Abstract
Using evidence from the Health and Retirement Study, we explore racial disparities in Alzheimer’s Disease (AD) onset risk. From a stress process perspective, there is substantial evidence in the literature that everyday discrimination is a chronic strain for Black individuals that acts as a social determinant of illness. However, few studies have examined specific relationships between this social stressor, race, and AD onset risk. Using Cox Proportional Hazard Models, we examined racial differences in exposure and vulnerability to everyday discrimination. Findings suggest that everyday discrimination predicts AD onset risk, and Black individuals experience more frequent exposure to everyday discrimination as a chronic strain. However, contrary to the stress process model, Black respondents were not more vulnerable to the effect of everyday discrimination on AD onset risk. Racial bias from medical professionals during the diagnostic process and mortality selection bias may explain this effect. Overall, the results of this study provide further evidence that discrimination is a key factor in predicting AD while also considering that many racial minorities with high rates of this type of social stress may not receive an unbiased diagnosis and/or survive to late life to develop AD.
Introduction
Since the Institute of Medicine’s (IOM) landmark report that identifies discrimination as a major source of racial disparities in health care (discrimination) (Nelson, 2002), it has become well-established in the literature that racial disparities are a pervasive public health threat (American Medical Association; O’Reilly, 2020). There is a wealth of evidence that demonstrates the existence of racial disparities across a range of adverse health concerns, including heart disease (Benjamin et al., 2017), coronavirus (Muñoz-Price et al., 2020), stroke (Gutierrez & Williams, 2014), and mortality (Farmer et al., 2019). In addition to these medical outcomes, researchers have asserted that there is an “immediate need” to further explore racial and ethnic disparities as it pertains to Alzheimer’s disease (AD) and cognition (Babulal et al., 2019).
AD and its related dementias represent a prominent challenge to the U.S. health system, and this burden is unevenly distributed across racial groups (Lin et al., 2021; Research Triangle Park; Lines et al., 2014). These racial disparities in AD exist for a variety of reasons, such as ambient fine particles (Younan et al., 2021), biomarkers (Morris et al., 2019), and stressful life events (Zuelsdorff et al., 2020). However, despite discrimination being a major source of stress among racial minorities that is related to a number of health concerns (e.g., Geronimus et al., 2006; Gonzales et al., 2021; Pascoe & Smart Richman, 2009; Williams & Mohammed, 2009), there are only a handful of studies that explore the relationship between everyday discrimination and health more broadly for those midlife and older (Sternthal et al., 2011; Williams & Mohammed, 2009; Zahodne et al., 2019), let alone studies exploring the specific relationships between race, discrimination as a chronic social stressor, and AD onset from a stress process perspective. For this reason, the purpose of this study is to understand how
The
Disadvantaged individuals (e.g., Black individuals) are more likely to experience greater exposure and vulnerability to social stress based on their social characteristics than their more privileged counterparts (e.g., whites), i.e.,
Our study contributes to the stress literature that explores how social factors may forecast AD in a number of ways. We utilize Health and Retirement Study (HRS) data to provide evidence on how the stress process explains the ways in which everyday discrimination as a social factor may predict AD onset risk in later life as well as the racial differences in this relationship. Thus, this study has two aims: (1) to test whether everyday discrimination acts as a chronic stressor that has a positive association with AD onset risk, and (2) explore racial differences in this relationship.
Methods
Data
The data for this study derive from the Health and Retirement Study (HRS). The HRS is a nationally representative, biennial longitudinal panel study of older Americans aged 51+ and now includes over 26,000 respondents. The Duke University Health System Institutional Review Board for Clinical Investigations approves the use of human subjects from this data source for this study (Institution: Duke University; FWA 00009025). The HRS first started collecting data in 1992 for the purposes of understanding the demographics, health transitions, career/retirement trajectories, and challenges associated with aging from an interdisciplinary perspective. However, in 2003, the HRS expanded to include psychosocial measures in order to better understand health disparities and offer research opportunities to other social scientific disciplines (e.g., sociology, gerontology, psychology) (Clarke et al., 2008).
The psychosocial measures are part of a leave-behind questionnaire that was completed by a subsample of respondents, meaning that 50% of rotating random HRS participants were given a self-administered questionnaire after completing the in-person Core Interview questions. The waves created after this time are particularly ideal for examining the research questions of this study, because they include psychosocial variables pertinent to the topic at hand, such as discrimination. Specifically, we will use the 2008 wave of the HRS as a baseline. This wave was the first to include scale items that inquire about chronic and acute discrimination from a medical professional—an important potential contributor to diagnosis timing and overall health-care-seeking behavior. We only selected participants from Wave 9 who completed this questionnaire and provided a response to both our chronic stress scale and all the other variables of interest to this study entirely (
Measures
Everyday discrimination
The HRS includes a measure for a widely recognized 6-item Everyday Discrimination Scale (EDS). Stress process researchers created the EDS to examine the chronic strains related to experiencing unjust and differential treatment on a daily basis (Williams et al., 1997). Everyday discrimination is a
Time to AD onset
The HRS includes onset information for Alzheimer’s Disease (AD), specifically codes 331.0 (ICD-9) and G30 (ICD-10). We use time-to-event censored data to examine AD onset starting from the 2008 wave and use age as the time-scale variable. Respondents who were lost at follow-up for other reasons unrelated to AD (e.g., going abroad or unrelated death) were censored.
Demographic variables
Race is included as a key predictor (coded as 1 = Black, 0 = white). We also include several demographic variables as control covariates, given that discrimination varies by social status (Lee & Turney, 2012). Specifically, we control for sex (coded as 1 = female, 0 = male), age (coded chronologically), education (coded as 1 = does not have a high school education, 0 = obtained a high school degree or more), and income (coded as 1 = lowest quintile of income with respect to total HRS sample, 0 = all other quintiles).
Exposure and vulnerability
We measure exposure as when respondents report higher scores on the Everyday Discrimination Scale (EDS), that is, more frequent experiences with everyday discrimination. We measure vulnerability as group differences in the effect size of the Everyday Discrimination Scale on AD onset risk.
Hypotheses and Analysis
Drawing from the stress process paradigm and in alignment with the goals of this study, we propose the following hypotheses:
H1: Everyday discrimination will have a positive association with AD onset risk.
H2: The relationship between more frequent everyday discrimination and AD onset risk will be stronger for Black individuals than their counterparts (differential vulnerability hypothesis),
Cox proportional hazard models also offer the appropriate analyses to examine some of the hypotheses, given that they assess for risk and allow for the ability to examine group differences through moderation. More specifically, to examine Hypothesis 1 and Hypothesis 2, we utilized Cox proportional hazard models to attain hazard ratios (HRs) for the relationship between everyday discrimination, race, and the risk of AD onset.
Results
Descriptives
Table 1 displays the descriptive statistics for the sample stratified by race. On average, results for the full sample suggest that respondents experience everyday discrimination relatively infrequently (
Sample Descriptives Stratified by Race.

Everyday Discrimination as a Race-Related Stressor That Predicts AD Onset Risk
Table 2 displays the results for the first Cox proportional hazard model, which tested the association between everyday discrimination and the risk of AD. Results confirm Hypothesis 1, which postulated that higher rates of everyday discrimination would be linked to greater AD onset risk. As shown, more frequent everyday discrimination was positively associated with AD onset risk (
Cox Proportional Hazard Model for Everyday Discrimination and Race Predicting AD Onset Risk Using Age as a Time-Scale Variable (

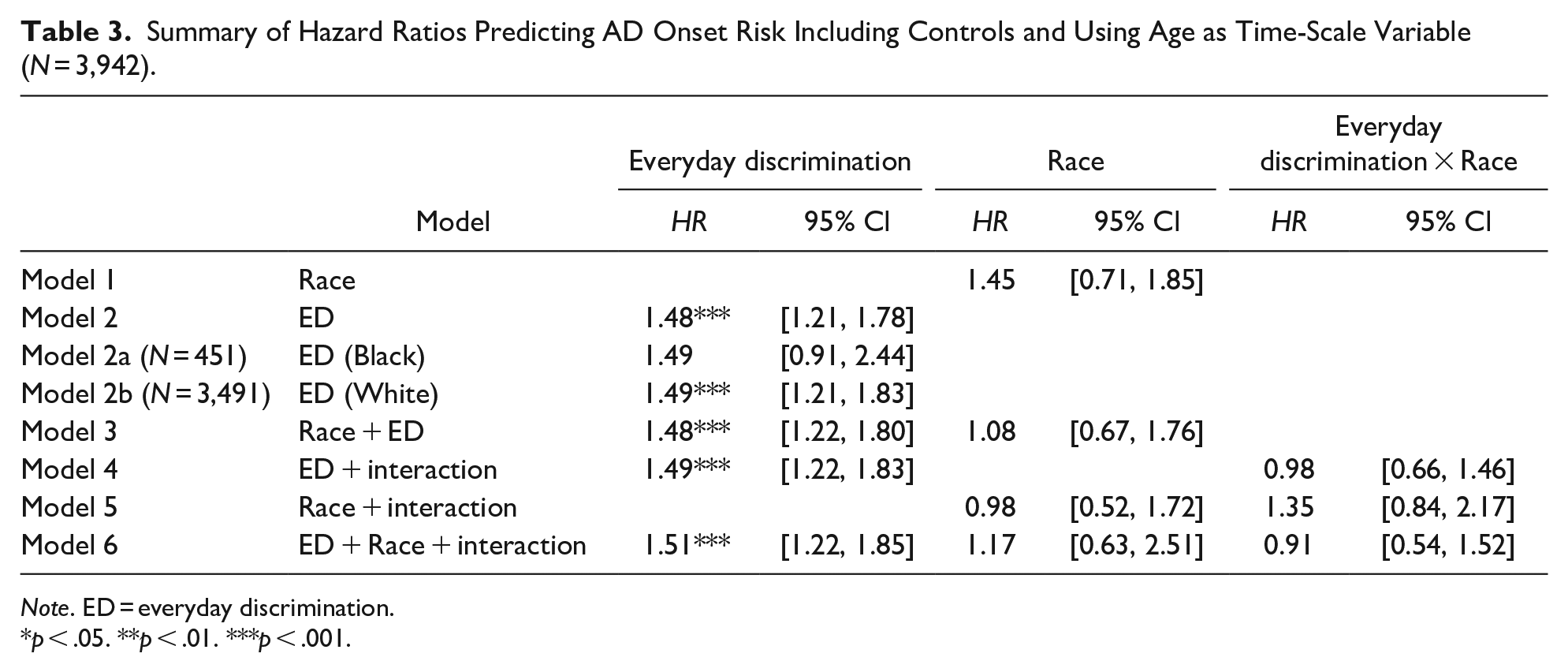
Table 3 displays a summary of the hazard ratios that predict AD onset risk with the inclusion of control covariates. This summary helped to test Hypothesis 2, which assessed whether everyday discrimination and AD onset risk is stronger for Black individuals. The HRs for everyday discrimination assess for vulnerability when comparing racial groups. Model 1 examined the effect of race on AD onset risk. Model 2 examined the effect of everyday discrimination on AD onset risk for the full sample, and Model 2a and 2b examined this relationship for the Black and white samples separately. Model 3 examined the effect of race and everyday discrimination on AD in the same model. Model 4 examined the interaction effect between race and everyday discrimination while also controlling for everyday discrimination in the same model. Model 5 examined the interaction effect between race and everyday discrimination while also controlling for race in the same model. Model 6 examined the interaction effect between race and everyday discrimination while also controlling for both everyday discrimination and race in the same model. Everyday discrimination had significant effect on AD onset risk for almost every model (
Summary of Hazard Ratios Predicting AD Onset Risk Including Controls and Using Age as Time-Scale Variable (
Discussion
Our study found that everyday discrimination is a chronic strain that predicts AD onset risk in later life, and Black individuals have higher exposure to this stressor than their white counterparts. These findings are consistent with the central stress process principles that social stress has a relationship with adverse well-being outcomes and that lower status individuals experience greater exposure to stressors (Thoits, 1995). Although the first set of findings were consistent with the stress process perspective, there were some that were contrary to this perspective. The stress process perspective articulates that low status individuals are more vulnerable to the effect of stress on illness (Brown et al., 2020; Thoits, 1995). Thus, we postulated that high rates of reported everyday discrimination would have a stronger association with AD onset risk for Black individuals. However, contrary to our hypothesis, we found that Black individuals were not more vulnerable to the effect of everyday discrimination in this respect. Although some research suggests that Black individuals are diagnosed with AD 1.33 to 1.50 times more often than whites (Akushevich et al., 2020; Matthews et al., 2019; Mayeda et al., 2016), other research suggests that Black individuals are 30% less likely to receive a diagnosis (e.g., Lennon et al., 2021). We found that there were not race differences in either of these directions regarding the percentage of respondents who had AD at follow-up and that everyday discrimination did not have a stronger effect for Black respondents.
There may be a couple reasons that explain the findings that did not support the stress process model. For instance, Black individuals who experience high rates of everyday discrimination may not receive a proper, unbiased diagnosis or be underdiagnosed, thus rendering a relationship between everyday discrimination and AD onset risk to be less robust in the data. There is evidence that medical professionals can have a racial bias when diagnosing psychological or psychiatric disorders (Garb, 2021; Schwartz & Blankenship, 2014). According to the Alzheimer’s Association (2021), 50% of Black individuals report experiencing discrimination as it relates to health care, and 36% view discrimination as a barrier to receiving care for AD, making it possible that they never receive a proper diagnosis. Perhaps some physicians may falsely assume that a Black individual’s cognitive decline is their normal functioning, especially when accounting for racial stereotypes that perpetuate the notion that Black individuals are less intelligent that whites (e.g., Gadson & Lewis, 2022).
Another explanation for the unexpected vulnerability results may be that Black individuals with high rates of everyday discrimination may not be living long enough to develop and be diagnosed with this disease, given that the majority of those with Alzheimer’s are older than 75-years-old (72%; Alzheimer’s Association, 2021). The average life expectancy of Black individuals (72-years-old) is significantly lower than that of white individuals (78-years-old; Arias et al., 2021). Respectively, more frequent everyday discrimination is associated with mortality risk (Farmer et al., 2019) and with other medical conditions that could result in death (e.g., Benjamin et al., 2017; Cobb et al., 2020). Thus, many Black individuals who report experiencing high rates of everyday discrimination may have been censored out of the data for deaths unrelated to AD, leaving only Black individuals left in our sample at follow-up who might be less vulnerable to the effects of stress than their similar others who died at an earlier age.
Although the results of this study contribute to a growing body of literature that discrimination has a significant role in explaining health disparities, it is not without limitations. As noted, there may be a mortality selection bias, in which those who report experiencing high rates of everyday discrimination may decease prior to reaching late-life and being diagnosed with AD or be censored out for other reasons leaving only the most stress-resilient individuals in the sample. Secondly, although the everyday discrimination scale (EDS) is one of the most used measures to study discrimination there has been some concern whether it exhaustively captures all aspects of discrimination and its severity (Harnois et al., 2019; Krieger, 1999; Williams & Mohammed, 2009). Similarly, we were not able to comparatively examine other racial and ethnic groups (e.g., Native Americans) as well as their intersections with other social statuses. Discrimination may be qualitatively different across and within certain social groups, and there may not be measurement equivalence across these groups using the everyday discrimination scale (Bastos & Harnois, 2020; Harnois et al., 2019). Another limitation is that our sample size for the Black sample is much smaller than the white sample, and this may explain why the relationship between everyday discrimination and AD was significant for the white respondents instead of their counterparts, given that larger sample sizes typically yield more significant results (Asiamah et al., 2017; VanVoorhis et al., 2007).
Although there were several limitations, this research study helps provide insight into necessary directions of future research. For instance, additional research is needed to examine racial bias in the diagnosis of AD. There are a number of healthcare barriers that may prevent a non-white individual from receiving an appropriate diagnosis of AD, such as mistrust of clinical trials, access of care, and medical professionals treating them as less smart (Alzheimer’s Association, 2021). Secondly, another direction for future research is to explore how other marginalized positionalities may intersect with race (e.g., gender, class, age, sexual orientation) that pose a risk to one’s well-being. Given that the average age of adults in this sample is about 72-years-old, it is possible that high status individuals (e.g., whites) could be experiencing high rates of discrimination for the first time related to ageism (Barnes et al., 2008; Chang et al., 2020) and not have the resiliency to deal with this type of chronic strain for this first time, thus leaving them more vulnerable to its effects.
Experience with discrimination and barriers to Alzheimer’s treatment also varies greatly across race and ethnic groups (Alzheimer’s Association, 2021), and additional research that oversamples smaller racial groups is needed. Respectively, within these racial and ethnic groups, further exploration is also needed to explore cohort differences, given that older adults who experienced the Civil Rights movement at different ages have different perceptions of discrimination (Barnes et al., 2008; Lewis & Van Dyke, 2018; Versey & Curtin, 2016). Another potential promising future direction is to explore how coping resources may explain the relationship between everyday discrimination and AD onset risk, such as common coping resources (e.g., self-esteem, social support) or coping strategies; Sivan-Donin et al., 2019; Thoits, 1995).
This study has a number of strengths. To begin, the results from this sample are nationally representative for midlife and older adults. This allows for the results to be generalizable to non-institutionalized adults in the United States aged over 50-years old. In addition, this study examined the relationship between everyday discrimination and AD onset risk over a lengthier follow-up period than prior studies by utilizing longitudinal data. Importantly, this study also expands the stress process in two ways. (1) It establishes everyday discrimination as a chronic strain that has an association with AD onset risk. (2) The results help to expand the stress process model by shedding light on the nuances of vulnerability as it relates to late-life illness and how exposure to an unhealthy stressor may be a better indicator of aging disorder disparities when accounting for mortality selection.
Conclusion
By exploring the specific relationships between race, everyday discrimination, and AD onset risk, this study offers important considerations when examining racial disparities in late-life disorders, given that everyday discrimination predicted risk of AD onset but race itself did not. Our results provide further evidence that the chronic stress of discrimination is a fundamental factor in forecasting health disparities in AD onset risk, while also taking into account existing literature that establishes how many racial minorities with considerable amounts of this form of stress may not have the privilege of surviving to late-life, that is, mortality selection bias, and/or the privilege of receiving an unbiased diagnosis (Alzheimer’s Association, 2021; Farmer et al., 2019). All in all, in keeping in mind these important considerations, the stress process paradigm helped to explain AD onset risk by demonstrating how social stress (i.e., everyday discrimination) can act as a predictor, suggesting that disproportionate social demands that vary by disadvantaged status, more generally, can play a significant role.
Footnotes
Appendix A
Acknowledgements
I would like to acknowledge Dr. Heather Farmer, University of Delaware, for providing insight on how to perform analysis using the Health and Retirement Study and for her existing body of work, particularly the paper previously submitted to
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Publication of this article was supported in part by the National Institute on Aging (grants R01-AG066133 and RF1-AG046860). The sponsors had no role in design and conduct of the study, collection, management, analysis, interpretation of the data, preparation, review, approval of the manuscript, nor decision to submit the manuscript for publication. The Health and Retirement Study data is sponsored by the National Institute on Aging (grant number U01-AG009740) and is conducted by the University of Michigan.
