Abstract
Introduction
Polypharmacy is common in the frail nursing home population and associated with an increased risk of adverse events, unplanned hospitalizations, and increased all-cause mortality. Deprescribing using a deprescribing algorithm might reduce unnecessary polypharmacy. This exploratory study was performed to determine the effect of this implicit deprescribing algorithm in deprescribing statins and proton pump inhibitors (PPIs) in nursing home residents.
Method
Multicenter, longitudinal, single-arm exploratory study. All participants received the same deprescribing intervention to identify and deprescribe potentially inappropriate statins and/or PPIs. Residents across 10 nursing homes in the Netherlands were included if they used a statin and/or PPI. Residents in hospices or short-stay wards were excluded. The intervention involved a deprescribing algorithm in which nursing home physicians identified and, if possible, deprescribed potentially inappropriate statins and/or PPIs.
Results
Sixty-seven residents participated in the study. At 3 months, deprescribing was successful in 52% of the residents. Six months after the intervention, all these residents still had their medication sustainably deprescribed.
Conclusion
Based on this study, deprescribing statins and PPIs using an implicit deprescribing algorithm is possible in a considerable number of nursing home residents.
Introduction
Polypharmacy is common in the frail nursing home population and associated with an increased risk of adverse drug events, unplanned hospitalizations, reduced functional capacity, and increased all-cause mortality (Page et al., 2016). With aging, the prevalence of chronic diseases and the number of potentially inappropriate medications taken increases (Morin et al., 2016). At the same time, medication is often less safe at higher age, with more side effects, drug interactions, and complications (Schenker et al., 2019). Nursing home residents, in particular, represent a group of very frail and disabled older persons, who use high numbers of medication, while extra vulnerable for negative consequences of polypharmacy (Schenker et al., 2019).
A possible solution for reducing inappropriate polypharmacy is called “deprescribing”: a planned and supervised process of dose reduction or stopping of medication that might be causing harm, or no longer be of benefit (Scott et al., 2015).
Kua et al. (2021) determined the effect of a deprescribing intervention by means of a stepped-wedge cluster-randomized controlled trial and showed that deprescribing was associated with reductions in mortality and number of hospitalized residents in nursing homes (Kua et al., 2021).
A systematic review performed by Dills et al. (2018) of 58 randomized controlled trials showed that deprescribing may be successful and effective in select classes of drugs. In contrast to the findings of Kua et al., Dills et al. suggested that deprescribing did not lead to expected outcomes such as lower hospital admission rate (Dills et al., 2018; Kua et al., 2021).
Nevertheless, it has been difficult for physicians to sustainably stop potentially inappropriate medication, mainly because guidelines often describe when to start medication and not when to stop (Doherty et al., 2020). Also, residents may experience barriers to cease their medication, such as disagreement with appropriateness of deprescribing or fear of deprescribing (Reeve et al., 2013). There is a demand for a more general deprescribing strategy to support nursing home physicians to deprescribe potentially inappropriate medication (Farrell et al., 2018). Medicines such as statins and proton pump inhibitors (PPIs) are widely used in nursing homes (Cook, 2020; Manzi, 2021). Main indications are primary or secondary prevention of respectively cardiovascular risk management (statins) and peptic ulcers, dyspepsia, and prophylaxis of gastrointestinal bleeding (PPIs). The appropriateness of statins and PPIs is often questionable: these medication groups mostly started years before nursing home admission; the indication is often unknown at time of admission and further guidelines have changed during the years.
We developed an evidence-based patient-centered implicit deprescribing algorithm for physicians working in nursing homes. This algorithm provides a systematic approach to review current medication to identify potentially inappropriate medicines to plan a deprescribing regimen, to create partnership with resident and family, and to monitor the sustainability of the deprescribing process (Visser et al., 2019). However, the effectiveness of this implicit deprescribing algorithm to reduce potentially inappropriate medication was not proven yet. Hence, this exploratory study was performed to determine the effect of this implicit deprescribing algorithm in deprescribing statins and proton pump inhibitors (PPIs) in nursing home residents.
Methods
A multicenter, longitudinal, single-arm exploratory study (NCT04204590, ClinicalTrials.gov) was conducted in 13 nursing homes of a nursing homes institution in the south of the Netherlands. All enrolled nursing home residents received the same deprescribing intervention (application of deprescribing algorithm) within 6 weeks after enrollment. The methodology of this trial has been described in more detail in a separate protocol article (Visser et al., 2021).
Study population: Residents of nursing home long-stay wards, using a statin and/or a PPI, were recruited. In the Netherlands, admission to nursing homes is only possible if the resident has permanent disabilities and comorbidities and consequently is care dependent for ADL and iADL and needs 24-hour long-term institutional care. Residents in hospices or short-stay (rehab) wards were excluded. Written informed consent was obtained according to ethical guidelines. For residents unable to provide written informed consent to participate, consent was sought from the person’s legal representative.
Intervention: The deprescribing intervention consisted of five steps including (1) reviewing the current medication; (2) identifying potentially inappropriate statins and/or PPIs, these were not deprescribed immediately; (3) planning a deprescribing regimen to present to the resident or his/her legal representative; (4) creating partnership with resident and family; and (5) actual deprescribing and monitoring the sustainability of the deprescribing process. The deprescribing intervention was implemented by training all nursing home physicians to execute the intervention in three workshops led by the researchers and a pharmacist. In the last workshop, the reliability was assessed with success. The actual intervention took place during the regular 6 weeks medication reviews conducted by the nursing home physicians. As part of the intervention, the nursing home physician discussed the advice with the resident and/or family. All nursing home physicians were aware of possible approaches to increase residents’ willingness to deprescribe. Direct care staff was informed about the result of the intervention and involved in monitoring the nursing home resident (for more detailed information about the intervention, see Visser et al. (2019).
Data collection: Data were collected at baseline as well as at three and 6 months after the deprescribing intervention. For each resident, demographic data (including gender and age), main diagnosis underlying reason for admission to the nursing home, which ward (somatic or psychogeriatric department), medication used since the day of enrollment (including total number of medicines, defined daily dose (DDD), and indication for starting statins and/or PPIs), hospital admissions since the day of enrollment, and reasons for hospital admission were recorded. Each nursing home physician was asked to indicate whether the deprescribing intervention was sustainably successful and if not, why it was not successful. These quotes were summarized and categorized by two researchers.
Outcome measures: The primary outcome measure was the percentage of enrolled residents whose medication has been successfully deprescribed 3 months after intervention. Deprescribing was defined as successful when stopping or a decrease in daily dose of either statins or PPI had been achieved. Deprescribing was defined as failed when the daily dose of either statins or PPI was the same or increased 3 months after intervention compared to baseline. Secondary measures included the deprescribing success rate 6 months after the intervention and all possible adverse events after deprescribing reported by the nursing home physician as harms due to, for instance, recurring original symptoms. Additionally, an outcome measure related to the physicians was obtained; after three and 6 months, the physicians answered for every participant whether deprescribing was sustainably successful and if not, why.
Sample size: Previous studies suggested that in 65% of residents using a PPI and 90% of residents using a statin, physicians can safely deprescribe this medication (Garfinkel et al., 2015). Assuming a significance level of 5% and a deprescribing success rate of 50% (conservative estimate), a sample size of 97 residents was required to be able to estimate this success rate with enough precision (margin of error of 10%).
Data analysis: Data were expressed as mean with (SD) for numerical variables, while number and percentage of residents were used for categorical variables. The proportion of residents in which medication was successfully deprescribed is presented together with a 95% confidence interval (CI). The differences in resident characteristics between residents who successfully deprescribed versus those who did not were compared using independent-samples t-tests for numerical variables and chi-square or Fisher’s exact tests for categorical variables. Normality assumption was checked using histograms and QQ-plots. For statins and PPI, the longitudinal trend in daily dose was assessed separately using a linear mixed model, with an unstructured covariance structure for repeated measures and a random intercept on nursing home level to account for potential correlation between residents within the same nursing home. Linear mixed model (LMM) analysis was performed on all residents who used PPI or statin at baseline. If a medication was discontinued, the dose was set equal to zero. If a resident died during the study period, the dose was missing for the next time points, but all available data were used in this LMM analysis. No missing outcome data were imputed because a likelihood-based approach was used which assumes missingness at random (MAR), that is, missingness may depend on observed variables. All analyses were performed using SPSS Statistics for Windows (version 26.0, Armonk, N.Y., USA, IBM Corp.). A two-sided p-value ≤ .05 was considered statistically significant.
Results
Group characteristics: From the 200 nursing home residents recruited for the study, in total 67 nursing home residents could be enrolled with a mean age of 83 years (SD 8) of whom 58% were women. On T1 (3 months), 66 residents were left for analyses; one resident moved to another nursing home facility and got lost to follow-up. On T2 (6 months), 57 residents were left for analyses, as nine residents died between 3 and 6 months.
Of the participating residents, 64% lived in psychogeriatric (PG) wards with a principal diagnosis of dementia and therefore all required third-party consent. The remaining residents (36%) lived in a somatic ward, a ward with a focus on cognitively intact residents with mainly physical (somatic) impairments, for instance stroke or Parkinson’s disease. Of all residents, 45 used a PPI only, 6 a statin only, and 15 both a PPI and a statin.
Baseline Characteristics.

One resident was lost to follow-up between baseline and 3 months, so it was unknown whether deprescribing at T1 (3 months) was successful or not.
Fisher’s exact test.
†Total N = 66.
PPI and statin use in successful deprescribing group (N = 34).

Regarding the defined daily dose (DDD) in all residents using a statin, respectively, a PPI at baseline, a statistically significant decrease of both PPI and statin dosage was observed over time. In statins, a DDD reduction of 8.1 mg (95% CI 2.8 mg–13.4 mg; p.005) at T1 and 11.9 mg (95% CI 5.2–18.6; p .001) at T2 was observed. For PPI, the DDD reduction was 11.0 mg (95% CI 7.3 mg–14.7 mg; p < .001) at T1 and 11.4 mg (95% CI 7.6 mg–15.2 mg; p < .001) at T2. Figure 1 shows the mean (SD) defined daily dose of PPI/statin over time. Defined daily dose (mean with standard deviation) of statins respectively PPIs in all statin resp. PPI users at baseline over time.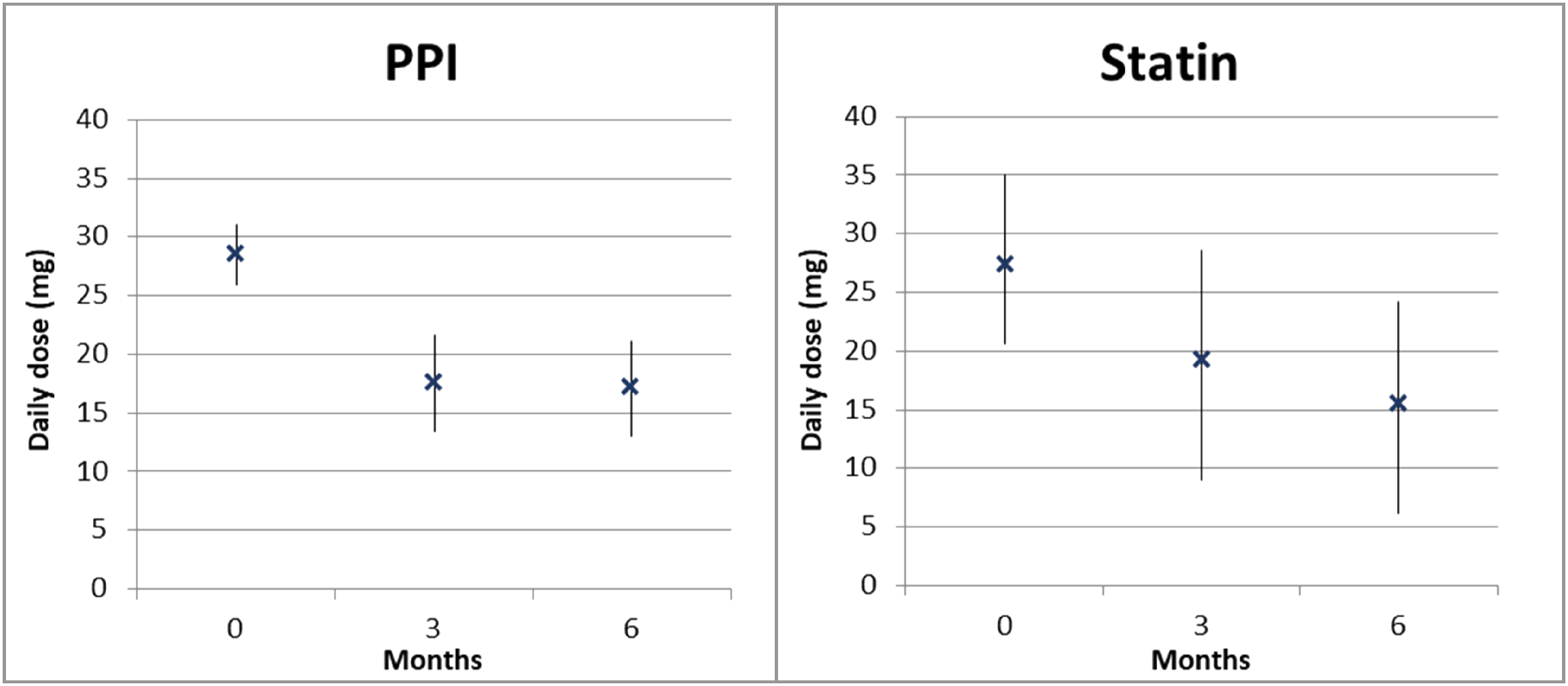
Deprescribing failed in 49% of the residents (N = 32). Participating physicians reported multiple reasons why deprescribing was unsuccessful, such as a residents’ unwillingness to deprescribe (N = 1) or the presence of a valid indication according to the nursing home physician (N = 26), such as prophylaxis of gastrointestinal bleeding. It was also reported that some nursing home physicians thought it was hard to deprescribe in case there used to be a valid indication before, which is not valid anymore at the moment of deprescribing.
Adverse events: In the deprescribing group, two residents experienced withdrawal effects after stopping PPI (=complaints of dyspepsia). These complaints completely vanished after restarting PPI at a lower dose than they initially had. Five residents experienced a hospital admission during the study period. Of these residents, four were in the failed deprescribing group and one was in the successful deprescribing group. According to their nursing home physician, hospital admission was not related to the deprescribing.
During the study, nine residents got lost to follow-up due to death, six residents were in the successful deprescribing group, and three in the failed deprescribing group. According to the nursing home physicians, causes of death were not related to the study intervention. The baseline characteristics of these nine residents were comparable to the other 57 residents, as shown in Supplementary Table 1.
Discussion
This longitudinal, single-arm, exploratory study was designed to determine the effect of an implicit deprescribing algorithm to cease potentially inappropriate medication—in this study statins and PPIs in nursing home residents.
We found that deprescribing statins and PPIs with the implicit deprescribing strategy is successful and sustainable in 52% of the residents. These findings confirm the outcomes of previous studies which stated that deprescribing interventions can be successful in the geriatric population (Garfinkel et al., 2015). A possible explanation for the higher success rate in other studies might be that these studies included less vulnerable participants and used an explicit deprescribing strategy.
It is interesting to note that in 26 of 32 residents in the failed deprescribing group, deprescribing failed because of a valid indication for statin and/or PPI use. It is unknown whether these numbers are an average or not because this has not previously been described.
The present study was not conducted to detect changes in health outcomes, but overall, residents did not experience negative consequences from the intervention such as hospital admission or death. This finding is also consistent with recent systematic reviews of deprescribing interventions (Dills et al., 2018).
Deprescribing is indeed possible without obvious negative effects. For the residents, this reduced medication use might be a benefit itself (Kua et al., 2021). Positive outcomes such as a reduction of pill burden, adverse events, hospital admissions, and even deaths might be possible, but further research is needed to confirm this (Kua et al., 2021).
There are some limitations of this study. Due to the explorative nature of the study, no control group was included and a relatively small number of residents, smaller than the calculated sample size, participated. In this study, we experienced that recruiting within nursing homes is difficult because residents are often in the last year(s) of their lives and are more reluctant to join a study. In addition, to reduce any risk of complications, legal representatives often prevent their clients from participating as well
Further research is necessary to test the generalizability of this deprescribing algorithm applied to other medications.
Conclusion
Based on this study, deprescribing statins and PPIs using an implicit deprescribing algorithm is possible in a considerable number of nursing home residents.
Supplemental Material
sj-xlsx-1-ggm-10.1177_23337214211050807 – Supplemental Material for Deprescribing Statins and Proton Pump Inhibitors in Nursing Home Residents; a Pragmatic Exploratory Study
Supplemental Material, sj-xlsx-1-ggm-10.1177_23337214211050807 for Deprescribing Statins and Proton Pump Inhibitors in Nursing Home Residents; a Pragmatic Exploratory Study by Anne G. R. Visser, Jos M. G. A. Schols, Mathieu A. L. M. Prevoo, Rob Janknegt and Bjorn Winkens in Gerontology and Geriatric Medicine
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the local Medical Ethical Committee (METC-Z number Z2018001).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
