Abstract
Background
Low mobility during hospitalization is a well-known problem imposing a considerable burden on patients and healthcare systems. It is particularly common in older patients and associated with adverse outcomes such as falls, functional decline, institutionalization and death (Brown et al., 2004, 2009; Kalisch et al., 2014). An estimated 80% of hospitalization time is spent lying in bed, and only 3% walking or standing, although most hospitalized patients would be able to ambulate independently (Brown et al., 2009). Improving hospital mobility could help reduce those adverse outcomes, such as falls, which are highly prevalent in the older population (28% to 35% of people aged over 64% and 23% to 42% of those over 70 years fall each year) and cause over half of injury-related hospitalizations in older adults (WHO, 2007).
Previous interventions that were prospectively evaluated in randomized or non-randomized studies, successfully increased hospital mobility under study conditions providing additional resources (Brown et al., 2016; Hamilton et al., 2019; Hastings et al., 2014; Raymond et al., 2017; Teodoro et al., 2016; Wood et al., 2014). However, they were hard to implement in clinical practice, so that broad-scale practice changes were not observed yet. This might be due to an incomplete consideration of barriers such as staff/cost resources, or facilitators, such as encouragement of patient self-efficacy or modification of the hospital environment (Brown et al., 2016; Hamilton et al., 2019; Hastings et al., 2014; Raymond et al., 2017).
The INTOMOB (INtervention TO increase MOBility of older hospitalized patients) project was developed to improve mobility of older acutely hospitalized patients, addressing the limitations of previous studies. This project includes a multilevel mobility intervention targeting patients, healthcare professionals (HCPs) and the hospital environment, developed after thoughtful assessment of barriers and facilitators to mobility, and designed so that it can be implemented without requiring resources unavailable in clinical practice.
We report the pilot study of the intervention that assessed its feasibility and acceptability, and compared two accelerometers, with the goal to adapt the intervention before testing it in a cluster randomized controlled trial (RCT), to ensure a smooth RCT conduction.
Methods
Study Design and Setting
In this mixed-methods pilot study, the INTOMOB intervention was implemented on acute general internal medicine wards of three hospitals, and its feasibility and acceptability were assessed. The intervention was implemented on one ward in each of the following hospitals of different sizes and regions of Switzerland: Bern University Hospital, Tiefenau Hospital, Fribourg Cantonal Hospital. Patients were followed up until discharge. Patient and HCP experience of the intervention were assessed through surveys and interviews. The study was planned over 3 months between December 2022 and March 2023. Results are reported according to the STROBE checklist.
The protocol of the INTOMOB study, including the present pilot study, was approved by the local ethical committees (“Ethikkommission für die Forschung am Menschen – Universität Bern” and “Commission cantonale d’éthique de la recherche sur l’être humain (CER-VD)”) and the study was registered before initiation on clinicaltrials.gov (NCT05639231) and in the Swiss National Clinical Trial Portal (SNCTP000005259).
Pilot-Study Participants and Sample Size
Patient inclusion criteria were: age ≥60 years, being ambulatory ≥2 weeks (self-report; no need of assessment by a physician/physiotherapist), living in the community for ≥30 days before admission, ability to understand French or German, possibility to start the study within 48 hr after admission, planned length of stay of ≥3 days after enrollment (based on ward HCP evaluation). Exclusion criteria were: medical contraindication to walk, wheelchair-bound, end-of-life, severe psychiatric disorder, delirium (according to the Confusion Assessment Method) (Inouye et al., 1990) and dementia (Mini-Cog < 3) (Borson et al., 2000). All patients admitted to a participating ward were screened for eligibility during workdays based on electronic health record and information provided by ward HCPs. Eligible patients were informed about the intervention and included after signing informed consent. We planned a sample of 20 patients for the pilot study.
All HCPs working on the included wards received the intervention and were asked to complete a survey. We planned interviews with 5 to 10 HCPs in each hospital with ≥2/3 of nurses which are the main target of HCP intervention (see intervention description). We planned to increase this number if we did not reach data saturation with the collected data.
We estimated that these sample sizes would allow collecting enough feedback to optimize the intervention for the RCT.
Intervention Description
The INTOMOB intervention was developed based on barriers and facilitators identified in the literature (Mani et al., 2022) and in a mixed method study (manuscript submitted and Herzog et al., 2023). To allow future implementation, the intervention had to require no additional resources. It targets patients, HCPs, and the hospital environment. The patient intervention includes: (1) an information booklet, (2) a customizable diary to set and document individual mobility goals, and (3) exercises in lying, sitting and standing position (pictures and explanations in a booklet, and videos on an iPad 10.2″).
The HCP intervention targets nursing staff and medical residents, with a focus on nursing staff to reduce contamination bias in the RCT that will be clustered at ward level (nursing staff rotation across wards being rare). It includes: (1) a 15-minute e-learning addressing knowledge and communications skills through text, multiple-choice questions, pictures and videos; (2) a mobility checklist to remind and help HCPs to assess mobility, provided as pocket card to the HCPs and hung up in offices and on visit carts (eFigure 1); and (3) a presentation provided orally and as PowerPoint slides to the HCPs. HCP role in the intervention was to discuss mobility goals, difficulties and results with the patients, to verify and discuss the checklist in order to assess and address barriers and facilitators to mobility, to prescribe physiotherapy and support the patients with the use of the iPad if needed.
The environment intervention includes posters and itineraries with quizzes (quiz on a poster, solution on the next poster) hung up in the hallways. There were posters on the following topics, chosen based on the mixed methods study mentioned above: sleep, why move at the hospital, moving after hospitalization, polypharmacy, eating when aging, hospital staff, auxiliary means, famous persons. There were four quiz itineraries, on the following topics: animals, flowers, landscapes, famous persons.
The detailed description of the intervention is in the study protocol (available on clinicaltrials.gov and in Mooser et al., manuscript in press).
Study Procedures
For patients, a baseline visit on the ward at study enrollment and a discharge assessment within 1 day of discharge were performed. During discharge assessment, we asked close- and open-ended questions (eTables 1 and 2) about patient experience of the intervention and with wearing the wrist-worn GENEActiv (Lim et al., 2018; Rowlands et al., 2014) and ankle-worn StepWatch (https://modushealth.com/. Accessed April 19 2023) accelerometers. For HCPs, a survey (close- and open-ended questions; eDocument 1) and interviews (open-ended questions to expand survey information; eTable 3) were conducted. Interviews were transcribed verbatim.
Data Analysis
Close-ended questions were analyzed descriptively and presented as percentages. For ease of interpretation, 5-point-Likert-scale answers were regrouped in 3-point-scale answers as follows: 1 = “totally disagree” or “disagree,” 2 = “neutral”, 3 = “agree” or “totally agree.” Data were analyzed for each hospital, and reported separately when relevant differences were observed.
Open-ended questions were analyzed using a deductive approach (DeCuir-Gunby et al., 2011). Domains and constructs were created based on the survey and interview guides. Domains classify the different parts of the intervention and experience/process aspects, and constructs the different aspects of the domains (e.g., content/format). Coding was conducted independently by two authors and iteratively discussed with the senior author. The following abbreviations were used to report quotes: I, interview; D, Doctor; N, nurse; P, patient; S, survey.
Quantitative and qualitative results were integrated using joint displays to draw meta-inferences from the mixed data, describing results as convergent, divergent, or expanding. The quotes cited in the manuscript were chosen to illustrate the different aspects of the domains and constructs.
Quantitative data analysis was conducted with Stata/MP 16.0 (StataCorp LP, College Station, Texas) and qualitative data analysis with MAXQDA2022 software (VERBI Software, Berlin, Germany).
Results
Between December 2022 and March 2023, 20/73 eligible patients were included (Figure 1). Main reason for non-eligibility was impossibility to start the study within 48 hr. Main reasons for refusal were lack of interest and feeling too weak. After pilot-study completion, 44 HCPs completed the survey and 27 participated in an interview, which allowed reaching data saturation. Description of pilot-study participants (mean patient age 74.1 years [SD 8.1] years, 7 [35%] female) is provided in eTables 4 to 5.
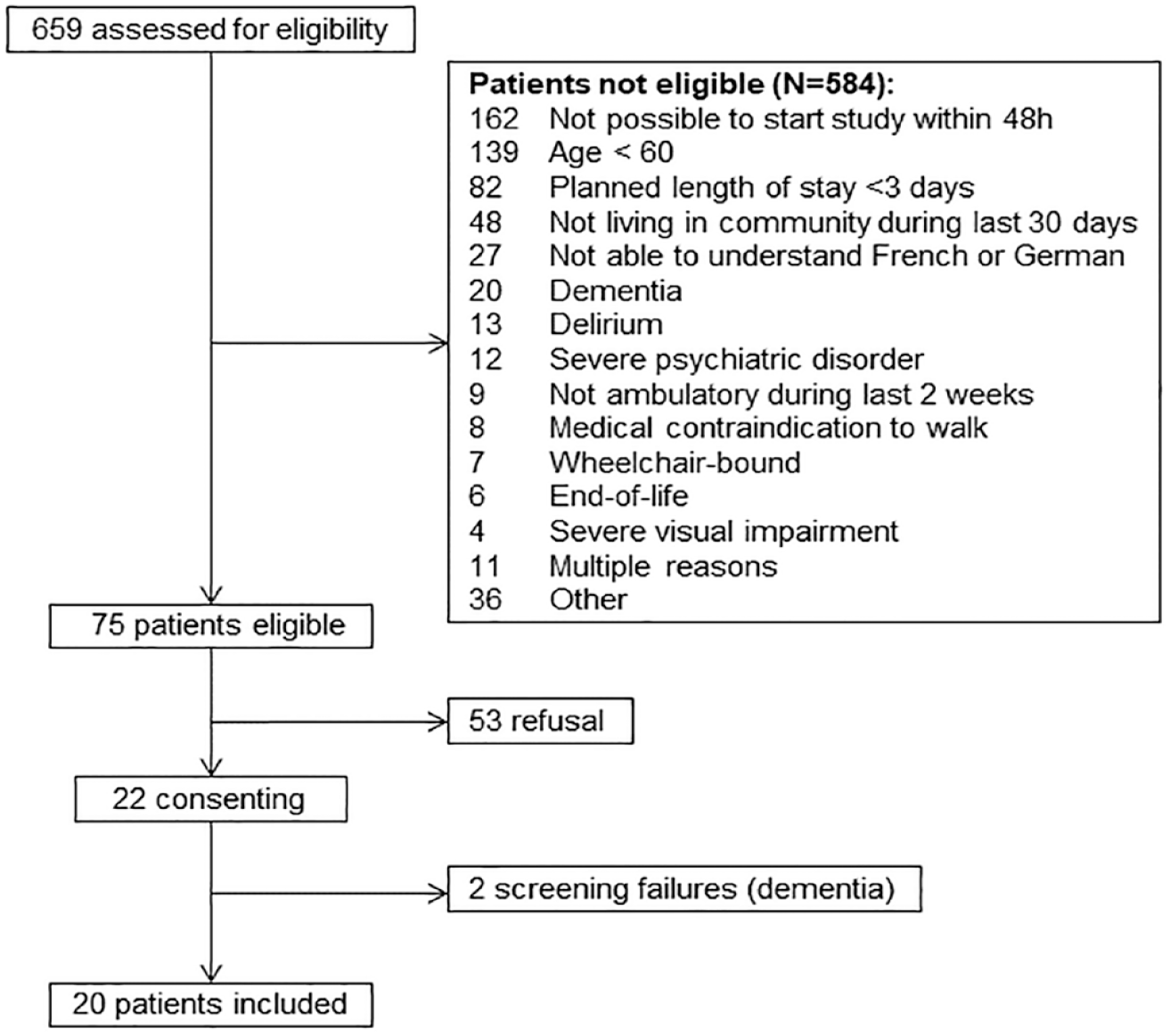
Flowchart.
Domains and constructs are presented in Table 1. Tables 2 and 3 present joint displays for the constructs where complementary information was provided by the quantitative and qualitative results. Table 4 presents the results on accelerometer assessment. Complete results of the quantitative analysis are provided in eTables 6 to 8. In the next sections, we synthesize the main results of the analyses.
Domains and Constructs of the Mixed Methods Analysis.

Includes clarity and utility.
Includes length.
Patient Evaluation of the Intervention.

Note. Only items that were not rated 100% positively are presented. Other items assessed are presented in eTable 7. Dom = domains; P = patient.
Includes length, colors, amount of information.
Healthcare Professional Evaluation of the Intervention.
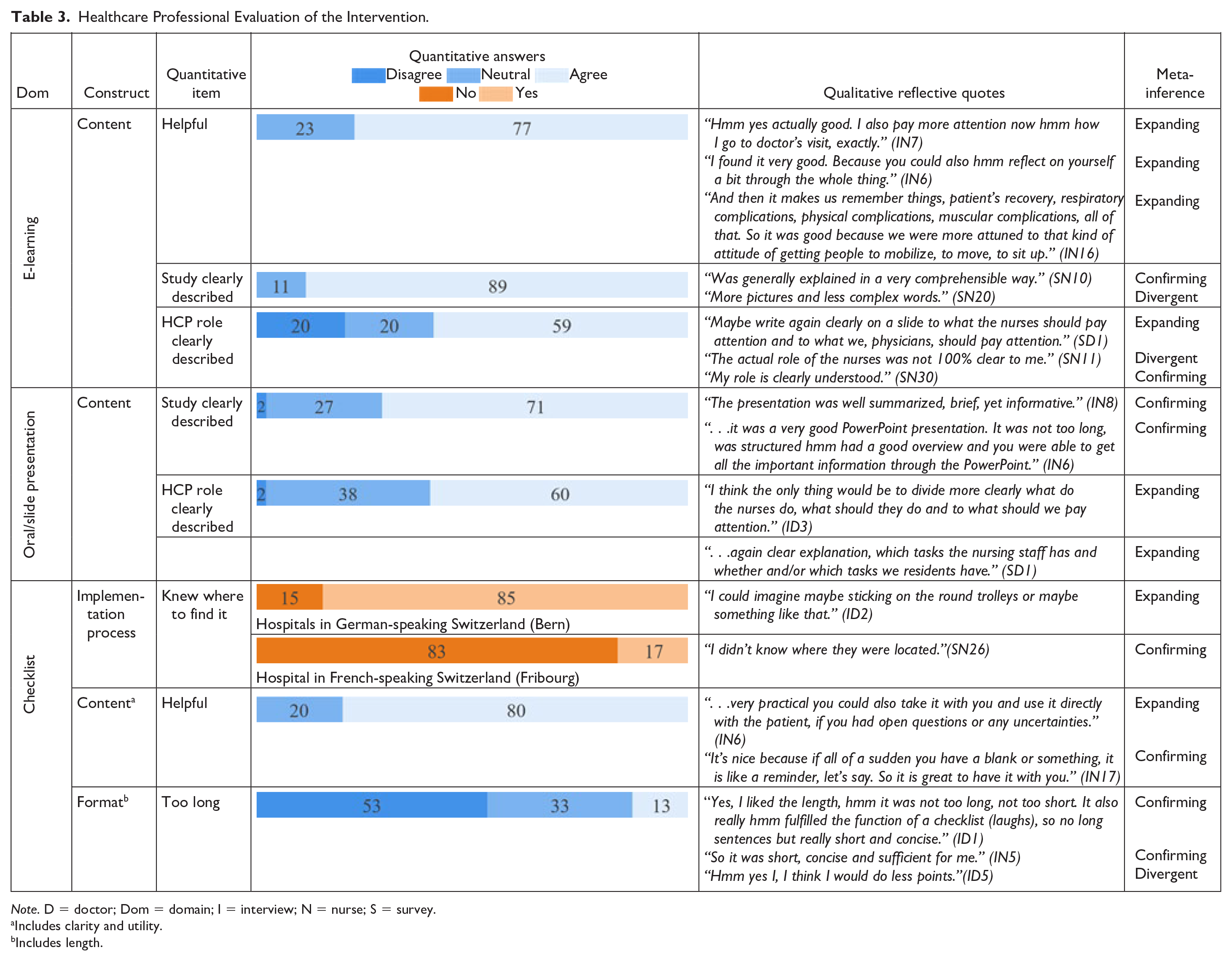
Note. D = doctor; Dom = domain; I = interview; N = nurse; S = survey.
Includes clarity and utility.
Includes length.
Healthcare Professional and Patient Evaluation of the Accelerometers.

Patient and Environment Intervention (Table 2, eTables 6-7)
Overall Experience
Patients described a positive experience of the intervention (P11): “I liked to do the exercises and moved more during hospitalization. I went walking in the hallway with the walking device with my son and looked at the posters.” (P12): “I liked it very much, for me, it was important to have a reminder, or a ‘wake up call’ to focus on moving more again and not getting too comfortable.” HCPs observed a positive effect from the intervention on patients (IN7): “They often said, ‘hmm, yes now that I am in this study, I have to move’. And I think that is already, it already motivates them to move.” (ID1): “What I have found very positive, for example, to be able to experience with the patient how certain progress can be made, how they suddenly improved their mobility, that’s been fun for both us and the patients.”
Booklets
Ninety-five percent of patients stated they used at least one of the booklets and 87% found them helpful (P9): “I liked the booklet as a start to know about the importance of mobility.” (P8): “The commitment to a journal was a positive motivation.” Eleven percent of HCPs reported that patients asked questions regarding the booklets (IN1): “. . .I just went through the brochure again with him and guided him through the exercises and then it worked out fine.”
Videos
Half of the patients said they used the exercise videos, among which 90% perceived them as useful (P7): “The iPad was easy to use, it was good to have the videos to understand how the exercises work.” All patients said the exercise videos were easy to understand and 91% stated they had no problem using the iPad. However, some HCPs reported problems with the use of the iPads (IN6): “Especially for older people, it was a bit more difficult to use the iPad, because some of them don’t even have a cell phone and then suddenly they get an iPad. So a few times, I had to explain a little bit how it works.” Technical issue with the iPads were however not reported.
Environment Intervention
Eighty-five percent of the patients perceived the posters as helpful and 94% found the topics interesting (P11): “I liked the posters very much, I also studied them when I had visits from my family.” The itineraries with quizzes were confusing for some patients (P9): “In the beginning, it was confusing that the posters didn’t provide the right information.” Sixty-eight percent of HCPs saw the patients reading the posters (IN1): “Many patients really followed these posters with great interest and, as a result, they were moving in the hallway more often than usual.” Eighty-two percent of HCPs reported their work routine was not disturbed by patients looking at the posters.
Healthcare Professional Intervention (Table 3, eTable 8)
Overall Experience
HCP experience of the intervention was mainly positive (ID3): “I think it’s really great and I mean it’s actually with little, with little effort you can do a lot there I think.” Eighteen percent of HCPs felt their workload was increased by the intervention, while some HCPs reported a beneficial effect on their workload (IN3): “I felt that the fact that people were more mobile actually made my job easier sometimes because it was prophylactic, for example, for thrombosis or contractures.” To further reduce the workload, HCPs suggested to “improve patient instruction by the study team” (SN15).
E-Learning and Oral/Slide Presentation
Seventy-six percent of HCPs attended the oral presentation or studied the slides. HCPs liked receiving the slides (IN13): “Doing it in PowerPoint, I think it was good, instead of just giving a document. I think, like this it was more pleasant to see.” HCPs suggested repeating the oral presentation to allow more people to attend (IN8): “But maybe for our large nursing department, it would have been better to do it two more times or one more time so that really enough people, hmm, could see this presentation.”
Seventy-seven percent of HCPs found the e-learning helpful, but suggested to send it earlier (ID6): “I did it a little late, I didn’t get it before the study. Maybe it [the intervention] would have been clearer if I had gotten it [the e-learning] before.” Nine percent of HCPs reported technical problems with the e-learning. For example (IN1): “For many who actually completed the e-learning, the result was not sent and they received a reminder to do it.”
Most HCPs perceived the study intervention as clearly described in the e-learning (89%) and oral/slide presentation (71%). HCP role was unclear for 20% after e-learning completion and 2% after the oral/slide presentation. HCPs suggested to specify nursing staff versus resident roles in the intervention more clearly (ID1): “That you get to know more precisely what you have to do on a function-specific basis.” Another idea was to provide key study information on paper (ID8): “I don’t know, maybe give a small pocket card (. . .) on, concretely, us, what it impacts in our daily life.”
Checklist
Eighty percent of HCPs found the checklist helpful (IN4): “I thought it was good that they were so small, because then, you could always take them with you on ward rounds.” Checklist implementation was different across hospitals. While 85% of HCPs in Bern hospitals knew where to find it, 83% of HCPs in Fribourg hospital did not. HCPs suggested to make the checklist more visible on the wards and suggested to clarify how it should be used (ID7): “You’d have to define something like, it has to be seen three times a week, discussed, and then you have to get two, three things in there, to validate it, at least, to make sure it’s been seen and validated.”
Process
Some HCPs felt the communication during the intervention could be improved. For example, it was not always clear which patients were included (ID8): “In fact, I wasn’t even particularly aware of which patients were included in the study.” Additionally, a regular reminder from the study team would have been appreciated (SN14): “Come by at the beginning of the week, remind the team in five minutes that the study is being done.” In order to make the study more visible for HCPs, a nurse (IN14) suggested to put “posters in the office so if a nurse is ever unaware, she can read herself what the study is all about.”
Accelerometers (Table 4)
Fifty-eight percent of HCPs and 63% of patients preferred the wrist-worn GENEActiv accelerometer. Patients found it more comfortable (89% vs. 65% for the ankle-worn accelerometer) and HCPs more natural (SN9): “It’s like a watch, most people are used to wearing something on their wrist.” However, HCPs experienced it as more disturbing for care (13% vs. 5%) (SN15): “. . . [the accelerometer] interferes with peripheral venous access catheter insertion and sometimes with blood sampling.” Patients and HCPs associated the ankle-worn StepWatch accelerometer with a prisoner ankle cuff (IN18): “They said it [the accelerometer] felt a bit like being a prisoner.”
Discussion
In this pilot study, we implemented the INTOMOB intervention, which goal is to improve mobility of older adults hospitalized on acute general internal medicine wards, and assessed its feasibility and acceptability. The intervention was experienced as helpful and motivating by patients and HCPs. It could be implemented without requiring resources unavailable in everyday practice, unlike in previous studies (Brown et al., 2016; Hamilton et al., 2019; Hastings et al., 2014; Raymond et al., 2017; Teodoro et al., 2016; Wood et al., 2014). The results allowed to improve the intervention and the implementation processes for the conduct of the RCT, as described in the next sections, and provides useful information for future mobility-fostering interventions.
Patient and HCP feedback on the intervention material allowed to identify three areas of improvement. Regarding the booklets, patients suggested some color changes, content simplification, adding in the diary the possibility to document the goals reached, and adding icons in the exercise booklet showing the body parts trained. These suggestions were implemented in the revised documents for the RCT, because they could improve acceptability and scalability of the patient intervention. For the posters, both patients and HCPs reported that the itineraries with quizzes were confusing and the pictures sometimes too unknown, so the environment intervention was simplified by removing the quizzes, while adding interesting facts about the pictures. For instructions, HCPs mentioned uncertainty, for example on management of the accelerometers for radiological imaging although technical issues were not reported. To address this, we developed instruction sheets for patients and HCPs. Patients and HCPs favored the wrist-worn accelerometer. A wrist-worn accelerometer will thus be used in the RCT.
Although the target patient sample size was reached within the 3-month planned time frame, some recruitment issues were identified. First, the most frequent non-eligibility reason was the impossibility to start the intervention within 48 hr, most often because of recruitment during workdays only. Since patients could still benefit from the intervention when started later during hospitalization, this criterion was removed for the RCT, while keeping the requirement of a planned length of stay of ≥3 days, to ensure patients stay long enough to benefit from the intervention. Second, some patients who proved good understanding of the study and of the intervention had to be excluded because they failed the Mini-Cog (Borson et al., 2000). It was emotionally hard for them not only to be unable to participate, but also to get a diagnosis of cognitive impairment from the study team. Since clinical judgment is used in similar studies with a low level of risk for patients (de Morton et al., 2007; Wood et al., 2014), the exclusion criterion was modified for the RCT accordingly.
The pilot study identified some implementation issues. First, some HCPs stated the information on the study and on HCP role in the intervention needed more specification. These points were adapted on the slides, e-learning and information sheets that will be implemented earlier before starting patient inclusion to ensure HCPs are trained. Second, some HCPs, mostly in Fribourg hospital, reported not being aware of the checklists, meaning that part of HCP intervention was not implemented. The issue was that the study team did not feel responsible for providing the checklist in those two sites. Study team training on this issue will be improved, including clear definition of each member’s role and responsibilities, and monitor implementation during the RCT.
Strengths and Limitations
The main strengths of this pilot study are the conduction of the intervention in three hospitals of different sizes and linguistic /cultural regions and feedback collection from both patients and HCPs through a mixed methods approach. The main limitations are the exclusion of patients with cognitive impairment (to include patients able to provide constructive feedback) and the rather short hospital stay that limited patient time to test the intervention.
Conclusion
In conclusion, this pilot study showed that the INTOMOB intervention is well accepted and feasible without requiring unavailable resources. The INTOMOB intervention thus offers a realistic way to improve older patient mobility during an acute hospitalization. This has an international relevance, given that low hospital mobility and related adverse outcomes are a world-wide problem. Low- and middle-income countries, which are particularly affected by adverse outcomes such as falls and related disability (Institute for Health Metrics and Evaluation, n.d; Stewart Williams et al., 2015; World Health Organization, n.d), but lack resources, could particularly benefit of the scalable INTOMOB intervention. The future RCT will show whether this intervention is effective to preserve patient functional mobility. The results of this mixed methods study provide important information to design future interventions to improve mobility during a hospitalization.
Supplemental Material
sj-docx-1-ggm-10.1177_23337214231202148 – Supplemental material for Feasibility and Acceptability of an INtervention TO Increase MOBility in Older Hospitalized Medical Patients (INTOMOB): A Mixed-Methods Pilot Study
Supplemental material, sj-docx-1-ggm-10.1177_23337214231202148 for Feasibility and Acceptability of an INtervention TO Increase MOBility in Older Hospitalized Medical Patients (INTOMOB): A Mixed-Methods Pilot Study by Dominique Bergsma, Claudia Panait, Pascal Leist, Blandine Mooser, Lynn Pantano, Fabian D. Liechti, Jenny Gentizon, Christine Baumgartner, Marco Mancinetti, Marie Méan, Joachim M. Schmidt Leuenberger and Carole E. Aubert in Gerontology and Geriatric Medicine
Supplemental Material
sj-docx-2-ggm-10.1177_23337214231202148 – Supplemental material for Feasibility and Acceptability of an INtervention TO Increase MOBility in Older Hospitalized Medical Patients (INTOMOB): A Mixed-Methods Pilot Study
Supplemental material, sj-docx-2-ggm-10.1177_23337214231202148 for Feasibility and Acceptability of an INtervention TO Increase MOBility in Older Hospitalized Medical Patients (INTOMOB): A Mixed-Methods Pilot Study by Dominique Bergsma, Claudia Panait, Pascal Leist, Blandine Mooser, Lynn Pantano, Fabian D. Liechti, Jenny Gentizon, Christine Baumgartner, Marco Mancinetti, Marie Méan, Joachim M. Schmidt Leuenberger and Carole E. Aubert in Gerontology and Geriatric Medicine
Footnotes
Acknowledgements
We thank Tabea Stoller and Yvonne Stauffer for their contributions in the development of the intervention.
Authors’ Contributions
Aubert conceived the project. Aubert, Mooser and Schmidt-Leuenberter contributed to the development of the intervention. Bergsma, Mancinetti, Panait, Pantano, Leist and Aubert contributed to the conduction of the pilot-study. Bergsma, Leist and Aubert analyzed the data. Mooser, Liechti, Gentizon, Baumgartner, Méan, Schmidt-Leuenberger, Bergsma and Aubert contributed to the adaptation of the intervention. Bergsma and Aubert wrote the first draft of the manuscript. All authors critically revised the manuscript and have approved its final version for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research of this article: This study is funded by the Ambizione Grant PZ00P3_201672 from the Swiss National Science Foundation (SNSF). The funders had no role in study design, data collection, data analysis, data interpretation, writing of the manuscript, and the decision to submit.
Ethics Approval and Consent to Participate
This study is conducted in compliance with the Declaration of Helsinki 16, the International Council on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use – Good Clinical Practice 17,18 and the Swiss human research legislation (Swiss Federal Human Research Act 810.30 and Ordinance on Clinical Trials with the exception of Clinical Trials of Medical Devices 810.305). Patient participation is voluntary and written informant consent was obtained by all participants. The protocol of the INTOMOB study has been approved by the local ethical committees (“Ethikkommission für die Forschung am Menschen – Universität Bern” and “Commission cantonale d’éthique de la recherche sur l’être humain (CER-VD)”) and the study has been registered before initiation on the Clinical Trials Registry platform of the National Institute of Health (NIH) – clinicaltrials.gov (NCT05639231) – as well as in the Swiss National Clinical Trial Portal (SNCTP000005259).
Consent for Publication
Study participants are informed that the results of the study will be published in peer-reviewed journals and provide written informed consent on that.
Availability of Data and Materials
Data available from the corresponding author on reasonable request.
Trial Registration
The INTOMOB study was registered before start of patient inclusion in the Clinical Trials Registry platform of the National Institute of Health (NIH) – clinicaltrials.gov (NCT05639231) – as well as in the Swiss National Clinical Trial Portal (SNCTP000005259).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



