Abstract
Aim
To investigate the efficacy of a new low-profile catheter on incidence of the catheter-associated urinary tract infections (CAUTI) in comatose patients admitted to the intensive care unit.
Background
Catheter-induced urothelial injury is a key component in the development of urinary tract infections in catheterized patients.
Methods
In this prospective randomized blinded clinical trial, 80 patients requiring indwelling urinary catheterization were equally randomized to either the standard Foley catheter (control) or the low-profile catheter (experimental) group. The signs of urinary tract infection for comatose patients were considered (ie, ≥105 of colony-forming unit/milliliter of urine, hematuria, serum leukocytes, and body temperature) and recorded at baseline and on days 3 and 5 after catheterization. The analysis of covariance was applied by the SPSS-20 software at a 95% confidence level.
Results
An increasing proportion of patients with elevated urinary colony counts were seen in the Foley catheter group compared with the low-profile catheter group (12.5% vs 5%). However, there were no between-group differences in the urinary colony counts and body temperature after controlling for antibiotic doses and fluid intake. Patients in the low-profile catheter group had significantly lower rates of hematuria and serum leukocytes than those in the Foley catheter group.
Conclusion
A newly designed low-profile urinary catheter has demonstrated a trend toward reducing the incidence of CAUTI in patients with indwelling urinary catheters. Further studies with larger sample sizes and follow-up are needed to confirm the benefits.
INTRODUCTION
Despite the efforts of limiting indwelling urinary catheter (IUC) insertion in determined cases and duration and fast removal, many patients still undergo urinary catheterization during their stay in the hospital,1,2 especially comatose patients 3 in intensive care units (ICUs).4–7
IUCs can lead to catheter-associated urinary tract infections (CAUTI) 8 in 70-80% of patients 9 within 48 h of catheterization. 1 The bacterial biofilm-based CAUTI is detected in 73.4% of patients with a mean duration of catheterization of 5.01 days. 10
IUCs change urodynamic and bladder epithelial defenses 2 and can provide a suitable environment for microbial biofilm formation which damages the urothelium. 11 If the urothelium is traumatized, for example, by the rounded closed tip of the catheter, host defenses are compromised and bacteria can invade the underlying tissues of the bladder resulting in colonization and proliferation.11,12
Using IUCs in comatose patients promote fast and continuous urine flow and bladder emptying. 3 However, this can lead to anatomical traumatization due to pressure on the catheter's tip when the bladder collapses after emptying. Negative pressure from fast voiding can also pull the inner bladder wall into the catheter's eyelets, leading to urothelial injury.3,13 Transurethral catheterization can also cause noninfectious complications such as urethral traumatization and hematuria. 4 To reduce urothelial damage, more attention needs to be given to the physical structure of urinary catheters. 14
To reduce the urothelium trauma resulting from the IUC structure, a bladder drainage system is designed with urine eyelets between the two balloons. The use of these dual-balloon urinary catheters compared to standard (Foley) catheters was able to reduce the incidence of CAUTI, although this reduction was not found to be significant. 15 However, the presence of two balloons complicates the process of catheter insertion and removal, occasionally leading to trauma and urinary tract infection, 16 and sometimes requiring invasive and non-invasive methods for removal. 17 In addition, this dual-balloon catheter tended to leave higher levels of residual urine in the bladder. 3
In 2015, a novel urinary catheter was designed to minimize catheter-induced urothelial trauma and improve drainage. The new catheter features a patented low-profile, open-ended rounded tip slightly above the doughnut-shaped balloon. This low-profile catheter was compared to the standard Foley catheter in ewes. Analysis of bladder images after 96 h of catheterization showed a 75% reduction in the percentage of damage and inflammation to the bladder with this new catheter compared to a Foley catheter. 13 Moreover, when the smaller-sized doughnut-shaped balloon was filled with 5 ml of water, there was no statistically significant difference in the force needed to forcibly remove it as compared with a Foley catheter filled with 10 ml in randomly assigned fresh female cadavers. 18 Building on these successes in reducing urothelial damage, the present study was designed with a primary outcome of evaluating the efficacy of the new low-profile urinary catheter in preventing of CAUTIs in comatose patients in ICU.
Methods
This prospective randomized controlled blinded clinical trial was conducted in the ICU in a hospital affiliated with a governmental Medical University. A prospective RCT allows patients to be followed over time, and prospective data collection allows for standardized collection of exposures and outcomes. To report the parallel group randomized trials, the CONSORT 2010 checklist was used (Supplement File 1).
All patients who were candidates for urinary catheterization and were admitted directly from the Emergency Department to the ICU, with a score of consciousness ≤9 on Glasgow Coma Scale (GCS), and 18 years or older, were screened for eligibility. Patients were included if they have had no recent history of UTI and use of aminoglycoside medications, and no history of diabetes, chronic kidney disease (CKD), and immune system diseases. Exclusion criteria were any planned or unplanned removal of the patient's urinary catheter before 3 days, patient's death, or transfer outside the ICU during the study.
The number of participants required in each study arm was about 40 (total = 80) based on a prior randomized trial, 19 at the level of 95% confidence, with 90% power, and 0.32 effect size.
Over the 10-month trial period (September 2019 to July 2020), all triaged patients prescribed catheterization by the Emergency Department physicians and were to be admitted to the ICU (n = 283), were assessed for eligibility. Of those meeting all eligibility criteria (n = 129), 80 patients completed the study protocol (Figure 1).

CONSORT 2010 flow diagram.
Patients were allocated to the standard Foley catheter (ie, control) group or the low-profile catheter (ie, experimental) group by a random, computer-generated randomization schedule (https://www.sealedenvelope.com). Patients’ masking was preserved because they were comatose.
The ICU nurses who took the patient's temperature were unaware of the patient's group assignment because the externally visible parts of the two types of catheters were similar and the only difference between catheters was the internal part inserted into the bladder. The laboratory specialists and the data analyst were blinded to the participants’ study group as well.
Urine sampling was sterilely collected through the tubing drainage port. Blood samples were obtained following standard sterile procedures. All samples were collected by laboratory specialists in the evenings immediately following (baseline) catheterization and on the third day and the fifth day after catheterization. The samples were analyzed at the same participating Hospital Clinical Laboratory.
Data on participants’ demographic and clinical characteristics were collected using a validated checklist 19 to review their medical records upon enrollment in the study.
In this study, the primary outcome was the incidence of CAUTI that was defined as a positive urine culture (≥105 CFU/ml) with urine-specific microorganisms isolated,1,19,20 positive microscopic hematuria, 21 and the presence of a fever ≥38° 22 after 2 days of catheterization.1,12,20,23 Because our patients were unconscious, they could not explain other UTI symptoms such as dysuria, frequent urination, urgent urination, and flank pain. Secondary outcomes included urinalysis and hematological parameters compared between the two groups of study.
Catheterization of the two groups of study patients was performed according to the standards of care and under aseptic conditions by the trained nurses. All patients in both groups were provided otherwise standard care for their medical conditions. The urinary catheters were either an AMSure® 2-Way Standard Tip 5 cc Balloon 16 Fr. Silicone Foley Catheter (Amsino International, China) or the low-profile Kohli Atraumatic 2-Way 5cc Balloon 16 Fr. Silicone Catheter (Emmy Medical, LLC USA). The remarkable difference was in the tip shape and size of the balloon as shown in Figure 2. Both catheters were attached to a McKesson Anti-Reflux Valve Sterile 2000 ml Vinyl Urinary Drainage Bag (McKesson Inc. USA).

16 Fr. Foley catheter (left); 16 Fr. low-profile catheter (right).
Various statistical tests were used to examine differences in patients’ demographic and clinical characteristics between the two groups. These tests included the t-test, Chi-square test, Fisher's exact test, or Fisher-Freeman-Halton test (for 2 × 3 contingency tables). The primary outcome of interest was urinary colony counts, and the Chi-square test and Cochran's Q test were employed to assess between-group and intragroup differences in urinary CFU/ml, respectively. A logistic regression was employed to evaluate the variation in our primary outcome between the groups over time adjusted for intravenous fluid therapy and prophylactic antibiotics as specific covariates. To compare intergroup differences in patients’ axillary body temperatures (°C), a repeated measures analysis of variances (RM-ANOVA) was conducted considered intravenous fluid therapy and prophylactic antibiotics as covariates. Data analysis was performed using SPSS software version 20.0 with a confidence interval of 95%.
The Research Ethics Committee at Hamadan University of Medical Sciences (ID: IR.UMSHA.REC.1397.766), approved the trial protocol in accordance with the Declaration of Helsinki. The trial was registered by Iranian Registry of Clinical Trials: http://en.irct.ir/trial/37227 (ID: IRCT20100816004578N7). Written informed consent was signed by each comatose patient's legal guardian.
Results
The demographic and clinical characteristics of participants were similar between groups (Table 1).
Comparison of the Patients’ Demographic and Clinical Characteristics Between the Two Groups (n = 80).

GCS = Glasgow Coma Scale (GCS); TBI = traumatic brain injuries; Polytrauma-TBI = a concurrent injury to the brain and one or more body areas. 24 HTN = hypertension; CAD = coronary artery diseases; IV fluids = intravenous fluid.
t-test.
Chi-square test.
Fisher–Freeman–Halton test.
Fisher's exact test.
The results related to urine CFU/ml for patients in both over time are shown in Table 2. There were more positive urine cultures on day 5 in the Foley catheter group (n = 5; 12.5%) as compared to the low-profile group (n = 2; 5%), but the result of the logistic regression analysis demonstrated that the risk of CAUTI in both groups was not significantly different (odds ratio, 3.22; p = 0.208). The bacteria species were similar in both groups (Table 2). The results of the analysis of covariance showed no significant differences between groups’ body temperature after controlling for the effects of antibiotic doses, and serum volume (Table 2).
Comparing Differences in Primary Outcomes of Trial Between the 2 Groups of Patients (n = 80).
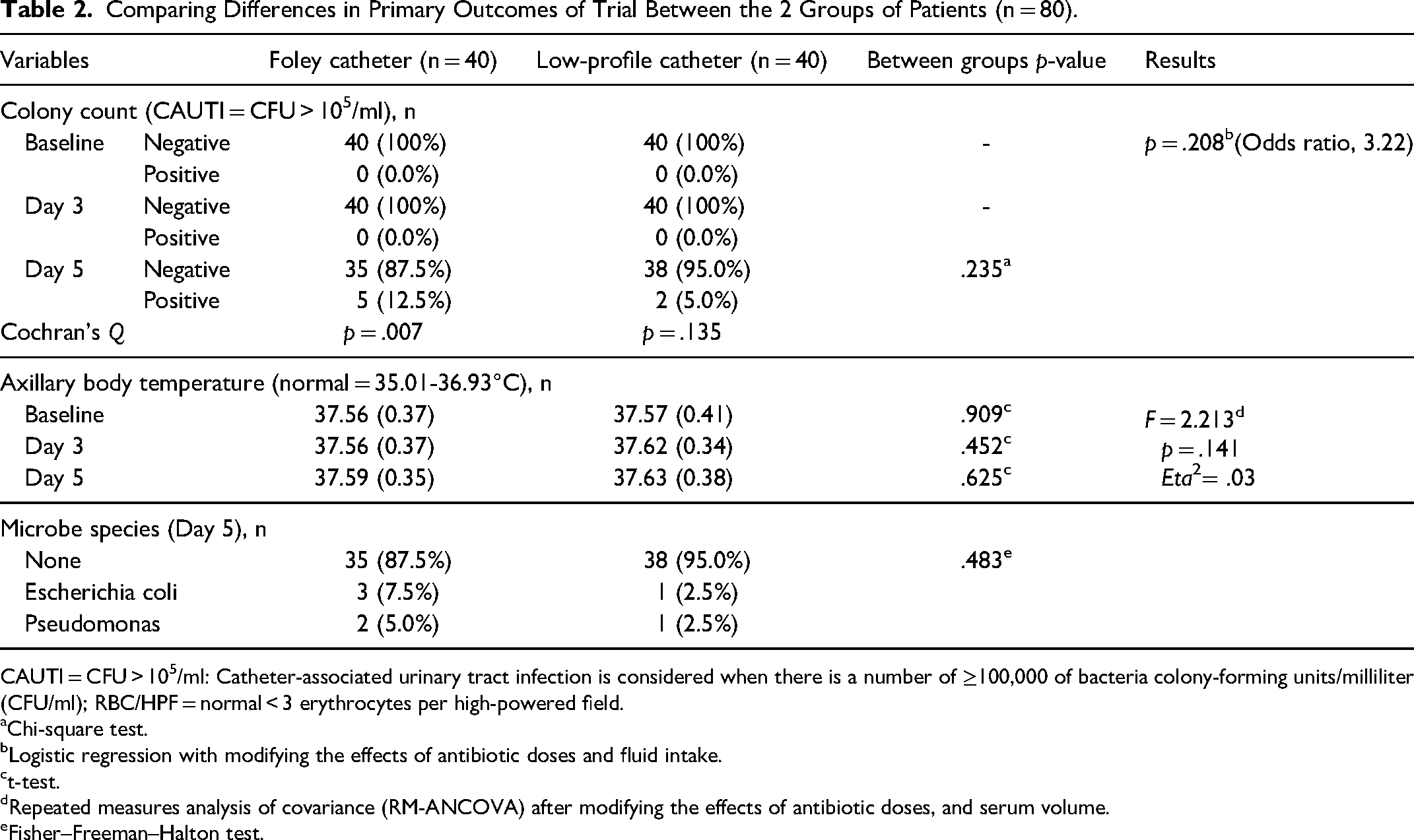
CAUTI = CFU > 105/ml: Catheter-associated urinary tract infection is considered when there is a number of ≥100,000 of bacteria colony-forming units/milliliter (CFU/ml); RBC/HPF = normal < 3 erythrocytes per high-powered field.
Chi-square test.
Logistic regression with modifying the effects of antibiotic doses and fluid intake.
t-test.
Repeated measures analysis of covariance (RM-ANCOVA) after modifying the effects of antibiotic doses, and serum volume.
Fisher–Freeman–Halton test.
A significant reduction (p < 0.05) in the associated proportions of hematuria was reported only in the low-profile catheter group. On day 5, we had six patients with normal urine RBCs versus two patients with normal urine RBCs in the Foley catheter group (Table 3). However, there were no statistically significant between-group differences in any of the other urinary parameters at baseline or at either follow-up visit.
Comparison of Differences in Urinalysis Parameters Between the Two Groups (n = 80).

RBC = erythrocytes; HPF = high-powered field; WBC = leukocytes; EP = epithelial cell.
Chi-square test.
Fisher's exact test.
Fisher–Freeman–Halton test.
After controlling for the effects of antibiotic doses and serum volume, the counts of WBCs in the blood sampling were significantly lower (p < 0.05) in the low-profile catheter group than in the Foley catheter group (Table 4). There were no statistically significant differences in all other blood parameters between the two groups.
Comparison of Differences in Hematological Parameters Between the Two Groups (n = 80).

WBC = leukocytes.
Intergroup comparisons = All continuous variables are compared using the t-test.
Repeated measures analysis of variances (RM-ANOVA) after modifying the effects of baseline measure, antibiotic doses, and serum volume.
Discussion
The results of this clinical trial present significant insights into the effectiveness of low-profile urinary catheters compared to standard Foley catheters in preventing CAUTIs in comatose patients. The study design was influenced by previous research in the field. Specifically, one study assessed the efficacy of bladder irrigation with latex Foley catheters in reducing CAUTIs in 60 comatose ICU patients, with measurements taken immediately after catheter insertion (baseline) and on day 5 of catheterization. 19 Our patients’ demographic characteristics and clinical conditions were comparable with a previous study. 19 In our study, we introduced a newly designed silicon catheter for 80 comatose patients in ICU and implemented two additional follow-up assessments, on days 3 and 5 of catheterization.
The incidence of positive urine cultures on day 5 was approximately two and a half times higher in the Foley catheter group compared to the low-profile group (12.5% vs 5%). However, after adjusting for administered fluids and antibiotics, the difference in CAUTI incidences between the two groups did not reach statistical significance. This finding is clinically significant due to the substantial additional cost incurred by each nosocomial infection in the ICU ($11,475 or €10,035.24). 25
Despite the promising nature of this observation, the lack of a significant difference between groups may be due to the relatively short follow-up period of 5 days after catheterization. It is worth noting that a previous study confirmed the CAUTI-reducing efficacy of noble metal alloy catheters after 3 days. 26 The Centers for Disease Control and Prevention and the European Centre for Disease Prevention and Control have also emphasized that CAUTIs can occur within 48 h of catheterization.1,20 Given the significant impact of ICU length of stay on CAUTI rates,11,27 even the development bacteriuria by 3-7% per day of catheterization, and the formation of mature biofilms on IUCs in two weeks, 28 further studies with longer follow-up are recommended to confirm the efficacy of low-profile catheters in preventing CAUTIs.
Hematuria is a side effect of catheterization due to urothelial injuries. A significant reduction in the number of patients with hematuria was reported in the low-profile catheter group only (p < 0.05). However, no significant difference was found between the two groups’ urine erythrocyte deposits. This may reflect the reduction of urothelium trauma due to less physical contact with the low-profile catheter balloon. This finding is consistent with results showing significant reductions in the percentage of ulceration and inflammation of the urothelial layers with the use of the low-profile catheter. 13 However, further studies are needed to support this hypothesis.
Although the blood WBC deposits were similar at the baseline, a significant reduction in WBCs was found in patients with the low-profile group over 5 days, after adjusting the effects of administrated fluid and antibiotics (11 vs 9 K). The observed decreased rate may be associated with reduced inflammation resulting from either the use of the low-profile catheter or expected improvement in injury and inflammation. Further studies are needed to determine factors associated with the change in WBCs in the short and the long term.
In both groups, patients’ axillary body temperatures were similarly higher than normal at the entrance and during the study which seems to be related to the patient's conditions as head trauma or multiple trauma rather than the CAUTI. No significant between-group differences were found in patients fevers after adjusting for the administrated fluid and antibiotic agents. Fevers may be a nonspecific symptom of CAUTI in ICU patients 19 and may be associated with other patient conditions 6 or neurogenic factors such as stroke. 29 The present trial was carried out in a trauma center where the patients suffered from head trauma or multiple trauma, which may explain the early onset and maintenance of high temperature.
Limitations
Although the prospective, randomized, controlled, blinded clinical trial design enhanced the validity and broader applicability of our study, there are some limitations. Our small sample size, a certain follow-up period of 5 days, and the selection of patients without a history of disease may have reduced the ability to detect statistically significant differences between the groups despite the notable clinical differences observed.
Conclusions
A newly designed low-profile urinary catheter has demonstrated a trend toward reducing the incidence of CAUTI in patients with IUCs. Further studies with larger sample sizes and follow-ups in diverse hospital populations are needed to confirm the benefits.
Supplemental Material
sj-doc-1-hme-10.1177_23333928231211410 - Supplemental material for Evaluating the Potential of a New Low-Profile Urinary Catheter in Preventing Catheter-Associated Urinary Tract Infections: A Prospective Randomized Blinded Clinical Trial
Supplemental material, sj-doc-1-hme-10.1177_23333928231211410 for Evaluating the Potential of a New Low-Profile Urinary Catheter in Preventing Catheter-Associated Urinary Tract Infections: A Prospective Randomized Blinded Clinical Trial by Farahnaz Ramezani, Mahnaz Khatiban, Farshid Rahimbashar, Ali Reza Soltanian, Seyed Habibollah Mousavi-bahar and Ensieh Elyasi in Health Services Research and Managerial Epidemiology
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vice Chancellor for Research and Technology, Hamadan University of Medical Sciences (grant number 9805013424).
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
