Abstract
Introduction
Dementia is a progressive, neurodegenerative disorder affecting millions of people in the United States (Goodman et al., 2017; Plassman et al., 2007). The most common subtypes are Alzheimer’s disease, vascular dementia, dementia with Lewy bodies, frontotemporal dementia, and dementia associated with Parkinson’s disease. Although the disorder is characterized by cognitive decline, nearly all patients (>90%) experience neuropsychiatric symptoms (NPS) at some point during the disease course (Preuss et al., 2016; Yunusa et al., 2019). NPS may manifest as psychosis (delusions or hallucinations), depression, anxiety, and agitation or aggression (Press & Alexander, 2018; Preuss et al., 2016; Steinberg & Lyketsos, 2012). Development of NPS is associated with increased institutionalization, hospitalization, morbidity, and mortality (Peters et al., 2015; Porter et al., 2016; Preuss et al., 2016).
Patients with dementia commonly experience dementia-related hallucinations and delusions, with prevalence varying across subtypes and frequency increasing as dementia progresses (Cummings et al., 2018; Peters et al., 2015; Selbaek et al., 2014). No US Food and Drug Administration (FDA)-approved pharmacologic treatment options for the hallucinations and delusions associated with dementia-related psychosis are currently available; therefore, patients are often prescribed one or more off-label medications, including antipsychotics (APs) (Reus et al., 2016). Commonly used APs are often ineffective or provide only a small benefit for patients who do respond to treatment (Reus et al., 2016; Schneider et al., 2006b; Yunusa et al., 2019). They can also worsen cognitive impairment (Schneider et al., 2006a) and increase the risk of cardiovascular events and mortality (1.6- to 1.7-fold) in older adults with dementia (American Geriatrics Society Beers Criteria Update Expert Panel, 2019; Reus et al., 2016; Schneider et al., 2005; US Food and Drug Administration, 2008; US Food and Drug Administration Center for Drug Evaluation and Research, 2005). Metabolic effects, sedation/fatigue, anticholinergic effects, postural hypotension, falls and fractures, and extrapyramidal symptoms may be associated with the use of APs in patients with dementia (Fraser et al., 2015; Reus et al., 2016; Reynolds, 2011; Schneider et al., 2006a; Trigoboff et al., 2013).
All APs carry a boxed warning regarding increased risk of mortality in elderly patients with dementia-related psychosis (US Food and Drug Administration, 2008; US Food and Drug Administration Center for Drug Evaluation and Research, 2005). The American Geriatrics Society recommends that APs (typical and atypical) be avoided in this population when possible (American Geriatrics Society Beers Criteria Update Expert Panel, 2019). The various risks associated with the use of APs for NPS in older adult patients with dementia have, in general, reserved their use to severe cases where nonpharmacological options have failed or are not possible, or for cases where the patient is threatening substantial harm to him or herself or others (American Geriatrics Society Beers Criteria Update Expert Panel, 2019; Reus et al., 2016). The US Centers for Medicare & Medicaid Services (CMS) has aimed to reduce the use of APs in long-term care (LTC) facilities, and guidelines require that residents only receive APs when necessary (CMS, 2017). When APs are clinically necessary, attempts at gradual dose reductions and behavioral intervention must be made and accrediting bodies expect compliance with the guidelines (CMS, 2016; Mathew et al., 2016). Potential alternatives include antiepileptics (e.g., valproic acid) or dextromethorphan/quinidine. However, supportive evidence for the efficacy of antiepileptics in this context is lacking and dextromethorphan/quinidine is approved specifically for pseudobulbar affect. Both of these options are also associated with their own risks, such as hepatotoxicity, blood disorders, somnolence, increased falls, and drug–drug interactions (American Geriatrics Society Beers Criteria Update Expert Panel, 2019; Depakene [Prescribing Information], 2013; Nuedexta [Prescribing Information], 2019; Sink et al., 2005).
There is a paucity of literature evaluating current medication use in residents with dementia-related psychosis in the LTC setting. This analysis of real-world data was conducted to better understand current medication treatment patterns and factors related to medication treatment changes in residents diagnosed with dementia-related psychosis in the LTC setting (i.e., skilled nursing facilities, nursing homes, LTC facilities).
Methods
Study Design and Data Source
This retrospective database cohort study was conducted using the national PharMerica® database, which contains demographic, diagnostic, and prescription information from residents within adult LTC facilities in the United States. The pharmacy claims data were used to identify all prescription information, which was available from approximately 15% of nursing homes across the United States. Additionally, the electronic medical records were used from the Minimum Data Set (MDS) where diagnosis codes were identified. The MDS includes information on entry, discharge, and periodic clinical screening/assessment for residents in all Medicare and Medicaid certified nursing homes. This study did not involve individually identifiable data; institutional review board approval and patient consent was not required.
Study Population
Residents with dementia in LTC settings from January 1, 2013, through May 30, 2017 were included in the study. Dementia was defined as ≥2 diagnostic codes for dementia at least 30 days apart, or one dementia diagnostic code plus one prescription for dementia therapy (i.e., rivastigmine, donepezil, memantine, galantamine, tacrine) during the study period. The following subtypes of dementia were included: Alzheimer’s disease, vascular dementia, dementia with Lewy bodies, Parkinson’s disease dementia, frontotemporal dementia, and Others, Not Specified. A full list of diagnostic codes used in this study is included in Supplemental File 1. Patients were excluded from the dementia-only cohort if they had a history of psychosis diagnosis and prescription for dementia-related psychosis therapy (i.e., AP, divalproex or valproic acid, dextromethorphan/quinidine) during the 12 months before or after the dementia date. The dementia-only index date was defined as the date of first dementia diagnosis during the study time period.
Dementia-related psychosis was defined as ≥2 prescriptions for dementia-related psychosis treatment ≤45 days apart with ≥45 days’ supply, or ≥2 diagnoses of psychosis (i.e., hallucinations, delusions, and other psychosis diagnostic codes) ≥30 days apart, or 1 diagnosis of psychosis and ≥1 prescription for dementia-related psychosis treatment during the study period. Full lists of diagnostic codes and prescriptions included in this study are shown in Supplemental File 2. Divalproex and valproic acid were combined for all analyses as they result in the same active moiety. The dementia-related psychosis index date was the date of the first psychosis diagnosis or dementia-related psychosis prescription fill, whichever came first. Patients did not have a history of psychosis during the 12 months prior.
Residents were excluded from the study if their record was missing a date of birth or included <12 months of follow-up after the index date (up to May 30, 2018). Residents were excluded from the dementia-related psychosis category if they had a diagnosis for a mental health condition that might manifest as psychosis (e.g., schizophrenia, bipolar disorder) or affect cognition (e.g., acute stroke), or if the diagnosis of psychosis or ≥2 pharmacy claims for dementia-related psychosis therapy preceded the initial dementia diagnosis. Patients with a past diagnosis of pseudobulbar affect, seizure, or epilepsy were also excluded in an attempt to identify only prescriptions intended to treat psychosis.
In order to identify and characterize dementia-related psychosis residents and to avoid also evaluating those with dementia-related agitation and aggression (in the absence of psychosis), residents with ≥2 diagnoses of agitation or aggression ≥30 days apart and no psychosis diagnosis or dementia-related psychosis therapy during the prior 12 months, were excluded from the analysis.
Study Outcomes
Baseline patient demographics and characteristics, concomitant comorbidities, and medications for dementia-related psychosis residents and dementia-only residents were identified and assessed during the 12 months prior to the index date. Treatment patterns for dementia-related psychosis patients were assessed during the 12 months post-index and were defined as treatment continuation, discontinuation, switching, or sporadic usage by evaluating a 45-day period between prescription fills (date of last index fill + days’ supply + 45 days). Continuation was defined as use of the same index therapy during the post-index period. Discontinuation was defined as no subsequent prescription fill during the 45-day period or at any time during the 12 months after the index date. Switching was identified by a prescription for a different or additional dementia-related psychosis therapy within the 45-day gap. Sporadic use was defined as any prescription of 30-day supply after the 45-day gap with an inconsistency in prescribing behavior.
Time to treatment change was calculated for residents who switched or discontinued the initial dementia-related psychosis therapy; the end date was calculated as the last prescription date plus the days’ supply. Doses for dementia-related psychosis therapy (APs, divalproex, and dextromethorphan/quinidine) were calculated in milligrams; mean daily dose was calculated as the sum of daily doses divided by the number of residents prescribed the respective therapy. Concomitant medications were evaluated descriptively during the 12 months pre- and post-index.
Statistical Analysis
Descriptive statistics were used to compare baseline characteristics and time with treatment change means, standard deviation (SD), and percentages. Differences between categorical variables were analyzed using chi-squared tests and differences between the means of continuous variables were analyzed using the Student’s t-test. A backward selection, multivariable, logistic regression model was used to identify factors associated with treatment change (i.e., switched, continued, or sporadic use versus continued antipsychotic therapy). First, we combined the variables that were used for similar indications, re-labeled them to remove duplicative variables, and placed all new variables into the logistic regression model. The probability of chi-square for each variable was evaluated. If the probability was greater than 0.35, the variable was removed from the model in a stepwise backward selection process. The model was run until there were no variables left with a probability >0.35. This method is robust and provided a good fit model with c-statistic. The model controlled for patient age, sex, comorbidities, and concomitant medications. P values <.05 were considered statistically significant. Statistical analyses were conducted using SAS version 9.4.
Results
Study Population
The study included 23,353 residents with dementia. Of these, 11,432 (49%) had dementia-only and 11,921 (51%) were identifed with dementia-related psychosis (Figure 1). Among both groups, most were female and residents with dementia-related psychosis were a mean 3.5 years younger than those with dementia-only (Table 1). Patients with dementia-related psychosis had higher rates of comorbidities than patients with dementia only (p < .05 for all comparisons; Table 1). Significantly more residents with dementia-related psychosis were prescribed dementia therapy and, despite a higher proportion of depression diagnoses compared with the dementia-only group, fewer residents with dementia-related psychosis were receiving selective serotonin reuptake inhibitors (SSRIs) or other antidepressants during baseline (p < .001 for all comparisons; Table 1).
Patient Demographics and Baseline Characteristics.

COPD = chronic obstructive pulmonary disease; HMO = health maintenance organization; SD = standard deviation; SSRI = selective serotonin reuptake inhibitor; UTI = urinary tract infection.
Except where specified, values are n (%).
P < .05, dementia-only versus dementia-related psychosis.
P < .05, dementia-only versus dementia-related psychosis for >40 to 55 years, >55 to 65 years; no significant difference between dementia-only versus dementia-related psychosis for ≤40 years.
Includes commercial, HMO/managed care, or Veterans Affairs insurance.

Patient cohort diagram.
Treatment Patterns
Among residents with dementia-related psychosis, 11,246 (94.3%) were prescribed ≥1 index medication to treat psychosis, including 77.3% who received ≥1 typical or atypical AP (Figure 2). Quetiapine was the most frequently prescribed index therapy (31.1%), with doses >100 and ≤100 mg prescribed for 60.7% and 39.3% of residents, respectively and a mean ± SD daily dose of 156.6 ± 71.2 mg (Table 2). Other prescribed therapies included risperidone, divalproex, olanzapine, haloperidol, and aripiprazole (Figure 2).
Treatment Patterns among Residents with Dementia-Related Psychosis.
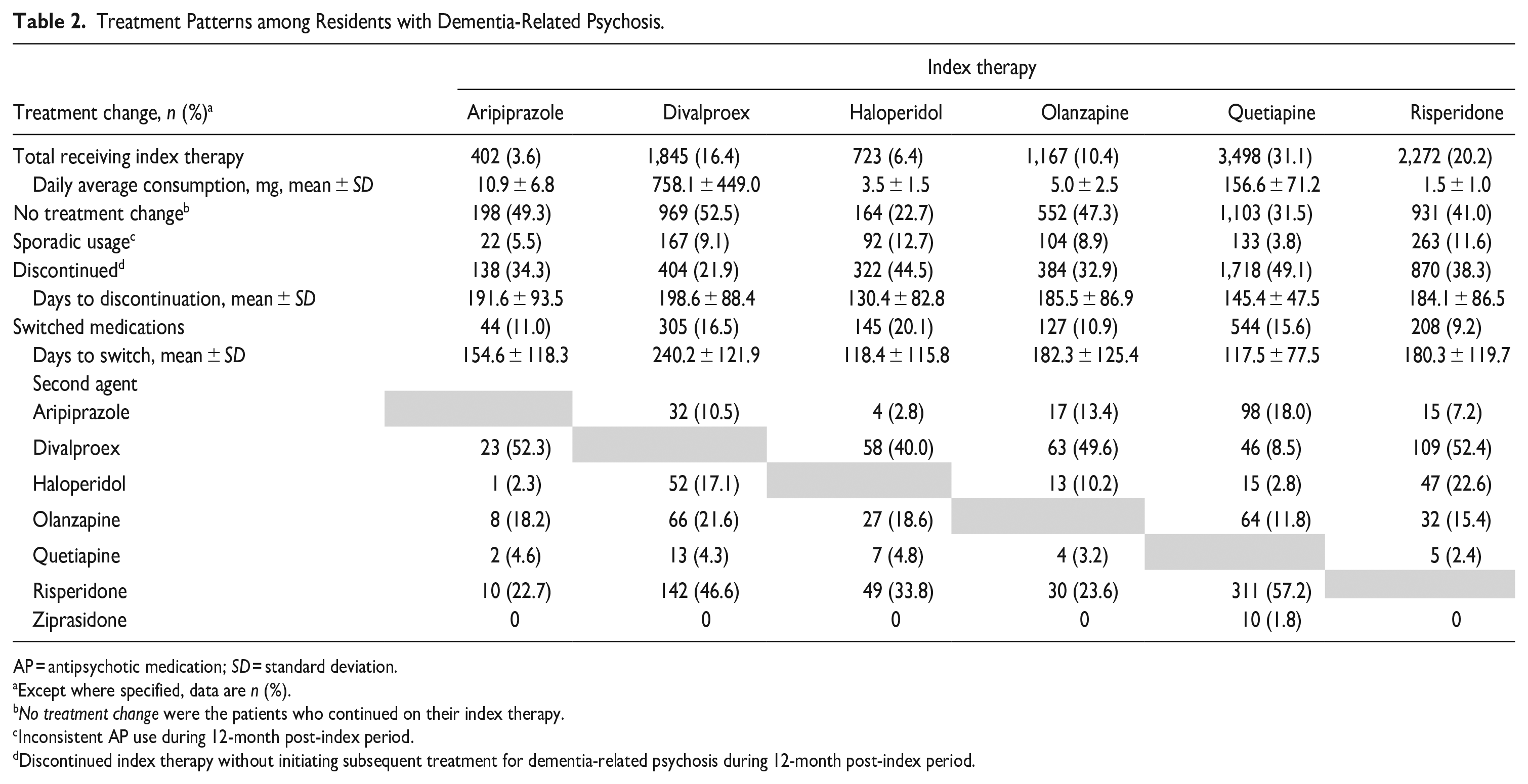
AP = antipsychotic medication; SD = standard deviation.
Except where specified, data are n (%).
No treatment change were the patients who continued on their index therapy.
Inconsistent AP use during 12-month post-index period.
Discontinued index therapy without initiating subsequent treatment for dementia-related psychosis during 12-month post-index period.
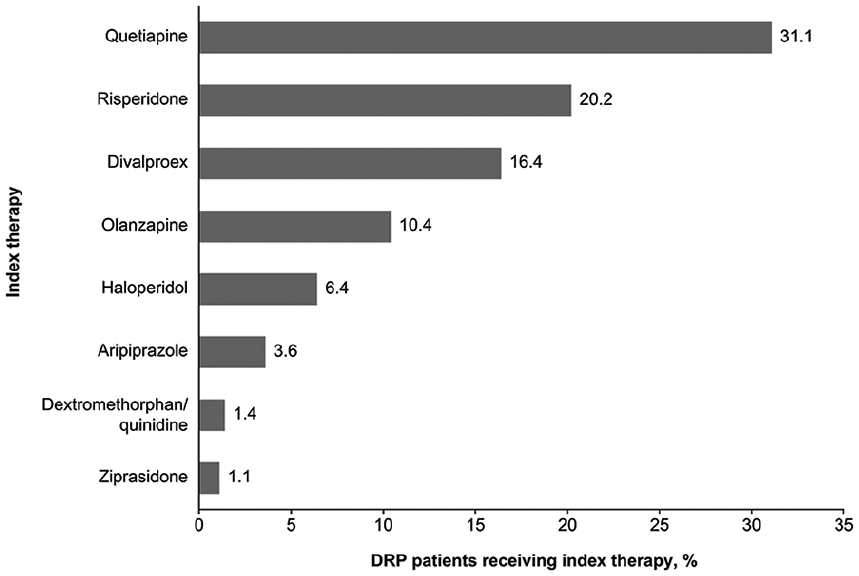
Index therapy prescriptions among residents with dementia-related psychosis.
Treatment change was assessed for residents who received aripiprazole, divalproex, haloperidol, olanzapine, quetiapine, and risperidone. These therapies were selected because they were used by ≥3% of patients. During the 12 months post-index, 39.5% of residents in the dementia-related psychosis group continued on the index therapy, whereas 38.7% discontinued treatment, 13.9% switched treatments, and 7.9% had sporadic use (Table 2). Divalproex had the highest rate of continuation (52.5%) and haloperidol had the lowest rate (22.7%). Mean time to discontinuation and to switch were longest for divalproex (198.6 and 240.2 days, respectively) and were shortest for haloperidol (130.4 and 118.4 days, respectively) and quetiapine (145.4 and 117.5 days, respectively). Except for residents who received quetiapine, all other index therapies were most likely to be switched to divalproex as the second agent. Residents who first received quetiapine were most likely to switch to risperidone. Residents who discontinued index therapy and did not switch to another medication received dementia-related psychosis treatment for a mean of 130 to 199 days.
Concomitant Medications during the Post-Index Period
Concomitant post-index therapies for the dementia-only and dementia-related psychosis groups included SSRIs (59.9% and 60.1%, respectively) and other antidepressants (58.1% and 66.7%), benzodiazepines (47.7% and 58.1%), and antiepileptics or lithium (47.1% and 38.3%) (Supplemental Table 1). In the dementia-related psychosis group, prescription rates for all of these therapies increased significantly (p < .001) from pre- to post-index. Prescriptions for antidepressants other than SSRIs increased most significantly from pre- to post-index. No significant change in concomitant medication was observed in the dementia-only group between the pre- and post-index periods.
Factors Associated with Treatment Change
Baseline patient and clinical characteristics from Table 1 were added into the multivarate logistic model to evaluate which factors would have an association with any treatment change. The model identified the following factors associated with treatment change: age ≥65 years, Medicare insurance, and comorbid conditions (or prescriptions for associated medications) of anemia, coronary heart disease, diabetes, falls, depression, hypertension, or hyperlipidemia (Table 3).
Multivariate Logistic Regression Analysis of Factors Associated with Changes to Dementia-Related Psychosis Index Therapy Versus Treatment Persistence.
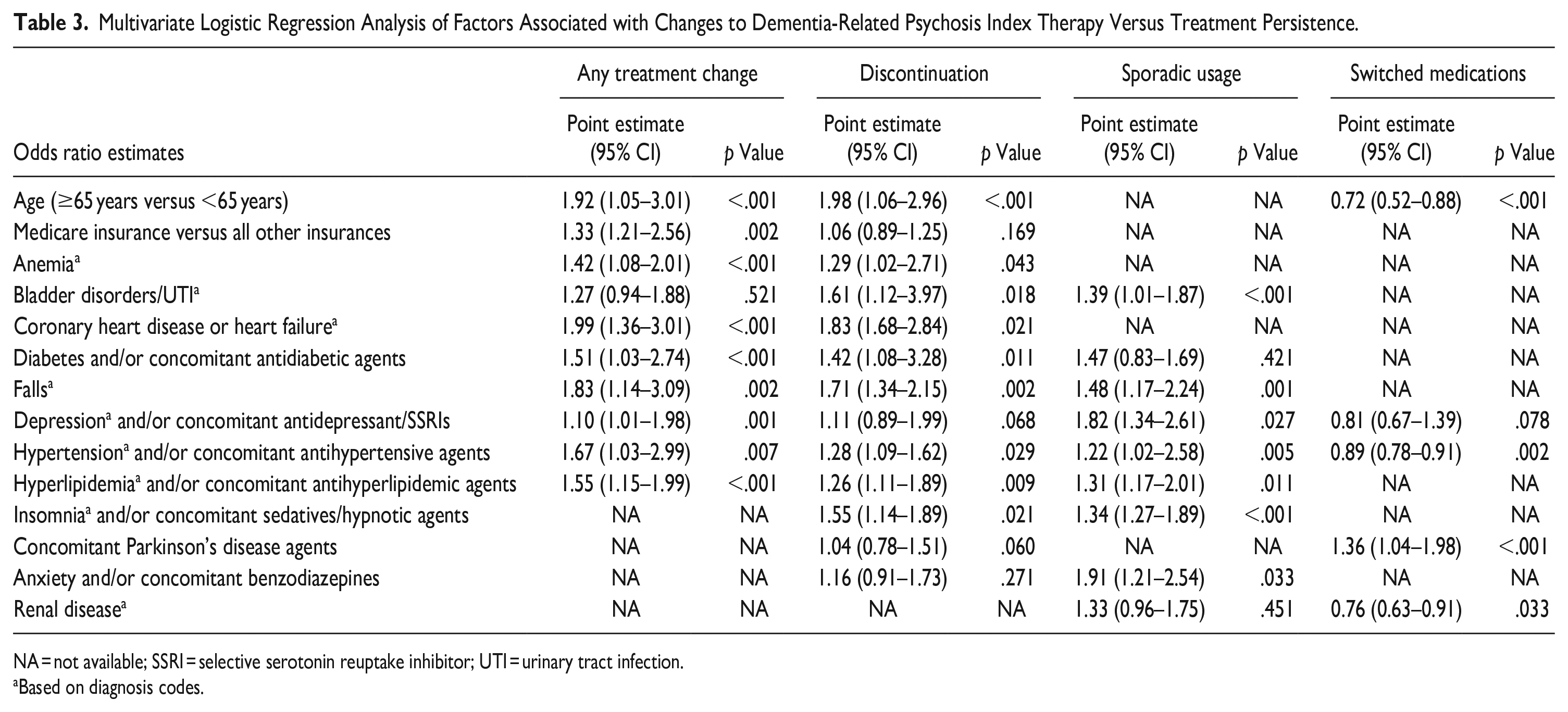
NA = not available; SSRI = selective serotonin reuptake inhibitor; UTI = urinary tract infection.
Based on diagnosis codes.
Discussion
This analysis of real-world data for residents of LTC facilities with dementia showed that 94% of residents with dementia-related psychosis were receiving treatments that are not approved by the FDA for this use, most commonly APs. APs are known to increase the risk of cardiovascular events, infection, and death among older adults with dementia and have generally demonstrated, at best, only modest efficacy in treating dementia-related hallucinations and delusions (Schneider et al., 2005; Schneider et al., 2006b; Tampi et al., 2016). Despite these negative risks and not being approved by the FDA, our analysis shows that these continue to be prescribed as the primary treatment approach for patients with dementia-related psychosis. The high rates of antipsychotic use identified in this study of LTC patients are consistent with prior research where dementia patients who live in skilled nursing facilities/LTC have significantly greater rates of AP prescriptions when compared with patients living in the community (Kuroda et al., 2019). Practice guidelines recommend the use of APs in this population only in the case of severe, dangerous, and/or distressful symptoms (Reus et al., 2016), yet our findings suggest that nearly all residents with a dementia-related psychosis diagnosis received treatment at some point during the 12-month post-index period.
During the 12-month post-index period, treatment changes were common, which suggests that initial medications either did not adequately treat symptoms, were associated with unacceptable adverse effects, or that regulatory requirements and/or access restrictions made their continued use no longer possible. Although reasons for treatment change were not captured in this study, high rates of discontinuation, switching, and sporadic usage may reflect attempts to minimize harms (i.e., known safety concerns such as increased risk for cardiovascular events, infection, and mortality [Schneider et al., 2005; Tampi et al., 2016]. Many patients may have tapered or discontinued treatment once symptoms improved and then restarted only if symptoms worsened again. These treatment changes may be explained by adherence to CMS guidelines, which require frequent reviews of drug regimens during LTC and gradual dose reductions in an effort to discontinue APs (CMS, 2016). Dose reductions must occur within the first year that a resident is admitted on an antipsychotic medication or within the first year that the facility has initiated an antipsychotic medication. The frequent treatment changes may suggest an awareness of the risks associated with AP usage among individuals with dementia-related psychosis and confirm that these medications continue to be prescribed to nearly all residents with dementia-related psychosis despite this risk. This highlights the existence of an unmet need for treatments that effectively and safely improve the symptoms of psychosis.
Quetiapine was the most commonly prescribed single agent (31%); however, it was not the highest in continuation (31.5%). Divalproex was found to be the most continued (52.5%). A majority of patients switched to divalproex, potentially reflecting clinicians’ discomfort with switching to another AP and desire for an alternative to APs. It may also reflect regulatory guidelines to reduce the use of APs. However, the safety profile of these non-AP medications is not well established in older adults with dementia. Interestingly, patients on quetiapine and divalproex switched to risperdone (57% and 47%), an agent that may be less sedating (Baillon et al., 2018; Yunusa et al., 2019).
The prevalence of comorbidities and concomitant medications among LTC residents with dementia-related psychosis further complicates the considerations of potential risks of these therapies. Further examining patients who were initiated on quetiapine, the mean daily dose was 157 mg and 40% of residents received ≤ 100 mg/day. Many residents may have been receiving quetiapine for its sedative effects, which are observed predominantly at low doses (Blaszczyk et al., 2015; Miller, 2004).
Baseline factors of older age, Medicare insurance, and high rates of comorbid conditions, were associated with treatment changes. This supports the conclusion that physicians attempt to limit exposure to APs in LTC residents with dementia-related psychosis who are older or whose risk of cardiovascular events, infection, or death is further increased by comorbidities. It is possible that, in line with American Psychiatric Association recommendations (Reus et al., 2016) some residents/caregivers and physicians accepted a certain degree of symptoms before deciding to initiate pharmacological treatment. Thus, some residents categorized as “dementia-only” may have experienced hallucinations or delusions without receiving a diagnosis or prescription. LTC or nursing home facilities dispense medications on a daily basis, which increases the likelihood that residents receive their prescribed treatments; however, there could be some patients who refuse their medicine.
In this study, psychosis diagnosis was associated with a substantial increase of benzodiazepines and antidepressants during the 12 months post-index. This may suggest that psychosis onset was accompanied by occurrence of other conditions or worsening of current comorbid conditions, or reflect an attempt to control unwanted behaviors in the dementia-related psychosis LTC residents. This raises concerns about polypharmacy in these older patients. Research was published evaluating the impact of benzodiazepines and antidepressants on the risk of death in patients with dementia initiating antipsychotic drug treatment. Even though the results of this research cannot prove causality, it was found that the risk of death in combination with the use of benzodiazepines, antidepressants, and antipsychotics was two-fold versus antipsychotics alone (Norgaard et al., 2020).
There are some important notes to highlight when interpreting these results. First, patients with dementia-related psychosis were identified through claims data. Particularly with no standardized diagnosis code or FDA-approved therapy for dementia-related psychosis, some patients may not have been identified. In addition, the pre-index period differed among residents with dementia-only and the duration of dementia was not known (i.e., the 12 months preceding dementia diagnosis). Between-group comparisons must be interpreted with caution because factors evaluated during the pre-index period do not necessarily indicate differences among individuals who do or do not develop dementia-related psychosis. It is possible that these differences may demonstrate progression of a disease that is frequently accompanied (and may be worsened) by certain comorbidities.
Residents with dementia-related agitation and aggression were excluded from the study as a way to better identify and characterize residents with dementia-related psychosis and to avoid evaluating those with dementia-related agitation and aggression in the absence of psychosis. The exclusion of these residents strengthens the study by helping to ensure that the findings are linked to psychosis and not to the associated aggression and agitation. As residents diagnosed with major depressive disorder were not excluded from the analysis, it is possible that some AP use can be attributed to the treatment of depression. In addition, some residents may have been receiving cholinesterase inhibitors to assist in the management of psychosis symptoms.
Although all analyses in this study are descriptive and hypothesis-generating, and data were collected from a medical claims database designed for operational purposes rather than to answer research questions, the results assist in understanding the frequency of use and the factors driving changes in medications used to treat dementia-related psychosis in LTC facilities. This is the first study to use a national US LTC pharmacy database linked with government electronic medical records that provides real-world data on the comorbidity burden for residents with dementia-related psychosis. Further analyses are ongoing to better understand the reasons why patients discontinued, switched, or had sporadic use with their therapies, using chart notes and other methods.
Conclusion
This analysis of real-world data suggests that nearly all residents with dementia-related psychosis are prescribed off-label medications associated with small benefit and known safety risks for the treatment of dementia-related psychosis. Having no approved therapy to treat dementia-related psychosis, residents discontinue, switch, or sporadically use these therapies off-label due to tolerability issues, side effects, or a lack of efficacy. Further studies need to be completed to address the reasons why patients have high rates of pharmacologic treatment changes. The higher rates of comorbidities among dementia-related psychosis versus dementia-only residents further increase the safety risk associated with dementia-related psychosis treatments. These data highlight the strong unmet need for medications with demonstrated efficacy and long-term safety in patients with dementia-related psychosis.
Supplemental Material
sj-pdf-1-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-1-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-2-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-2-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-3-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-3-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-4-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-4-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-5-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-5-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-6-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-6-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-7-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-7-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Supplemental Material
sj-pdf-8-ggm-10.1177_23337214211016565 – Supplemental material for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis
Supplemental material, sj-pdf-8-ggm-10.1177_23337214211016565 for Real-World Medication Treatment Patterns for Long-Term Care Residents with Dementia-Related Psychosis by Nazia Rashid, Victor Abler, Sherry Andes and Leslie Citrome in Gerontology and Geriatric Medicine
Footnotes
Acknowledgements
Nicole Fowler, PhD, (Ashfield Healthcare Communications, Middletown, CT) provided writing support based on input from authors, and Dena McWain (Ashfield Healthcare Communications) copyedited and styled the manuscript per journal requirements.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.R., S.A., and V.A. are employees of Acadia Pharmaceuticals Inc. L.C. in the past 12 months, served as a consultant for AbbVie, Acadia, Alkermes, Allergan, Avanir, Axsome, BioXcel, Cadent Therapeutics, Eisai, Impel, Intra-Cellular Therapies, Janssen, Karuna, Lundbeck, Luye, Merck, Neurocrine, Noven, Osmotica, Otsuka, Sage, Shire, Sunovion, Takeda, Teva; speaker for AbbVie, Acadia, Alkermes, Allergan, Eisai, Intra-Cellular Therapies, Janssen, Lundbeck, Merck, Neurocrine, Noven, Otsuka, Sage, Shire, Sunovion, Takeda, Teva; owns stocks (small number of shares of common stock) in Bristol-Myers Squibb, Eli Lilly, J & J, Merck, Pfizer purchased >10 years ago; and has received royalties from Wiley (Editor-in-Chief, International Journal of Clinical Practice, through end of 2019), UpToDate (reviewer), Springer Healthcare (book), Elsevier (Topic Editor, Psychiatry, Clinical Therapeutics).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Acadia Pharmaceuticals Inc.
Data Availability Statement
Data available on request from authors: The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
