Abstract
Introduction
Depression occurs among 8 to 20% of adults in primary care (Barry et al., 1998; Tarricone et al., 2012). One hallmark of depression is impaired cognitive performance, most prominently characterized by difficulty concentrating (Park & Zarate, 2019). However, impairments may occur across all cognitive domains (Ismail et al., 2014). Research has suggested a bidirectional association between depression and cognitive impairment, with increasing evidence they share pathophysiological pathways, such as volume loss in the hippocampus (Ismail et al., 2014). Studies demonstrating the high co-occurrence rates of depression and cognitive impairment support this hypothesis. For example, depression rates of 32% (Ismail et al., 2017) and 37 to 41% (Leung et al., 2021) have been reported for persons with mild cognitive impairment (MCI) and dementia, respectively. Depression also confers a greater risk for cognitive decline in cognitively intact individuals, as well as more rapid progression from MCI to dementia (Ismail et al., 2014; Kessing, 2012).
Although depression and cognitive impairment may not be easily disentangled (Ismail et al., 2014), identifying one in the context of the other is clinically relevant, particularly when providing care for older adults, in whom the probability of cognitive impairment is higher. For instance, when a person is found to have depression, screening for cognitive impairment could add information about the individual’s risk for progression to dementia or guide medication selection or decisions about reducing polypharmacy.
While many studies have examined the associations of depression and cognitive impairment (Ismail et al., 2017; Li et al., 2001), they have largely involved patients in specialty practices, such as neurology and memory clinics or in research cohorts comprised of patients with MCI or dementia. Estimates of the prevalence of these comorbidities are likely to be higher in such settings than in primary care practices. In this study, we sought to identify the prevalence of previously undiagnosed cognitive impairment in the context of comorbid depression among adults ages 55 and older in primary care.
Methods
Study Participants and Settings
We recruited patients from five primary care practices affiliated with two academic medical centers, Mount Sinai Hospital in New York City, NY and Northwestern Hospital in Chicago, IL from August 2020 through December 2021. These practices were selected because they are part of large urban health care systems serving diverse patient populations. Patients were eligible to participate if they were ages 55 years and older, conversant in English and could provide informed consent. Patients were excluded if they had a diagnosis of dementia or mild cognitive impairment in their electronic medical record. All study procedures were approved by the institutional review boards of the Icahn School of Medicine at Mount Sinai and the Northwestern University Feinberg School of Medicine.
Recruitment, Interviews, and Data Collection
Research assistants identified a pool of patients by medical record review then obtained permission from their primary care providers for recruitment outreach. Selection of patients for recruitment from the pool was random, though 9 months after the start of recruitment, we began oversampling white and Black male patients to achieve balanced representation of the sexes, race and Latinx ethnicity. Patients were mailed a recruitment letter, received a recruitment call 7 to 14 days later and underwent final eligibility screening by phone. Informed consent was obtained at the time of the 30-minute in-person interview, which was conducted in a clinical exam room.
Measures
We selected the Patient Health Questionnaire (PHQ)-8 to measure depression because it is a validated depression screening tool commonly used in US primary care settings (Kroenke et al., 2001, 2009; Kroenke & Spitzer, 2002) and is briefer than and performs similarly to the Geriatric Depression Scale (GDS) among older adults (area under the receiver operating curve for major depression, 0.87 vs. GDS, 0.81) (Pellas & Damberg, 2021; Phelan et al., 2010; Zhang et al., 2020). It consists of eight of the nine diagnostic criteria for depression, based on the Diagnostic and Statistical Manual IV. The PHQ-8 omits a question about suicidal ideation and performs similarly to the PHQ-9 (score range 0–24) (Kroenke et al., 2009). Depression was scored as absent (score <5), mild (5–9), moderate (10–14), or severe (≥15) (Kroenke et al., 2009; Kroenke & Spitzer, 2002). PHQ-8 scores ≥10 have 88% sensitivity and 88% specificity for major depression (Kroenke & Spitzer, 2002).
Cognitive function was assessed with the Montreal Cognitive Assessment (MoCA), a screener for MCI and dementia widely used in clinical care and research (Davis et al., 2015; Freitas et al., 2013; Nasreddine et al., 2005; Rossetti et al., 2011). It consists of 12 tasks covering visuospatial/executive functioning, naming, memory, attention, language, delayed recall and orientation. Scores range from 0 to 30. A single cutoff score of 26 is typically recommended to differentiate MCI or dementia from “normal”; however, this cutoff is sample-dependent (Dautzenberg et al., 2020) and may lack specificity in diverse cohorts (Milani et al., 2018). Instead, we calculated age and education adjusted z-scores and defined mild impairment as z-scores between 1.0 and 1.5 standard deviations below published norms, and moderate to severe impairment as ≥1.5 standard deviations below the norm (Langa & Levine, 2014; Pugh et al., 2018; Rossetti et al., 2011).
Research assistants were trained by a neuropsychologist in the administration of the PHQ-8 and MoCA and were periodically observed for quality and fidelity.
Data were also collected for variables associated with performance on the MoCA for inclusion in multivariate models, including age, education, race and ethnicity, English language proficiency (ELP), country of birth and physical functioning. ELP was assessed with a single item, “How would you describe your ability to speak and understand English?” with six response options ranging from very poor to excellent. Low ELP was defined as a response of very poor, poor or fair. Activities of daily living (ADL) included bathing or showering, dressing, eating, getting in or out of bed or chair, walking, and toileting. Instrumental activities of daily living (IADL) included ability to use a telephone, shop, prepare food, perform housekeeping chores, do laundry, use public transportation, self-manage medications, and handle finances. Summary scores for ADL and IADL impairments were the sum of impairments for the 6 and 8 items of the measures, respectively.
Analysis
We examined the unadjusted associations of patient characteristics with having any depression (mild, moderate, or severe) using t-tests and chi-square tests as indicated. Next, we compared differences in levels of cognitive impairment (none, mild, moderate-severe) and MoCA z-scores by levels of depression using the chi-square test and analysis of variance, respectively. We compared differences in raw scores for each of the seven domains of the MoCA by levels of depression using the Wilcoxon Rank Sum test. Pearson correlations were calculated for the associations of PHQ-8 scores and MoCA raw and adjusted z-scores. Because low ELP may affect performance on cognitive assessments, we repeated the aforementioned analyses in the subgroup that excluded persons with low ELP. Finally, we tested the associations of depression with raw cognitive screening scores in the full cohort in a series of linear regression models that serially adjusted for age and education, followed by race, sex, country of origin (U.S. vs. other) and self-reported ELP, then basic and instrumental activities of daily living. For the linear regressions, we report standardized regression coefficients. All analyses were conducted in SAS, version 9.4 (SAS Institute, Cary, N.C.).
Results
Research coordinators mailed recruitment letters to 4,130 persons and reached 3,209 (77.7%) by telephone. Of these, 522 (16.3%) were ineligible, 1,724 (53.7%) declined to be interviewed and 949 (29.6%) agreed to participate. Signed consent was provided by 941 individuals; 872 (92.7%) completed the in-person interview and the depression and cognitive assessments were completed by 855 (90.9%).
The mean age of participants was 66.8 (8.0) years, 45.3% were male, 32.7% Black and 29.2% were Latinx (Table 1). More than half reported some college (37.8%) or graduate education (19.4%). One quarter (25.1%) were born outside the U.S. and 7.1% reported low ELP. ADL and IADL impairments were identified for 17.1 and 30.5%, respectively.
Demographic Characteristics of Study Participants, Stratified by Presence (PHQ-8 ≥ 5) and Absence of Depression (PHQ-8 < 5).

Note. PHQ = Patient Health Questionnaire; ELP = English language proficiency; ADL = activities of daily living; IADL = instrumental activities of daily living.
Depression and Cognitive Impairment
Depression was identified in 39.0% of study participants (mild depression, 22.6%; moderate, 9.9%; severe, 6.5%). Depression of any severity was more common among middle aged adults than older adults (ages 55–64 years, 46.0%, 65–74 years, 34.9% and 75 and older, 31.0%; p = .0009) (Table 1). Depression rates were also higher among women, Black and Latinx patients, and those with low educational attainment, low English language proficiency and functional impairments. Cognitive impairment of any severity was observed in 20.6% of participants; 10.3% had mild impairment and 10.4% had moderate-severe impairment (Table 2).
Prevalence of Cognitive Impairment by Severity of Depression Among Primary Care Patients Ages ≥55 Years.

Note. Mild cognitive impairment, MoCA Z-score <1.0 and >1.5 standard deviations below age and education adjusted norms; Moderate-severe, <1.5 standard deviations below norm.
p = .0002 for differences in level of cognitive impairment by depression severity.
Previously undiagnosed cognitive impairment was observed in 20.6% of patients overall, in 26.0% of patients with depression and 17.2% of patients without it (p = 0.002) (Table 2). Its prevalence increased with increasing severity of depression: 22.9% in persons with mild depression, 27.4% in moderate depression and 41.8% in severe depression (p = .0002). Rates of mild and moderate to severe cognitive impairment were 12.4 and 13.6%, respectively, among patients with depression and 8.9 and 8.3% in those without depression (p = .007).
The mean of raw MoCA scores was 21.8 (4.5) and of the age and education adjusted z-score, −0.12 (1.02). The adjusted MoCA z-scores declined significantly across the four levels of depression: no depression, −0.02 (0.97); mild depression, −0.11 (0.97); moderate depression, −0.34 (1.11) and severe depression, −0.79 (1.22) (p < .0001; Figure 1). PHQ-8 scores were significantly correlated with both raw MoCA scores and adjusted MoCA z-scores (r = −0.25, p < .0001 and r = −0.18, p < .0001, respectively).
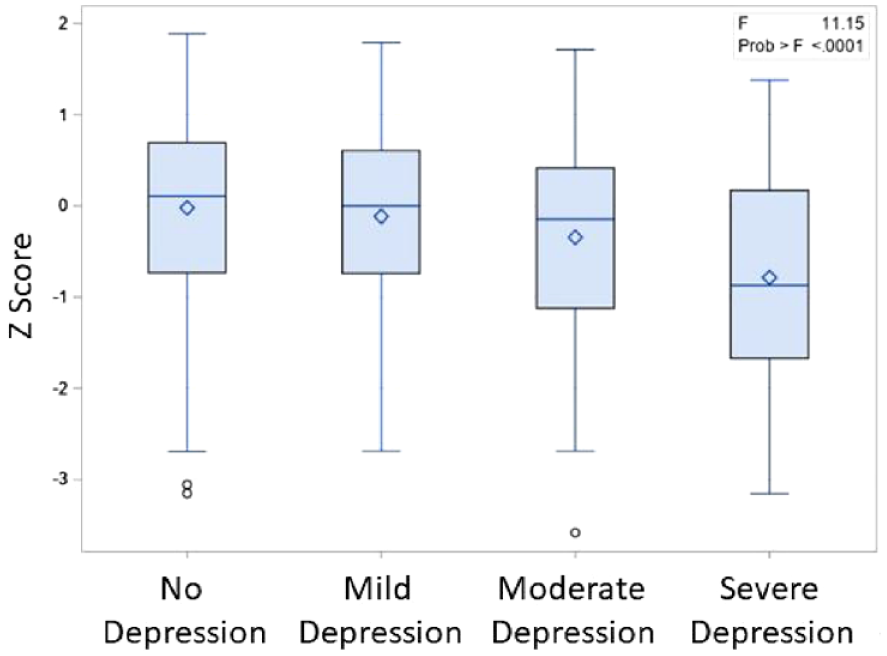
Age- and education-normed Z scores for the Montreal Cognitive Assessment, by depression severity.
Raw scores on several subdomains of the MoCA worsened significantly with increasing severity of depression (Table 3). These were visuospatial/executive functioning (p < .0001), attention (p < .0001), language (p < .0001), abstraction (p = .007), and delayed recall (p = .0003). There were no significant associations of depression with naming and orientation (p > .05).
Mean Montreal Cognitive Assessment Domain Scores, by Severity of Depression, Primary Care Patients Ages ≥55 Years.
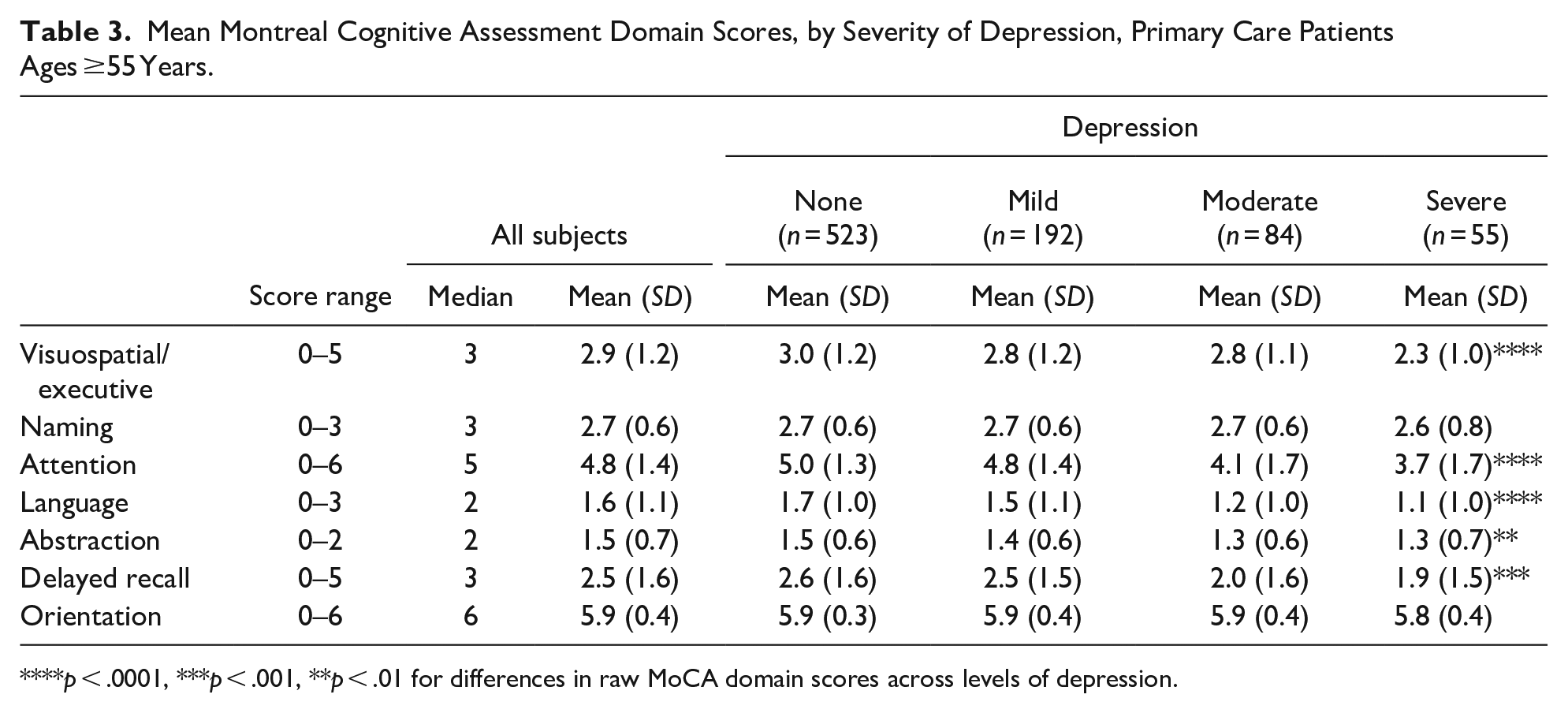
p < .0001, ***p < .001, **p < .01 for differences in raw MoCA domain scores across levels of depression.
Subgroup Analysis of Patients Without Low English Language Proficiency
Results for the subgroup of patients without low ELP were similar to those for the full cohort. Mild and moderate-severe cognitive impairment occurred in 8.7 and 7.9% of patients without depression, respectively, and in 13.0 and 23.9% of those with severe depression (Supplemental Table S1). The adjusted MoCA z-scores also declined significantly across the four levels of depression: no depression, −0.003 (0.97); mild depression, −0.08 (0.98); moderate depression, −0.25 (1.08), and severe depression, −0.66 (1.20) (p < .0001). The pattern of associations of depression with individual domains of the MoCA were consistent with those of the full cohort (Supplemental Table S2).
Adjusted Associations of Depression With Raw Montreal Cognitive Assessment Scores
Depression was significantly associated with raw MoCA scores in a series of models that adjusted for patient characteristics (Table 4). In the unadjusted model, the standardized β coefficient for the association of severe depression with MoCA scores was −0.22 (0.33) (p < .0001, R2 = 0.07). Severe depression remained significantly associated with MoCA scores in the fully specified model (β = −.11 [0.33], p < .001; R2 = 0.44).
Adjusted Associations of Patient Characteristics With Raw Montreal Cognitive Assessment Scores Among Primary Care Patients Aged ≥ 55 Years.

Note. Linear regression. Regression coefficients are standardized. SE = standard error; ELP = English language proficiency; ADL = activities of daily living; IADL = instrumental activities of daily living.
Discussion
In this sample of primary care patients ages 55 years and older, we found that 49% of cases of previously undetected cognitive impairment occurred among persons with depression, and rates increased with depression severity. The study findings demonstrate that depression and cognitive impairment are common co-occurring problems among older adults in primary care settings and suggest clinical value for assessing cognitive function when depression is identified or suspected, or depression when cognitive impairment is identified.
This study adds to the existing literature on the associations of depression and cognitive impairment by providing data on patients in primary care and reporting on previously undetected cognitive impairment. Studies of this nature are conspicuously absent in the literature even though depression and cognitive impairment are both very common in primary care (Barry et al., 1998; Federman et al., 2023; Tarricone et al., 2012; Weyerer et al., 2008). Research to date has mostly drawn samples from specialty practice settings, such as neurology and memory clinics, or used data from selected cohort studies or registries like the National Alzheimer’s Coordinating Center (Ismail et al., 2017; Leung et al., 2021; Mourao et al., 2016). Primary care clinicians may be challenged in extrapolating findings from these studies to the primary care setting where the prevalence of cognitive impairment is considerably lower, or unknown. In a systematic review of the prevalence of depression in patients with MCI, Ismail and colleagues reported a pooled prevalence rate for depression of 40% (95% CI [0.32, 0.48]) in studies conducted in specialized clinical settings, none of which were primary care (Ismail et al., 2017). In contrast, community-based studies had a pooled prevalence rate of 25% (95% CI [19%, 30%]). In our study, the prevalence of depression among persons with cognitive impairment was closer to that of community based studies, at 26.0%.
The frequent co-occurrence and bidirectional relationship (Ismail et al., 2014) of depression and cognitive impairment should serve as an impetus for screening for both conditions, as identifying one condition may guide evaluation and treatment of the other and inform prognosis. Depression impairs alertness and attention, executive functioning, psychomotor speed and working and long term memory—consistent with the impairments in the subdomains of the MoCA observed among patients with depression in our study. Such deficits may persist beyond resolution of major depressive episodes (Semkovska et al., 2019). One theory holds that depression, as an internalizing disorder, reduces a patient’s cognitive control capacity resulting in difficulty allocating cognitive resources to a task (Jones et al., 2010; Kircanski et al., 2012). In turn, symptoms that commonly appear in depression, like apathy, inattention and sleep disturbance, frequently accompany cognitive impairment resulting from neurodegenerative conditions, especially in dementia. Moreover, depression itself may be a symptom of cognitive impairment (Ismail et al., 2014).
Depression does not simply diminish cognitive performance but is a risk factor for incident MCI and dementia, as well as for faster progression from MCI to dementia (Donovan et al., 2014; Mourao et al., 2016). Progression to dementia from MCI is 28% higher in patients with depressive symptoms (Donovan et al., 2014; Mourao et al., 2016). For these reasons, screening for both depression and cognitive impairment could provide clinicians valuable information for prognostication and guiding medical treatment and clinical management. Clinicians might also consider monitoring cognitive function over the long term in older adults with depression because of the increased risk for progression to dementia (Ismail et al., 2014). Importantly, awareness of these comorbidities is critical knowledge for primary care clinicians because each strongly limits the ability of patients to manage their chronic conditions (Lovett et al., 2020).
We note that controversy exists over routine screening for cognitive impairment. In 2020, the U.S. Preventative Services Task Force’s updated its 2014 recommendations, still finding insufficient evidence to assess the balance of benefits and harms of screening for cognitive impairment in older adults (Owens et al., 2020). Others have argued strongly for screening, suggesting opportunities to identify treatable causes, reduce risk of adverse events and provide patient and family support (Alzheimer’s Disease Association, 2022; Morley et al., 2015). In MCI, for example, treatment of depression with selective serotonin reuptake inhibitors (SSRI) may reduce depressive symptoms and delay progression to dementia (Bartels et al., 2018; Brendel et al., 2018). Clinicians can also reduce risk of cognitive decline by optimizing management of cardiovascular disease risks, avoiding medications with anticholinergic properties and reducing the burden of polypharmacy, among other interventions (Langa & Levine, 2014).
Although there may be benefit for screening for both conditions, screening rates for either condition alone may not yet be optimized. Screening for depression has increased in ambulatory care practices in recent years (Bhattacharjee et al., 2018) and is nearly universal in some health systems (Garcia et al., 2022), but rates remain low elsewhere. Data from the nationally representative Medical Expenditure Panel Survey in 2017 showed that just 50% of primary care patients ages 35 years and older were assessed for depression (Kato et al., 2018). Even in a large scale pragmatic trial of self-screening through an electronic health record portal or a clinic-based desktop computer application screening rates only reached 43% and 33%, respectively (Franco et al., 2023). Screening for cognitive impairment ostensibly occurs less frequently, though few studies have examined it directly (Bradford et al., 2009; Chodosh et al., 2004; Kotagal et al., 2015; Sabbagh et al., 2020). This is likely due, in part, to the U.S. Preventative Services Task Force’s 2014 and 2020 reports (Owens et al., 2020). Widespread adoption of cognitive impairment screening may not occur without more research on outcomes of screening, better supports to assist primary care clinicians with screening, diagnosis and management, and better treatments for MCI and dementia.
Finally, clinicians should be aware that depression and cognitive impairment may affect performance on screening assessments for these conditions. The PHQ-9 has a specificity of 0.71 for depression when administered to persons with cognitive impairment compared to 0.89 among those without it (Boyle et al., 2011). Similarly, the Mini Mental Status Exam has lower sensitivity for cognitive impairment when administered to patients with depression (Rajji et al., 2009). Data on sensitivity and specificity for the MoCA in the context of depression are lacking, though a small study (n = 57) reported that MoCA scores correlated highly with a composite score of five standard neuropsychological tests in patients with major depressive disorder (r = 0.78) (Srisurapanont et al., 2017).
Study Strengths and Limitations
Strengths of this study include its large sample size and sociodemographic diversity, and use of the PHQ-8 and MoCA, screening tools for depression and cognitive impairment that are readily available for use in primary care. A number of limitations also warrant mention. This study excluded persons with previously diagnosed cognitive impairment, thus the rates of cognitive impairment we reported are lower than the actual overall rates of cognitive impairment in the participating study sites. As noted above, brief assessments of cognition and depression may have diminished performance characteristics, including sensitivity and specificity, when these conditions are co-occurring. If specificity of the PHQ-8 was reduced when administered to patients in this study, we might have mislabeled some patients with depression as not depressed. We did not estimate pre-morbid intelligence, which can affect performance on cognitive assessments even when accounting for education (Gerstenecker & Mast, 2014). We used a single-item self-reported measure of ELP that may lack the sensitivity and specificity of more rigorous measures. The MoCA and PHQ-8 are screening tools and are not diagnostic for MCI, dementia and depression and we did not perform comprehensive neuropsychological evaluations. Finally, study participants were recruited from only two urban healthcare systems, which may limit generalizability.
Conclusion
Cognitive impairment and depression commonly co-occur among older adults in primary care. Primary care clinicians should consider screening, or expand their screening, for both conditions.
Supplemental Material
sj-docx-1-ggm-10.1177_23337214231214217 – Supplemental material for Relationship Between Cognitive Impairment and Depression Among Middle Aged and Older Adults in Primary Care
Supplemental material, sj-docx-1-ggm-10.1177_23337214231214217 for Relationship Between Cognitive Impairment and Depression Among Middle Aged and Older Adults in Primary Care by Alex D. Federman, Jacqueline Becker, Fernando Carnavali, Monica Rivera Mindt, Dayeon Cho, Gaurav Pandey, Lili Chan, Laura Curtis, Michael S. Wolf and Juan P. Wisnivesky in Gerontology and Geriatric Medicine
Footnotes
Author Contributions
Dr. Federman had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Concept and design: Federman, Becker
Acquisition, analysis, or interpretation of data: All authors.
Drafting of the manuscript: Federman.
Critical revision of the manuscript for important intellectual content: All authors.
Statistical analysis: Federman, Wisnivesky.
Obtained funding: Federman, Wisnivesky, Pandey, Rivera Mindt, Wolf.
Administrative, technical, or material support: Cho.
Supervision: Federman.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Wisnivesky received consulting honoraria from Sanofi, PPD, Banook, and Prospero and research grants from Sanofi, Regeneron, Axella, and Arnold Consultants. Dr. Wolf reports grants from the Gordon and Betty Moore Foundation, and Eli Lilly, and personal fees from Pfizer, Sanofi, Luto UK, University of Westminster, and Lundbeck outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the National Institute Aging of the National Institutes of Health (grant number R01AG066471).
Role of the Funder/Sponsor
The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Anonymized Text
All study procedures were approved by the institutional review boards of the Icahn School of Medicine at Mount Sinai and the Northwestern University Feinberg School of Medicine.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
