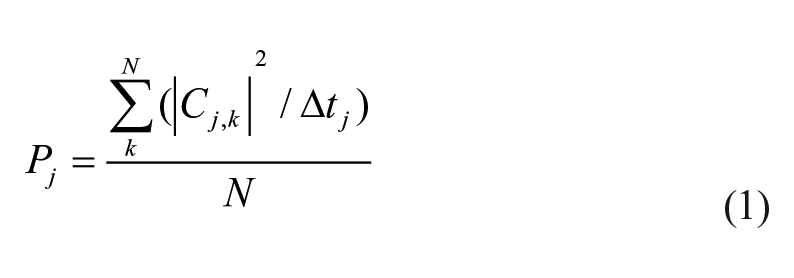Abstract
Skin autofluorescence is a useful index to estimate the accumulation of advanced glycation end-products in human tissues. Elderly persons with higher skin autofluorescence have lower muscle mass, muscle strength and muscle power, however, little is known about the relationship between the skin autofluorescence level and each muscle activity. We measured the values of skin autofluorescence from five places on a lower limb, and the signals of surface electromyogram during isometric contractions from five muscles on that, simultaneously. The waveforms of surface electromyogram were analyzed by Daubechies-4 wavelet transformation. The value of skin autofluorescence was increased in the proximal part of the lower limb compared with the value of the distal part. The principal component of surface electromyogram activity in a time-frequency domain was lower in the proximal part compared with that of the distal part. There was a weak negative correlation between the value of skin autofluorescence on the gluteal region and the value of the mean wavelet coefficient of the surface electromyogram signals within the gluteus maximus muscle. The higher accumulation of advanced glycation end-products on the gluteal region might suggest the lower muscle activity in aging without disease and disability.
Keywords
Introduction
The accumulation of advanced glycation end-products (AGEs) on tissue proteins has been implicated in the aging of proteins and the progression of chronic, age-related diseases, and it results from a combination of hyperglycemia, oxidative/carbonyl stress, and/or decreased renal clearance of AGEs precursors (Schleicher et al., 1997). Skin autofluorescence (AF) is one of the markers to evaluate the accumulation of AGEs in human tissues (Meerwaldt et al., 2004, 2005). Over the past few years a considerable number of studies have been made on the relationship between skin AF and various diseases, for example, type 1 or type 2 diabetes (Fokkens & Smit, 2016; Osawa et al., 2017; Temma et al., 2015), cardiovascular disease (Yamagishi et al., 2015), chronic kidney disease (Furuya et al., 2015), peripheral artery disease (de Vos et al., 2014; Schutte et al., 2016), and chronic obstructive pulmonary disease (Gopal et al., 2014; Hoonhorst et al., 2014). These pathological studies have shown that skin AF is a useful index to estimate these diseases as a simple and non-invasive method.
In recent years there has been interest in the relationship between the value of skin AF and reduced functions of skeletal muscles among aging and elderly persons from the viewpoint of sarcopenia. The higher value of skin AF is one of the reasons of impaired lower limb muscle function in patients with type 1 diabetes (Mori et al., 2017). Middle-aged and elderly persons with a higher value of skin AF had lower muscle strength and muscle power (Momma et al., 2011) and low muscle mass (Kato et al., 2017; Tabara et al., 2019). Although, little is known about the relationship between the values of skin AF on various body parts and the reduced activities of various skeletal muscles in aging with nonpathologic states: successful aging (Rowe & Kahn, 1997). In this study, we focused on the muscle activities of the lower limb which are strongly related to locomotion activities, as an evaluation scale for sarcopenia (L. K. Chen et al., 2014; Cruz-Jentoft et al., 2019; Rosenberg, 1989). It was hypothesized that higher accumulation of AGEs into the skin decreased for activity rather than lower limb muscles in young adults and elderly persons without disease and disability.
Materials and Methods
The values of skin AF representing the accumulation of AGEs on the lower limb and the muscle activities of the lower limb during isometric contractions were compared between healthy young persons and elderly persons without disease and disability. The muscle activities were estimated using a wavelet analysis of surface electromyogram (EMG) signals to determine the muscle functions on the time-frequency domain, and we compared them among the subjective groups. Also, the relation between skin AF level and the activity of surface EMG on the lower limbs were analyzed in successful aging.
Participants
The study included 20 elderly healthy male volunteers (age: 71.2 ± 2.5 years) and 18 young healthy volunteers (age: 21.3 ± 4.2 years). The skin AF measurements in subjects with darker skin colors typically resulted in lower values than in subjects with fair skin colors (Koetsier et al., 2010; Mulder et al., 2006), therefore, the northern Japanese with intermediate skin color were used in this study. Normal health status was previously established by medical history, physical examination, biochemical and hematologic screening, and electrocardiogram. All volunteers had undergone this examination to exclude diabetes mellitus, hypertension, cardiac disease, and other symptoms pertaining to the cardiovascular system. They were of average physical fitness, and had not taken any medication during the previous month before entry into the study. All participants provided written informed consent before participating in the study as approved by the ethics committee of the University Institutional Review Board.
Measurement of Skin AF
The skin AF representing the accumulation of AGEs was measured using of the AGE Reader™ (DiagnOptics Technologies, the Netherlands). The AGE Reader™ estimated the accumulation of AGEs as skin AF that was defined as the ratio of the average emitted fluorescence intensity per nm in the range of 420 to 600 nm and the average reflected fluorescence intensity per nm in the range of 300 to 420 nm (Meerwaldt et al., 2004). Recently, it has been widely accepted to measure the skin AGEs as a simple non-invasive method. In this study, the values of skin AF were measured from five regions of the right lower limb which included the anterior region of the leg, the posterior region of the leg, the anterior region of the thigh, the posterior region of the thigh, and the gluteal region. These skin AF values were recorded with the subject in a lying position on an experimental bed.
Measurement of Surface EMG Signal
Surface EMG signals were recorded from five muscles of the right lower limb: tibialis anterior (TA) muscle, gastrocnemius medialis (GA) muscle, rectus femoris (RF) muscle, biceps femoris (BF) muscle, and gluteus maximus (GM) muscle by using a wireless SEMG system (Trigno Wireless System, DELSYS, USA) during the knee joint extension and flexion movement with maximum voluntary isometric contraction. The wireless surface EMG electrodes were placed longitudinally with respect to the underlying muscle fiber arrangement and were located according to the recommendations of SENIAN (Hermens et al., 2000). The frequency range and the amplifier gain were set from 20 to 450 Hz and 80 dB, respectively. In order to remove motion artifacts, the low cut frequency was set at 20 Hz (De Luca et al., 2010). The SEMG signals were stored on a personal computer through an A/D converter (PowerLab, ADInstruments, New Zealand) with a sampling frequency of 1,000 Hz.
Data Processing
First of all, the values of skin AF from five regions on the lower limbs were standardized according to the value from the palmar side of the right forearm of each individual. The mean values of standardized skin AF were compared among each recording region and subject group.
Secondly, the normal distribution was confirmed for all EMG data by Anderson-Darling Normality test. The original EMG data were applied using a discrete wavelet transform (DWT). The DWT is becoming a standard tool for analyzing EMG data (Koenig et al., 2018). The method of DWT has been described in our previous paper (Kaneko et al., 2020). In short, the DWT is defined as the convolution between the signal x(t), which has its wavelet transform coefficients Cj,k and the wavelet functions Ψj,k(t). In this study, Cj,k was estimated by using the Daubechies-4 (Daubechies, 1990) wavelet algorism and EMG data of 512 points in relation to the knee joint extension and/or flexion movement. The EMG data were decomposed into five separate scales (i.e., j = 1: 450–250 Hz, j = 2: 250–125 Hz, j = 3: 125–62.5 Hz, j = 4: 62.5–31.25 Hz, and j = 5: 31.25–20 Hz, respectively), by analysis software (MATLAB MathWorks, USA). To examine the mean power of each frequency band, Pj was quantified as
(Δtj was sampling time). Reduction of the influence of the period of analyzing time was considered. The proportion of frequency component for the total value of the Pj was evaluated as a % Pj. The values of % Pj were compared in each frequency band and subject group to estimate functional changes of lower limb muscles as an effect of aging.
Finally, the relation between z-scores of skin AF and z-scores of Pj were analyzed. The z-scores of Pj were integrated into three frequency bands (i.e., high-frequency band: j = 1 and 2, middle-frequency band: j = 3, and low-frequency band: j = 4 and 5, respectively).
Statistical Analysis
For a multiple comparison procedure, the homogeneity of variance was assessed by Leven test. An analysis of variance (ANOVA) in a two-way layout without repeated measurements in standardized skin AF for both subject groups was performed to investigate which factors contributed to the parameter. Significances of individual differences were evaluated by using the post-hoc comparisons if ANOVA was significant. Tukey’s honestly significant difference (Tukey’s HSD) was applied for post-hoc comparisons with significance levels of .01. For comparisons of the proportion of Pj (%Pj), a chi-square (χ2) test for independence was performed to investigate how independence changes with significance levels of .01 and .05. Pearson’s product-moment correlation coefficient (γ) was used to study the relationship between the z-score of skin AF and the z-score of Pj. Statistical significance was defined as a p < .01. All statistical analysis was completed using IBM SPSS Statistics for Windows, Version 25.0.
Results
The standardized skin AF was increased in the proximal side of the lower limb compared with that of the distal side in both young adults and elderly persons. The values of standardized skin AF in elderly persons were higher than that in young adults at all five recording positions of the right lower limb (Figure 1). The two-way ANOVA revealed significant effects of recording-position (F(4,140) = 41.06, p < .001, η2 = .24) and of aging (F(1,140) = 356.79, p < .001, η2 = .53) for the standardized value of skin AF.

Comparison of young adults and elderly persons in standardized skin AF. Mean values of standardized skin AF (±SE) on lower limb were compared among five recording positions in young adults and elderly persons. The mark ** indicates a significantly different level of 1% of post-hoc test.
Surface EMG activities of the five muscles in each subject group were estimated as the values of %Pj and compared in five frequency bands. In Figure 2, TA muscle and GA muscle were activated with higher frequency bands (450–125 Hz) of surface EMG than RF muscle and GM muscle in both subject groups. On the contrary, the surface EMG activities with lower frequency bands (62.5–20 Hz) were larger within TA muscle and GA muscle rather compared to RF muscle and GM muscle. For the BF muscle, values of %Pj differed in each subject group (χ2 = 13.24; p < .01, Cramér’s V = 0.66). The principle frequency components of %Pj of BF muscle was higher in young adults compared to that in elderly persons, thus these values represented the features of respective muscle activities. A similar tendency was seen among the %Pj of GA muscle as well, though there was no statistical significance (χ2 = 7.58; p = .11, Cramér’s V = 0.50).

Values of proportion of Pj (%Pj) were compared between the elderly persons and the young adults for five muscles (a–e). The EMG activities were decomposed into five separate scales as frequency bands; j = 1: 450–250 Hz, j = 2: 250–125 Hz, j = 3: 125–62.5 Hz, j = 4: 62.5–31.25 Hz, and j = 5: 31.25–20 Hz).
The relationships between the z-scores of skin AF and the z-scores of Pj were analyzed by Pearson’s correlation coefficient (γ) as shown in Table 1. Overall, there was a moderate negative correlation between the standardized values of Pj with three kinds of frequency bands and the z-scores of skin AF on the lower limb without the set of TA versus Leg (anterior). The values of γ were lower in the activities of surface EMG with low frequency band than that with high frequency band in the set of GA versus Leg (posterior), BF versus Thigh (posterior), and GM versus Gluteal Region (Table 1). The lowest value of γ was observed in the relation between the skin AF of gluteal region and activities with the low frequency band of surface EMG of GM muscle. Linear regression analysis was performed to investigate the associations between skin AF and frequency characteristics of surface EMG activities as aging effects. The z-score of skin AF at the gluteal region was associated with the z-score of P1+2 (γ = −.39, p < .01), P3 (γ = −.54, p < .001), and P4+5 (γ = −.57, p < .001) at GM muscle (Figure 3(a)–(c)). In addition, Figure 3(d) showed negative correlations between the z-scores of skin AF on the posterior thigh and the z-scores of P4+5 from BF muscle in the total population (γ = −.46, p < .01). These four graphs showed large differences in distribution of the value of skin AF and the activities of surface EMG between the young adults and the elderly persons.
Summary of Pearson’s Correlation Coefficient (γ).

Note. The relationships between surface EMG activities during knee joint flexion and/or extension movements with the maximum voluntary efforts and the standardized values (z-score) of skin AF at rest are presented above as Pearson’s correlation coefficient (γ). The z-scores of Pj were separated into three frequency bands (i.e., high, middle, and low) because we have speculated a relationship between the values of Pj and the activity of muscle fiber types.
Significant at p < .05, N = 30.
Significant at p < .01.

Association of the z-scores of skin AF at the gluteal region and (a) muscle activities of GM muscle with high frequency band (P1 + P2: 450–125 Hz), (b) with middle frequency band (P3: 125–62.5 Hz), and (c) with low frequency band (P4 + P5: 62.5–20 Hz), respectively. The relationship between the z-scores of skin AF at posterior region of leg and the muscle activities with low frequency band (P4 + P5: 62.5–20 Hz) of BF muscle is shown in (d). R2 shows correlation coefficient.
Discussion
It has been established that skeletal muscle aging is characterized by a structural and functional changes, as age-related alterations in muscle metabolism, including insulin sensitivity and mitochondrial capacity. The present study estimated the accumulation of AGEs by the use of the skin AF to assess the relationship between accumulated level of AGEs within various body parts of the lower limb and aspects of muscle activities of the lower limb due to aging without disease and disability. Research on the relationship between the skin AF and sarcopenia was studied by Mori et al., (2019), Eguchi et al., (2021), and Waqas et al., (2022). These studies showed that the value of skin AF was significantly negatively associated with the skeletal muscle mass, skeletal muscle index and knee extension strength, although these studies did not recognize the direct relationship between the values of skin AF from various body parts of the lower limbs and the muscle activities of the lower limbs because the skin AF was recorded from only one fixed region. In our study, the skin AF was recorded from five regions on the lower limbs. The skin AF from various recording regions on the lower limbs showed different values which were increased in the proximal side of the lower limbs compared with the values of the distal side and the same disposition in both subject groups as shown in Figure 1. These results suggested the accumulated level of AGEs on the lower limb depended on each body part. The values of skin AF were calculated by the assessment of the AGEs accumulation of the skin tissues (Meerwaldt et al., 2004, 2005), these were associated with collagen-linked fluorescence, pentosidine, N ε -(carboxyemethyl)-lysine, and N ε -(carboxyethyl)-lysine in the dermal tissue. The excitation light of the AGE Reader™ was emitted to 1 to 2 mm under the skin. It was not known if the AGE Reader™ detected the accumulated AGEs in the dermal tissue and/or the muscle tissues, although, the highest value of skin AF was observed at the gluteal region of lower limb in both subject groups. The values of skin AF might be influenced by the AGEs accumulation of subcutaneous tissues of each lower limb part.
As Distefano and Goodpaster (2018) pointed out, there is no agreement on these points concerning the roles of both mass and function, along with the underlying mechanisms of sarcopenia and the bioenergetics of the aged muscle. In our study, the muscle activities of lower limb muscles were assessed using surface EMG. The data of surface EMG were decomposed into five frequency bands using the DWT method. The mean amplitude information of surface EMG was observed as the Pjs which was the power of surface EMG activities at the frequency level of j. The calculated values of %Pj as a proportion of Pj (j = 1, 2, 3, 4, and 5, respectively) to the entire Pj values shows that the principal frequency components of surface EMG activities were significantly different between the young adults and the elderly persons in the BF muscle. A similar tendency was seen among the %Pj of GA muscle as well. The principal frequency components of %Pj in the elderly persons was higher than that in the young adults, thus these values represented the features of respective muscle activities in relation to the physiologic aging. In the previous studies, it was assumed that the spectral property of the surface EMG was altered by the volume conductor effects of the muscle fibers and the number of activated motor units (MUs). Recruitment strategies that resulted in a greater proportion of faster MUs being active had a significantly higher mean frequency (Wakeling, 2009). The increase in surface EMG amplitude might represent MUs recruitment and/or MUs firing frequency modulation, whereas the increase of mean frequency of the power spectrum might represent the additional recruitment of superficial high threshold MUs that most likely possess large and sharp spikes affecting high frequency bands of the surface EMG power spectrum (Moritani et al., 2004). The median frequency of power spectrum increased with age and showed negative correlation with the forced vital capacity. The higher median frequency of the power spectrum may be due to early recruitment of larger MUs (R. Chen et al., 1996). In our results regarding DWT analysis of surface EMG data, BF muscle activities during maximum voluntary contraction resulted from the recruitment of high threshold MUs in elderly persons in comparison to that of the young adults because of the lower muscle mass and/or the increased intermuscular adipocytes due to aging.
The present data showed significant negative correlations between the skin AF on gluteal region and the values of Pj from the GM muscle (Figure 3(a)–(c)). These results suggested that the activities of GM muscle for maximal voluntary contraction effort were reduced in subjects with higher skin AF on the gluteal region. The distributions of skin AF and the values of Pj were significantly different between the young adults and the elderly persons from the viewpoint of three frequency bands (high-frequency band: P1+2, middle-frequency band: P3, and low-frequency band: P4+5, respectively).
In general, muscle size is reduced with aging, and this quantitative loss of muscle affects the generation of force (Distefano & Goodpaster, 2018; Narici et al., 1991). Our results of surface EMG supported that the activities of lower limb muscles with aging were reduced (Figure 3). Elderly persons also exhibit a decrease in MU discharge rate, an increase in variability of MU discharge at high force levels (Roos et al., 1997), and the occurrence of several other age-related adaptive mechanisms which influence fiber sizes and fiber number, as well as enzyme histochemical fiber characteristics (Lexell et al., 1988; Sato et al., 1984). The loss of muscle tissue in elderly people has been attributed to reduced numbers of both slow-twitch fibers and fast-twitch fibers, plus a reduction in the cross-sectional area of single fibers, especially of type II. Older muscle is not only atrophied, but is also slower and tetanizes at lower fusion frequencies (Macaluso & De Vito, 2004). Gerdle et al., (1991) showed that the mean power frequency of the surface EMG was dependent on the fiber-type proportion. A low proportion of type-II fibers might have lower intercepts than those with a high proportion of type-II fibers. These results of the relationship of the frequency aspects of surface EMG activities and the characteristics of muscle fiber types differ from ours. The characteristics of muscle fibers were not different among the participants in this study. Namely, our finding of the values of Pj can be thought to differ depending on the recruitment strategies in each subject group, as mentioned above. More research is needed about the relationship between the skin AF and the accumulation of AGEs into muscle fibers in a future study.
Conclusion
The physiological change of muscle activities of lower limbs due to the aging with nonpathologic states was experimentally evaluated as the relationship between the skin AF and mean wavelet coefficient of surface EMG. The value of skin AF at the gluteal region was associated negatively with the values of P1+2, P3, and P4+5 at GL muscle. The higher accumulation of advanced glycation end-products on the gluteal region suggested the lower muscle activity in aging without disease and disability. The increase of skin AF might be a useful index for estimation of muscle hypofunction and/or cooperative activity as the recruitment strategies of elderly persons with nonpahtologic aging.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number 20H01709.