Abstract
During the last decade, there has been a move towards consumer-centric hearing healthcare. This is a direct result of technological advancements (e.g., merger of consumer grade hearing aids with consumer grade earphones creating a wide range of hearing devices) as well as policy changes (e.g., the U.S. Food and Drug Administration creating a new over-the-counter [OTC] hearing aid category). In addition to various direct-to-consumer (DTC) hearing devices available on the market, there are also several validated tools for the self-assessment of auditory function and the detection of ear disease, as well as tools for education about hearing loss, hearing devices, and communication strategies. Further, all can be made easily available to a wide range of people. This perspective provides a framework and identifies tools to improve and maintain optimal auditory wellness across the adult life course. A broadly available and accessible set of tools that can be made available on a digital platform to aid adults in the assessment and as needed, the improvement, of auditory wellness is discussed.
Introduction
Auditory wellness is conceived here as the effective processing of sound that enables the individual to fully participate in a wide range of desired daily listening activities without limitations or restrictions. The specific listening and communication activities that are desirable and deemed important are determined by the individual and often include communication with others, listening to music, and an appreciation of the sounds of nature, among others. For the first time in the past century, adults with nonmedically treatable hearing loss have been afforded the opportunity to assess and manage their own auditory wellness, including the acquisition of over-the-counter (OTC) hearing aids, without the need to consult with healthcare professionals for assessment or treatment. This option grew from a decade-long movement toward affordable and accessible consumer-centric hearing healthcare. Technological advancements (e.g., the merger of consumer-grade hearing aids with consumer-grade earphones creating a wide range of hearing devices) and policy changes (e.g., the U.S. Food and Drug Administration has created a new OTC hearing aid category; National Archives, 2022) have facilitated this re-direction of hearing healthcare for adults with perceived mild-to-moderate hearing difficulties.
Although the door to this new self-managed pathway to improved auditory wellness has been opened, few tools have been provided to adults with less-than-optimal wellness to assist them along this pathway. Several validated tools for the self-assessment of auditory function, detection of ear disease, and education about hearing loss, hearing devices, and communication strategies exist, but have not been made easily available to a wide range of people. Thus, the purpose of this article is to provide a framework and tools to assist those adults with less-than-optimal auditory wellness on their journey to achieve the highest possible auditory wellness. The set of tools to enable the adult to self-assess auditory wellness and, if needed, to improve auditory wellness can be made broadly available on a digital platform. These tools are described and discussed here.
Before describing auditory wellness in more detail and the tools available to self-manage auditory wellness, this perspective begins with an overview of general information about health, wellness, and well-being. The main goal is to present a framework for the maintenance of auditory wellness that integrates several tools validated for different purposes (e.g., detection of ear disease, measuring the level of hearing disability, improvement strategies, etc.) to facilitate the achievement of optimal lifetime auditory wellness. After this general overview of health, wellness and well-being, the remainder of this perspective is divided into three main sections which: (a) describe the importance of auditory wellness to other wellness domains and well-being, including a comparison of auditory-wellness and hearing-wellness concepts; (b) provide a rationale for moving away from auditory impairment and toward auditory wellness; and (c) outline an approach to maintaining auditory wellness throughout the adult life course. The latter section proposes one of many ways in which such an auditory-wellness self-management system could be realized. The proposed system is U.S.-centric in that the availability of OTC hearing aids is assumed to be an option among the intervention alternatives, although with slight modification this approach can be relevant to other areas and contexts as most of the world has access to direct-to-consumer (DTC) hearing devices (e.g., hearables).
General Overview of Health, Wellness, and Well-Being
Over the past half century, high-resource countries have focused on maintenance of high well-being, good quality of life, or attainment of a thriving life as the goal for all members of society. More recently, organizations, businesses, communities, states, and nations have even branded and marketed their positive well-being (Grénman, 2019; Grénman et al., 2019), recognizing its importance to individuals as well as society at large.
Well-being is synonymous with good quality of life and happiness (Diener, 1984; Diener et al., 1999). Prior to this focus on well-being, societies, often of necessity, were preoccupied with the prevention of disease, illness, and death. This changed to a focus on well-being in the mid-20th-century for many nations and is reflected in the constitution of the World Health Organization (WHO; Miller, 2005; Oliver & Baldwin, 2018). Specifically, the 1948 WHO constitution defined health as “a state of complete physical, mental and social well-being and not merely the absence of disease or infirmity.” Reflecting the importance of well-being to society, measures of national well-being were developed and proposed (e.g., Kahneman et al., 2004). Increasingly, when available resources allowed, both high- and middle-resource countries shifted their focus to wellness and away from health as the absence of illness.
Wellness, a part of attaining a thriving well-being, describes a healthy lifestyle, one which enables people to do what they want in life without suffering or restriction (Pendell, 2021). “Wellness” is sometimes used in a broader sense, akin to well-being, which has created some confusion (Kauppi et al., 2023). Wellness, however, is probably most often used in a somewhat narrower health context and is considered a part of well-being. Whereas individual wellness is actively caring for oneself to achieve a thriving well-being, both a community's wellness and the individual's wellness influence the state of well-being (https://www.unh.edu/health/wellbeing-wheel). A terminally ill cancer patient, for example, can have poor wellness but surrounded by family and friends and satisfied with life, could still have a very positive well-being. Likewise, an individual may maintain high wellness across several domains but be hampered in achieving a thriving well-being due to poor community wellness.
Travis (1972) illustrated the important conceptual shift from health as illness prevention to health as attainment of high-level wellness (Figure 1). From the inception of the wellness movement, one focusing on health as wellness rather than lack of illness, adults were made responsible for achieving their own high-level wellness (Dunn, 1959, 1961). As shown in Figure 1, Travis (1972) emphasized this through the critical importance of self-actualization to the attainment of high-level wellness, with self-actualization realized through awareness, education, and personal growth.

Model of wellness continua from Travis (1972) as presented in Miller (2005).
Although objective measures of well-being (number of positive and negative life events, annual income, etc.) and wellness (measures of autonomic nervous system function, levels of various hormones, etc.) have been described or suggested (e.g., Oliver & Baldwin, 2018), most measures of both constructs have been based on self-reports. This is because well-being and wellness are defined individually as perceived by the person, varying across individuals and time (Grénman, 2019; Kauppi et al., 2023; Linton et al., 2016; Naci & Ioannidis, 2015; Oliver & Baldwin, 2018). In many conceptualizations, one has an image of an ideal self and well-being is assessed by evaluating the current self against that ideal. The closer the match, the higher the perceived well-being and satisfaction with life. As a result, well-being is inherently subjective and is unique to an individual. Most models of well-being, however, include measures of positive and negative affect together with a measure of life satisfaction (Diener, 1984).
A myriad of models of well-being and wellness have been developed over the past several decades based on analyses of self-report measures. Systematic reviews of measures of well-being have revealed numerous scales grounded in a wide range of theories (Linton et al., 2016), including, unfortunately, considerable confusion in the definition of well-being. Nonetheless, most self-report measures of well-being tap multiple dimensions assumed to underlie the complex psychological constructs of positive and negative affect, and life satisfaction. For wellness, a recent systematic review by Kauppi et al. (2023) identified 44 models of self-reported wellness that described 379 domains clustered into 14 themes. They noted that the most commonly occurring domains among these 44 models were centered on six themes addressing physical, psychological, social, emotional, spiritual, and environmental wellness. Figure 2 illustrates the connection of each wellness domain to overall well-being. Auditory wellness (AW) is also shown in this schematic illustration. As discussed in more detail below, research has demonstrated many connections between auditory wellness and emotional, social, psychological, and physical wellness. Auditory wellness is conceptualized in Figure 2 as being linked to several domains of wellness, each of which has direct connections to well-being, as well as a separate domain with its own direct connections to well-being (Humes, 2021b).

The primary domains of wellness, as identified by Kauppi et al. (2023), are shown interconnected with overall well-being. Auditory wellness (AW) is viewed as another form of wellness with direct links to well-being as well as indirect links to well-being through other domains of wellness. The inset at the bottom is from Figure 4.
High-level wellness in multiple domains is considered critical to a thriving well-being, that is, achieving a well-lived or “happy” life. It was out of this context of health as wellness that the WHO developed their well-known and widely applied framework of healthy function, the International Classification of Functioning, Disability and Health that is often referred to as the ICF (WHO, 2001). The ICF framework is based on the more comprehensive bio-psycho-social model of functioning, disability, and health. As noted, the WHO constitution focuses on physical, mental/psychological, and social well-being (WHO, 1948). Here, as noted, we consider overall subjective well-being to be a unitary construct shaped by wellness in each of several domains (Figure 2). Within this framework then, the “physical, mental/psychological, and social well-being” in WHO (1948) are recast as “physical, mental/psychological, and social wellness” with each contributing to a thriving well-being.
The basic components of the ICF framework indicate that optimal wellness in each domain can be attained by minimizing the effects of three factors that limit the realization of one's ideal self: (a) bodily impairments; (b) activity limitations; and (c) participation restrictions. Optimal wellness is achieved through absence of bodily impairments, limitations on desired activities, and restrictions on participation in society. Each of these three aspects of healthy function or wellness is modulated by contextual factors in the WHO (2001) model, such as environmental and personal factors. According to the WHO-ICF framework (see Figure 3), the interaction between disease and wellness is not straightforward and can be influenced by internal biological factors as well as external contextual factors (Manchaiah & Stephens, 2013). For instance, the same disease may cause different levels of impairment that could result in different levels of activity limitations and participation restrictions, ultimately resulting in variations in the individual's state of wellness. Likewise, those with the same disease and level of impairment may experience different impacts on daily activities and participation in society. Various environmental and personal contextual factors such as personality, mental state, social support, and financial resources play a key role in influencing this interaction. It is noteworthy that this framework recognizes that people could experience reduced wellness in a domain and less-than-optimal well-being in the absence of disease or impairment. Alternatively, people with a disease or impairment could experience limited or no reduction in subjective well-being. Further, poor wellness and well-being could exacerbate impairments. Consider obesity as an example. Obesity can lead to poor physical wellness which, in turn, limits physical activity, resulting in loss of muscle mass, possible diabetes, or other additional physical impairments. Hence, this framework makes a strong case for moving away from the standard medical model focused solely on treating disease and resulting impairment, and instead focuses efforts on optimizing well-being through optimized wellness.

Modified WHO-ICF model showing interaction between disease, impairment, disability, participation restrictions, and wellness (modified from Manchaiah & Stephens, 2013).
Despite several decades of recognition of the importance of wellness to well-being for members of society, medical professionals have long continued to focus on illness prevention rather than wellness optimization (Naci & Ioannidis, 2015). The illness-driven medical model has been supported in the United States by the most prevalent system of third-party reimbursement based on fee-for-service payments rather than the quality of the patient's outcomes resulting from those services. Although the Affordable Care Act (ACA) of 2010 incentivized shifts in the reimbursement structure from fee-for-service to models that are value driven, implementation of value-based reimbursement systems has been slow with nonoptimal results to date (McDonough & Adashi, 2022; Smith, 2021). Moreover, per the ACA, the focus on healthcare value and quality has been at the institution or healthcare-system level, not at the patient level. Ideally, the ultimate outcome for the patient in a high-quality, high-value, healthcare system would be improved wellness and well-being. At least a part of the challenge in transitioning to a system focused on the wellness of the individual has been the lack of efficient, valid, and reliable tools to assess wellness and well-being in a clinical setting (Bart et al., 2018).
From this broader context for wellness and well-being, the remainder of this article focuses on the concept of auditory wellness. Methods for measuring auditory wellness and maintaining auditory wellness to facilitate the individual's quest for optimal well-being over the adult life course are described.
The Importance of Auditory Wellness to Other Wellness Domains and Well-Being
In the listening hierarchy of Erber (1982), the perceptual processing of sound by human listeners is complex and involves much more than the detection of pure tones as quantified by the audiogram. Kidd et al. (2007), for example, evaluated the performance of 340 young adults with normal hearing on 19 sound-discrimination and sound-identification tasks. A wide range of sounds and tasks were examined with four auditory abilities identified: (a) loudness and duration (energy) discrimination; (b) detection of temporal envelope variation; (c) identification of familiar sounds (speech and nonspeech); and (d) discrimination of unfamiliar simple and complex spectral and temporal sound patterns. Importantly, sound-localization abilities in space and musical abilities were not examined in detail by Kidd et al. (2007) and could increase the number of dimensions or abilities underpinning listening and auditory perception. Nonetheless, auditory perception by human listeners clearly is multidimensional.
The detection of pure-tone stimuli (the audiogram) represents an important pre-requisite for the hearing of sounds but captures only a small part of broader auditory perception and listening. Excellent auditory perception requires a healthy auditory periphery from the outer ear through the cochlea, a system that is at least partially evaluated by pure-tone audiometry, healthy higher-level auditory processing from the auditory nerve to the auditory portions of the brainstem and cortex, and, frequently, healthy cognitive and linguistic processing. The systems involved in auditory perception from the periphery through the cortex, moreover, are often intertwined. Performance on measures of higher-level auditory processing probably best reflect this with performance on these measures often confounded by deficits in peripheral auditory function, cognition, or both (e.g., Davidson & Souza, 2024; Gallun et al., 2020; Humes, 2021c; Humes et al., 2012). As a result, auditory wellness was chosen here over “hearing wellness” to better reflect the full breadth of auditory perception and listening. This choice is supported in more detail in the next section.
Auditory wellness, however, is more than auditory perception. Sound is the basis for much of the communication among human beings, not just messages exchanged between the talker and the listener, but indexical and prosodic information that may identify the participants’ gender, age, emotional state, and ethnicity, among other things. Auditory/oral communication is critical to social wellness too. Thus, maintaining good auditory wellness throughout life is important for the maintenance of high physical, psychological, social, and emotional wellness, as was previously illustrated schematically in Figure 2. This is reflected in numerous research studies that found associations between the most basic form of auditory function, detection of pure tones, and physical health, including falls (Jiam et al., 2016; Martinez-Amezcua et al., 2021), psychological wellness, especially anxiety, depression and fatigue (Cosh et al., 2019; Pichora-Fuller et al., 2016), social wellness (Mick et al., 2014; Timmer et al., 2023), and cognitive function (Amieva & Ouvrard, 2020; Livingston et al., 2020).
The Case for “Auditory Wellness” Over “Hearing Wellness”
Pure-tone audiometry has been adopted as the preferred measure of “hearing wellness” in the recently adopted Consumer Technology Association (CTA) national standard, ANSI/CTA-2118 (2023). Consistent with WHO (2021) criteria for normal hearing, the CTA standard describes good hearing wellness as having PTA4, the average hearing thresholds at four frequencies (500, 1,000, 2,000 and 4,000 Hz), in the better ear < 20 dB HL. Good “hearing wellness,” however, is not equivalent to good “auditory wellness.” Many individuals without significant pure-tone hearing loss report hearing trouble or reduced auditory wellness (e.g., Humes, 2021a, 2023a; Ventry & Weinstein, 1982, 1983). Humes (2021a), for example, analyzed a large dataset of clinical and population data from almost 11,000 adults 50 to 89 years of age that included PTA4, and a self-report hearing handicap inventory (HHI), and found an overall correlation between the PTA4 and HHI score of r = 0.6. This represents an overlap of 36% between the audiometric PTA4 and the self-reported HHI score, the latter representing auditory wellness and its connection to social and emotional wellness (Humes, 2021a). Essentially, Humes (2021a) argued that auditory wellness includes measures like the CTA's PTA4-based measure of hearing wellness, but also encompasses many more aspects of sound perception and listening that are critical to social, emotional, psychological, and physical wellness as shown previously in the conceptual model in Figure 2. This conceptualization of the connection between hearing wellness and auditory wellness is illustrated schematically in Figure 4. Importantly, hearing wellness is just a part of auditory wellness, albeit an important part, with about one-third overlap between them as noted previously.

Conceptual model of auditory wellness which shows the multiple dimensions of auditory perception and listening. Note that “hearing wellness,” based on pure-tone audiometry, is one component of “auditory wellness.”
Among older adults, generally 50 years of age and older, it has been well documented that those with good “hearing wellness” (or the absence of pure-tone hearing loss) can experience deficits in many aspects of auditory processing. The review of 132 peer-reviewed research articles by the American Academy of Audiology's Task Force on Central Presbycusis (Humes et al., 2012) concluded that for measures of speech understanding, a total of 76 studies, “…significant negative effects of age, unconfounded by hearing loss, were observed in most of the studies of speech in competing speech, time-compressed speech, and binaural speech perception…”. Further, for 36 studies of mainly temporal processing of non-speech stimuli, “…hearing loss was seldom a significant factor … and negative effects of age were almost always observed.” In summary, as auditory wellness is conceptualized in Figure 2, good “hearing wellness” based on PTA4 < 20 dB HL does not guarantee good auditory wellness. There are age-associated changes in auditory processing that appear to be independent of hearing loss and can diminish auditory wellness.
In many of the 132 studies reviewed in Humes et al. (2012), the way in which the presence of absence of hearing loss was described varied markedly across studies. In some cases, this was based on correlational analyses which included pure-tone threshold as a continuous variable and, in others, groups were compared with differences in hearing thresholds, including both some with hearing loss and others with normal hearing. Exactly how normal hearing was defined varied considerably as well. As a result, data from an extensive laboratory-based study of auditory perceptual processing in older adults published previously by Humes et al. (2013) were reexamined here. Importantly, controls were implemented in that study to minimize the role of audibility on any of these tasks. Consistent with the 2023 CTA standard and WHO (2021), normal hearing or good hearing wellness was defined as test-ear PTA4 < 20 dB HL. Among the 245 young, middle-aged, and older adults in that study, 130 had PTA4 < 20 dB HL.
Figure 5 shows the thresholds obtained for each of three age groups and for each of six measures of auditory temporal processing. The values plotted in Figure 5 are the boot-strapped (N = 1000) means and 95% confidence intervals for each measure and age group. The two leftmost temporal-processing measures are gap-detection thresholds for a noise band centered at either 1000 or 3500 Hz. The remaining four tasks in Figure 5 all made use of four brief vowel segments and the task in each case was the correct identification of the sequence presented. For two tasks, MonoTO2 and MonoTO4, the vowel sequence was presented to the same ear and was comprised of 2 or 4 vowels, respectively. For the right-most measures in Figure 5, the stimuli were presented dichotically, different sounds to each ear, with the identification of the vowel sequence required for DichTOsylID and the identification of the first ear stimulated (right or left) for DichTOsylLOC. The asterisks mark significant effects (all three significant F > 5.3, p < .01) and Bonferroni-adjusted post hoc t-tests found the young group to have significantly lower temporal-processing values than both the other age groups (MonoTO2, DichTO sylID) or just the older group (MonoTO4). There were no significant age-group differences for gap detection and none for the dichotic location/ear task. Only the three temporal-processing tasks that used sequences of brief speech stimuli showed significant differences with one or both older age groups showing poorer temporal-processing of speech sequences than young adults. Recall that all subjects in these analyses had good hearing wellness (PTA4 < 20 dB HL). These reanalyses of the data from Humes et al. (2013) further establish that good “hearing wellness” does not mean good “auditory wellness.” This may be especially true for older adults. Cross-sectional (Fischer et al., 2017, 2019) and longitudinal (Dillard et al., 2020) population studies have also found age-related declines in older adults for dichotic-digit identification independent of the effects of hearing loss.

Boot-strapped means and 95% confidence intervals for measures of auditory temporal processing for each of three age groups. Data from Humes et al. (2013) but for PTA4 < 20 dB HL only.
It is well known that aging results in declines in many aspects of cognition, especially process-related cognitive measures (e.g., Salthouse, 2010). This is true even in adults with very little audiometric hearing loss (e.g., Golub et al., 2020). It has also been noted that performance on complex perceptual tasks with complex stimuli, including those used in Humes et al. (2013) and shown in Figure 5, can be associated with individual differences in cognitive processing (Davidson & Souza, 2024; Humes et al., 2013, 2022; Lentz et al., 2022). Thus, it is not possible to identify the causes underlying the age-group differences in temporal processing shown in Figure 5. Regardless of the cause, however, having good hearing does not ensure good processing of sequences of speech sounds, especially for older adults.
How many individuals with good “hearing wellness” based on PTA4 have reduced auditory wellness? The large dataset of almost 11,000 adults analyzed by Humes (2021a) was further explored to estimate the prevalence of reduced auditory wellness among those with PTA4 < 20 dB HL. The top panel of Figure 6 provides the results of these additional analyses. In that dataset, there were 3,396 adults 50 years of age and older for whom the better-ear PTA4 was less than 20 dB HL; that is, who had good “hearing wellness.” Roughly, one-third of those in the dataset with good “hearing wellness” had poor “auditory wellness” as defined by the self-reported HHI score. If this dataset was restricted to just the population data in Humes (2021a), excluding the clinical datasets for which poor auditory wellness may be expected to be more commonplace, the results from the largest of the population datasets, the Blue Mountains Eye Study (BMES; Sindhusake et al., 2001), also shown in the top panel of Figure 6, reveal that about 20% of those with good hearing wellness had less-than-optimal auditory wellness based on the HHI. The top panel of Figure 6 also indicates that the prevalence of poor auditory wellness among those with good hearing wellness is fairly constant from 50 through 79 years of age.
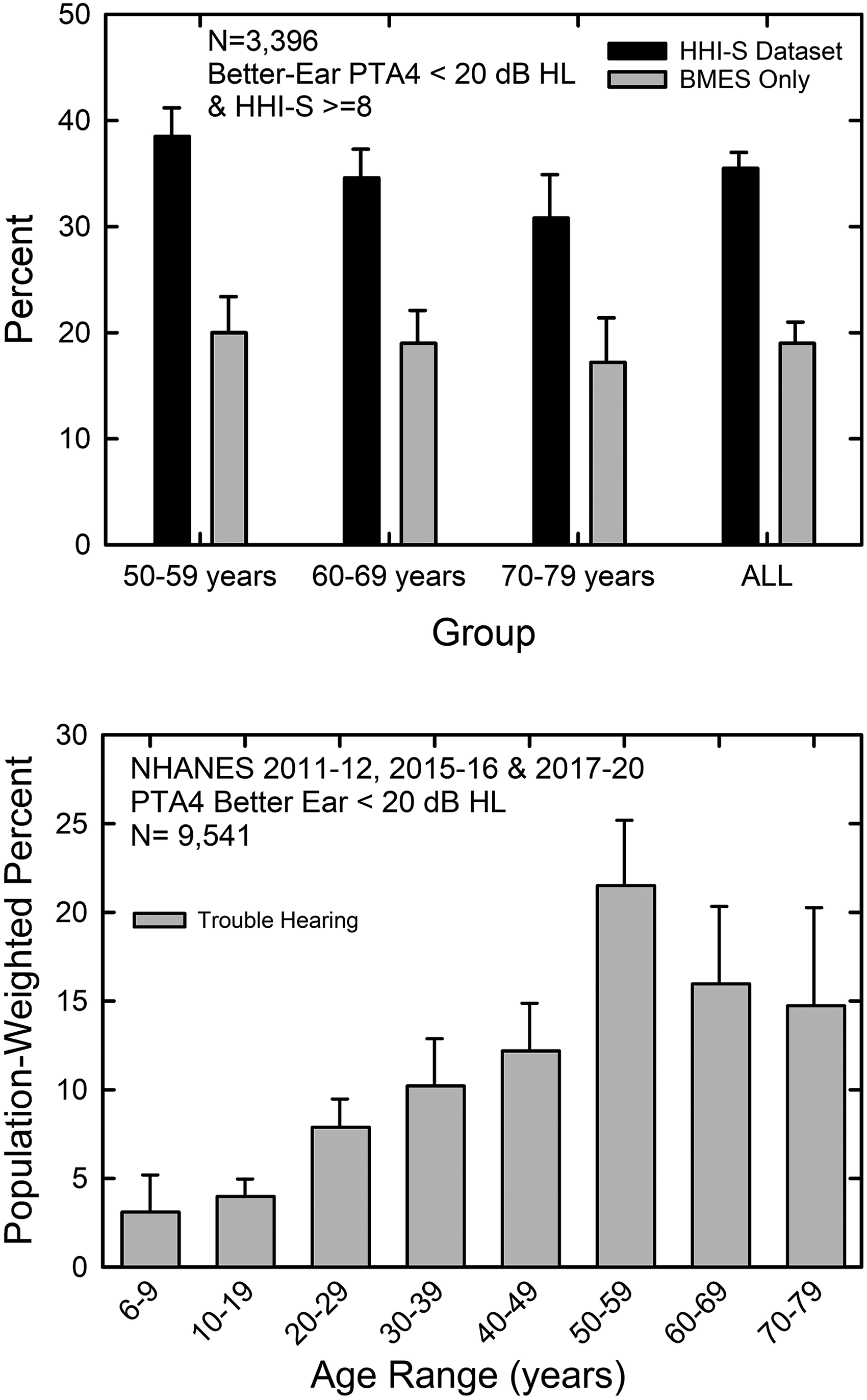
Prevalence estimates for reduced auditory wellness among those with good hearing wellness based on a large HHI dataset (Humes, 2021a), top, and NHANES data (bottom).
The lower panel of Figure 6 shows similar data from the National Health and Nutrition Examination Survey (NHANES) for 2011–2012, 2015–2016, and 2017–2020. These are the three most recent NHANES surveys that included audiograms. The HHI was not included in NHANES but a general question about hearing was included in each of the surveys. This question asks whether “…you would say that your hearing is excellent, good, or that you have a little trouble, moderate trouble, a lot of trouble, or are deaf?” Responses to this question for the same NHANES surveys have been analyzed previously (Humes, 2023a) and the functional significance of responses of “a little trouble” or greater have been validated against hearing-related limitations in everyday life (Humes, 2023b). In addition, the correlation between PTA4 and trouble hearing in NHANES was 0.6 (Humes, 2023a), again reflecting about one-third overlap between “hearing wellness” (PTA4) and “auditory wellness” (self-reported trouble hearing).
The prevalence of those having at least a little trouble hearing among those with better-ear PTA4 < 20 dB HL from the NHANES surveys is shown in the bottom panel of Figure 6 for each of several age groups from 6 to 79 years of age. The prevalence of hearing trouble among those with good hearing wellness increases steadily with advancing age, reaching a maximum of about 20% for 50–59-year-olds and then declining somewhat to about 15% for 60–79-year-olds. Note that for the same age range as the population BMES data in the top panel of Figure 6, the NHANES data show similar prevalence rates for hearing trouble or reduced auditory wellness of about 15–20% among those with good hearing wellness.
Using population data from the Beaver Dam, Wisconsin community, Tremblay et al. (2015) reported an overall prevalence of reduced auditory wellness (based on just four items from the HHI) of 12% among those with normal hearing. In that case, however, “hearing wellness” was defined as all pure-tone thresholds from 500 through 8,000 Hz < 20 dB HL in both ears. The stricter the criterion for normal hearing, the lower the prevalence of those with reduced auditory wellness among those with normal hearing. For example, for the NHANES 2011–2012 and 2015–2016 surveys for 20–69-year-olds, when the same strict criterion for normal hearing used by Tremblay et al. (2015) was applied, only 6.3% (95% CI [5.2%, 7.5%]) of those with normal hearing have self-reported trouble hearing.
Overall, the NHANES data showed that 85.9%, [84.4%, 87.4%] of U.S. adults 20–69-years-old had good hearing wellness, better-ear PTA4 < 20 dB HL, with 12.9%, [11.7%, 14.1%] of those in this age range self-reporting trouble hearing despite good hearing wellness. This amounts to a U.S. population estimate of 19.2 million adults 20–69 years of age with good hearing wellness but poor auditory wellness (trouble hearing). For those 70–80+ years of age, only 29.5%, [25.3%, 33.7%] have good hearing wellness and 14.5%, [9.8%, 19.1%] of those with good hearing wellness self-report trouble hearing. This translates to an estimated 1.1 million adults in this older age group with good hearing wellness measured via PTA4 who have poor auditory wellness as documented by self-reported trouble hearing. All told, among those with good hearing wellness as demonstrated by better-ear PTA4 < 20 dB HL, 20.3 million adults have trouble hearing or reduced auditory wellness. These individuals would not be identified as needing hearing help using the 2023 CTA guideline for hearing wellness.
Evidence for reduced auditory wellness in the presence of good hearing wellness is not restricted to the HHI or to the single-item survey of hearing condition used in the recent NHANES surveys. Figure 7 provides reanalyses of data from Humes and Dubno (2021) in which the comprehensive 163-item communication profile of the hearing impaired (CPHI; Demorest & Erdman, 1986, 1987) was used to measure self-reported communication problems. The responses to the large number of items in the CPHI are reduced to four factor scores that have means of 0 and standard deviations of 1 (Demorest & Erdman, 1989; Humes & Dubno, 2021). These factor scores are plotted in the left-hand portion of Figure 7 for two groups, those with HHI scores reflecting excellent or good auditory wellness and those with HHI scores representative of fair, poor, or very poor auditory wellness using the scale proposed in Humes (2021a). Importantly, all participants in these analyses had better-ear PTA4 < 20 dB HL; that is, good “hearing wellness.” Of the 596 individuals 50–89 years of age included in Humes and Dubno (2021), 139 (23.3%) had better-ear PTA4 < 20 dB HL. Of these 139 with good hearing wellness, 24 (17.3%) reported reduced auditory wellness based on the HHI. These prevalence estimates from this smaller sample of individuals are very similar to the population estimates noted previously (Figure 6).

Boot-strapped means and 95% confidence intervals for CPHI (left) and WIN (right) for those with either excellent/good auditory wellness or fair-to-very-poor auditory wellness based on HHI and the scale of Humes (2021a). All individuals included in these analyses had good “hearing wellness.”
Boot-strapped means and 95% confidence intervals for the CPHI factor scores are plotted for both auditory-wellness groups from the Humes and Dubno (2021) dataset in the left-hand portion Figure 7. Those with poorer auditory wellness had significantly lower factors scores for three of the four CPHI scales (for all three significant effects [F(1,137) > 15.2, p < .001; for the lone nonsignificant effect, F(1,137) = 1.66, p = .200]). Despite having good hearing wellness (PTA4), those with poor auditory wellness (HHI) had significantly poorer personal adjustment to hearing problems (CPHI-PA), worse communication performance (CPHI-CP), and poorer interactions with others (CPHI-IntrxOth).
One could argue that the reason that those with lower self-reported HHI scores have lower self-reported CPHI factor scores is simply due to the shared or common response mode of these measures. This is generally referred to as common method variance or common method bias (e.g., Podsakoff et al., 2003, 2012). The data shown in the right-hand portion of Figure 7 from Wilson (2011), however, indicate that differences in self-reported auditory wellness (HHI) result in statistically significant differences in behaviorally measured speech-in-noise performance. The dataset of Wilson (2011) included 3,391 adults, 20–93 years of age (M = 62.3 years; SD = 12.8 years) with 3,191 having complete audiograms, HHI scores, and behavioral measures of speech-in-noise performance. Of these 3,191, only 466 (14.6%) had better-ear PTA4 < 20 dB HL or good hearing wellness. Of the 466 with good hearing wellness, 323 (69.3%) had poor auditory wellness as measured by the HHI. Both the low percentage of those with better-ear PTA4 < 20 dB HL and the high percentage of those with good hearing wellness who have reduced auditory wellness are not too surprising given the clinical nature of this dataset from a medical center of the Department of Veterans Affairs. The boot-strapped means and 95% confidence intervals for the signal-to-noise ratios obtained for speech-in-noise performance, the words-in-noise (WIN) test (Wilson et al., 2007), are plotted in the right-hand portion of Figure 7. These results show that, among those with good hearing wellness as reflected in better-ear PTA4 < 20 dB HL, the subgroup of those individuals with excellent/good auditory wellness has significantly lower (better) WIN signal-to-noise ratios than those with reduced auditory wellness and for both the right and left ears (both F(1,464) > 4.57, p < .05). Among those with good “hearing wellness,” reduced auditory wellness resulted in poorer speech-in-noise performance.
The main point from the analyses described above and presented in Figure 7 is that adults with good hearing wellness, defined as better-ear PTA4, often have reduced auditory wellness and there are functional consequences associated with reduced auditory wellness. This was manifest both as lower CPHI scores and poorer (higher) signal to noise ratios (SNRs) among those with good hearing wellness but differing in auditory wellness.
A recent study by Shepherd et al. (2022) reported results from the analyses of 1,314 adults in the Jackson Heart Study for whom audiological data were available. Of these 1,314 adults, 911 had normal hearing specified by Shepherd et al. (2022) as PTA4 in both ears ≤ 25 dB HL. Using an alternative definition, which required all thresholds from 250 through 8,000 Hz to be ≤ 25 dB HL in both ears, referred to by the study's authors as PT-AF (all frequencies) ≤ 25 dB HL, 516 of the 1,314 individuals met this criterion for normal audiometric hearing. These normal-hearing groups were each then split into two groups based on their responses of “yes” or “no” to the question, “Do you feel like you have a hearing problem?” When performance of the normal-hearing group with perceived hearing problems was compared to the normal-hearing group without such problems, significant differences in performance were found for a speech-in-noise measure (QuickSIN; Killion et al., 2004) and a measure of higher-level auditory processing, the Dichotic Digits-Double Pairs Test (DDT2; Musiek et al., 1991). Generally, regardless of how normal hearing was defined by Shepherd et al. (2022), using PTA4 or PT-AF, those with normal hearing thresholds who had perceived hearing difficulties performed significantly (p < .05) worse than those without such difficulties on all the speech-in-noise and dichotic-processing measures. Most of these significant effects, moreover, remained after adjustment for age, sex, and education. Interestingly, although not used to partition the groups according to perceived hearing difficulties, all those in the group without such difficulties had an HHI score of 0 whereas those with perceived hearing difficulties had a mean HHI score of 9.7 (SD = 7.3). This suggests that many in the group with perceived hearing difficulties would have reduced auditory wellness based on the HHI ≥ 8 criterion of Humes (2021a).
In another recent study, Kamerer et al. (2022) evaluated 111 adults, 19–74 years of age, who had normal hearing, defined using the same PT-AF ≤ 25 dB HL criterion as in Shepherd et al. (2022). Here, however, the 12-item version of the Speech, Spatial, and Qualities of Hearing (SSQ) scale (Gatehouse & Noble, 2004), the SSQ-12 (Noble et al., 2013), was used to identify those with perceived hearing difficulties. The top and bottom quartiles of SSQ12 scores were used to identify those with and without perceived hearing difficulties. Among this group of 111 adults with normal audiometric hearing, three measures proved useful in segregating those with perceived hearing difficulties from those without: (a) the audibility of speech; (b) a history of exposure to impulse noise; and (c) performance on a behavioral measure of frequency-modulation detection, considered to be an auditory temporal-processing measure. This study provides further evidence that many of those with good “hearing wellness” have differences in auditory wellness that are systematic and related to differences in auditory perception; in this case, frequency-modulation detection.
In summary, “hearing wellness” is not “auditory wellness.” Whereas hearing wellness is an important part of auditory wellness, it has been demonstrated here and in recent studies that as many as 20% of those with good hearing wellness have reduced auditory wellness as measured by several different self-report measures, including established scales such as the HHI, the CPHI, and the SSQ12, as well as single-item questions about hearing trouble used in NHANES and the Jackson Heart Study. Many older adults with normal hearing have deficits in the temporal processing of vowel sequences with the size of the deficit increasing with advancing age. Moreover, those with audiometrically normal hearing and reduced auditory wellness show significant deficits in speech-in-noise performance, as well as in the processing of complex stimuli, whether dichotic digits or frequency-modulated tones, compared to normal-hearing adults without reduced auditory wellness.
Moving Away From Auditory Impairment and Toward Auditory Wellness
To date, the prevailing model of hearing healthcare (HHC) has been driven by the identification of auditory disease or hearing impairment with the focus being the pure-tone audiogram obtained by hearing healthcare professionals. The prevailing impairment-driven model of HHC has failed in many high-resource countries. In the United States, for example, 80–85% of those with self-identified trouble hearing have not acquired devices to improve their hearing (e.g., Humes, 2023a, 2023b). Consistent with the findings of Nash et al. (2013) and Zelaya et al. (2015) from population studies in the United States, Figure 8 shows that the poor performance of the disease-based approach to HHC has persisted for many years. In addition, the professional-driven HHC model is not practical for many middle- and low-resource countries that lack the HHC workforce necessary to meet the needs of their citizens with poor auditory wellness (WHO, 2021).

Percentage (and 95% confidence intervals, CIs) of unmet hearing healthcare needs from the National Health Interview Survey (NHIS) for years 2007 through 2018 and overall (total). Unmet hearing healthcare need defined as the percentage of those who self-reported at least a little trouble hearing but were not currently using hearing aids. These could also be considered estimates of “unaddressed” needs.
As noted above, associations have been reported by many others between hearing wellness, a component of auditory wellness, and wellness in other domains and overall well-being. Based on this, and other emerging evidence linking self-report measures of hearing difficulty to cognition (e.g., Kawata et al., 2021), the authors suggest here that maintenance of auditory wellness across the life course is critical to wellness in other domains and overall well-being. There have been other recent calls to embrace hearing health as a part of healthy living and the maintenance of high social-emotional wellness (e.g., Humes, 2021a; Saunders et al., 2021; Timmer et al., 2023; Vercammen et al., 2020). In most of these recent calls to action, however, the focus has remained on the audiologist as the service provider who identifies the problems and then manages them for the patient (at least in the early stages of identification and intervention). As noted above, a basic tenet of well-being and wellness is the empowerment of the individual through education and growth to manage his or her own wellness.
The failure of the professional-driven impairment-based model of HHC is not only apparent in the poor uptake of hearing aids but can also be found in the infrequent evaluation of hearing by U.S. adults. Attaining a hearing test has been the entry point into the prevailing HHC system for decades. NHANES 2011–2012, 2015–2016 and 2017–2020 asked the participant how long it had been since his or her last hearing test. A hearing test was defined as “…one that is done in a soundproof booth or room, or with headphones” (and performed by) “…audiologists, ear nose and throat doctors, and trained technicians or occupational nurses.” Data from NHANES 2011–2012, 2015–2016, and 2017–2020 indicate that, among U.S. adults with at least a little trouble hearing, it has been 5 or more years since their last hearing test for about 66% of 20–69-year-olds and for about 45% of those 70–80+ years of age. These data compare favorably to those from Nash et al. (2013) and Zelaya et al. (2015), both finding over 50% in the population studies analyzed had not had a hearing test in the past 5 years. Obtaining a hearing test and retaining HHC professionals as the gatekeepers to the successful management of an individual's auditory wellness has not proven to be successful for many of those in need of help. Adults must be empowered to assess and manage their own auditory wellness (Humes, 2021a).
Heretofore, self-management of auditory wellness was simply not possible within the U.S. HHC system. Over the past 14 years, however, a movement has been underway to improve the affordability and accessibility of HHC for millions of U.S. adults (Donahue et al., 2010; NASEM, 2016). These efforts culminated in the passage of the over the counter (OTC) Hearing Aid Act of 2017 and the adoption of new guidelines for hearing aids by the Food and Drug Administration (FDA) in October 2022. The new guidelines divide the types of hearing aids into OTC and prescription devices with candidacy for OTC hearing aids restricted to adults with perceived mild-to-moderate hearing loss. Those adults with hearing trouble exceeding perceived mild-to-moderate difficulties would pursue the conventional path to help with the assistance of HHC professionals. It is important to note that HHC professionals using audiology best practices have demonstrated excellent outcomes for adults with pure-tone hearing loss of moderate or greater severity when fitted with prescription hearing aids. This has been confirmed recently in several RCTs (e.g., De Sousa et al., 2023; Humes et al., 2017; Lin et al., 2023; Sabin et al., 2020).
For the first time since their inception, hearing aids that are regulated by the FDA can now be pursued directly by adults with perceived mild-to-moderate hearing difficulties. This empowers adults to manage their own auditory wellness and to do so directly.
How many people might benefit from the creation of this new pathway to hearing help? Humes (2023a, 2023b) recently analyzed data from NHANES 2011–2012, 2015–2016, and 2017–2020 and the annual National Health Interview Survey (NHIS) from 2007–2018 to answer this question. Both surveys used the same single-item measure of perceived hearing trouble which inquired about the condition of the respondent's hearing as described above. The recent analyses of Humes (2023a, 2023b) focused on the group who self-identified themselves as having at least a little trouble which includes those who have “a lot of trouble” as well as those who consider themselves “deaf,” neither of which are the intended target groups for OTC hearing aids. When these datasets were reanalyzed here with a focus on just those having perceived mild-to-moderate hearing difficulty, interpreted as those responding either “a little trouble” or “moderate trouble,” then 13.6% (NHIS) to 19.4% (NHANES) of all U.S. adults would be considered candidates for OTC hearing aids. Of these potential candidates, only 11.6% (NHIS) to 13.1% (NHANES) were currently using hearing aids. Applying these prevalence estimates to the U.S. Census Bureau's estimates for the number of people 18+ years of age residing in the United States in 2022 (260.8 million) yields 31.3 to 43.9 million Americans who could improve their auditory wellness through the acquisition of OTC hearing aids. Importantly, as supported by the low percentages for the uptake of what are now labeled prescription hearing aids (see Figure 8), the prevailing HHC system has not met the auditory wellness needs of those with perceived mild-to-moderate hearing loss.
A pathway has been opened in the United States for adults with perceived mild-to-moderate hearing loss to gain control of their auditory wellness. However, availability of devices alone is insufficient. As was noted in the discussion of wellness, education, and growth are keys to self-actualization. The same holds true for the attainment of high auditory wellness. It is important to educate all Americans about the need to maintain good auditory wellness throughout adulthood and about options for help available to those with less-than-optimal auditory wellness. These options should include not just the devices, but support systems to help the individual manage his or her auditory wellness, including support systems designed originally for use by HHC professionals (Timmer et al., 2023). Reliable, valid, and simple-to-use digital tools should be widely available to assist with the measurement of auditory wellness and to guide the individual with less-than-optimal auditory wellness to viable options for improvement. The acquisition of a hearing device to help compensate for the loss of audibility, such as a personal sound amplification product (PSAP), hearable, or OTC hearing aid, must be seen as just one step, albeit for many an important one, on a pathway to achieving high auditory wellness (Humes, 2021a; Saunders et al., 2021; Timmer et al., 2023).
The opportunity for adults with “perceived mild-to-moderate hearing loss,” characterized here as fair or poor auditory wellness or a little or moderate hearing trouble, to self-manage and improve their auditory wellness is unprecedented in the U.S. OTC hearing aids have exploded onto the market immediately prior to and following the release of the FDA's revised guidelines in late 2022. A myriad of companies and devices are already available. On the other hand, tools to assist adults with less-than-optimal auditory wellness along their journey to improved auditory wellness are sorely lacking (Allen et al., 2022; Humes, 2021a). Although at least one reliable and valid measure of auditory-related subjective well-being has been developed (Humes, 2021b), it was only evaluated in a group of 173 older adults, 60–88 years of age. To our knowledge, no other measures that directly assess auditory-related well-being have been developed. These assessment tools, moreover, are just for the front-end determination of candidacy for devices, and tools supporting all aspects of the person's journey to improved auditory wellness are needed. Given the explosion of devices onto the market, early indications that returns-for-credit for OTC hearing aids may be twice that of prescription hearing aids (∼35%; Taylor, 2023), and the lack of tools developed specifically with auditory wellness in mind, tools that already exist should be made broadly available quickly to minimize further chaos in the market and the poor outcomes that may follow. The set of tools to enable the adult to self-assess auditory wellness and, if needed, to improve auditory wellness can be made broadly available on a digital platform and are described next.
An Approach to Maintaining Auditory Wellness Throughout the Adult Life Course
This approach to maintaining auditory wellness is informed by what Stephens and Kramer (2009) referred to as the “audiological enablement/rehabilitation model” and is referred to here as the “auditory-wellness enablement model.” This model includes four steps: (a) evaluation; (b) integration and decision making; (c) short-term remediation; and (d) ongoing remediation. Table 1 provides a summary of possible actions that could be taken to improve the auditory wellness and tools available for each of these actions. The tools presented here can be made available online making it easily accessible to large populations with limited resources. The importance of auditory wellness to healthy everyday function should be broadly promoted through the development and rollout of ad campaigns in partnership with organizations such as the American Association of Retired Persons (AARP), the Hearing Loss Association of America (HLAA), and via publications like Consumer Reports. However, this section will not describe these broad awareness campaigns in detail.
An Auditory Wellness Enablement Model With Specific Actions and Tools.

HHC = hearing healthcare.
Stage 1: Evaluation
In the absence of measures designed specifically to assess auditory wellness, existing valid, reliable, and efficient tools may be substituted as proxy measures for auditory wellness. Historically, there have been three versions of the HHI tools: (a) the 25-item HHIE; (b) a shortened 10-item screener, the HHIE-S; and (c) the 25-item HHIA, a slightly modified version of the original HHIE designed for adults under the age of 50 years. Here, for convenience, these have been referred to simply as “HHI” measures. Humes (2021a) made the case for the use of the HHI for all adults as a proxy measure for the measurement of auditory wellness, a recommendation supported recently by Timmer et al. (2023). As noted, HHI scores have been found to be moderately correlated to measures of pure-tone hearing loss, such as PTA4, and the HHI items directly assess connections to both social and emotional wellness, a key linkage of auditory wellness to each of these wellness domains and to well-being (Figure 2).
HHI score ranges were established and were used to assign auditory-wellness grades ranging from excellent through very poor in Humes (2021a). Based on the psychometric analyses of the HHIE by Cassarly et al. (2020), the 18-item Revised Hearing Handicap Inventory (RHHI) was developed and is recommended for use here. The 18-item RHHI is more efficient than the full 25-item HHIE, yet provides the same information (Cassarly et al., 2020) and has been mapped to the HHI-based auditory-wellness scale (Humes, 2021a). The RHHI should be made broadly available in a web-format or as an app for completion on mobile devices and personal computer, devices widely available in the United States to adults of all ages, genders, races, and ethnicities. In addition to providing a score at completion, brief feedback aiding the interpretation of that score and suggesting next steps should be included. The resulting database for this tool should be designed to record de-identified demographic information in addition to the score and the time required for completion of the tool. Furthermore, a digital platform with the ability to create a profile (with login credentials or similar) and to store the previous results could help individuals track their performance over time as desired. Low (good) RHHI scores may be commonplace due either to denial or unawareness of less-than-optimal auditory wellness. As a result, an additional tool, the hearWHO digits-in-noise (DIN) test (De Sousa et al., 2020, 2022), can be used to validate the responses of those reporting excellent auditory wellness. For those with good RHHI scores but poor hearWHO results, the individual would be pointed to helpful online educational resources about hearing and its importance to wellness and well-being, then encouraged to revisit the test site soon.
To have the best impact, individuals should be encouraged to complete the auditory wellness check annually, using the RHHI and hearWHO. This could be tied to a key date each year (e.g., May 1st as it is the first day of Better Speech and Hearing Month; or March 3rd as it is the World Hearing Day), with broad marketing-campaign reminders, including pointers to easy and free access to the web-platform or app.
An important consideration for the broad application of these two tools for the assessment of auditory wellness by U.S. adults is that both are available in English and in Spanish (Carrillo et al., 2019; De Sousa et al., 2022). In addition, many of the helpful online resources for understanding hearing loss and its consequences from organizations such as the HLAA and the AARP are available in both English and Spanish.
Auditory Wellness Check Results
The first step is the efficient, reliable, and valid assessment of auditory wellness and the determination of candidacy for improved auditory wellness. As noted, the 18-item RHHI, a more robust psychometrically valid HHI measure, is the specific candidacy measure recommended here. Because the recommendations or suggested interventions are similar for those falling into the “excellent” and “good” auditory-wellness categories and those falling into the “fair” and “poor” categories, these categories were combined here to produce three RHHI-based auditory-wellness classifications: excellent/good, fair/poor, and very poor. Corresponding RHHI score ranges are as follows: (a) excellent/good, 0 to 6; (b) fair/poor, 8 to 28; and (c) very poor, 30–72.
The hearWHO DIN test (De Sousa et al., 2020, 2022) represents the other candidacy measure. As noted, two measures are needed, including a behavioral measure like the hearWHO DIN test, because broadly administered self-report measures only capture those who are aware of their hearing difficulties and are not in denial of those difficulties (Humes, 2021a, 2023a; Humes & Dubno, 2021; Pronk et al., 2018). The hearWHO DIN is a test patterned after the original Dutch version developed by Smits et al. (2004). This is the second online measure to be applied to check auditory wellness and to aid in the interpretation of the RHHI scores. The hearWHO DIN test is a 3-min test, automated for remote administration via telephone or other electronic devices (DeSousa et al., 2022). The hearWHO is now endorsed by the WHO and used nationally and internationally as a hearing screening system (De Sousa et al., 2022; Kwak et al., 2022). The hearWHO, as with other DIN tests, was designed as a surrogate for pure-tone hearing loss with correlations between hearWHO SNR and better-ear pure-tone average of four frequencies (PTA4) of r = 0.85 (De Sousa et al., 2020). As such, it may be considered a surrogate for pure-tone audiometry and “hearing wellness.”
An interesting feature of the hearWHO DIN test, however, is that it makes use of antiphasic stimulus presentation to both ears which also taps lower-level binaural processing (dichotic) abilities (De Sousa et al., 2020). Humes et al. (2013) found associations between dichotic processing and aided speech understanding, Lentz et al. (2022) found these same binaural measures to be related to cognitive function, Humes (2021c) reported significant correlations between longitudinal changes in cognition and longitudinal changes in dichotic syllable identification, and the longitudinal population study of Dillard et al. (2020) reported age-related declines in dichotic processing of digits independent of hearing loss. Thus, this version of the DIN test, the hearWHO (antiphasic), may tap broader abilities than just the detection of simple sounds, including some aspects of dichotic processing. This version of the hearWHO (antiphasic) was also shown to be sensitive to conductive and unilateral hearing impairments in De Sousa et al. (2020).
Given the sensitivity of the hearWHO to unilateral or asymmetric hearing loss (between-ear difference in PTA4 values>10 dB), De Sousa et al. (2020) used worse-ear PTA4 to categorize the severity of hearing loss. The hearing-loss scale used by De Sousa et al. (2020) was as follows: (a) excellent-hearing-to-minimal hearing loss, worse-ear PTA4 of 0 to 25 dB HL; (b) mild-to-moderate hearing loss, worse-ear PTA4 from 26 to 55 dB HL; and (c) severe-to-profound hearing loss, worse-ear PTA4 ≥ 56 dB HL. In terms of the corresponding SNR values from the hearWHO, an SNR <−16 dB identified those with excellent hearing or minimal pure-tone hearing loss, an SNR of −16 to −10.5 dB identified those with mild/moderate pure-tone hearing loss, and an SNR value of >−10.5 dB identified those with severe/profound pure-tone hearing loss.
To summarize, there are three RHHI-based auditory-wellness categories and three hearWHO SNR-based hearing loss categories of interest. The results from everyone for these two tests can then be categorized into one of nine cells in the 3 × 3 grid shown in Figure 9. Figure 9 makes use of the boundary RHHI and boundary SNR values specified above for each test and then, based on that pattern, identifies the targeted pathway believed to be the best choice for improved auditory wellness. Ultimately, the choice of intervention is up to the individual completing the two measures, the RHHI and the hearWHO. In addition to showing the targeted treatment pathway for each of the nine cells in this 3 × 3 grid, the estimated prevalence of each combination is also shown in Figure 9. These prevalence estimates were derived from data in Humes (2021a) which included both the HHI and worse-ear PTA4 measures. Specifically, data from 1,619 adults (52.5% females) ranging in age from 50 to 90 years from a large community convenience sample (N = 1,186) from Charleston, SC, and two clinical studies (total N = 433) from Bloomington, IN with a range of hearing loss and RHHI values were used to derive these prevalence estimates. Those with fair/poor auditory wellness (8 ≤ RHHI ≤ 28), or “perceived mild-to-moderate hearing loss” are the primary targets for OTC hearing aids, as noted previously, whereas those with very poor auditory wellness would generally be considered best-suited to prescription hearing aids, especially in the presence of at least mild-to-moderate audiometric hearing loss (red labels and boxes in the right-most column of Figure 9). However, analyses of these data suggest that a small percentage of older adults with fair/poor auditory wellness may have severe/profound audiometric hearing loss as estimated by the hearWHO. These individuals would be referred to area HHC professionals for follow-up evaluation. Note that those who have excellent/good auditory wellness and excellent hearing/minimal hearing loss, shown in green in the lower left corner of Figure 9, would be considered to have optimal auditory wellness and would simply be asked to return for reevaluation in a year, sooner if experiencing listening difficulties.

The 3 × 3 candidacy grid and suggested interventions for various combinations of hearWHO and HHI scores.
Several cells in the grid in Figure 9 have been labeled, “counsel and educate” (blue text and boxes). In these cases, there is a sizable mismatch between the two candidacy assessments and online educational materials should be provided for individuals with these conflicting results. The blue cells to the left of Figure 9 reflect a pattern consistent with either poor awareness or denial of hearing difficulties and educational materials should be tailored to address those potential issues. For the blue cell in Figure 9 at the lower right corner, the individual is experiencing a considerable reduction in auditory wellness despite little estimated audiometric hearing loss. In this case, the counseling and education would be centered on providing online aural rehabilitation including use of communication strategies, such as the Hearing Loss Toolkit (Arnold et al., 2019), and the use of assistive listening technologies that target the improvement of SNR given the apparent lack of audiometric hearing loss.
In addition to the RHHI and hearWHO, all respondents are also asked to complete the Consumer Ear Disease Risk Assessment (CEDRA; Klyn et al., 2019), a validated tool designed to help consumers evaluate the risk for the presence of ear disease that might contra-indicate use of hearing aids. CEDRA is a 15-item questionnaire with yes/no and multiple-choice questions. Most questions relate to hearing and balance, one question relates to general health, and the remainder query things such as vision impairment or recurring fever that may co-occur with hearing issues. Participants must score <4 on the CEDRA to be considered eligible for self-management using hearing devices without the need for referral to a hearing healthcare professional for follow-up. Results from broad use of the CEDRA by a former DTC hearing-aid company, Lively (now Jabra Enhance), for a population like that targeted here, indicated ∼18% failure rate for the CEDRA (personal communication). This rate is similar to the prevalence of ear disease in a small unscreened sample of OTC hearing-aid candidates (Humes et al., 2019).
Once respondents have had their auditory wellness assessed using the RHHI and hearWHO and have been screened for medical concerns with CEDRA, the individual will be presented with the results and helpful information on the next possible steps. Those failing the CEDRA will be informed of their assessed auditory wellness and referred to appropriate local HHC professionals for possible assistance based on the items failed.
Although the focus here is on those with “fair” or “poor” auditory wellness, as noted, feedback will also be provided to those who received other auditory wellness grades (or, as noted above, who failed CEDRA regardless of wellness grade). These individuals can proceed to the next steps toward improved auditory wellness using a proposed decision aid (see next section). As noted, those who have an assessed auditory wellness of excellent/good (RHHI, 0 to 6) will simply be informed of their results and asked to return for evaluation annually, or sooner if they experience listening difficulties or declines in auditory wellness. They will also be pointed to online resources such as the HLAA's Hearing Loss Basics—How to Tell If You Have Hearing Loss and the AARP's 10 Signs That You Could Have Hearing Loss and six Habits for Healthy Ears at Any Age. Links to information on hearing conservation will also be provided to help them preserve their current auditory wellness. As noted, for those with self-assessed “very poor” auditory wellness, the information accompanying the results will indicate that the respondent would most likely benefit from prescription hearing aids and the help of an HHC professional should be sought. If they would like a list of nearby HHC professionals, including contact information, they would be instructed to enter their zip code and a list would be provided to them for review and printing, as desired.
Stage 2: Integration and Decision Making
Individuals may have different motivations and expectations about how and when to manage their auditory wellness. For this reason, it is necessary to evaluate the individual's motivation and expectations in relation to the assessed auditory-wellness score, as well as their willingness (and ability) to pursue possible next steps on their path to improved auditory wellness. The respondent will be advised of the recommended pathway to improved auditory wellness based on their pattern of RHHI and hearWHO results as shown in the 3 × 3 candidacy grid in Figure 9. The individual may not wish to pursue the recommended pathway, however, and a decision aid can help guide them to next steps in their journey to improved auditory wellness. An example of such a web-based decision tool can be found at https://hearing.tools/tool. Such an approach complements healthcare movements such as “no decision about me without me” (O'Brien, 2022) and fosters the shared-decision making elements of patient-centered care. The decision aid should include options (both device and non-device options) that are available to improve auditory wellness, as well as benefits/harms associated with each of these options, and will help an individual find congruence between decisions, personal values, and motivations (Drug and Therapeutics Bulletin, 2013).
Stages 3 and 4: Short-Term and Ongoing Remediation
The short-term and ongoing remediation includes device and non-device options. For instance, individuals may choose to use hearing devices such as OTC hearing aids, PSAPs, or hearables to improve their auditory wellness. On the other hand, they may opt for non-device interventions such as hearing health education or communication training. Individuals are also free to pursue both device and non-device interventions and may choose to do so separately (asynchronously) or at the same time (synchronously).
For those with fair-to-poor auditory wellness who have passed CEDRA and do not appear to have severe-to-profound hearing loss based on the hearWHO (Figure 9), general information about hearing-device options (e.g., HLAA: Hearing Loss Basics-What Treatments and Devices Can Help?), with a focus on OTC hearing aids (e.g., AARP: How to Shop for an Over-the-Counter Hearing Aid), will be presented first. Those who are interested in perusing DTC hearing devices such as OTC hearing aids, PSAPs or hearables will be encouraged to review objective information on the performance and quality of hearing devices on platforms, such as the Hearing Advisor (https://hearadvisor.com/), or to read consumer reviews from websites, such as the Hearing Tracker (https://www.hearingtracker.com/) or Soundly (https://www.soundly.com/), before making the choice of a hearing device. The Hearing Advisor initiative (Manchaiah et al., 2023; Sabin et al., 2023) provides results from extensive standardized testing, distilled down to a novel consumer-centric metric referred to as a “SoundScore.” This single score will help a consumer compare the sound quality between different devices and make an informed choice. In addition, those who are interested in perusing more detailed communication education (e.g., ACE; Hickson et al., 2007) and/or the hearing device education (e.g., C2Hear; https://www.c2hearonline.com/) can be directed to relevant resources, including the Hearing Loss Toolkit (Arnold et al., 2019). It is also possible to develop web-based or app-based platforms (Beukes et al., 2019) to offer such education programs virtually for a large population with limited access. Most likely, however, such tools will need to be well integrated into the digital delivery platform to make access seamless and to help guide the individual through the various tools available for their chosen pathway to improved auditory wellness.
Concluding Remarks
Tens of millions of U.S. adults with perceived mild-to-moderate hearing difficulties, or alternately, with fair-to-poor auditory wellness, have been presented with the opportunity to self-manage their auditory wellness. Solutions to improve auditory wellness, including electroacoustic devices such as FDA-approved OTC hearing aids, are now broadly available. The tools needed by the consumer to both assess and manage their own auditory wellness, however, are not readily available or accessible to consumers. This perspective placed this need in a broad context of wellness and well-being and provided a guide to the development of a widely available digital platform to help fill this void in hopes of improving auditory wellness, and in turn, well-being.
Figure 10 provides a broad overview of the self-management of auditory wellness throughout the life course as proposed here. It begins with awareness and education of all adults about the importance of auditory wellness to well-being. It then proceeds to the self-assessment of auditory wellness followed by decisions to be made by those with less-than-optimal auditory wellness. It concludes either with the return to the beginning at a later point in time for those with excellent or good auditory wellness or those with less-than-optimal wellness who decide to do nothing about it now or to the measurement of post-intervention outcomes for those who opt for intervention.
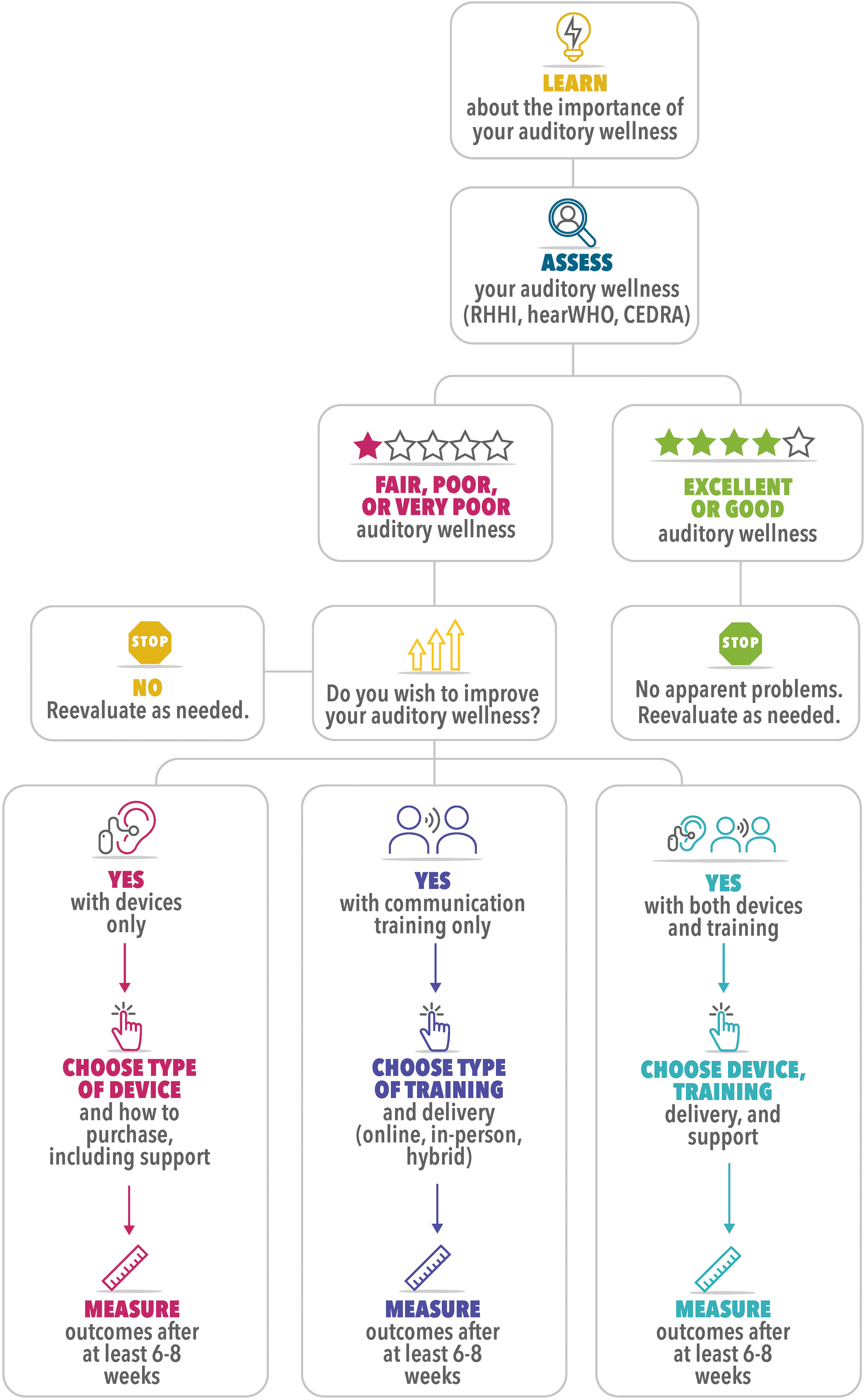
Flow chart summarizing proposed self-managed auditory-wellness-improvement pathway.
Although improvement of auditory wellness has been described in this perspective as a fully self-managed process, a single approach likely will not be suitable for all. For example, whereas there is little burden imposed on the individual in the early stages in Figure 10 that focus on education and self-assessment, the responsibility for the selection and completion of intervention at the later stages may be perceived as too challenging for some. Consultation with others, including hearing healthcare professionals, for support with the selected intervention or even with the process of selecting the desired intervention may be appropriate for some. We suspect that age or generational differences may drive some of the perceived need for assistance. Given that OTC hearing aids have only recently made possible a fully self-managed auditory-wellness-improvement process, nobody can be expected to rely on their prior experience, or even that of friends and family, in navigating through these experiences. Early adopters of this approach may need support even more than others who pursue this pathway in the future.
Although this perspective makes a case for the overarching importance of auditory wellness, encouraging self-assessed hearing wellness through online or in-person pure-tone audiometry is certainly a step in the right direction. Recall that 80–85% of adults who have documented hearing needs (either through self-report or pure-tone audiometry) currently do not have those needs met with hearing devices (either hearing aids or ALDs; Humes, 2023a). There is a lot of room for improvement and multiple pathways to improved auditory wellness should be made available to minimize end-user confusion and to reduce barriers to obtaining assistance.
Likewise, this perspective recommends the use of specific tools at various stages of the self-managed auditory-wellness-improvement process (Figure 10). For example, the RHHI and hearWHO were the tests chosen to assess wellness and establish candidacy for intervention. These tools were selected because of their reliability, validity, widespread usage (including availability of both in Spanish), and the large volume of data available for each tool. This does not preclude the use of other similarly vetted tools at various stages shown in Figure 10. Some may opt for the use of the shorter RHHI-S instead of the RHHI or for the use of alternate digits-in-noise tests, such as the National Hearing Test (NHT; Watson et al., 2012; Williams-Sanchez et al., 2014), instead of the hearWHO. In addition, after the identification of less-than-optimal auditory wellness in the initial stages, some may choose to leave the self-managed pathway outlined in Figure 10 entirely and seek help from hearing healthcare professionals, including the use of prescription hearing aids. A myriad of potential branches are possible variations in the default self-managed auditory-wellness-improvement pathway outlined in Figure 10 and, as long as improved auditory wellness is the outcome, all options should be considered viable. We leave it to others to document those alternatives and to provide the evidence in support of each alternative.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
