Abstract
Cognitive testing situations can be stressful for both younger and older adults, but threats of cognitive evaluation may be particularly salient among anxious older individuals as they tend to be more concerned than younger adults about their cognitive abilities and age-related cognitive decline. We examined age-related differences in the effect of anxiety on cortisol responses during cognitive testing in a sample of 27 younger (M = 19.8) and 29 older (M = 71.2) adults. Older adults with higher anxiety also had higher during-task cortisol (suggesting higher reactivity to testing) than older adults with lower anxiety and young adults. There was no effect of anxiety on cortisol for younger adults. Simultaneously examining subjective (state anxiety) and physiological (cortisol response) indicators of threat during cognitive testing appears to be especially important for older adults with higher state anxiety. The results are important for understanding cortisol reactivity, particularly in older adults. Researchers who administer cognitive tests to older adults and clinicians who work with older adults with cognitive concerns and/or anxiety may want to consider how they present their material.
Cognitive evaluation is often associated with elevated arousal and stress response, regardless of age. Younger adults (Seeman et al., 2001) as well as middle-aged and older adults (Armstrong et al., 2017; Kelly et al., 1997; Neupert et al., 2006; Wetherell et al., 2002) find cognitive testing situations stressful. However, threat of cognitive evaluation may be particularly salient among older individuals as they tend to be more concerned than younger adults about their cognitive abilities (Hess, 2014) and age-related cognitive decline (Lachman, 2004). Older individuals are also more likely to attribute memory issues to uncontrollable factors, such as innate ability (Blatt-Eisengart & Lachman, 2004), and may overestimate age-related decline in performance measures (Schaie et al., 1994).
Studies on stereotype threat have also found that older adults are more likely than the young to be primed by cognitive-related tasks (Armstrong et al., 2017; Barber et al., 2015), revealing potential vulnerabilities to age-based stereotypes common in Western societies (Horton et al., 2008). Given the importance of perceived self-efficacy as well as beliefs about performance on how people respond to testing (Bandura, 1989; Jones et al., 2009), there is reason to believe that subjective and physiological measures of the stress response associated with cognitive evaluation will be different for older compared with younger adults.
Research studies examining the stress response to perceived threats (e.g., cognitive testing) often include subjective measures of emotion, state anxiety (an “in-the-moment” measure of anxiety as described subjectively by an individual), as well as physiological reactivity, such as changes in cortisol (a glucocorticoid stress hormone). Although previous research has investigated the effects of state anxiety and cortisol reactivity on performance (e.g., S. D. Hood et al., 2006; Robinson et al., 2008), few studies have examined age-related differences in arousal due to the threat of cognitive evaluation.
The hypothalamic–pituitary–adrenal (HPA) axis is an important endocrine system that responds to stress with a release of cortisol. This system is activated when a person is challenged, either mentally or physically. Elevated cortisol is a common consequence of written (Lovallo et al., 1986; Preuß et al., 2010) and oral (Herbert et al., 1986; Lacey et al., 2000; Merz & Wolf, 2015; Preuß et al., 2010; Schoofs et al., 2008) examinations in academic settings and is also a common consequence of cognitive testing within laboratory settings (Leininger & Skeel, 2012). Because of the known deleterious effects of cortisol, such as inflammation and brain volume decreases, in both the short term (Hidalgo et al., 2014; Pulopulos et al., 2015) and the long term (Ouanes et al., 2017; Sudheimer et al., 2014), understanding factors that lead to an elevation of this stress hormone is an important task.
Research has also demonstrated that older adults tend to be more reactive in terms of cortisol secretion (Otte et al., 2005). For example, Gotthardt et al. (1995) demonstrated that a cognitive challenge resulted in an increase in stress-induced hormonal secretion that was more pronounced in older adults than in younger adults. Researchers have also previously shown that older adults had higher cortisol reactivity to cognitive challenges (Otte et al., 2005) as well as longer recoveries (i.e., slower return to baseline cortisol) after those challenges (Seeman & Robbins, 1994). Neupert et al. (2006) found a positive relationship between older age and cortisol reactivity to cognitive testing such that older adults’ cortisol secretions increased throughout cognitive tasks, whereas younger adults’ cortisol secretions decreased. It has been suggested that older adults tend to be more invested in cognitive tasks due to a heightened sense of relevance or a social-evaluative threat (Dickerson & Kemeny, 2004; Hess, 2014). Relatedly, in a study focused specifically on anxious older adults, Kelly et al. (1997) found that cortisol reactivity was negatively associated with intellectual self-efficacy when completing a fluid intelligence task. Such findings could suggest that although older adults have an increased physiological stress response relative to younger adults, additional individual differences, such as anxiety, may also play a key role in cognitive evaluation–related arousal (Dickerson & Kemeny, 2004).
State anxiety is an “in-the-moment” measure of anxiety as described subjectively by an individual. The deleterious effects of anxiety on general functioning in adults are well established inside and outside of the laboratory. Although previous research has investigated the effects of state anxiety and cortisol reactivity on performance (e.g., S. D. Hood et al., 2006; Robinson et al., 2008), few studies have examined whether there are age-related differences in anxiety and arousal response due to the experience of cognitive evaluation.
Research findings have been mixed regarding the relationship between self-report and physiological measures of arousal (Avero & Calvo, 1999). In an academic setting with younger adults, Merz and Wolf (2015) found a positive relationship between state anxiety and cortisol reactivity. In the laboratory, more inconsistent results have been observed, including experiments which did not find associations between state anxiety and cortisol reactivity (Childs et al., 2014; Leininger & Skeel, 2012; Oswald et al., 2006). All of these studies, however, comprised exclusively younger adults and/or did not examine potential age differences in response. To the authors’ knowledge, this study will be the first to examine the role of state anxiety on cortisol response to cognitive evaluation in a sample that includes both younger and older adults. Gaining a better understanding of how cognitive evaluation influences stress response in older adults is important from both a research and a clinical perspective. For instance, if anxious older adults have a stress response to cognitive testing, clinicians may want to change the way they introduce tests to these individuals or may want to work with that anxiety in a therapeutic modality, such as cognitive behavioral therapy. In addition, as mentioned previously, the known negative effects of cortisol elevation on physical and cognitive functioning, particularly in older adults, make examining factors related to cortisol elevation an important one.
This Study
This study measured physiological indicators of arousal before, during, and after cognitive testing as well as state anxiety prior to the testing in both younger and older adults. The study was designed to examine age-related differences in the effect of state anxiety on stress responses (as measured by cortisol) to cognitive testing situations. We hypothesized that (a) older participants would have large cortisol increases in response to the cognitive tasks in comparison with the younger adults, (b) participants higher in state anxiety would have larger cortisol increases in response to the cognitive tasks in comparison with participants lower in state anxiety, and (c) older adults higher in state anxiety would show the largest cortisol response to the cognitive tasks. Exploratory analyses were also therefore conducted on cognitive performance to examine any potential relationships with age, cortisol reactivity, and state anxiety.
Method
Participants
Volunteers were recruited from fliers around campus, notices in local senior centers, and fliers mailed to an older adult volunteer database. Interested volunteers called the laboratory and spoke with a research assistant who provided more information and conducted the telephone screening procedure. Potential participants were excluded from the study if they reported one or more of the following: (a) a stroke or serious head injury in the last 5 years, (b) a dementia diagnosis, (c) less than a high school education (to match young and older samples), (d) prescription stimulant or steroid use, or (e) if they made more than two errors on a modified-for-telephone version of the Short Portable Mental Status Questionnaire (SPMSQ; Pfeiffer, 1975). The SPMSQ was used to assess global cognitive functioning as the study was designed for participants without gross cognitive impairment. The medication exclusions were those known to impact resting cortisol levels. These exclusion criteria are commonly used in this type of physiological cognitive aging study. The sample consisted of 27 university undergraduates (nine men, 18 women) and 29 community-dwelling older adults and all participants were compensated US$25. Brandeis University’s institutional review board (IRB) approved this study.
Measures
Self-reported anxiety
A 10-item version of the state measure of the State-Trait Anxiety Inventory (Spielberger, 1983) was used to evaluate anxiety immediately prior to the cognitive tasks. Individuals rated their current anxiety levels on four-point Likert-type scales. Total scores can range from 10 to 40 with higher scores indicating higher levels of state anxiety (Cronbach’s α = .83).
Salivary cortisol
Salivary cortisol, a commonly used measure of acute cortisol, was collected. Cotton salivettes were purchased from Sarstedt, Inc. and were stored at room temperature prior to the study. Participants chewed on cotton salivettes for 45 s at each time of measurement. Six salivary cortisol samples were collected over the course of the in-laboratory study. After collection, the samples were stored at −20°C. Briefly before assaying, the samples were thawed and spun at 3,000 rpm for 4 min to obtain samples with low viscosity. One thousand microliters of clear saliva was removed for duplicate analysis of cortisol levels using a time-resolved immunoassay with fluorescence detection. The assay process has been described previously (Dressendörfer et al., 1992; Kirschbaum & Hellhammer, 1989). Cortisol is reported in nanomoles/liter (nmol/L).
Covariates
Cognitive tasks
There were six cognitive tasks administered as stressors during the testing phase of the study, including two attention span tasks (Backward Digit Span [BDS]; Wechsler, 1997; and the n-back; Kirchner, 1958), two declarative memory tasks (word list recall; Hertzog et al., 1990; and story recall; Wechsler, 1997), and two processing speed tasks (Digit Symbol Substitution and Digit Symbol Copy; Wechsler, 1997). To create a measure of overall cognitive performance, z scores of all of the cognitive tests were obtained and then a mean score was computed. Reliability analysis revealed that a high Cronbach’s alpha (.89) for the combined measure indicated that the tests were highly correlated with one another.
Health
Because of the known effect of poor health on cortisol levels and cortisol reactivity (see Dahlgren et al., 2009; De Rooij & Roseboom, 2010), several subjective indicators of poor health, including number of chronic illnesses, number of prescription medications, functional limitations, self-reported poor health, and impairments in daily functioning, were collected. As a measure of overall health, these five variables were standardized and a mean poor health variable composite was computed (Cronbach’s α = .81) with higher scores indicating worse health.
Time of testing
Because of the diurnal fluctuations of cortisol throughout the day, time of testing is also included as a covariate in all cortisol analyses.
Caffeine intake
Because of the known effect of caffeine on cortisol levels, participants were encouraged to not drink caffeine immediately prior to the study. However, the amount of caffeine consumed on the day of testing (in cups) was measured at the start of the study. This number is used as a covariate in the cortisol analyses.
Depression
Given the possible blunting effect of depression on cortisol response, the 15-item Geriatric Depression Scale (GDS; Sheikh & Yesavage, 1986) was used to measure depressive symptoms. The GDS is a self-report depression measure specifically designed to avoid classification errors due to somatic complaints common in older adults. Participants answer 15 yes/no questions about their mood over the past 2 weeks. Scores can range from 0 to 15 with higher scores indicating more symptoms. The GDS has a good internal consistency with an alpha coefficient of .61. Although this scale is designed for older adults, it measures the same information as other commonly used depression inventories (with the exception of somatic symptoms) and has been used previously in this type of study (e.g., Ferraro & Chelminski, 1996; Pearman, 2009; Pearman & Storandt, 2004).
Procedure
After potential volunteers were screened by telephone (see “Participants” section), they were mailed a welcome letter which further described the study, explained the use of the cortisol measurements, and gave detailed parking information. Participants were tested individually by trained research assistants in a laboratory room at a small private university. Upon arrival, participants were given written informed consent by the research assistant conducting the study. They were explained the nature of the study and briefly oriented to the tasks they were to undergo. After written informed consent was obtained, the first salivary cortisol sample was taken. Participants then filled out a series of questionnaires, which took approximately 15 min. They then provided their second saliva sample. After the second saliva sample, participants began the cognitive testing. Saliva samples were taken three times during the 45-min cognitive testing session. At the end of the cognitive testing, participants filled out another series of questionnaires (approximately 15 min) and then provided the posttest saliva sample. Figure 1 illustrates the timeline of the series of measurements.

Time progression of the experimental protocol.
Statistical Analyses
The first two cortisol measurements, which were both administered prior to any cognitive tests, were averaged to represent baseline cortisol. The three cortisol measurements taken during the testing were averaged and called task cortisol. The final cortisol measurement is labeled posttest cortisol. To determine age differences in cortisol trajectories during the testing session, a 2 × 3 (age group by time) repeated-measures analysis of covariance (RM-ANCOVA) was run with time tested, caffeine intake, depression, poor health, mean performance of the cognitive tasks, and pretest anxiety included as covariates. To further understand the observed cortisol change, age and pretest anxiety were examined using regression analyses with cortisol reactivity and recovery as the outcome variables. For reactivity, the mean of the task cortisol controlling for baseline cortisol was used as the dependent variable. For recovery, posttest cortisol controlling for task cortisol was used as the dependent variable. In both regressions, age group, anxiety, and the age-group-by-anxiety interaction term were entered as hierarchical steps. In both analyses, the time tested, caffeine intake, depression, poor health, and the mean performance of the cognitive tasks were included as covariates. Finally, a regression analysis using the mean performance of the cognitive tasks as the outcome variable was conducted for our exploratory analysis to examine relationships between age, cortisol, and anxiety on performance. Specifically, using mean cognitive performance as the dependent variable, both baseline and task cortisol, state anxiety, and age (along with covariates) were used as predictor variables.
Results
Sample Characteristics
One younger adult (prescription stimulant use) and four older adults (one with stroke, three with gross cognitive impairment) were excluded from the study. In addition, one participant’s mean cortisol score was more than three standard deviations above the mean and was therefore omitted from the final sample (see Miller et al., 2016). The final sample consisted of 27 university undergraduates (nine men, 18 women) and 29 community-dwelling older adults (12 men, 17 women). The mean age of the young adult sample is 19.8 (SD = 1.2) ranging from 18 to 23 years. The mean age of the older adult sample is 71.2 (SD = 6.6) ranging from 60 to 85 years. The means, standard deviations, and zero-order correlations of primary study variables are shown in Table 1. In terms of education, the older adult sample was highly educated with all participants having graduated from high school and 60% of participants having a bachelor’s degree or higher. Because the younger adult sample consisted entirely of undergraduate students, we did not conduct an age group comparison of education (i.e., all students had high school diplomas but none had bachelor’s degrees). Because of the restricted range of educational attainment in the younger adult sample, we did not include education as a covariate. 1 There were significant age group differences between cognitive performance and baseline cortisol with younger adults having higher baseline cortisol and higher cognitive performance than older adults. For older adults, pretest anxiety was positively correlated with during-task cortisol levels.
Sample Description.
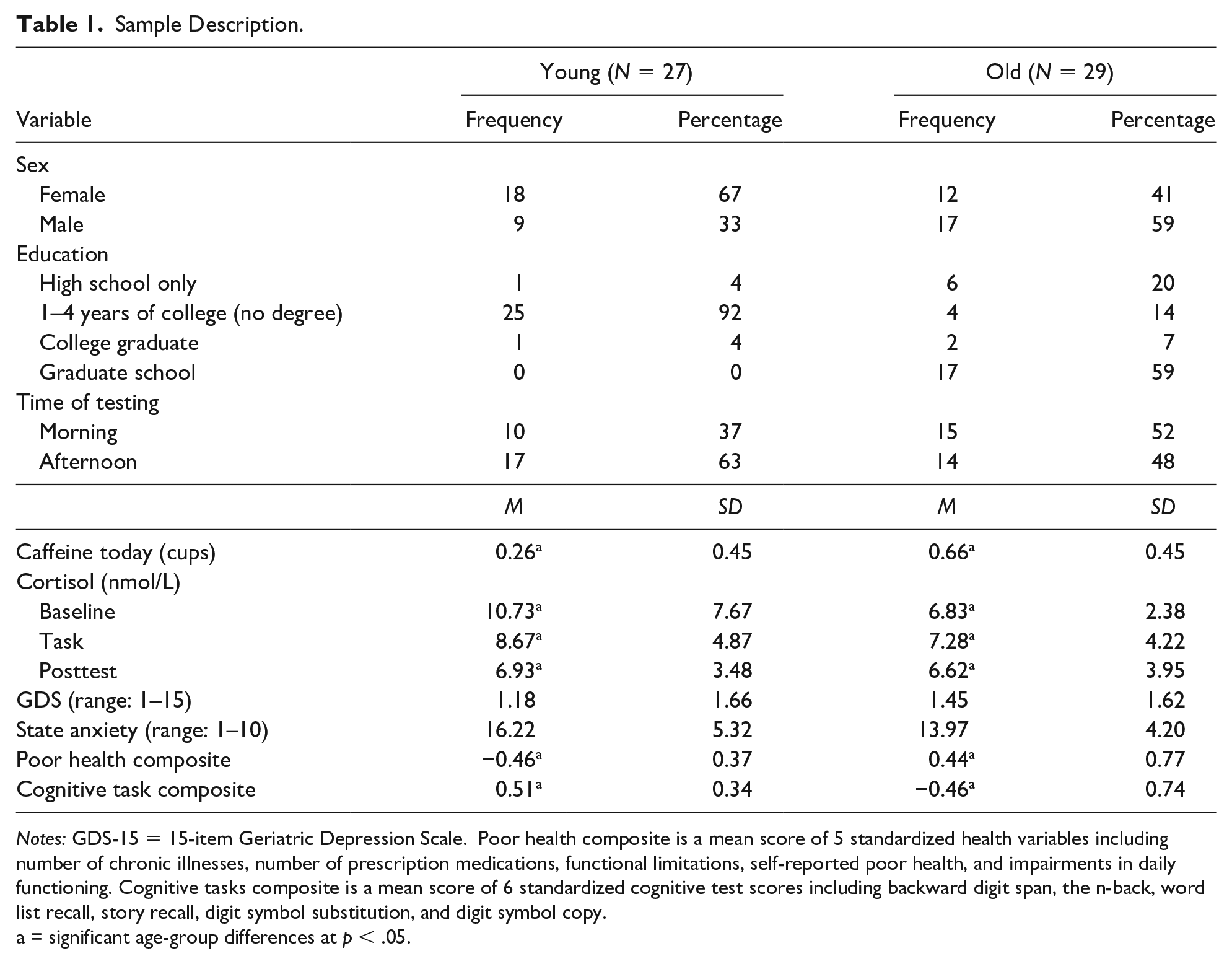
Notes: GDS-15 = 15-item Geriatric Depression Scale. Poor health composite is a mean score of 5 standardized health variables including number of chronic illnesses, number of prescription medications, functional limitations, self-reported poor health, and impairments in daily functioning. Cognitive tasks composite is a mean score of 6 standardized cognitive test scores including backward digit span, the n-back, word list recall, story recall, digit symbol substitution, and digit symbol copy.
a = significant age-group differences at p < .05.
RM-ANCOVA
The RM-ANCOVA for cortisol revealed a significant main effect of time, F(2, 96) = 9.26, p = .002, partial η2 = .16, such that overall cortisol decreased across the assessments. In addition, there was an interactive effect of age group and time, F(2, 96) = 4.36, p = .02, partial η2 = .08. Within younger adults, there was a significant linear decrease in cortisol over the session, F(1, 20) = 15.89, p < .001, partial η2 = .35, as well as a quadratic change, F(1, 20) = 8.99, p = .017, partial η2 = .24. There were not similar significant trends for the older adults. For the between-person covariates, there was a significant effect of time of testing, F(1, 48) = 8.78, p = .004, partial η2 = .15, but not for caffeine intake, state anxiety, depression, or poor health.
Regression Analyses
The multiple regression for cortisol reactivity (i.e., using task cortisol as the dependent variable controlling for baseline cortisol in Step 1) was significant overall (see Table 2) such that higher baseline cortisol was positively related to higher during-task cortisol. There were no significant main effects of age or anxiety. However, there was a significant age-by-anxiety interaction. The interaction was such that older adults with higher anxiety had the highest task cortisol (suggesting higher reactivity to testing) than the older adults with lower anxiety as well as the young adults (see Figure 2). There was no effect of anxiety on during-task cortisol for younger adults. The multiple regression with posttest cortisol as the dependent variable controlling for task cortisol (cortisol recovery) was significant overall, R2 = .83, F(9, 46) = 24.96, p < .001. However, only task cortisol was significant in the steps with a final beta value of .96 (p < .001), such that higher posttest cortisol was related to higher during-task cortisol.
Pearson Product Moment Correlations Between Study Variables Split by Age Group (N = 56).

p < .05; **p < .01.
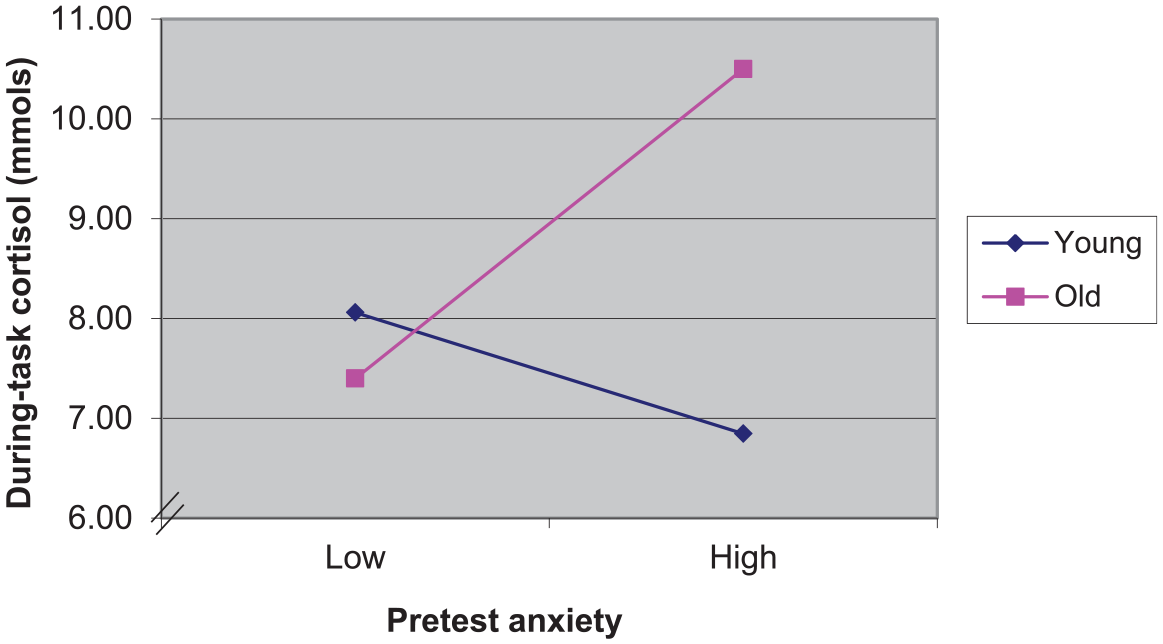
Cortisol during cognitive testing by age group and pretest anxiety.
For our exploratory analysis to examine if cortisol or anxiety were related to overall cognitive performance above and beyond age effects, a regression analysis was conducted with overall cognitive performance mean 2 as the dependent variable and baseline and during-task cortisol along with the other study variables (age, anxiety, poor health, education, caffeine intake, depression) as predictors. Only age was significantly related to cognitive performance, such that younger adults outperformed older adults (final β = −.69, p < .001, overall R2 = .51).
Discussion
The primary aim of this study was to examine the role of anxiety on physiological response (i.e., cortisol) to a stressor (i.e., series of cognitive tests) in a sample of younger and older adults. We comprehensively assessed age group differences in the role of anxiety on physiological arousal before, during, and after cognitive performance. We were able to further differentiate the relationship of anxiety and cortisol reactivity from that of anxiety and cortisol recovery. Older adults’ anxiety was directly related to cortisol reactivity (see Table 3 and Figure 2) but not recovery.
Summary of Multiple Regression Coefficients for Cortisol Reactivity (N = 56).

Notes: Cortisol measured in nmol/L. GDS-15 = 15-item Geriatric Depression Scale. Poor health composite is a mean score of 5 standardized health variables including number of chronic illnesses, number of prescription medications, functional limitations, self-reported poor health, and impairments in daily functioning. Cognitive tasks composite is a mean score of 6 standardized cognitive test scores including backward digit span, the n-back, word list recall, story recall, digit symbol substitution, and digit symbol copy. Age group is coded as 0 = young, 1 = old.
As predicted, the primary finding suggests that older adults who report higher state anxiety are more physiologically responsive during cognitive testing than younger adults. Of note, the older adults did not report more state anxiety prior to testing, and they just had an increased cortisol response in relation to that anxiety. This finding has several related possible explanations. The first is in line with the stereotype threat literature that older adults who are more anxious are going to be more threatened by cognitive testing than less anxious older adults. Given that older adults that are higher in neuroticism and state anxiety tend to have more memory concerns than older adults lower in neuroticism (Pearman & Storandt, 2004), it is likely that these individuals are also more reactive to taking cognitive tests than older adults lower in anxiety. Furthermore, the role of social self-preservation may be especially relevant for older adults who are higher in anxiety and value their cognitive performance (Dickerson & Kemeny, 2004). Because sustained cortisol elevation is associated with impairment in cognitive functioning, looking at particular subgroups of older adults (i.e., higher anxiety, higher memory complaints, and/or higher cognitive achievement motivation) and their stress reactivity to cognitive testing could provide further insight into the role of the HPA axis and cognitive decline (Lupien et al., 2005; McEwen, 2002). Our sample was not large enough to examine these other subgroups of participants.
Our exploratory analyses showed that changes in cortisol were unrelated to performance. That is, in terms of performance on the cognitive tasks, we were unable to replicate previous findings showing that anxiety and cortisol were related to performance (A. Hood et al., 2015; Merz & Wolf, 2015; ó Hartaigh et al., 2012). One reason for this lack of significant findings could be that we used a composite mean for the test performance outcome, whereas previous studies have looked at response to individual tests. However, we ran the same analyses for each individual cognitive test (see Note 2) and were not able to detect relationships other than the age differences in performance on each test. The cognitive tasks also may not have been stress inducing enough, particularly for the younger adults, to see a relationship between cortisol and performance. In addition, we may have been underpowered to detect the cortisol–performance relationship in the older adults who did show cortisol increases during the session.
There are several limitations to this study. The first limitation is that the younger adults came into the laboratory with very elevated cortisol levels compared with similar studies (Kudielka et al., 2004) and showed significant decreases in cortisol from baseline to posttest. It appears that, in this study, the younger adults got calmer during the session (according to cortisol response). We posit that the decrease in cortisol is due to the cognitive tests not being stressful for the younger adults. However, it could be that heightened levels of starting cortisol automatically lead to decreases when college students sit down in a quiet room for a couple of hours, even with the testing. To try to address these wide ranges of cortisol levels, we included baseline cortisol in the regression analysis predicting the during-task cortisol. Another limitation was that although participants were instructed to report all medications and caffeine intake (as well as to refrain from caffeine prior to the study), it was not possible to effectively enforce or measure this adherence. However, we did ask participants to report their caffeine intake for the day and used it in the analyses. Finally, this study used a small and select nonrepresentative sample of highly educated younger and older adults, which makes generalizability more of a challenge.
Overall, this study furthers understanding of age differences in anxiety and arousal associated with cognitive testing. Self-reports of anxiety were related to hormonal changes and levels in the older adults but not the young. Given that under most “stressful” scenarios younger adults do show the expected increases in cortisol (Blatt-Eisengart & Lachman, 2004), this study suggests that cognitive testing such as the type completed in this study is not anxiety producing for younger adults but is for older adults, particularly those who are higher in anxiety. These findings support previous research that suggests older adults who are nervous about cognitive testing tend to show increases in cortisol (Neupert et al., 2006) and also supports previous findings on stereotype threat response in older adults (Barber et al., 2015). These findings are an important step in identifying noncognitive factors that may contribute to the memory and cognitive testing experiences in older adults. This is of importance to both researchers who administer cognitive tests to older adults as well as clinicians who work with older adults who may have cognitive concerns and/or anxiety. Specifically, understanding that older adults who are anxious upon presentation for a cognitive evaluation may in turn show a heightened stress response to that testing is important for both clinicians and researchers. Finding ways to interact and counteract this anxiety may also play an important role in the well-being of the patients.
Footnotes
Acknowledgements
The authors thank Margie E. Lachman for the use of the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from the National Institute on Aging R01 AG17920 to Margie E. Lachman.
