Abstract
Introduction
The rise in life expectancy and the decrease in the birth rate lead us toward an increasingly aging population. The prevalence of neurodegenerative diseases such as dementia is also growing (Sen et al., 2017). Dementia has become the biggest health problem facing the health systems in the 21st century. In 2015 about 47 million people worldwide live with some kind of dementia (Alzheimer’s Disease International, 2015), it is expected that this figure will double every 20 years, reaching 74.7 million in 2030 and 131.5 million in 2050 (Alzheimer’s Disease International, 2015; Livingston et al., 2017).
Dementia is a syndrome involving a decrease in cognitive function beyond the normal aging process. The term dementia indicates a loss of several brain functions interfering in the activities of people’s daily lives. The most common form of dementia is Alzheimer’s disease (AD), accounting for 60% to 70% of all dementias (Kovacs, 2018; Livingston et al., 2017; Sandilyan & Dening, 2015; World Heath Organization, 2019).
Dementia produces alterations in memory, language, comprehension, thinking, orientation, and learning ability. In addition, people living with dementia usually present sleep disturbances that affect the mood and quality of life of both people living with dementia themselves and their caregivers (Sadeghi et al., 2019; Webster et al., 2020). Prevalence data are variable due to the different types of disorders and research modalities, but it is estimated that between 25% and 60% of people living with dementia suffer some type of sleep disorder (Chwiszczuk et al., 2016; Guarnieri et al, 2012; Rongve et al., 2010), the most frequent alterations are insomnia, fragmentation of sleep with prolonged nocturnal awakenings and, in more advanced stages, excessive daytime sleepiness (Livingston et al., 2017; Lyketsos et al., 2002; Ownby et al., 2014).
However, the relationship between sleep disorders and dementia is not entirely clear. Some studies suggest that sleep disorders or alterations in sleep quality may appear before dementia itself, so that they would be associated with an increased risk of suffering dementia (Brzecka et al., 2018; Galbiati et al., 2019; Shi et al., 2018; Westwood et al., 2017).
The causes of sleep disturbances are diverse. Aging, the lack of light exposure, physical exercise, or meal timing play a very important role regulating of circadian rhythms. Medical or psychiatric morbidity and the drugs used to treat various diseases (dementia, hypertension, depression) are also some of the factors that can cause sleep disturbances (Cipriani et al., 2015; Ju et al., 2014; Shi et al., 2018). In addition to these causes, dementia is added to the neurodegenerative changes that alter sleep-wakefulness (Rongve et al., 2010), and predispose to suffer certain sleep disorders (restless legs syndrome, nightfall syndrome) (Troussière et al., 2014). The frequency, type and severity of sleep disorders vary from case to case depending on the variety and severity of the dementia (Ooms & Ju, 2016). It should be noted that unbalanced nutritional values (high consumption of refined sugar, animal products, high-calorie foods, and saturated fats) can also influence the circadian rhythm by altering the release of cortisol and disturbing the quality of sleep (Pistollato et al., 2016).
As we have described, dementia and other factors can influence sleep quality. Therefore, we aim to analyze the effect of dementia, gender, age, BMI, drug and coffee consumption and psychiatric diseases.
The proposed objectives of this study were (i) to determine the influence of dementia on sleep quality and (ii) to analyze if age, drug consumption, BMI, coffee, or psychiatric diseases influence the quality of sleep in subjects aged ≥65 years with and without dementia.
Materials and Methods
The population of the study consisted of 31 subjects, which ages ranged from 65 to 91 years old, divided into two groups: 15 participants with dementia (mean age 81.8 ± 8.1, 9 women) who attended the day care center of the Association of Relatives and People with Alzheimer disease in Murcia, Spain (AFAMUR); and a control group with 16 subjects without dementia (mean age 76.81 ± 6.25, 10 women) attending social centers in Murcia (Spain). The people living with dementia who participated in the study had previously been diagnosed by their neurologist. They, or their caregivers, signed the informed consent. We excluded those who did not agree to fill in the data to be collected and those in the control group who presented mild cognitive impairment.
The study was approved by the Ethics Commission of the University of Murcia. All participants signed informed consent and were assigned a numerical code to ensure confidentiality.
The Pittsburgh Sleep Quality Index (PSQI) was used to assess sleep quality (Buysse et al., 1989). This index is a self-administered questionnaire that consists of 19 questions that refer to how the person has slept normally during the last month. These questions include seven scoring areas (subjective quality of sleep, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of hypnotic medication, and diurnal dysfunction) with a range between 0 and 3 points. A score of 0 indicates that there is no difficulty, while a score of 3 indicates severe difficulty. Of the sum of the seven components is obtained the overall score of this index, which has a range of 0 to 21 points, therefore, a score of 0 would indicate the absence of difficulties and 21 would indicate severe difficulty in all the studied areas. Buysse et al. (1989) established the cut-off point to identify subjects with sleep disturbances from subjects without disturbances when the total score of the questionnaire was greater than 5 points. The Spanish version of the PSQI has been described as valid and reliable in various population groups (Hita-Contreras et al., 2014; Zhong et al., 2015).
The questionnaire was explained and given to the participants to complete (in the case of the group with dementia, the questionnaire was filled in by the of the person living with dementia’s relative and/or caregiver to avoid the possible biases derived from dementia itself).
We also collected information about gender, age, weight, height, psychiatric diseases, and drug and coffee consumption. For the group with dementia, the score on the Reisberg Global Deterioration Scale (GDS) (Reisberg et al., 1982, 1988) was collected. The GDS is a 7 point rating instrument for the staging of the magnitude of cognitive and functional capacity from normal aging capacities to very severe cognitive decline. It is applied to adults with cognitive symptomatology. This scale is valid and reliable (Ohayon et al., 2004).
All analyses were performed with the SPSS Version 24. The normality of the variables was checked with the Shapiro-Wilk test. The Student’s t-test was used for independent samples. The size effect was calculated with Cohen’s d for groups with different sample size. The correlations between the quantitative variables were made with Pearson’s correlation coefficient r and the correlations between qualitative variables with Pearson’s chi-square (χ2). As independent variables we used gender, age, body mass index (BMI), coffee consumption, number of drugs taken daily, psychiatric diseases for both groups, and GDS only for the dementia group. The PQSI score was analyzed in relation to these variables.
All results were expressed as mean ± standard deviation.
Results
The distribution of the data was found to be normal.
In Table 1 we can see the sample characteristics. No differences were observed between the groups in the distribution of gender, age, BMI, number of daily medications, or psychiatric diseases. In our sample, fewer people living with dementia (13.33%) consumed coffee compared to the other group (62.5%) and this difference was significant (p = .005).
Sample Characteristics.

Note. BMI = Body mass index; PSQI = Pittsburgh Quality of Sleep Questionnaire; GDS = Global Deterioration Scale.
p < .05.
We can observe in Table 2 the mean scores obtained in the PSQI by both groups, as well as the mean scores in every area of the questionnaire. There is no difference between the groups in the total score nor the subscores of the questionnaire.
PSQI Score and Subsores in Each Group.
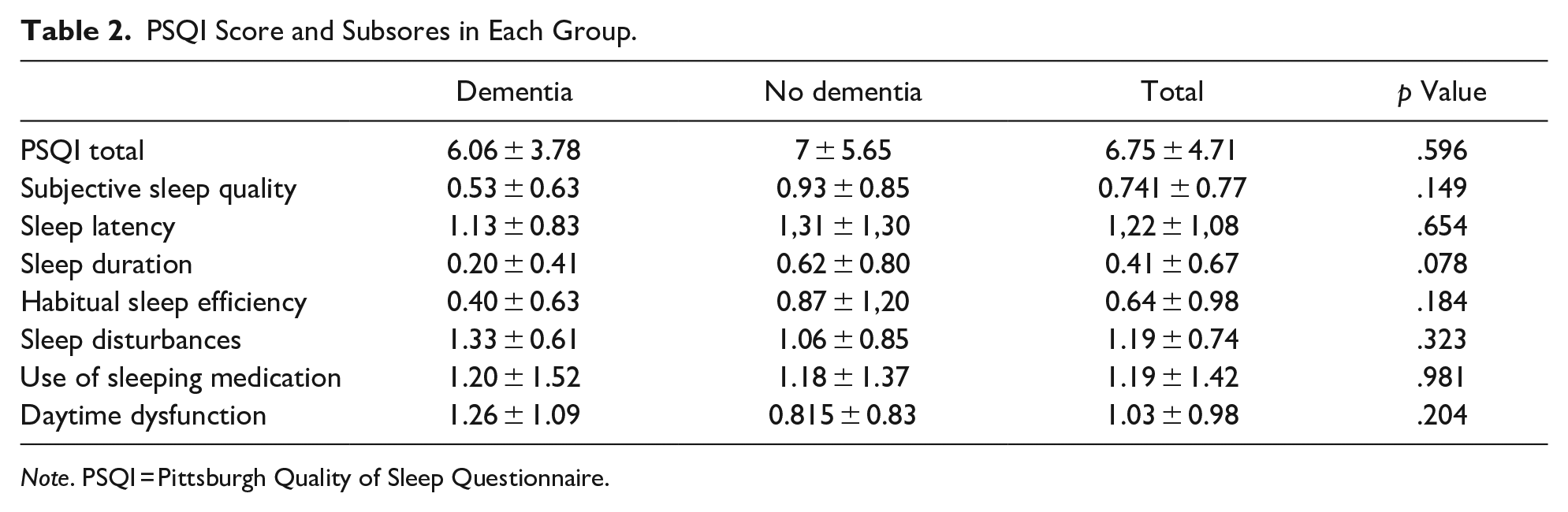
Note. PSQI = Pittsburgh Quality of Sleep Questionnaire.
In Table 3 we analyze the PSQI score in relation to the other variables. We observe some differences in this analysis. In our sample, in the group of people living with dementia, those who had other psychiatric diseases had higher scores in PSQI (8.83 ± 4.40) than those who did not have other psychiatric disease (4.22 ± 1.85) (p = .014). (Cohen’s d: 1.49). This difference is also observed in the total sample; those with psychiatric diseases scored 9.54 ± 5.39 and those without psychiatric diseases scored 4.9 ± 3.58 (p = .007) (Cohen’s d: 1.08). This difference can also be seen in Figure 1. In the analysis we found correlation between number of medications consumed daily and PSQI score in the group without dementia (r = .785, p < .001) and the total sample (r = .6, p < .001) (Figure 2). We observe that the PSQI score in the participants without dementia who do not consume coffee (9.50 ± 6.64) is higher than the score in the participants living with dementia who do not consume coffee (6.62 ± 3.77), but this difference is not significant (p = .229).
Comparison of PSQI Scores and Correlations.

Note. BMI = Body mass index; GDS = Global Deterioration Scale; PSQI = Pittsburgh Quality of Sleep Questionnaire.
p < .05.

Comparison of the mean PSQI score obtained by subjects with psychiatric diseases and those who do not suffer from psychiatric diseases in both groups and in the total sample.

Association between the PSQI score and the number of daily medications in the total sample.
In the dementia group, 46.7% had sleep disturbances, while in the group without dementia this percentage was 56.25%. We observe no differences in this aspect between the groups (χ2 = .285; p = .594). We have divided the groups according to whether or not participants had sleep disturbances (PSQI ≤ 5 = without sleep disturbances, PSQI > 5 = with sleep disturbances). The analysis of the variables in the participants with and without sleep difficulties is shown in Table 4. In the group of people living with dementia, we observed that sleeping disturbances were more frequent in people with other psychiatric diseases (83.33%, p = .02). This was also observed in the total sample (81.81%, p = .013). Analyzing the variable of number of drugs consumed daily differences were observed in both groups. In the group of people living with dementia, the number of drugs consumed daily is higher in those who have sleeping disturbances (9.57 ± 3.15) than in those who scored 5 points or less in the PSQI (5.57 ± 2.6) (p = .02) (Cohen’s d: 1.31). This is also observed in the group without dementia (7.66 ± 5.47, 2.43 ± 2.5, p = .026) (Cohen’s d: 1.17) and in the total sample (8.5 ± 4.57, 4.2 ± 3, p = .005) (Cohen’s d: 1.1).
Prevalence of Sleep Disturbances According to the Variables Analyzed.

Note. PSQI ≤ 5 = Without sleep disturbances; PSQI > 5 = With sleep disturbances; BMI = Body mass index; PSQI = Pittsburgh Quality of Sleep Questionnaire; GDS = Global Deterioration Scale.
p < .05.
Discussion
This study assessed the prevalence of sleep disturbances in subjects with dementia aged 65 years or older as compared to age-matched healthy people. We included the highest possible number of people living with dementia without excluding any type of dementia or advanced dementia. In the group without dementia, we only excluded subjects with cognitive impairment in order to make a comparison of this variable. The variables analyzed were age, gender, BMI, number of daily drugs, coffee consumption, and diagnosis of psychiatric diseases, and were compared with sleep quality by the PQSI in both groups. We have found that people who consume more drugs or have another psychiatric disease tend to have worse sleep quality.
There are studies that evaluated the quality of sleep and excluded those who presented a diagnosis of dementia (Keskin et al., 2018) due to the bias that this may imply in the results; therefore, we thought it would be interesting to have the collaboration of the caregivers and/or relatives of the people living with dementia to obtain the information, this fact was reflected in studies such as Rongve et al. (2010) or Ubis-Díez and Olivera-Pueyo (2012).
According to Guarnieri et al. (2012), sleep disturbances were more frequent in dementia due to Lewy bodies, affecting approximately 90% of people living with this type of dementia.
However, we can not affirm that there is a direct relationship between dementia and alterations in sleep quality, although 46.7% of the subjects with dementia presented sleep disturbances since no differences were observed in relation to the total PSQI score compared to the group without dementia. Similarly, in the study by Ubis-Díez and Olivera-Pueyo (2012) no significant differences were found between the two groups.
To quantify the cognitive impairment of the subjects, we used the score on the global deterioration scale of Reisberg (GDS). Based on this scale, we have compared the different scores obtained in the PSQI. No correlation was found between the cognitive deterioration measured with the GDS and a decrease in the quality of sleep measured with the PSQI. In contrast, authors such as Jirong et al. (2013) did find a correlation between the PSQI and the scores on the Mini-Mental State Examination. Differences were probably due to the fact that we worked with a small sample of subjects with dementia (n = 15) in relation to the study by Jirong et al. (2013), which consists of 409. However, not having found a relationship between these variables does not mean that the impact of dementia on sleep quality is not a factor to be taken into account when addressing and studying this population. In addition, we consider that it is very important to consider the assessment of the quality of sleep in the old people since studies such as that of Brzecka et al. (2018) have associated sleep disturbances with an increased risk of living with dementia.
In our sample, no significant differences were found in the age between the groups. However, we must consider that it is possible that this difference would have been obtained if the sample had been larger.
We did not find a correlation between age and the total score of the PSQI. On the other hand, studies such as Ohayon et al. (2004) and Redline et al. (2004), showed that age influences sleep. In addition, in the study by Li et al. (2020) a significant correlation was found between age and the score in the PSQI.
When analyzing the correlation between drug consumption and the total score in the PSQI in the group without dementia, a positive correlation was obtained so that the higher the consumption of drugs, the poorer quality of sleep, with a large size effect. In both groups, the intake of drugs was higher in people with sleep disturbances. Some studies also correlated polypharmacy and inappropriate medication use with sleep disturbances and poor sleep quality (Hamzaet al., 2019; Kumar et al., 2019).
In our study, we have also correlated BMI and PSQI. A previous study by Badran et al. (2015) demonstrated that a high BMI increased the possibility of having a poor restorative sleep. In addition, this factor must be taken into account since Badran et al. (2015) identified poor quality sleep as a risk factor for obesity. However, our study results did not show a significant correlation between BMI and sleep quality, unlike a study by Escobar et al. (2013) in which subjects with a higher BMI showed a poorer quality of sleep. The results in relation to BMI seemed to be contradictory. In the study by Öztürk and Ayhan (2018), and Yan et al. (2012), a relationship between the two was not obtained when correlating both parameters.
The consumption of coffee was also taken into account, and we obtained that the average score in the PSQI tended to be higher for non-consumers: the differences were not significant. This may be due to the small sample and the difference in the number of subjects who did not consume coffee between the groups (6 in the group without dementia, 13 in the group of people living with dementia). Still, several studies indicated that coffee consumption being psycho-stimulant promotes pathological dream (Loucraf-Hernández et al., 2006; Watson et al., 2016). In contrast, a previous study by other authors, such as López et al. (2016) has shown that coffee did not negatively affect sleep quality.
There was a difference in PQSI mean score between people with and without psychiatric diseases in the dementia group. A negative relationship was found between the quality of sleep and the presence of psychiatric diseases, with large size effect. This fact is underlined by a previous study by Doi et al. (2005) in which, as in our study, it was found that mean PSQI scores were significantly higher in subjects with psychiatric illnesses. Another study by Sun et al. (2018) has shown a bidirectional relationship between sleep quality and psychiatric disorders such as depression, so alterations in sleep quality can induce these diseases, as well as psychiatric disorders, and can produce changes in the architecture of sleep.
Finally, regarding the limitations of the study, it was very difficult to reach a larger enough sample size. Still, we consider it important to study subjects from the same day-care center to make factors such as feeding, exercise, and activities as similar as possible; although these were factors we could not control in the group without dementia. It would have been interesting to compare light exposure and physical exercise and meal timing outside the center. We acknowledge the limitation that the small sample implies, but we found large size effect in the differences we found to be significant. This study can be the precursor to other similar ones that can gather a larger sample, because the significant correlations we found have small power given the small sample size. In addition, note that questions about sleep quality referred to moments before the completion of the questionnaire (concerning the last month), which could cause errors when producing biased responses. Moreover, in the fulfillment of any questionnaire, personal perception comes into play, which is totally subjective. However, we consider that this type of tool can be a complementary tool versus more objective forms of evaluation. We believe it would be interesting for future studies to assess sleep quality in different types of dementia.
Conclusions
No differences are observed in relation to the quality of sleep between subjects with dementia and those who do not live with it.
High consumption of daily drugs and psychiatric diseases are factors that influence the quality of sleep in subjects older than or equal to 65 years.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Ethics
Subjects were informed and their caregivers/relatives signed the informed consent. The study had a favorable report from the Ethics Commission of the University of Murcia. The research was conducted ethically in accordance with the Declaration of Helsinki.
