Abstract
In this study, near infrared spectroscopy (NIRS) was used to measure activity in Broca’s area during a visual short-term memory task in young and older adults, comparing brain activity during three types of stimuli (numbers, easy-to-name shapes, and hard-to-name shapes) and examining how additive language functions are involved. We hypothesized that the Broca’s area would be activated in both age groups when memorizing numbers, but not when memorizing hard-to-name shapes. The results showed that visual memory capacity is reduced in the elderly, but not when easy-to-name stimuli are used. This indicates that adding a verbal function to the input of visual information by the elderly seemingly suppresses the effects of aging on memory capacity. This result indicates that the addition of verbal functions may complement other types of cognitive functions that have declined.
In studies of cognition and aging, it is generally considered that aging primarily affects the long-term memory and that it has little impact on the short-term memory (Nyberg et al., 2012; Park et al., 2002). However, as demonstrated by a study by Kunimi and Matsukawa (2009) elderly individuals have poorer short-term retention of matrix patterns immediately after display if the shapes are difficult to name. Kunimi (2010) examined how various properties of stimulus affect the impact of aging by comparing the memory capacity for visual stimuli that does or does not allow verbal encoding by different age groups. The study demonstrated that age only affected the memory capacity for shapes that are difficult to name, and that age did not affect the memory capacity if the shapes are easy to name. This demonstrates that while the visual short-term memory capacity is reduced in the elderly population, the impact of aging was seemingly suppressed if the visual stimuli is aided by verbal encoding or articulatory rehearsal. However, the role of these “additional verbal functions” is only hypothetical as it has not been examined experimentally. In the present study, we aim to provide neuroscientific evidence to support this hypothesis. Previous studies in the field of neuroscience demonstrated that the left inferior frontal gyrus (Brodmann area 44, Broca’s area) is involved in articulatory rehearsal (Ishkhanyan et al., 2020; Rogalsky & Hickok, 2011). Furthermore, studies using functional magnetic resonance imaging (fMRI) demonstrated that the activity level of the same region is elevated during semantic encoding of stimuli (Demb et al., 1995). In the present study, we used near infrared spectroscopy (NIRS) to measure the activity in Broca’s area during visual short-term memory tasks in younger and older subjects. Using three types of stimuli (numbers, shapes that are easy to name, shapes that are difficult to name), we compared the brain activities to determine how the additional verbal functions are involved in the process. We hypothesized that in both age groups, Broca’s area would be activated when memorizing numbers but not when memorizing shapes that are difficult to name. We also hypothesized that Broca’s area would be activated in the older subjects when memorizing shapes that are easy to name as they would employ the additional verbal functions.
Methods
Study subjects: We recruited 26 subjects aged between 20 and 22 years and 26 subjects aged between 65 and 74 years. All of the participants in the young group were college students. The older group was comprised of elderly people from Ishikawa and the surrounding suburbs who volunteered for this study. They were recruited through several local community associations. All potential subjects underwent medical check-ups (performed by physicians), and those who were considered to be healthy and to have normal or corrected-to-normal vision were enrolled in the study. Subjects who felt ill or had problems with their daily lives during the study period were excluded. We also used the mini-mental state examination (MMSE; Folstein et al., 1975) to screen for possible cognitive impairment such as apraxia or amnesia. All of the participants scored over 26 on the MMSE. The study procedures were approved by the ethics committee of the Kanazawa Institute of Technology, Japan, and all participants gave their written informed consent in accordance with the Declaration of Helsinki.
Devices: A 15-inch laptop (MacBook Pro, Apple) was used to display stimuli. Brain activity during the memory task was measured using a 24-channel near infrared imaging device (ETG-4000, Hitachi Medical Corporation). Probes were placed in a 3 × 5 arrangement in the left frontal-temporal region where Broca’s area is located according to the international 10-20 method.
Stimuli: Three types of stimuli were used for the memory tasks: “2-digit numbers,” “shapes that are difficult to name,” and “shapes that are easy to name.” The stimulus set of shapes created in the previous study (Kunimi, 2010). In the previous study, randomly arranged 4 × 4 matrix figures were divided into two groups: those that were judged to look like something (linguistic naming possible) and those that were judged not to look like anything (linguistic naming not possible). From that matrix, 40 shapes that no one judged to be “linguistically nameable” were selected and used as the “shapes that are difficult to name set.” The 40 figures that all participants judged to be “linguistically nameable” were used as the “shapes that are easy to name set.”
Procedures: The study subjects completed visual recognition tasks. A randomized block design was used for all experiments. The subjects completed a set of sequences 5 times; the set of sequences consisted of (1) input of stimuli that are presented in a sequence (task A, 15 s), (2) rest (30 s), and (3) output (task B, 15 s). The task A blocks were the input section, in which the study figure was presented continuously for 2,000 ms with a 1,000 ms gazing point in between. In the rest blocks, a mask was presented for 30,000 ms. The task B blocks were the output section. Participants judged on each trial whether the continuously presented test figure was the same as the training figure presented before the mask. All of these were arranged so that they were presented in the center of the screen. Task A and task B were repeated five times alternately, with rest blocks inserted at the beginning and end and between task blocks. Three sets of these procedures were performed for each stimulus condition. The order of implementation was counterbalanced.
Analysis design: Change in the level of oxyhemoglobin (OxyHb) in channel 16 during the execution of the task was analyzed. A two-factor experimental design was used for age group (young, old) × Stimuli (“2-digit numbers,” “shapes that are difficult to name,” and “shapes that are easy to name”; within-subject). The behavioral index was d’, calculated based on signal detection theory, and the physiological index was mMmm (ΔOxy Hb × optical pathlength; relative change of Oxy Hb) as the dependent variable. The two-factor analysis of variance was used in the analysis.
Results and Discussion
As shown in the top row of Table 1, the results of the two-factor analysis of variance with the behavioral index d' as the dependent variable showed an interaction between age and stimulus (F(2,100) = 4.15, p < .05). The results of the Bonferroni method-based subtest showed age differences only for figures that were difficult to name linguistically (p < .01). In agreement with the previous study (Kunimi, 2010), we also demonstrated that the older subjects had significantly poorer recognition of shapes that are difficult to name while the difference in age did not have an impact on the recognition of numbers and shapes that are easy to name. Since we focused on the language processing abilities at the time of stimuli input, we analyzed the brain activity during task A (Figure 1). Change in OxyHb (mM•mm) was used as the dependent variable, and a two-way analysis of variance was performed for the age group (younger vs. older) and types of stimuli (number vs. easy-to-name shape vs. difficult-to-name shape). As shown in the bottom row of Table 1, we demonstrated that both age (F(1, 50) = 15.76, p < .01) and the types of stimuli (F(2, 100) = 18.54, p < .01) had main effects on the outcome, and that age and stimuli had interaction effects (F(2, 100) = 12.54, p < .01). Lower-order analysis further demonstrated that in younger subjects, there were significant differences among numbers, shapes that are easy to name, and shapes that are difficult to name (p < .01 for all). In contrast, in the older subjects, there were significant differences between numbers and shapes that are difficult to name (p < .01) and between shapes that are easy to name and those that are difficult to name (p < .05).
Two-way ANOVA Table With Behavioral Indicators (Top Row) and Change in OxyHb (Bottom Row) as the Dependent Variable.

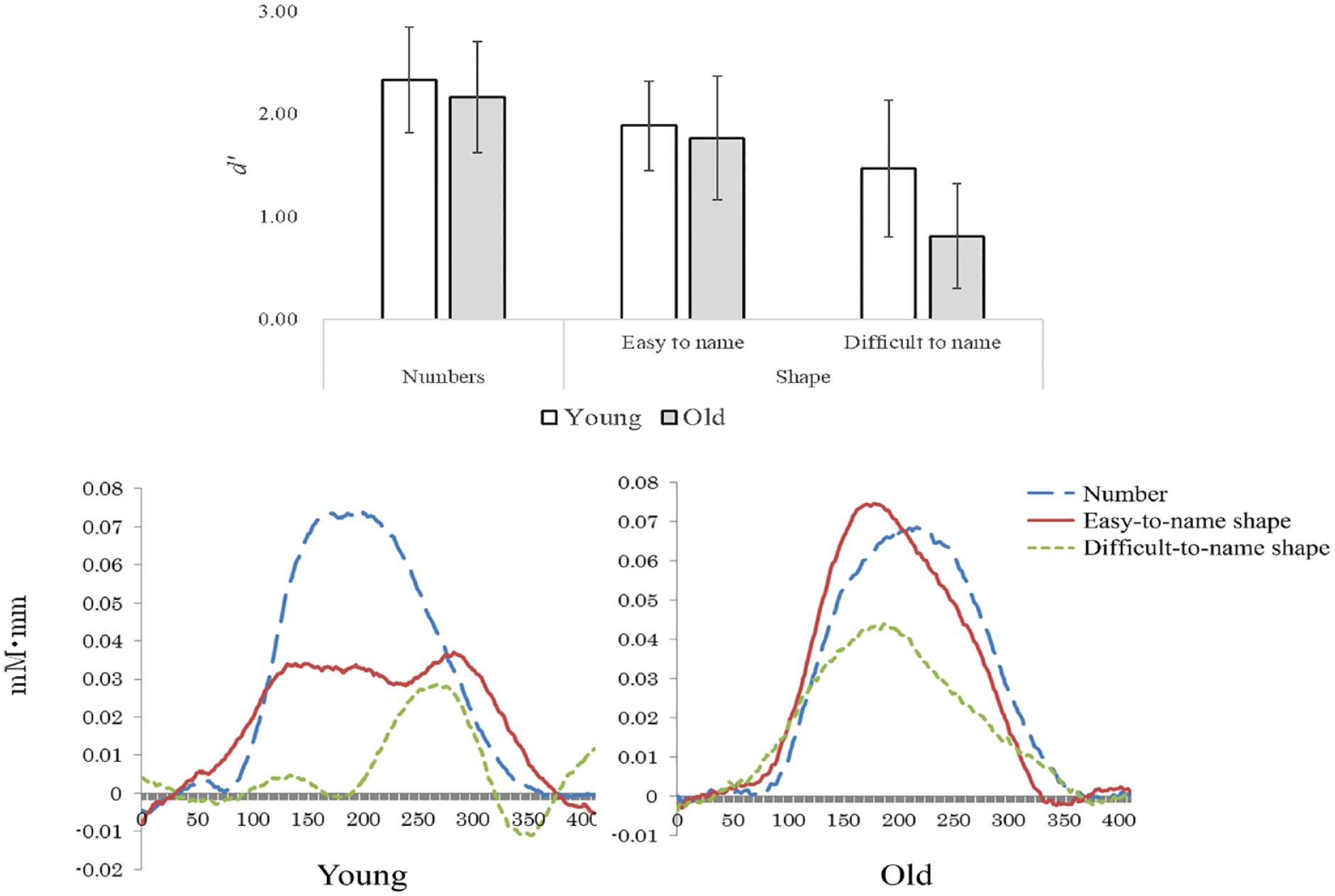
Results of behavioral indicators (top row) and change in OxyHb during the memory task (bottom row).
Although the visual memory capacity is reduced in older individuals, we demonstrated that the memory capacity is not reduced when stimuli that are easy to name are used. This demonstrates that the impact of aging on memory capacity was seemingly suppressed as older subjects employed additional verbal functions to input visual information. Our findings indicate that additional verbal functions may complement other types of cognitive functions that have been reduced.
These results are more reasonable to interpret that “the elderly have reduced short-term retention of nonsense figures” rather than “the elderly have reduced visual short-term memory but improved memory performance with the aid of verbal encoding, if available.” This interpretation is consistent with Bäckman and Dixon's (1992) argument for the existence of a compensation mechanism for loss as a process that reduces or eliminates the gap between currently available skills and what the environment requires. The results of this study demonstrate the need to interpret cognitive aging not only as a quantitative decline, but also as a qualitative difference. The results of this study may provide a scientific basis for our empirical approach to aging in the medical field, such as “providing information one by one in an easy-to-understand manner so that it is easy to encode” when dealing with the elderly.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Grant-in-Aid for Scientific Research (KAKENHI) Grant Number 20K03396.
