Abstract
Introduction
Cognitive function, particularly executive function, declines with normal aging (Buckner, 2004). Executive function is largely dependent on the prefrontal brain region and is defined as cognitive function to perform complex, goal-directed, and self-serving behaviors independently (Malloy & Richardson, 1994). A previous longitudinal study reported that executive function predicts a decline in instrumental activities of daily living, physical impairment, and mortality (Cahn-Weiner et al., 2007; Vazzana et al., 2010). Thus, preserving the executive function is vital for everyday functioning as well as to maintain autonomy and quality of life, which is of great public health importance. There is an urgent need for prevention of cognitive decline.
Several studies have reported the association of pain with executive function (Cherry et al., 2014; Lee et al., 2010; Murata et al., 2017; van der Leeuw et al., 2016; Weiner, Rudy, Morrow, Slaboda, & Lieber, 2006). More than 50% of older adults reported chronic pain (Andorsen, Ahmed, Emaus, & Klouman, 2014), which led to adverse outcomes, such as depression, disability, and decreased quality of life (Denkinger et al., 2014; Eggermont et al., 2014; Laslett et al., 2012). Some researchers have suggested that chronic pain affected executive function decline (Cherry et al., 2014; Lee et al., 2010; Murata et al., 2017; van der Leeuw et al., 2016; Weiner et al., 2006), while other studies have suggested that high executive function suppresses pain and leads to high pain tolerance (Attal et al., 2014; Zhou, Kemp, Despres, Pebayle, & Dufour, 2015). High executive function may reflect a high ability of coping with pain and high cognitive regulation of emotions to deal with chronic pain (Solberg Nes, Roach, & Segerstrom, 2009). One study also suggested that high cognitive inhibition, an element of executive function, is associated with high pain tolerance (Zhou et al., 2015). However, most of these studies were of cross-sectional design, and only one longitudinal study suggested that preoperative low executive function is associated with postoperative transition to chronic pain and high pain severity (Attal et al., 2014). The reciprocal relationship between chronic pain and executive function is not well understood. Additional evidence from longitudinal design is needed to investigate the effect of chronic pain on subsequent decline in executive function, or that of declined executive function on worsening of chronic pain.
The purpose of this study was to explore the reciprocal relationship between pain severity and executive function in community-dwelling older adults.
Method
Subjects and Procedure
In this prospective cohort study, baseline data were collected from 133 Japanese community-dwelling older adults aged 60 years or older who were recruited from community clubs at 2014. The exclusion criteria for this study were scoring below 22 on the Mini-Mental State Examination (MMSE; n = 2; Weiner et al., 2006) and not completing baseline examinations (n = 5). The remaining 126 participants were interviewed again 1 year after the baseline assessment, and we also excluded those who did not complete the survey on pain and evaluation of executive function at the 1-year follow-up assessment. Therefore, 64 people met the criteria and were available for the final analysis in this study (mean age ± standard deviation, 72.8 ± 6.5 years; women, 68.8%). The Research Ethics Committee of Kobe University Graduate School of Health Sciences approved the study (authorization number 181-1), and informed consent was obtained from all subjects before participation.
Assessment of Pain
Chronic pain, according to previous report, was defined as persistent pain lasting longer than 3 months (Treede et al., 2015). We assessed the presence of chronic pain in various musculoskeletal locations: neck, low back, shoulder, elbow, wrist, hand, hip, knee, ankle, and foot. When the participants had chronic musculoskeletal pain, pain intensity was evaluated at each site using the 11-point numeric rating scale (NRS), ranging from 0 (no pain) to 10 (worst pain), with higher scores indicating worse pain. For participants with chronic multisite musculoskeletal pain, we assessed NRS at each pain site and the highest value of NRS score was used in the analysis.
Evaluation of Executive Function
We assessed executive function using the Trail Making Test (TMT; Lezak, 1995), which consisted of two parts providing information on processing speed (Part A [TMT-A]; Army Individual Test Battery, 1944) and set shifting (Part B [TMT-B]; Sánchez-Cubillo et al., 2009). Processing speed and set shifting are components of executive function. Processing speed has been defined as how fast one can execute the mental operations needed to complete a task at hand (Salthouse, 2000). Set shifting is described as the process of shifting back and forth between multiple tasks, operations, or mental sets (Miyake et al., 2000). TMT-A required participants to draw a line sequentially connecting numbers (1-2-3) as quickly as possible without lifting the pencil off the paper. If an error was made, the examiner indicated the error and allowed the participant to correct the mistake and continue the task. TMT-B was similar, except that numbers and letters were alternated in a progressive sequence (1-A-2-B). In the Japanese version of TMT-B, the test has been revised by changing letters from the Roman alphabet to Kana characters. Completion times were measured for TMT-A and TMT-B.
Other Variables
Demographic data were recorded, including age, sex, years of education (≤9, 10-12, or ≥13 years), body mass index (BMI), and analgesic drug use. BMI was calculated as weight (kg) divided by height (m) squared. Global cognitive function was assessed using the MMSE (Folstein, Folstein, & McHugh, 1975). Depressive symptoms were assessed using the Geriatric Depression Scale–15 (GDS; a 15-item inventory with a yes/no format; Yesavage, 1988). Depressive symptoms were screened using a cutoff value of ≥6 to indicate depressive symptoms (Friedman, Heisel, & Delavan, 2005). Orthopedic diseases were assessed by self-reporting (hip and knee osteoarthritis, rheumatoid arthritis, and spinal disease, including spinal disk herniation, spinal canal stenosis, and compression fracture of the spine). The gait speed was assessed using a stopwatch. Participants were asked to walk 6.4 m (divided into two 2.0-m zones at each end and a 2.4-m middle zone) at their usual pace. We measured the time required (in seconds) to complete the 2.4-m middle zone over two trials to calculate the mean gait speed (meter per second, m/s).
Statistical Analysis
Baseline characteristics, pain severity, and executive function with and without follow-up assessment were compared. Whereas continuous variables with normal distribution (age and BMI) were analyzed by an unpaired t test, continuous variables with nonnormal distribution (NRS, MMSE, TMT-A, TMT-B, and gait speed) were analyzed using the Wilcoxon rank sum test. Sex was analyzed using the χ2 test. The other categorical variables were analyzed using Fisher’s exact test. A mixed effect model was applied to analyze the reciprocal relationship between pain severity and executive function. First, to investigate the effect of baseline executive function on change in pain severity, the mixed effect mode was conducted with pain severity as the dependent variable and time (year), baseline executive function, and interactions of time with baseline executive function as the independent variables. Next, to investigate the effect of baseline pain severity on change in executive function, the mixed effect model was conducted with executive function as the dependent variable and time (year), baseline pain severity, and interactions of time with baseline pain severity as the independent variables. The random effects included participant identification (ID) in all models. Crude and adjusted models were constructed. The following variables were included as confounding factors in the adjusted model: age, sex, years of education, depressive symptoms, and analgesic medication use. Statistical significance was set at p < .05, and all analyses were conducted using STATA version 14.2 (StataCorp, College Station, TX, USA).
Results
Mean (standard deviation) values at baseline and at 1-year follow-up were 71.1 (17.9) and 69.1 (20.1), respectively, for TMT-A; 102.6 (42.8) and 118.5 (61.4), respectively, for TMT-B; and 2.3 (2.8) and 2.1 (2.5), respectively, for NRS. Table 1 shows baseline characteristics of participants with and without follow-up assessment. There was no significant difference of baseline data between the two groups.
Baseline Characteristics of the Participants.

Note. Data of age, BMI, numeric rating scale, MMSE, TMT-A, TMT-B, and gait speed are expressed as mean (standard deviation), and the others are expressed as n (%). In analyzed participants, the total number of data points for knee osteoarthritis, hip osteoarthritis, and spinal disease is 63, 63, and 62, respectively, due to some missing data. In dropout participants, the total number of data points for spinal disease is 61 due to some missing data. BMI = body mass index; MMSE = Mini-Mental State Examination; TMT-A = Trail Making Test Part A; TMT-B = Trail Making Test Part B.
Table 2 shows the relationship between baseline executive function and change in pain severity. The effects of time, baseline TMT-A, baseline TMT-B, and their interactions on NRS were not significant after adjustment.
Effect of Executive Function on Pain Severity: Mixed Effect Model.
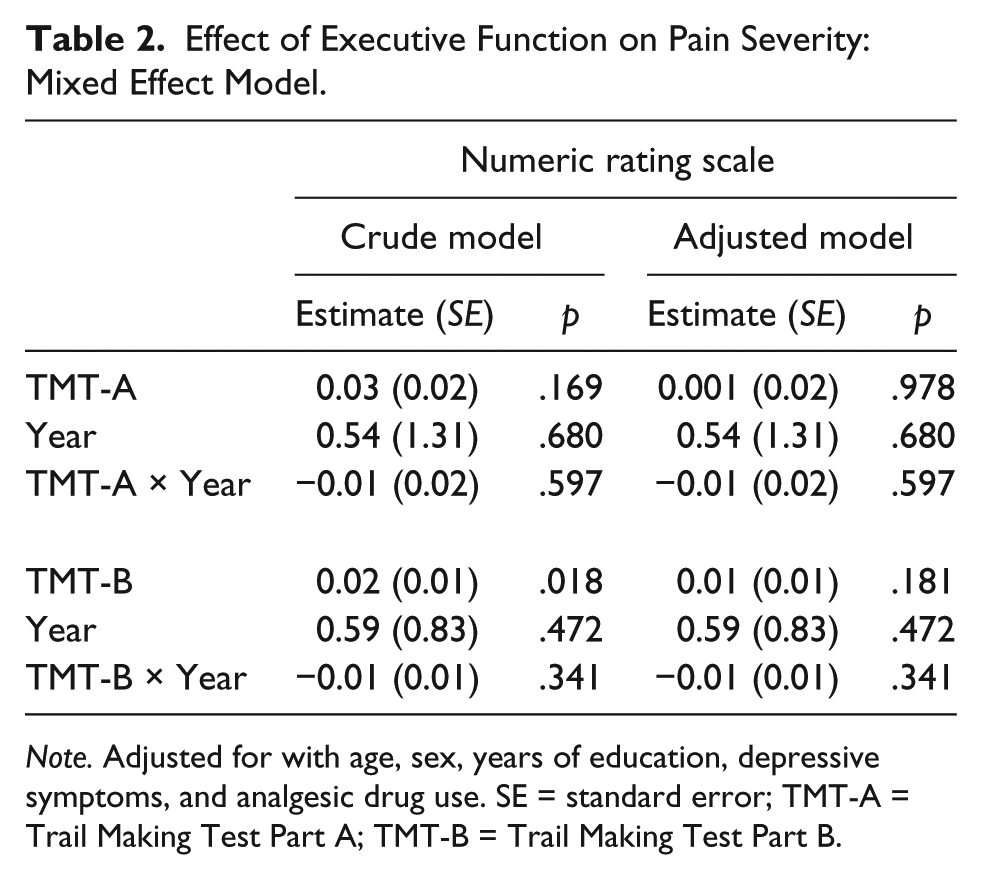
Note. Adjusted for with age, sex, years of education, depressive symptoms, and analgesic drug use. SE = standard error; TMT-A = Trail Making Test Part A; TMT-B = Trail Making Test Part B.
Table 3 shows the relationship between baseline pain severity and change in executive function. There was a significant effect of the interaction of time with pain severity on TMT-B even after adjustment (estimate [standard error] = 5.72 [2.04], p = .005). The effects of time, baseline NRS, and their interaction on TMT-A were not significant after adjustment. No significant effect of time and baseline pain severity on TMT-B was observed.
Effect of Pain Severity on Executive Function: Mixed Effect Model.

Note. Adjusted for age, sex, years of education, depressive symptoms, and analgesic drug use. SE = standard error; TMT-A = Trail Making Test Part A; TMT-B = Trail Making Test Part B; NRS = numeric rating scale.
Discussion
We investigated the reciprocal relationship between pain severity and executive function in community-dwelling older adults. No significant association between baseline executive function and change in pain severity was observed. On the contrary, baseline higher pain severity was associated with a greater decline in set shifting. Our findings suggested that exposure to high pain severity predicted a decline in set shifting.
Contrary to our expectation, no significant association between baseline executive function and change in pain severity was observed in our study. Attal et al. (2014) hypothesized that worse cognitive function predicts chronic pain after surgery, and suggested that preoperative low executive function was associated with postoperative transition to chronic pain and high pain severity in patients undergoing total knee arthroplasty or breast cancer surgery. Pain in their participants undergoing surgery drastically changed compared with that in older adults. Actually, pain severity in the patients undergoing total knee arthroplasty was significantly decreased after 1 year, and the incidence of pain in those undergoing breast cancer surgery was 17.7% at one year postoperatively (all patients had no pain before breast cancer surgery; Attal et al., 2014). In our study, no significant change in pain severity was observed from baseline assessment to 1-year follow-up (Data not shown). Their study participants were much different from those in our study. Therefore, this difference in participants might contribute to inconsistent results between the studies. Further study with longer follow-up should be conducted to elucidate the effect of executive function on chronic pain.
Our study suggested that baseline pain severity affected the decline in set shifting in community-dwelling older adults. To our knowledge, ours is the first study to show an effect of pain severity on decline in set shifting with a longitudinal design. Further insight was provided into understanding the effect of chronic pain on executive function decline and in determining the prevention strategy of cognitive decline. Chronic pain often is coexistent with physical inactivity and depression (Denkinger et al., 2014; Stubbs et al., 2013), which are large risk factors for cognitive decline (Barnes & Yaffe, 2011). In addition, previous studies reported that depressive symptoms and physical inactivity were associated with a low TMT-B score (Avila et al., 2009; Kerr et al., 2013). Hence, the effect of pain severity may reach set shifting decline.
Our results indicated a greater decline in set shifting in participants with higher than with lower pain severity. Some studies have suggested that set shifting assessed using TMT-B is a predictor of Alzheimer’s disease, disability, and mortality (Johnson, Lui, & Yaffe, 2007; Rapp & Reischies, 2005). Therefore, set shifting, as a component of executive function, can be also crucial for older adults’ health maintenance. When treating older adults with high pain severity, chronic pain management can be efficacious in keeping set shifting. As described above, depressive symptoms and physical inactivity may be related to the effect of chronic pain on cognitive decline. Accordingly, intervention for physical inactivity and depressive symptoms due to chronic pain also might be efficacious in keeping cognitive function. However, our study cannot show why high pain severity led to a decline in set shifting. Therefore, further study is required to examine the mechanism and to determine a prevention strategy for executive function decline in older adults with severe pain.
Although this study has provided a number of important contributions, its limitations should be considered. First, our study had a small sample size and low follow-up rate. However, there were no significant differences between analyzed participants and those who dropped out of the study; therefore, migration bias did not occur. Effects of pain location and number of chronic musculoskeletal pain sites on executive function decline should be investigated in further studies with larger samples. Finally, a detailed assessment of analgesic medication was not conducted. Nonsteroidal anti-inflammatory drugs (NSAIDs) can protect against Alzheimer’s disease. NSAIDs and opioids should be included as confounding factors (in t’ Veld et al., 2001).
Conclusion
Higher baseline pain severity was associated with greater executive function decline in community-dwelling older adults. Executive function decline due to severe pain should be focused on as well as pain itself.
Footnotes
Acknowledgements
The authors would like to thank the staff members and all other people involved in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Grant-in-Aid for Japan Society for the Promotion of Science Fellows (Grant No. 17J05200).
