Abstract
Introduction
The normal aging process is accompanied by a gradual decline in physical functioning, and previous research has reported a potentially “bi-directional” inverse relationship between chronic inflammation and physical functioning in older adults (Brinkley et al., 2009; Cesari et al., 2010; Cummings, King, & Mainous, 2003; Levine & Crimmins, 2012; Marenberg, 2003; Tomey, Sowers, Zheng, & Jackson, 2009). Among suggested mechanisms, diminished physical functioning can result from a prolonged state of inflammatory response, which in turn can compromise strength, aerobic capacity, balance, and coordination (Tomey et al., 2009). In this case, chronic inflammation is a determinant of physical functioning. Alternatively, diminished physical functioning may co-occur with a sedentary lifestyle leading to excess body weight and subsequently chronic inflammation (Tomey et al., 2009). Thus, physical functioning can determine inflammation through the mediating effect of excess body weight.
Previously conducted studies have associated two biomarkers of inflammation, C-reactive protein (CRP) and fibrinogen, with cardiovascular morbidity and mortality (Cummings et al., 2003; Nguyen, Lane, Smith, & Nguyen, 2009). Produced by hepatocytes in response to infection, injury, or obesity, CRP is a marker for nonspecific inflammation (Nguyen et al., 2009; Tomey et al., 2009) with levels increasing thousandfold in response to inflammatory disorders and decreasing in response to statins, aspirin, and nonsteroidal anti-inflammatory drugs (NSAIDs; Cummings et al., 2003; Tomey et al., 2009). A less clearly established biomarker of chronic inflammation is fibrinogen, which is known to be a precursor to fibrin and a determinant of platelet aggregation and plasma viscosity (Nguyen et al., 2009).
Chronic conditions, including type 2 diabetes, hypertension, dyslipidemia, metabolic syndrome, atherosclerosis, chronic inflammatory bowel disease, periodontal disease, obstructive sleep apnea, cancer, and arthritis were linked to excessive weight, with chronic inflammation as a well-established intermediate factor (Christian et al., 2011; Nguyen et al., 2009).
Specifically, physician-diagnosed arthritis is a condition encompassing 100 degenerative joint diseases, affecting 1 in 5 Americans (~46 million U.S. adults), and costing ~US$128 billion in health care costs in 2003. Two commonly diagnosed forms of arthritis are “osteoarthritis” (OA; degeneration of the cartilage and underlying bone within a joint, characterized by bony overgrowth) and “rheumatoid arthritis” (RA; systemic inflammatory disease manifesting in multiple joints caused by rheumatic diseases). Nearly 41% of individuals who are physician-diagnosed with arthritis report impaired physical functioning. Arthritis increases in prevalence with age and is more common in women. Furthermore, impaired physical functioning accompanying arthritis is more prevalent in Blacks and Hispanics versus Whites, with comorbid conditions including diabetes (52.4%), heart disease (57.8%), and hypertension (47.6%; Battle, 2008).
To date, few studies have examined the interrelationship of sex, physician-diagnosed RA and/or OA, chronic inflammation, and physical functioning using a nationally representative sample of older adults. In a cross-sectional study using National Health and Nutrition Examination Survey (NHANES) III data, we examined whether serum concentrations of CRP and fibrinogen were associated with total score on a validated 12-item physical functioning scale and whether the magnitude and direction of these associations differed according to sex and presence and type of physician-diagnosed arthritis.
Method
Study Population
The National Center for Health Statistics (NCHS) of the Centers for Disease Control and Prevention (CDC) implemented the NHANES III between 1988 and 1994 in two phases (Phase I: 1988-1991, Phase II: 1991-1994) using a complex multistage probability sample design that can provide national estimates of health and nutritional status of the civilian noninstitutionalized population (NCHS, 1994). In this study, questionnaire, physical examination, and laboratory data from both phases of NHANES III were utilized. The NHANES III protocol is compliant with the ethical rules for human experimentation that are stated in the Declaration of Helsinki, including approval of an institutional review board and informed consent. Of 33,199 NHANES III participants, 26,774 were excluded for being less than 60 years of age and 15,786 were excluded for not having data on physician-diagnosed arthritis, for a total of 28,593 exclusions. Out of 4,606 remaining NHANES III participants (2,302 men and 2,304 women), 907 had missing data on CRP and 989 had missing data on fibrinogen, as they were not part of the NHANES III Mobile Examination Center (MEC) subsamples, and 715 had missing data on physical functioning. Accordingly, complete-subject analyses consisted of subsamples of 3,173 individuals with data available on physical functioning and CRP concentration and subsamples of 3,107 individuals with data available on physical functioning and fibrinogen concentration, taking into account missing data from other covariates, which range between 0% and 12.4%.
Measures
Physician-diagnosed arthritis
In the Household Adult Questionnaire, older adults 60 years and older were identified as having ever been diagnosed with any type of arthritis, OA or RA, as follows: “Has a doctor ever told you that you had arthritis?” [“yes” or “no”] and “Which type of arthritis was it? Was it rheumatoid arthritis or osteoarthritis?” [“Rheumatoid arthritis” or “Osteoarthritis”]. We classified study participants as having “no arthritis,” “OA” or “RA.” OA and RA were mutually exclusive conditions that subsequently were combined into “any arthritis” [OA or RA] versus none.
Chronic inflammation
As part of the MEC of the NHANES III, serum specimens were obtained using standard phlebotomy techniques and frozen at −20 °C until analyzed (King, Mainous, Buchanan, & Pearson, 2003). For CRP (Behring Nephelometer Analyzer System; Behring Diagnostics, Somerville, NJ), a lower detection limit of 0.30 mg/dl was used and a value of 0.21 mg/dl was assigned to participants with CRP <0.3 mg/dl (King et al., 2003). The coefficient of variation for CRP ranged between 3.2% and 16.1% (Ford & Erlinger, 2004). Fibrinogen level, in mg/dL, was measured using Coag-A-Mate XC Plus by Organon-Teknika and General Diagnostics (Kritchevsky, Bush, Pahor, & Gross, 2000). Both CRP and fibrinogen concentrations presented skewed distributions and were therefore loge-transformed.
Physical functioning
A 12-item validated physical functioning scale that measured self-reported difficulty in activities of daily living (ADL; “difficulty walking a quarter of a mile,” “difficulty walking 10 steps without rest,” “difficulty stooping, crouching, kneeling,” “difficulty lifting or carrying 10 pounds,” “difficulty doing chores around the house,” “difficulty preparing own meals,” “difficulty managing your money,” “difficulty walking room to room, 1 level,” “difficulty standing from armless chair,” “difficulty getting in or out of bed,” “difficulty eating,” “difficulty dressing yourself”) was used in NHANES III (NCHS, 1994). For each item, respondents were asked whether they had no, some, or much difficulty or were unable to perform the task, with scores for each item ranging from 1 to 4, and the sum ranging from 12 to 48. Cronbach’s alpha, used to evaluate internal consistency, was estimated to be .93 for the physical functioning scale. We defined individual items as ordinal variables and “overall physical functioning” as a continuous variable.
Covariates
The hypothesized relationships between chronic inflammation and overall physical functioning score were evaluated before and after adjustment for a priori confounding factors, namely, sex (male, female), age (continuous; <65, 65-69, 70-79, 80+ years), education (less than high school, high school, more than high school), race/ethnicity (non-Hispanic White, non-Hispanic Black, Hispanic, Other), area of residence (metropolitan, other), poverty-income ratio (<100%, 100%-<200%, ≥200%), marital status (married/cohabiting, not married), smoking status (current smoker, ex-smoker, never smoker), and body mass index (BMI; continuous; <25, 25-<30, 30+ kg/m2) and physician-diagnosed arthritis status (OA, RA, no arthritis). Sex and physician-diagnosed arthritis status were also examined as potential effect modifiers for the hypothesized relationships between inflammatory biomarkers and physical functioning.
Statistical Analysis
Statistical analyses were performed using STATA Version 12 (STATA Corporation, College Station, TX). Whereas frequencies and percentages were computed for categorical variables, means and standard errors of the mean (SEM) were computed for continuous variables. We used design-based F tests (svy:tab) for examining bivariate associations between categorical variables. Bivariate associations involving a categorical and a continuous variable were also examined using design-based F tests (svy:reg). Crude and adjusted beta coefficients and their 95% confidence intervals (CI) were calculated using ordinary least squares regression by applying the “svy:reg” command. Several aspects of the NHANES III design were taken into account in the data analysis, including the sampling weights and the complex survey design (i.e., primary sampling unit and strata). Three full-sample and four subsample weights are available in NHANES III. For the current analyses, we applied the MEC examination weights pertaining to Phases I and II of NHANES III (i.e., 6 years). Sampling weights were used to produce correct population estimates of prevalence rates, means, and other statistics accounting for differential probabilities of selection and adjusting for noncoverage, nonresponse, and oversampling of subpopulations. Two-sided statistical tests were performed at an alpha level of .05.
Results
Out of 4,606 NHANES III participants, 60 years and older with known physician-diagnosed arthritis status, 3,540 reported no arthritis, 570 reported OA, and 496 reported RA. Physician-diagnosed OA and RA were significantly more prevalent among women compared with men. On average, individuals with OA (but not those with RA) were significantly older than individuals with no arthritis. Furthermore, OA prevalence differed according to education, race/ethnicity, and BMI categories and differences in RA prevalence were noted according to marital status, poverty-income ratio, and BMI categories (Table 1).
History of Physician-Diagnosed Arthritis by Demographic, Socioeconomic, and Health Characteristics—National Health and Nutrition Examination Study III.

Note. OA = osteoarthritis; RA = rheumatoid arthritis; POA represents statistical significance using design-based F test (svy:reg or svy:tab), comparing OA with no arthritis; PRA represents statistical significance using design-based F test (svy:reg or svy:tab), comparing RA with no arthritis; SEM = standard error of the mean; HS = high school.
Column percentage.
Weighted.
Row percentage.
Table 2 presents levels of loge-transformed inflammatory biomarkers, namely, CRP and fibrinogen, according to physician-diagnosed arthritis status and sex. Whereas NHANES III participants were evenly distributed according to fibrinogen level, a total of 2,195 participants had CRP levels below the limit of detection. CRP level was significantly higher among women with a self-reported OA diagnosis compared with women who did not report arthritis. Similarly, CRP and fibrinogen were significantly higher among women with a self-reported RA diagnosis compared with women who did not report arthritis. Neither CRP nor fibrinogen levels differed according to physician-diagnosed arthritis status among men.
CRP and Fibrinogen Concentration by History of Physician-Diagnosed Arthritis and Sex—National Health and Nutrition Examination Study III.

Note. CRP = C-reactive protein; SEM = standard error of the mean; POA represents statistical significance of design-based F test (svy:reg), comparing OA with no arthritis, after loge-transformation of CRP or fibrinogen levels; PRA represents statistical significance of design-based F test (svy:reg), comparing RA with no arthritis, after loge-transformation of CRP or fibrinogen levels; OA = osteoarthritis; RA = rheumatoid arthritis.
Table 3 summarizes indicators of physical functioning according to self-reported OA or RA diagnosis. With few exceptions, significantly higher scores on physical functioning items suggesting more difficulties in performing specific tasks were observed among individuals with OA or RA compared with individuals with no arthritis. The total physical functioning score was also significantly higher among arthritis-diagnosed individuals compared with those with no arthritis diagnosis.
Physical Functioning by History of Physician-Diagnosed Osteoarthritis or Rheumatoid Arthritis—National Health and Nutrition Examination Study III.
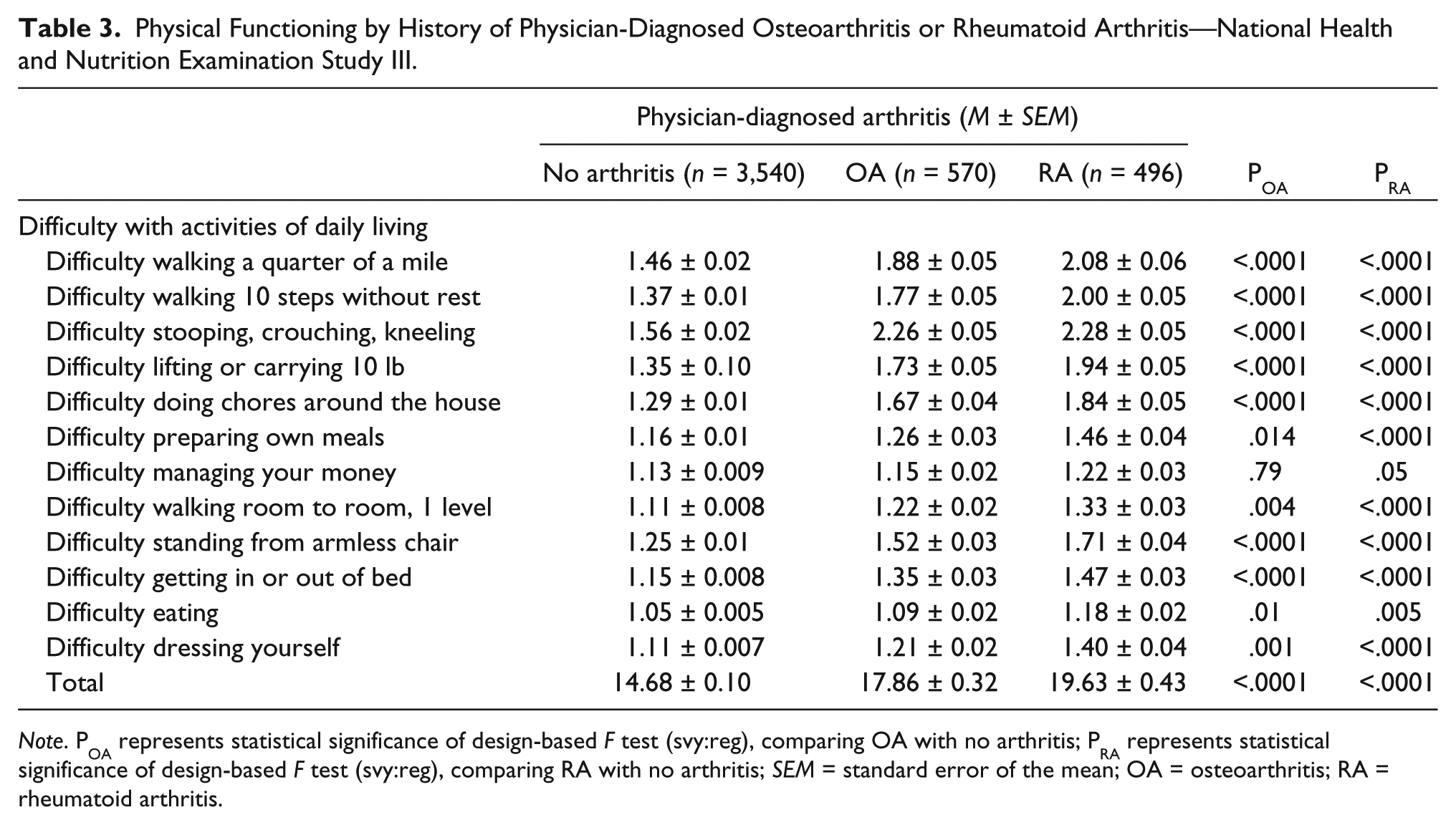
Note. POA represents statistical significance of design-based F test (svy:reg), comparing OA with no arthritis; PRA represents statistical significance of design-based F test (svy:reg), comparing RA with no arthritis; SEM = standard error of the mean; OA = osteoarthritis; RA = rheumatoid arthritis.
The results of simple and multiple linear regression models for loge-transformed CRP and fibrinogen concentrations as correlates of physical functioning scores are displayed in Table 4. After adjustment for key confounders, the overall score on physical functioning was strongly associated with loge-transformed CRP (adjusted β = +0.68, 95% CI = [+0.42, +0.94]) and fibrinogen (adjusted β = +1.66, 95% CI = [+0.89, +2.42]) levels.
Linear Regression Models for CRP and Fibrinogen Concentrations as a Predictor of Physical Functioning Score—National Health and Nutrition Examination Study III.

Note. CRP = C-reactive protein; CI = confidence interval; OA = osteoarthritis; RA = rheumatoid arthritis.
Multivariate models were adjusted for sex, age (continuous), education, race/ethnicity, area of residence, poverty-income ratio, marital status, smoking status, body mass index (continuous), and arthritis status (OA, RA, no arthritis).
The inflammatory biomarkers in relation to overall physical functioning were examined in multivariable linear regression models that stratified according to or included interaction terms for sex (Model 1) and physician-diagnosed arthritis status (Model 2). Results suggested no significant effect modification of the hypothesized relationships by sex. In contrast, physician-diagnosed arthritis status appears to interact with CRP and fibrinogen in relation to physical functioning. Specifically, the hypothesized relationships were significant among individuals with RA or no arthritis, but not among those with OA (Table 5 and Figure 1).
Interaction of Sex and Physician-Diagnosed Arthritis With Inflammatory Biomarkers in Relation to Physical Functioning Score—National Health and Nutrition Examination Study III.

Note. CI = confidence interval; CRP = C-reactive protein.
Multivariate models were adjusted for the following a priori confounders: sex (male, female), age (continuous), education (less than high school, high school, more than high school), race/ethnicity (non-Hispanic White, non-Hispanic Black, Mexican American, Other), area of residence (metropolitan, other), poverty-income ratio (<100%, 100%-<200%, ≥200%), marital status (married/cohabiting, not married), smoking status (current smoker, ex-smoker, never smoker), body mass index (continuous), and arthritis status (OA, RA, no arthritis).

Physical functioning overall score with 95% confidence intervals according to levels of C-reactive protein and fibrinogen by physician-diagnosed arthritis status.
Discussion
In this population-based cross-sectional study, we evaluated whether CRP and fibrinogen levels were associated with an index of physical functioning among older adults, 60 years and older, according to sex and physician-diagnosed arthritis. Whereas CRP and fibrinogen levels were linked to arthritis only in women, arthritis-diagnosed men and women had difficulty performing ADL to a greater extent than those not diagnosed with arthritis. In general, CRP and fibrinogen concentrations were consistently associated with poorer physical functioning as depicted by more difficulty in undertaking ADL. Although CRP and fibrinogen concentrations were more strongly associated with a poorer physical functioning among men than women, there were no statistically significant interaction effects between inflammatory biomarkers and sex. In contrast, arthritis status interacts with CRP and fibrinogen in relation to overall physical functioning, with stronger associations observed in individuals diagnosed with RA or no arthritis.
The finding of a distinct pattern of relationships between inflammatory biomarkers and the two forms of arthritis is expected because of different anatomical structures that may be involved in each condition and the greater significance of inflammation in RA than in OA. Specifically, the impact of RA on physical functioning is probably due to involvement of the hand while the impact of OA on physical functioning is likely linked to the knee or back. Also, RA is more likely to be associated with inflammatory biomarkers as it is an active inflammation as compared with OA, which is more of a low-grade inflammatory process.
To our knowledge, this is one of few studies using a nationally representative sample to evaluate these two biomarkers of chronic inflammation as they correlate with physical functioning as an outcome rather than specific chronic conditions that can reduce mobility or result in disability. Nevertheless, several limitations should be taken into account for a valid interpretation of study findings. First, the cross-sectional design precludes establishment of observed relationships between the exposure (CRP or fibrinogen) and outcome (physical functioning) of interest as being cause and effect. Second, information bias may be an issue with self-reported arthritis diagnosis, perceived (subjective) as opposed to performance-based (objective) physical functioning and measurement of biomarkers that are subject to variability over time such as CRP and fibrinogen. Third, alternative measures of chronic inflammation including interleukin-1 [IL-1] and interleukin-6 [IL-6] were not available for assessment in NHANES III. Finally, selection bias resulting from missing data and confounding bias resulting from unmeasured or uncontrolled factors may constitute alternative explanations for the relationships identified in this observational study.
Our study findings are consistent with a growing body of the literature addressing similar hypotheses. For instance, Tomey et al. investigated whether CRP and fibrinogen are related to measures of physical functioning using a sample of 543 midlife Michigan women from the Study of Women’s Health across the Nation (Tomey et al., 2009). Physical functioning was measured using performance-based outcomes (gait, hand grip strength, flexibility, stair climb, 40-ft walk, and chair rise) and perceptions (Medical Outcomes Study Short-Form 36 questionnaire). The authors found distinct associations for CRP and fibrinogen in relation to indicators of physical functioning, performance-based and perceived, as well as different results depending on whether the analysis was cross-sectional or longitudinal. However, consistent with our study findings, higher CRP and fibrinogen were associated with poorer perceived functioning in cross-sectional analyses.
A recent study by Levine & Crimmins (2012) analyzed data on 2,287 nondiabetic 1999-2004 NHANES participants, aged 60 and older, to examine whether insulin resistance and inflammation (CRP) can mediate the relationship between physical functioning (self-reported difficulty performing six activities) and sarcopenic obesity (healthy, sarcopenic nonobese, nonsarcopenic obese, and sarcopenic obese; Levine & Crimmins, 2012). Investigators found that issues related to physical functioning were exacerbated in those with sarcopenic obesity, sarcopenic nonobesity, and nonsarcopenic obesity, and that these associations were mediated by insulin resistance but not by inflammation (Levine & Crimmins, 2012).
In a cross-sectional study involving 329 community-dwelling Italian older adults aged ≥75 years, Cesari et al. (2010) investigated the possible interaction of high-density lipoprotein cholesterol (HDL-C) on inflammation (erythrocyte sedimentation rate [ESR], CRP, and IL-6) and physical function as defined by ADL, instrumental ADL (IADL), and 4-m walking speed. In participants with normal HDL-C concentrations, only IL-6 showed a significant association with IADL. In participants with low HDL-C concentrations, all three inflammatory biomarkers were significantly associated with 4-m walking speed and IADL. IL-6 was also significantly associated with ADL, whereas borderline significances were reported for CRP and ESR (Cesari et al., 2010).
Brinkley et al. (2009) examined associations between physical functioning and inflammatory biomarkers in 542 older adults, enrolled in four clinical studies at Wake Forest University between 2001 and 2006, who were at least 55 years and had chronic obstructive pulmonary disease, congestive heart failure, high cardiovascular risk, or self-reported physical disability. Uniform clinical assessments were used across studies, including grip strength; a Short Physical Performance Battery (SPPB); inflammatory biomarker assays for IL-6, tumor necrosis factor alpha (TNF-α), and CRP; and anthropometric measures. Investigators found higher levels of CRP and IL-6, but not TNF-α, were associated with SPPB scores, independent of extraneous factors such as age, gender, race, disease status, as well as fat mass, lean mass, and percent body fat (Brinkley et al., 2009).
In Conclusion, the present study is among the largest to have examined the cross-sectional link between two biomarkers of chronic inflammation (CRP and fibrinogen) and physical functioning in older adults. Consistent with previous reports, we found that, regardless of sex, chronic inflammation may be linked to worse physical functioning and that the relationship between CRP (but not fibrinogen) and physical functioning may be modified by physician-diagnosed arthritis. Further research is needed to confirm these findings using longitudinal data and a larger sample size for the examination of interactions with other demographic, socioeconomic, and clinical characteristics.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
