE Discovery 750 3.0T MRI scanner was employed for routine T1WI, T2WI, and FLAIR imaging to rule out organic brain lesions, followed by SWI for detailed analysis. The Microbleed Anatomical Rating Scale (MARS) was used for grading the location and quantity of CMBs (Gregoire et al., 2009). Cerebral microbleeds were categorized based on location into cortical-subcortical, deep, and mixed types. The count of CMBs in each region was tabulated, and cases were further classified into mild (≤4 lesions), moderate (5–9 lesions), and severe (≥10 lesions) CMB groups.
Statistical Methods
Data processing and statistical analyses were performed using SPSS version 29.0. For quantitative data, one-way ANOVA was applied to compare differences among multiple groups, while independent sample t-tests were utilized for pairwise group comparisons. Pearson correlation analysis was employed to examine the relationships between various indicators. A p-value of less than .05 was deemed to indicate statistical significance.
Results
Comparison of Basic Clinical Data Between Two Groups of Cases
Patients were stratified into two cohorts according to the presence or absence of CMBs: a non-CMBs group (112 cases) and a CMBs group (124 cases). Notable statistical differences were observed between these groups concerning gender, age, hypertension, diabetes, history of cerebral infarction, and history of alcohol consumption (p < .05). Further details can be found in Table 1.
Basic Clinical Data Comparison Between Patients With and Without CMBs.
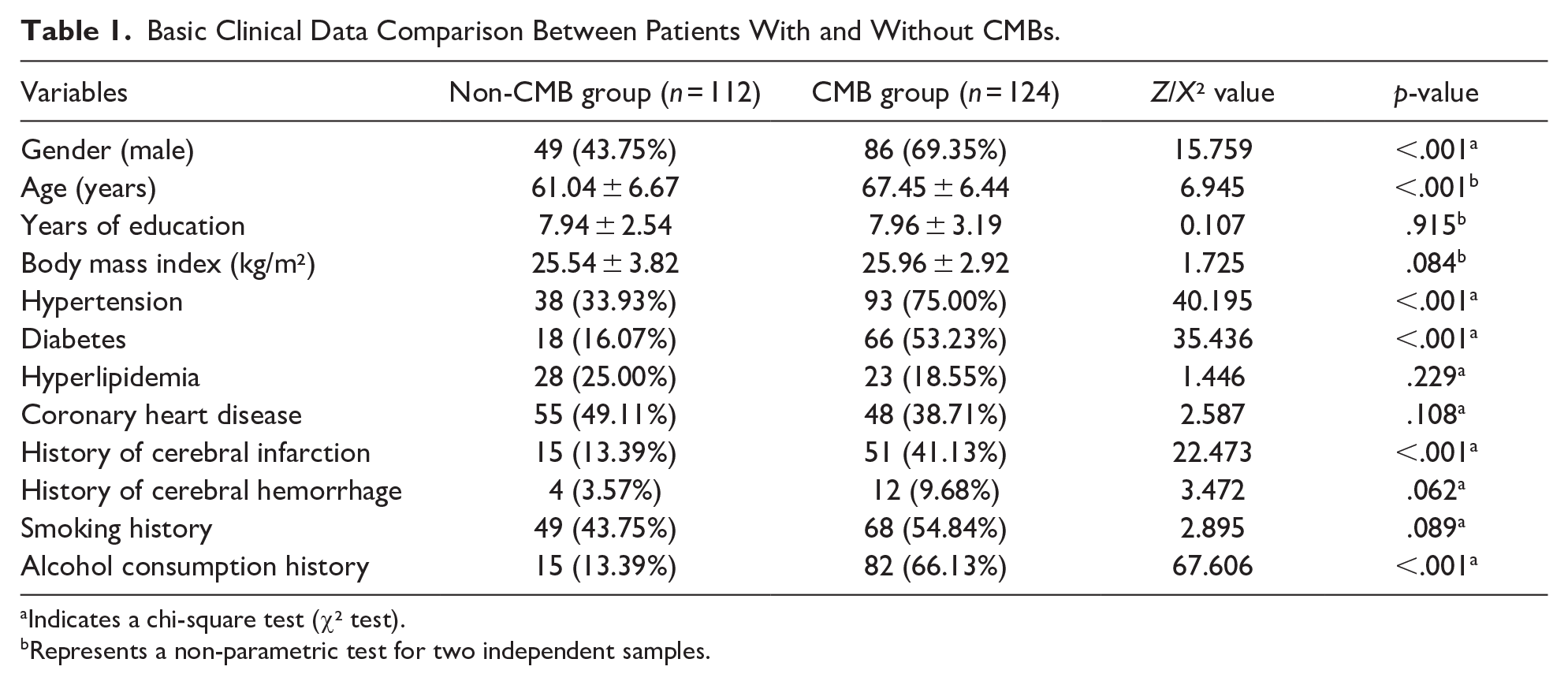
Indicates a chi-square test (χ² test).
Represents a non-parametric test for two independent samples.
Comparison of Laboratory Indicators Between Two Patient Groups
Significant differences in levels of glycosylated hemoglobin, low-density lipoprotein, and homocysteine were found when comparing patients without CMBs to those with CMBs (p < .05). For more specifics, see Table 2.
Laboratory Indicators Comparison Between Patients With and Without CMBs.

Represents a t-test.
Represents a non-parametric test.
Analysis of Independent Risk Factors for CMBs
Multifactorial logistic regression analysis was employed to evaluate the risk factors exhibiting statistically significant differences between the two groups. This analysis pinpointed age, hypertension, diabetes, history of alcohol consumption, and elevated homocysteine levels as independent risk factors for CMBs. Subsequently, binary logistic regression analysis was utilized to ascertain the independent risk factors influencing the occurrence of CMBs. The validity of the model was confirmed by the goodness-of-fit test (χ² = 160.401, p < .001). The analysis identified age, a history of hypertension, diabetes, alcohol consumption, and homocysteine (HCY) levels as distinct risk factors for CMBs (p < .05). Specifically, the incidence of CMBs escalates by 1.099 times with each additional year of age; the risk of developing CMBs is 0.399 times lower in individuals without hypertension compared to those with hypertension; the risk is 0.181 times lower in individuals without diabetes compared to those with diabetes; absence of a history of alcohol consumption reduces the risk by 0.064 times compared to those with such a history; and each 1 umol/L increase in HCY corresponds to a 1.271 times higher risk of developing CMBs. Comprehensive details are available in Table 3.
Binary Logistic Regression Analysis of Independent Risk Factors for CMBs.

Among patients exhibiting cognitive impairment, the average number of CMBs was recorded as 11.54 ± 6.510, and their mean MoCA score was 23.03 ± 2.375. As depicted in scatter plot Figure 1, Spearman correlation analysis demonstrated a negative linear relationship between the quantity of CMBs and the total MoCA score (r = −.837, p < .001). This correlation suggests that an increase in the number of CMBs is typically associated with lower MoCA total scores.

The scatter diagram of the number of CMBs and MoCA scores.
Relationship between the Severity of CMBs and MoCA Cognitive Domains
The severity of illness in patients was evaluated based on the quantity of CMBs, leading to their classification into mild (48 cases), moderate (44 cases), and severe (32 cases) categories. Relative to the cohort without CMBs, those with CMBs exhibited notable reductions in visuospatial execution function and delayed recall abilities, and these differences were found to be statistically significant (p < .05). Refer to Table 4 for detailed information.
Relationship Between Severity of CMBs and MoCA Cognitive Domains.

Significant difference compared to non-CMB group (p < .05).
Comparison of MoCA Cognitive Domains with the Distribution of CMBs
Based on the localization of CMBs, patients in the CMBs group were further subdivided into deep (56 cases), cortical-subcortical (45 cases), and mixed (23 cases) categories. The findings of the study revealed that although the location of CMBs did not correlate with the total MoCA score, it was significantly associated with impairments in various cognitive domains. Specifically, deep CMBs were associated with deficits in visuospatial execution function, naming, attention, calculation, language, delayed recall, and orientation (p < .05). Cortical-subcortical CMBs were linked to impairments in visuospatial execution function, attention, calculation, and delayed recall (p < .05). In the case of mixed CMBs, the related impairments were in visuospatial execution function and naming ability (p < .05). Refer to Tables 5 and 6 for detailed information.
Differentiated Cognitive Domain Scores of MoCA in Various CMBs Locations.

The Correlation Between the Quantity of CMBs in Different Locations and the Scores in Various Cognitive Domains of the MoCA.

Correlation is significant at the .01 level (2-tailed).
Correlation is significant at the .05 level (2-tailed).
Discussion
Independent Risk Factors for CMBs
The findings from this research established age, hypertension, diabetes, history of alcohol consumption, and elevated homocysteine levels as independent risk factors for CMBs. In a related study, Lu et al. analyzed basic demographic data and cranial SWI results from 8,159 individuals in the UK Biobank, corroborating that age is a significant independent risk factor for CMBs (Lu et al., 2021). The prevalence of CMBs shows a progressive increase with advancing age. The incidence rate is observed at 6.5% in the population aged between 45 to 50 years, and this rate significantly escalates to 35.7% in individuals who are 80 years and older (Poels et al., 2010).
Elmståhl et al. conducted a comprehensive longitudinal study involving 2,931 participants, concluding that hypertension is a significant risk factor for CMBs. This study particularly highlighted a strong link between hypertension and non-lobar CMBs (Elmståhl et al., 2019). Additionally, Lyu et al. established that hypertension not only predisposes individuals to CMBs but also indicated that the classification of hypertension is directly correlated with the severity of CMBs (Lyu et al., 2020). Moreover, research by Liang et al. revealed that among patients with Grade 1 and Grade 2 hypertension persisting for over 20 years, approximately 69% displayed CMBs. Their findings also suggested that the probability of developing CMBs escalates with both the severity and the duration of hypertension (Liang et al., 2022).
Diabetes is now widely acknowledged as an independent risk factor for cognitive impairment, with individuals suffering from diabetes having a 1.5 to 2.0 times higher likelihood of developing cognitive impairment compared to those with normal blood sugar levels. Emerging research also suggests an increased susceptibility to CMBs among diabetic patients (Cukierman-Yaffe et al., 2020). This study aligns with these findings, further affirming that diabetes is indeed a risk factor for CMBs (Cordonnier et al., 2007; Qiu et al., 2008). In related research, Yu and colleagues discovered that in populations without a history of stroke or transient ischemic attacks, there is a notable association between glycosylated hemoglobin levels and CMBs. Notably, they found that effectively managing glycosylated hemoglobin levels in diabetic patients can mitigate the risk of developing CMBs (Yu et al., 2022).
While this study did not perform a stratified analysis on alcohol consumption, the criteria for inclusion based on a history of alcohol consumption were defined as surpassing moderate or social drinking levels. The findings indicate that such a history of alcohol consumption is an independent risk factor for CMBs, a conclusion that is in harmony with Lu’s research findings (Lu et al., 2021). Furthermore, a cross-sectional Mendelian randomization analysis by Takashi et al. indicates a positive correlation between alcohol consumption and the risk of cerebral small vessel disease, which encompasses CMBs.
The findings of this study underscore that elevated homocysteine levels serve as an independent risk factor for CMBs. In a comprehensive multivariate analysis by Yoo et al., plasma homocysteine levels were confirmed as an independent predictive factor for CMBs, even after adjusting for variables such as age, gender, hypertension, and other potential confounders (Yoo et al., 2020). Similarly, Cao et al., utilizing the Mendelian randomization method in their research, established a link between plasma homocysteine levels and the overall radiological burden of cerebral small vessel diseases, including CMBs. Their findings particularly emphasize the need for reducing plasma homocysteine levels in patients genetically predisposed to hyperhomocysteinemia (Cao et al., 2021).
Age, hypertension, diabetes, alcohol consumption history, and elevated homocysteine levels are independent risk factors for CMBs. These factors are also vascular risk factors in elderly patients. Studies have found that they impact the instrumental activities of daily living (IADL) abilities of elderly patients, particularly impairing their financial management skills (Giannouli & Tsolaki, 2022). In our subsequent research, we will conduct a more extensive and in-depth study on the relationship between vascular risk factors and cognitive impairment.
The MICON cohort study included 7,839 subjects and found that among atrial fibrillation patients receiving antiplatelet and anticoagulant therapy as secondary prevention after an ischemic stroke, the presence of CMBs was more strongly associated with subsequent intracranial hemorrhage (Soo et al., 2023). Additionally, patients with a history of stroke or atrial fibrillation who require long-term use of antiplatelet and anticoagulant medications have been found in some studies to have an association between the use of these medications and the occurrence of CMBs (Graff-Radford et al., 2021; Yu et al., 2022). In this study, patients who were not explicitly excluded from taking antiplatelet drugs, anticoagulants, and those with atrial fibrillation were included. The presence of these factors may influence the incidence of CMBs, leading to potential bias in the study results. However, completely eliminating all potential confounding factors in clinical research is challenging. Despite these potential influences, this study still provides valuable information about CMBs. To reduce such bias, future research should consider more rigorous selection of subjects to improve the accuracy of the results.
Relationship Between CMBs and Cognitive Impairment
Recent research increasingly points to a correlation between CMBs in patients with cerebral small vessel disease and cognitive impairment. This study delves into analyzing this relationship, focusing on the variance in quantities and locations of CMBs (Nannoni et al., 2022). To facilitate this analysis, the Microbleed Anatomical Rating Scale (MARS) was employed. MARS offers a straightforward and expedient approach for evaluating CMBs, making it an ideal tool for classifying their severity and pinpointing their locations within the brain. The Montreal Cognitive Assessment (MoCA) is a widely validated comprehensive screening tool for cognitive impairment, specifically designed to detect mild cognitive impairment across different cognitive domains. Its sensitivity and specificity are well-documented in the literature, making it suitable for detecting early cognitive changes related to CMBs in this study. Additionally, some authors of this paper are clinicians, and the simplicity of the MoCA scale makes it suitable for use in clinical settings. Therefore, the MoCA scale was employed for cognitive assessment of the enrolled cases in this study.
In this research, a linear relationship was identified between the number of CMBs and the overall MoCA score, where a higher count of CMBs correlated with lower MoCA scores. When compared to participants without CMBs, those with CMBs exhibited significant reductions in visuospatial execution function and delayed recall abilities. This suggests that the severity of CMBs is closely linked to cognitive impairment, particularly affecting visuospatial execution function and delayed recall skills. Nannoni et al. confirmed this independent relationship between the presence of CMBs and cognitive impairment, a finding that remained significant even after adjusting for the number of lacunar infarcts and the severity of white matter lesions. This underscores the independent contribution of CMBs to cognitive decline (Nannoni et al., 2022). Furthermore, Zhang et al.’s research on primary hypertension patients without a history of transient ischemic attacks or strokes revealed that the number of CMBs is independently correlated with cognitive impairment. Notably, a higher count of CMBs (≥5) was associated with lower total MoCA scores and reduced performance in specific cognitive areas, including overall cognitive function, information processing speed, and motor speed (Zhang et al., 2018). Similarly, Gyanwali et al. conducted a 3-year follow-up study with 428 participants, which demonstrated that an increase in the baseline number of CMBs corresponded with a general cognitive decline, as well as specific reductions in executive function and memory. This aligns with the findings of our study (Gyanwali et al., 2021).
Cognitive functions are governed by various brain regions, suggesting a potential link between the locations of CMBs and cognitive impairment. This study explored the association between CMB distribution and cognitive domain impairments. We found that deep CMBs correlate with impairments in visuospatial execution function, naming, attention, calculation, language, delayed recall, and orientation. Yang et al. demonstrated that in ischemic cerebrovascular disease, CMB locations affect different cognitive domains, with thalamic and basal ganglia CMBs impacting memory and visuospatial executive function, and subcortical CMBs influencing abstract thinking (J. Yang et al., 2018). Our findings also reveal that cortical-subcortical CMBs compromise visuospatial execution function, attention, calculation, and delayed recall. A Rotterdam study with 4,841 participants showed lobar CMBs lead to declines in executive function, information processing, and memory (Akoudad et al., 2016). Similarly, Mitakid et al.’s longitudinal study on 769 individuals over 7.3 ± 3.5 years found a causal link between lobar CMBs and declining executive function. Our study further indicates that mixed CMBs impair visuospatial execution function and naming ability (Mitaki et al., 2023). Moreover, Valenti et al. discovered that both deep and cortical CMBs affect attention, executive function, and fluency, consistent with our observations (Valenti et al., 2016).
Currently, consensus is lacking regarding the relationship between CMBs in different brain locations and their impact on various cognitive domains. This variability may stem from factors like the age, education level, and ethnicity of study populations, alongside the distinct pathophysiological characteristics of CMBs depending on their location. Deep CMBs, commonly linked to atherosclerosis in deep penetrating arteries, are often associated with hypertension and smoking. The pathophysiological mechanism is believed to involve hyalinization of the small artery walls. Lobar CMBs, on the other hand, are connected with β-amyloid deposition in cerebral cortex or leptomeningeal blood vessels, influenced by factors such as the ApoE4 allele and body mass index. Asymptomatic lobar CMBs might also indicate cerebral amyloid angiopathy (Graff-Radford et al., 2019; Lu et al., 2021). Consequently, in individuals with well-controlled cerebrovascular risk factors, β-amyloid deposition might exert a greater influence on cognitive function than atherosclerosis. Moreover, a meta-analysis indicates that Eastern populations have a higher prevalence of deep and/or infratentorial CMBs compared to Western populations, suggesting a potential genetic basis for CMB incidence sites. This insight paves the way for future research targeting diverse populations (Yakushiji et al., 2019).
The exact pathogenic mechanism through which CMBs contribute to cognitive impairment remains unclear. Histopathological studies have shown that oxidative stress, inflammation, and degenerative changes, which weaken microvascular structures and cause endothelial dysfunction, are instrumental in damaging cerebral small arteries. Contributing factors to the development of CMBs include hypertension, amyloid deposition, and perivascular glial proliferation. These elements may release bioactive substances like hemosiderin, which in turn inflict direct damage on brain tissue adjacent to small vessels. Such damage can cause ultrastructural harm to cortical-subcortical neural fiber bundles and disrupt neural networks, leading to cognitive dysfunction (Ungvari et al., 2017; Zhang et al., 2018). Additionally, CMBs can interfere with the function of nearby neurons and potentially compromise the blood-brain barrier (Charidimou & Werring, 2012; Moran et al., 2017). Moreover, pathological alterations in the small blood vessels that result in CMBs, along with chronic diffuse hypoperfusion and the release of biologically active substances from blood breakdown products, can cause focal or diffuse damage to adjacent brain tissue. This damage is linked to impairments in specific cognitive domains and can extend to multiple cognitive areas (Cao et al., 2021). The association between the distribution of CMBs and specific cognitive domain impairments may stem from the direct impact of CMBs on brain tissue, as well as from damage to frontal-subcortical circuits and white matter fiber tracts (Wu et al., 2014).
This study is limited by its single-center design, relatively small sample size, and potential variability arising from differences in age, population demographics, detection methodologies, and evaluation techniques. These factors may contribute to inconsistencies in the findings. To gain a deeper understanding of the link between the locations of CMBs and cognitive impairment, future studies should encompass multi-center, large-sample prospective cohort research. Moreover, cognitive impairment is influenced by a range of cerebrovascular conditions beyond CMBs, including white matter hyperintensities, lacunar infarctions, perivascular spaces, and brain atrophy. Research such as Akouda et al.’s indicates that CMBs are not isolated determinants of cognitive decline but are secondary to severe vascular and neurodegenerative diseases (Akoudad et al., 2016). Depression is also a significant factor affecting cognitive impairment. Numerous studies have shown that patients with depression experience changes in perception, attention, memory, and executive function (Giannouli & Tsolaki, 2021).
Future research should incorporate approaches from neuropsychology and neuropathology to enhance early detection and intervention strategies for CMBs, ultimately aiming to improve cognitive functions in patients.
Conclusion
In conclusion, this study identifies age, hypertension, diabetes, a history of alcohol use, and elevated homocysteine levels as independent risk factors for CMBs. There is a linear correlation between the severity of CMBs and cognitive impairment, with patients exhibiting declines in visuospatial execution function and delayed recall abilities. Additionally, the location of CMBs is linked to impairments in different cognitive domains.
Based on the research findings, clinical management should emphasize early screening and diagnosis of CMBs in patients with high-risk factors. Personalized treatment plans should be implemented, including strict control of blood pressure and blood glucose, reduction or cessation of alcohol consumption, promotion of a healthy lifestyle, and regular assessment and intervention for cognitive function. Additionally, patient education and follow-up should be strengthened to effectively manage CMBs, slow cognitive decline, and improve patients’ quality of life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work were supported by funding from Shandong Province Medical and Health Technology Project (No.202309010656), Tai’an Science and Technology Innovation Development Project (No. 2023NS366, No.2022NS176, No.2022NS305).
Ethical Approval
The experimental protocol was established, according to the ethical guidelines of the Helsinki Declaration and was approved by the Human Ethics Committee of The Second Affiliated Hospital of Shandong First Medical University. Written informed consent was obtained from individual or guardian participants.
Data Access Statement
The data that support the findings of this study are available from The Second Affiliated Hospital of Shandong First Medical University, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of The Second Affiliated Hospital of Shandong First Medical University.
