Abstract
To reveal the associated factors of quality of life (QoL) in children with inborn errors of metabolism (IEM), their siblings, and their primary caregivers and partners, we conducted an anonymous questionnaire survey in Japan. Descriptive, correlation, and multiple regression analyses were performed. Fifty-six children with IEM, 35 siblings, 143 primary caregivers, and 86 partners completed our questionnaires. There were significant positive correlations between higher QoL in children with IEM and lower disease influence (r = 0.46) and higher perceived emotional support (r = 0.67). We could not find any associated factor of siblings’ QoL. Lower parental distress, higher family empowerment, and higher household income contributed to higher QoL in primary caregivers (adjusted R 2 = 0.636). Higher household income, lower anxiety about childrearing, and higher satisfaction in the relationship with the child and entire family contributed to higher QoL of partners (adjusted R 2 = 0.398). We concluded that developing effective interventions to improve QoL is needed for the entire family in future outpatient settings.
Introduction
In 2013, around 19 000 cases of patients with inborn errors of metabolism (IEM) were reported in Japan. 1 Although there have been recent improvements in the early detection and treatment of these conditions in Japan, 2 patients will still have to live with this condition for the rest of their lives, even in cases in which early diagnosis and treatment were successful. During the process of coping with their conditions, patients will face the same variety of challenges as their parents. 3 As with other chronic childhood conditions, such as food allergies, 4 the different forms of dietary restriction involved in the treatment of IEM will affect not only child’s quality of life (QoL) but the QoL of the entire family. Before considering effective means of support for these children and their families, it is essential to first understand their current QoL and associated factors.
In other countries, some studies on the QoL of patients with IEM and their parents have been conducted. 5 –7 In Turkey, the researcher reported that children with IEM and poor dietary compliance had lower scores in 2 aspects of QoL: school life and disease perception. 5 In our previous literature review, we found that Japanese children with IEM and their parents faced various difficulties in daily life, through infancy to adolescence and young adulthood. 6 We then hypothesized that QoL would be affected and impaired by IEM for both children and their families.
On the other hand, Simon et al in Germany and Cazzorla et al in Italy reported that QoL scores among young adult and adult patients with phenylketonuria (PKU) were not significantly different when compared to those of healthy individuals. 7,8 This may imply that children with IEM somehow overcome their difficulties during maturation. During childhood, children with IEM and their families should be able to get support from the people around them. There have been some qualitative studies on QoL of these individuals, 9 –11 but currently, there are no quantitative studies assessing QoL and its associated factors among families impacted by IEM in Japan. We investigated the actual situation of the QOL of children with IEM and their entire families in Japan as the first step, although it is known that investigation of individual diseases is important. It would be helpful to clarify the common associated factors of QoL of such children and their families for policy making for supporting these families. Like in other international studies, it is important to report the QoL in every diagnosis; however, we believed that investigating the actual situation of the QOL of children with IEM and their entire family in Japan is the first step. It will be helpful to clarify the common associated factors of QoL of those children and families for policy making, regarding the support for those families at a national level.
Therefore, the purpose of our study was to report the QoL of children with IEM as well as that of their immediate family they live with and reveal the factors associated with QoL of these individuals. These findings will be the basis for providing effective support to the entire family from the perspective of medical professionals.
Methods
We conducted cross-sectional questionnaire survey among children with IEM, their siblings, their primary caregivers, and their partners. The study was conducted in Japan from August to October 2015. We have used the terms, “primary caregiver” and “partner” in this study rather than “mother” and “father” because the role of a primary caregiver might be undertaken by either the mother or father, depending on the family. Anonymous questionnaires were sent to 532 eligible families across Japan in July 2015. Participants sent the completed questionnaires back to our university voluntarily.
Participants
We recruited children with IEM and their families from Japan registration System for Metabolic & Inherited Diseases (JaSMIn). This voluntary self-registration system was established in 2013 as a way to improve the diagnosis and treatment of IEM by the National Center of Child Health and Development (http://jasmin-mcbank.com). Patients and families were registered in this system in order to participate in the studies aiming to improve the care they receive and to obtain information about the studies such as clinical trials. Patients and their families who are willing to cooperate with studies on IEM enroll in this system. There were 532 children aged between 0 and 20 years registered with JaSMIn in July 2015. The participants involved in this study were children with IEM aged 7 to 20 years, siblings aged 7 to 20 years, primary caregivers of children with IEM aged 0 to 20 years, and the partners of the primary caregivers. Children aged 0 to 20 years with special chronic diseases, including IEM, received medical aid in Japan. Even if a child with IEM was below 7 years of age, their older siblings and primary caregivers were included if they volunteered to participate in this study. We included only children and their families living together at the time of the investigation and excluded the data of siblings having IEM (n = 3) in our analysis.
Questionnaires
Questionnaire sheets and survey contents were prepared separately for each participant: children with IEM, siblings, primary caregivers, and partners of primary caregivers. All existing scales were calculated along with each instruction and analyzed by using raw scores without any transformation.
Children With IEM and Their Siblings
We used the Japanese version of the Kinder Lebensqualitats Fragebogen (KINDL) scale for assessing QoL of children. The scale was developed in Germany, and its reliability and validity have been confirmed in Japanese samples. 12,13 This scale has 24 items and consists of 6 subscales: physical, emotional, self-esteem, family, friends, and school. Total scores ranged between 20 and 100 in the KINDL scale. We also used the additional disease subscale of the KINDL, which comprises 6 general items related to the disease. The disease subscale of KINDL is conceptually divided from other subscales, and assesses how much their disease affects their daily lives. 12 The 6 items of the disease scales are “I was afraid that my illness might get worse,” “I was sad because of my illness,” “I was able to cope well with my illness,” “My parents treated me like a baby because of my illness,” “I wanted nobody to notice my illness,” and “I missed something at school because of my illness.” For the purpose of this study, we added 4 original items to assess the quality of emotional support received in daily life. Items included, “I have difficulty,” “I have someone who helps me when I have a difficulty,” “I have someone who I can talk to when I have a difficulty,” and “I can talk to my parents when I have a difficulty.” These study-specific items were scored the same way as the items of the original KINDL scale. If there were several siblings in a family, only 1 sibling answered the questionnaire. General characteristics were also recorded for children and their siblings.
Primary Caregivers
We used the Japanese version of the 26-item short form of the World Health Organization QoL (WHOQoL-26) questionnaire for assessing the QoL of primary caregivers. 14 The Japanese version’s reliability and validity have previously been confirmed. 15 The WHOQOL-26 contains 24 items covering the 4 subscales of physical health, psychological health, social relationships, and environment and contains 2 items assessing global QoL.
For assessing parenting stress, we used the Parenting Stress Index–Short Form (PSI-SF). 16 The Japanese version was developed, and its reliability and validity have been established. 17,18 The Japanese version has 19 items and consists of 2 subscales: difficult child and parental distress. For assessing family empowerment, we used the Family Empowerment Scale (FES). 19 The concept of family empowerment represents a family’s ability to raise a child with disabilities independently and control their own lives and its process. 19 The reliability and validity of the Japanese version of Family Empowerment Scale (J-FES) have been established. 20 This scale has 34 items and consists of 3 subscales: family, service systems, and social politics. The J-FES was completed by the primary caregiver, and their answers were considered to represent the feelings of empowerment in the entire family. 20
The questionnaire consisted of (1) characteristics of the primary caregivers and (2) predictive factors of their QoL. To assess the characteristics of primary caregivers and the factors predicting their QoL, our survey included items devised from both previous literature, studying QoL of other chronic conditions, 21 –23 and our own literature review. 6 The characteristics of primary caregivers (1) are shown in Table 3. Predictive factors of primary caregivers’ QoL (2) were described by the following 12 items: the age of IEM onset in children, number of IEM-related hospital visits per year, difficulties related to raising a child with IEM, anxieties related to raising a child with IEM, degree of distress related to caring for a child with IEM, information resource of their child’s disease and treatment, satisfaction in the relationship with the child with IEM, satisfaction in the relationships within the family, household income, and degree of social acceptance and understanding shown to the child with IEM.
Partners of Primary Caregivers
We used the same WHOQOL-26 scale for assessing the QoL of partners. 15 The questionnaire consisted of (1) characteristics of partners and (2) predictive factors of QoL. Characteristics of partners (1) were the same 4 items as primary caregivers. Predictive factors of partners’ QoL (2) were the following 7 items: current age of children with IEM, anxieties related to raising a child with IEM, degree of distress related to caring for a child with IEM, satisfaction in the relationship with the child with IEM, satisfaction in the relationships within family, household income, and the degree of social acceptance and understanding shown to the child with IEM.
Data Analysis
Data analysis was conducted using SPSS 21 for Mac (SPSS Inc). For inclusion in the analysis, all questionnaire scales and subscales were required to have been completed fully (KINDL, WHOQOL-26, PSI-SF, and FES), and additional sociodemographic information, such as age and sex, were required to be 90% complete. We excluded the responses from participants who were in the hospital at the time of the investigation, as their physical or mental condition while in the hospital would not be reflective of their daily life experience while living at home, which may have affected their answers. The descriptive statistics, means, standard deviations (SDs), and response weighting were calculated for each variable. Spearman rank-correlation coefficients were used to evaluate the relationships between QoL of children and predictive factors of QoL separately for both children with IEM and their siblings. Multiple regression analysis was conducted by stepwise selection for revealing the associated factors of QoL of primary caregivers and partners. We set WHOQOL-26 scores as the dependent variable and each of the 17 factors (primary caregivers) and 10 factors (partners) as explanatory variables. There was no multicollinearity.
Ethical Considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the medical ethics review board of the University of Tsukuba (no 1101). Consent was implied if children with IEM, siblings, primary caregivers, and partners answered and returned the questionnaires. Participants only supplied their full names and addresses if they wanted to receive a completed report of this study after analysis, and at this point, participants were informed that their information would not be used for any other purpose.
Results
Of the 532 families who were sent the questionnaires, 143 participated in this study (response rate: 27.0%). Tables 2 and 3 show participant characteristics, Tables 4 and 5 show QoL scores, and Tables 1 and 6 show QoL associated factors. Details of predictive factors are available in Supplemental Tables 1 and 2.
Correlations Between QoL in Children With IEM and Their Siblings and Its Predictive Factors.
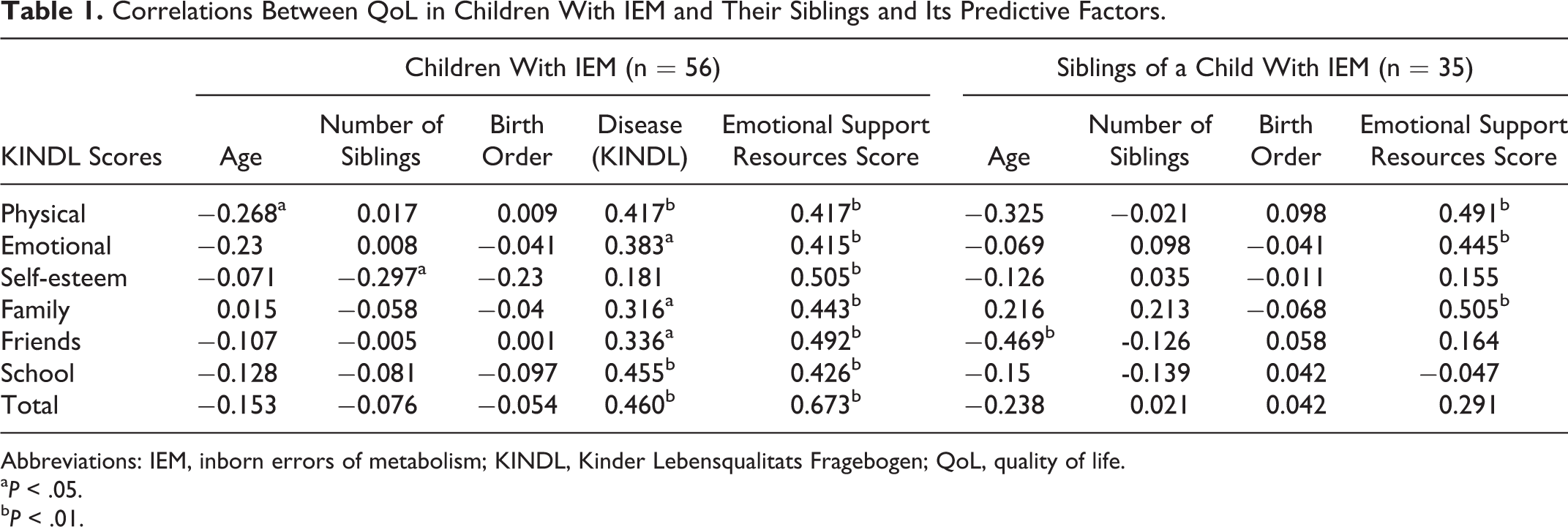
Abbreviations: IEM, inborn errors of metabolism; KINDL, Kinder Lebensqualitats Fragebogen; QoL, quality of life.
a P < .05.
b P < .01.
Children With IEM
In total, 56 children with IEM answered the questionnaires. Table 2 shows their characteristics. To prevent the respondents being identifiable, we put the disease category names instead of disease names, for example, “amino acid disorders” instead of PKU and Maple Syrup Urine Disease (MSUD) (Table 2). The mean KINDL scores, additional disease subscale scores, and emotional support in daily life scores are shown in Table 4.
Characteristics of Children With IEM and Their Siblings.
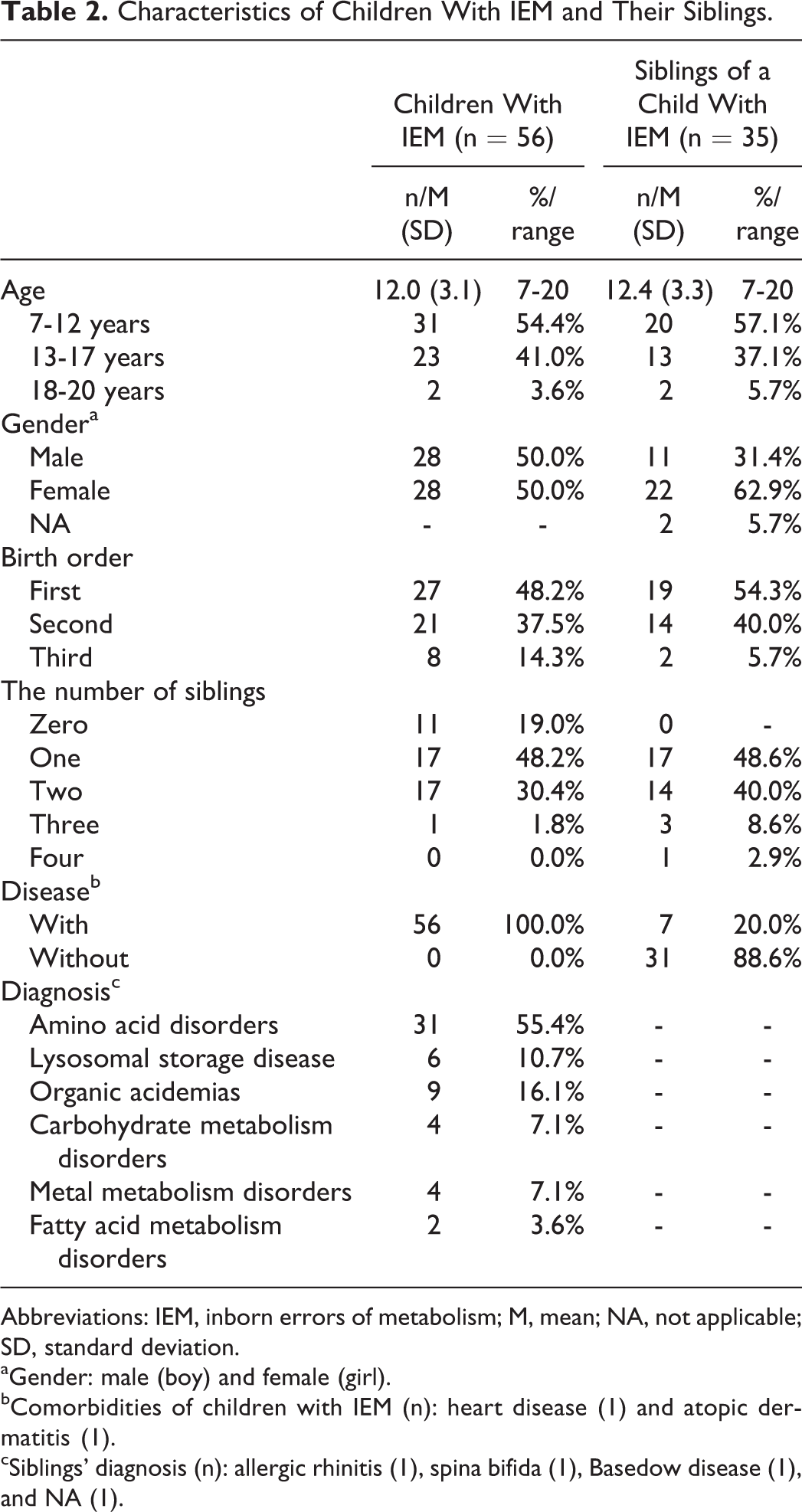
Abbreviations: IEM, inborn errors of metabolism; M, mean; NA, not applicable; SD, standard deviation.
aGender: male (boy) and female (girl).
bComorbidities of children with IEM (n): heart disease (1) and atopic dermatitis (1).
cSiblings’ diagnosis (n): allergic rhinitis (1), spina bifida (1), Basedow disease (1), and NA (1).
The mean age of the children was 12.0 ± 3.1 years (Table 2), and the mean score of their QoL was 69.2 ± 14.4 points (Table 4). There were significant positive correlations between QoL (total KINDL scores) and additional disease subscale scores (r = 0.460, P < .01) and between QoL and emotional support resources (r = 0.673, P < .001). All KINDL subscales were significantly and positively correlated with emotional support resources (r = 0.415-0.505, P < .01). The disease subscale was significantly correlated with the following 5 subscales of the KINDL: physical (r = 0.417, P < .01), emotional (r = 0.383, P < .05), family (r = 0.316, P < .05), friends (r = 0.336, P < .05), and school (r = 0.455, P < .001). There were negative correlations between the age of children with IEM and the physical subscale (r= −0.268, P < .05; Table 1).
Siblings of Children With IEM
In total, 35 siblings answered the questionnaires. Their characteristics are described in Table 2. Table 4 shows their mean KINDL scores, disease subscale scores, and our original scoring of emotional support resources in daily life.
The mean age of siblings was 12.4 ± 3.3 years (Table 2), and the mean score of their QoL was 71.7 ± 12.6 points (Table 4). There was no significant correlations between QoL (total KINDL scores) and other factors. Three KINDL subscales were significantly positively correlated with emotional support resources: physical (r = 0.491, P < .01), emotional (r = 0.445, P < .01), and family (r = 0.505, P < .01). There were negative correlations between the friends subscale and their age (r = −0.469, P < .01; Table 1).
Primary Caregivers of Children With IEM
A total of 143 primary caregivers answered the questionnaires, 92.3% of them were mothers. Table 3 shows their characteristics, and Table 5 shows the scores of each scale. Supplemental Table 1 shows the details of predictive factors of primary caregivers’ QoL.
Characteristics of Primary Caregivers of Children With IEM and Their Partners.
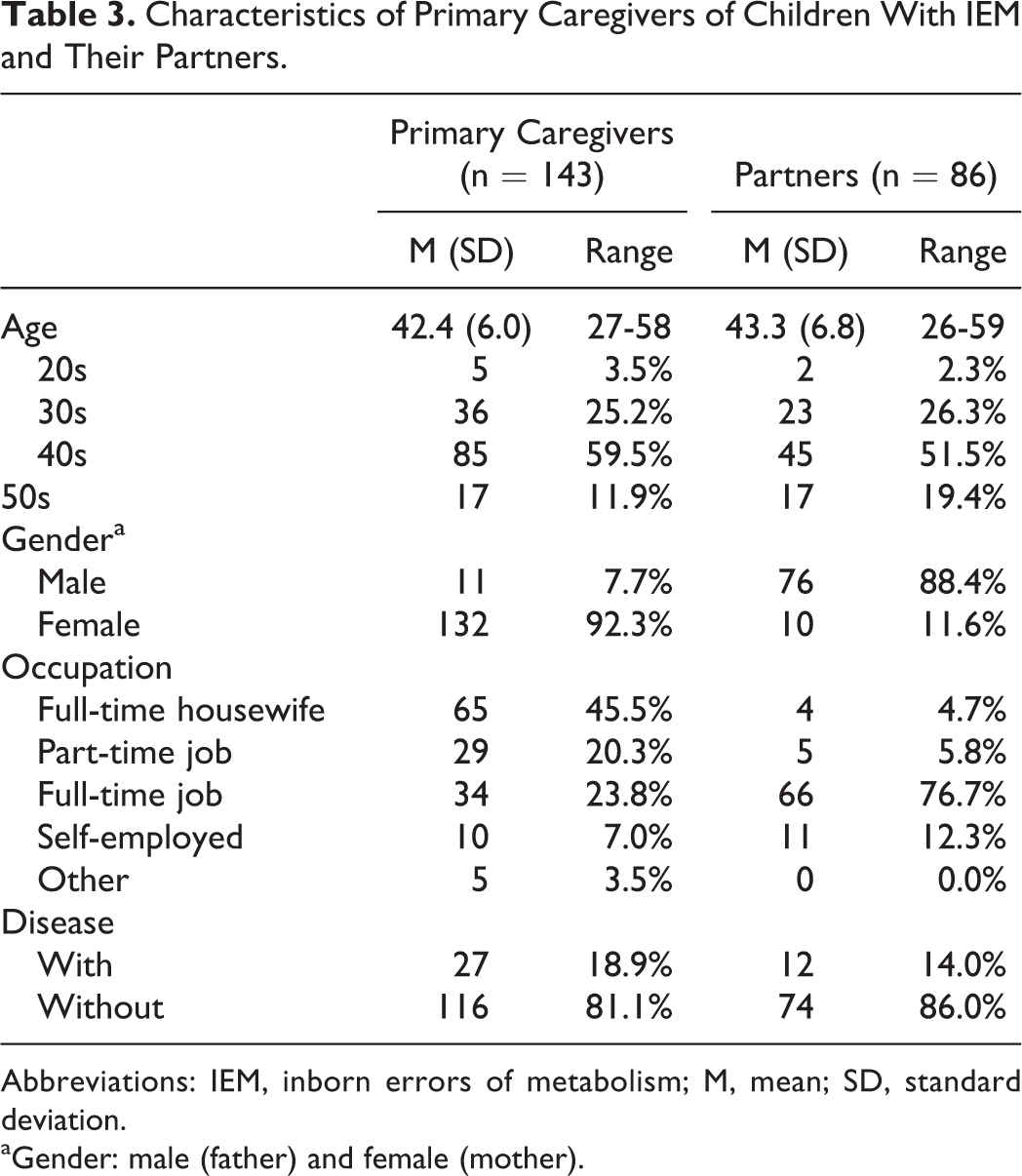
Abbreviations: IEM, inborn errors of metabolism; M, mean; SD, standard deviation.
aGender: male (father) and female (mother).
Current Status of QoL in Children With IEM and Their Siblings.a
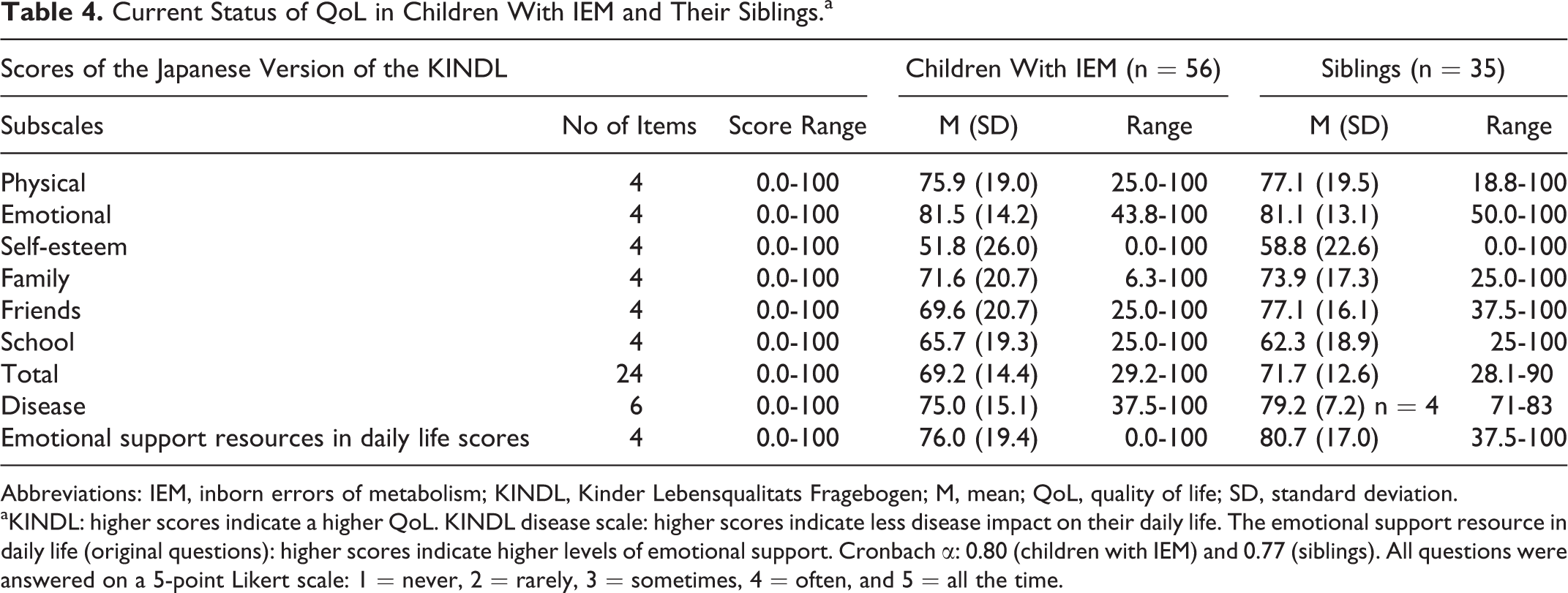
Abbreviations: IEM, inborn errors of metabolism; KINDL, Kinder Lebensqualitats Fragebogen; M, mean; QoL, quality of life; SD, standard deviation.
aKINDL: higher scores indicate a higher QoL. KINDL disease scale: higher scores indicate less disease impact on their daily life. The emotional support resource in daily life (original questions): higher scores indicate higher levels of emotional support. Cronbach α: 0.80 (children with IEM) and 0.77 (siblings). All questions were answered on a 5-point Likert scale: 1 = never, 2 = rarely, 3 = sometimes, 4 = often, and 5 = all the time.
The mean age of primary caregivers was 42.4 ± 6.0 years (Table 3), and the mean score of their QoL was 3.29 ± 0.62 points. The mean score of PSI-SF was 45.2 ± 10.6 points and that of J-FES was 103.3 ± 0.7 points. The mean score of WHOQOL-26 was 3.29 ± 0.62 points (Table 5). Multiple regression analysis was performed with the total WHOQOL-26 score as the dependent variable. Lower parental distress scores (standard partial regression coefficient [sb] = −0.586, P < .001), higher scores on the J-FES family subscale (sb = 0.248, P < .001), and higher levels of household income (sb = 0.147, P = .006) contributed to higher QoL of primary caregivers (adjusted R 2 = 0.636), as shown in Table 6.
Current Status of QoL in Primary Caregivers and Partners, Parental Stress of Primary Caregivers, and Family Empowerment.a
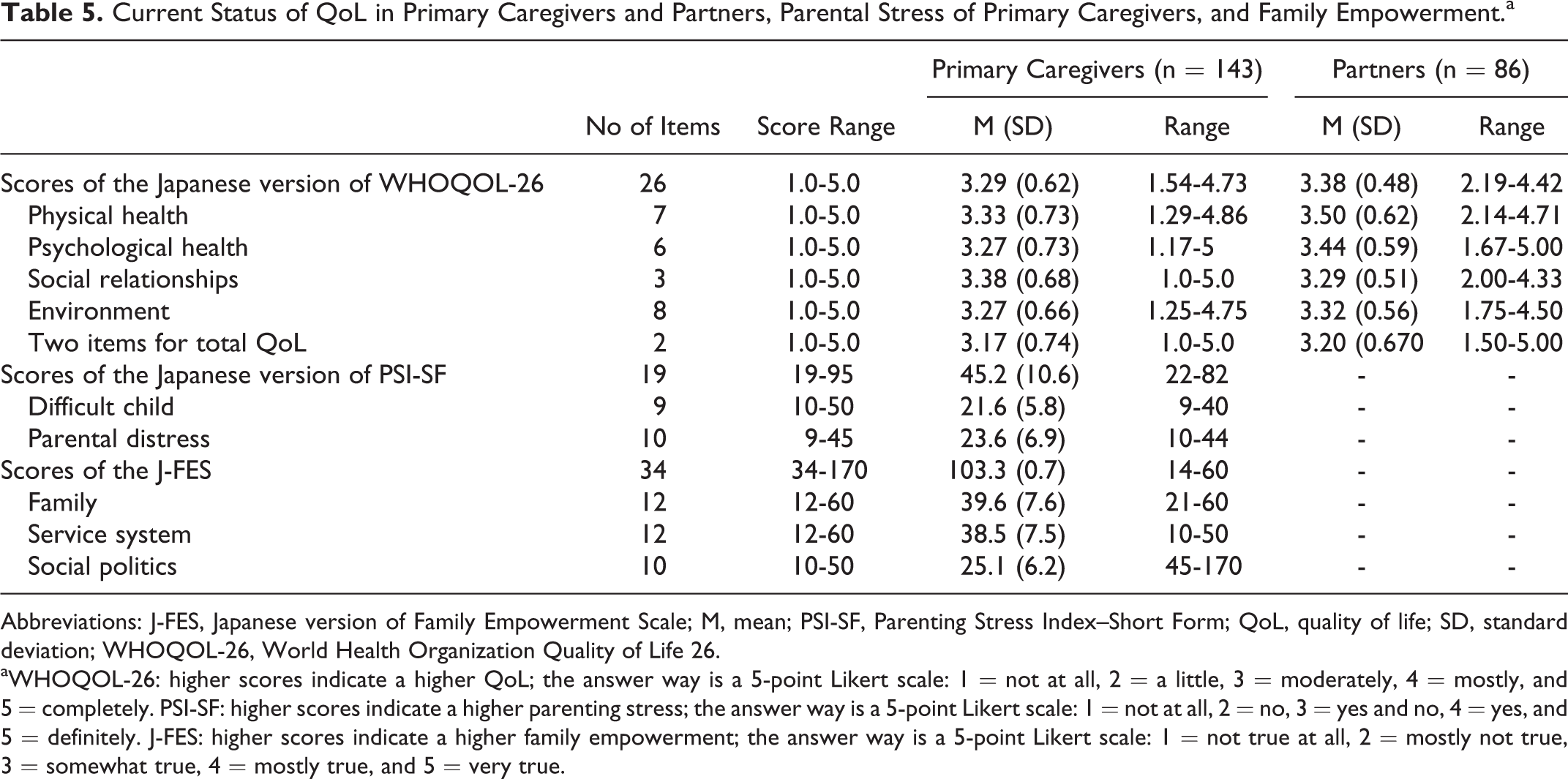
Abbreviations: J-FES, Japanese version of Family Empowerment Scale; M, mean; PSI-SF, Parenting Stress Index–Short Form; QoL, quality of life; SD, standard deviation; WHOQOL-26, World Health Organization Quality of Life 26.
aWHOQOL-26: higher scores indicate a higher QoL; the answer way is a 5-point Likert scale: 1 = not at all, 2 = a little, 3 = moderately, 4 = mostly, and 5 = completely. PSI-SF: higher scores indicate a higher parenting stress; the answer way is a 5-point Likert scale: 1 = not at all, 2 = no, 3 = yes and no, 4 = yes, and 5 = definitely. J-FES: higher scores indicate a higher family empowerment; the answer way is a 5-point Likert scale: 1 = not true at all, 2 = mostly not true, 3 = somewhat true, 4 = mostly true, and 5 = very true.
Factors Associated With Primary Caregivers’ and Partners’ QoL While a Child With IEM.
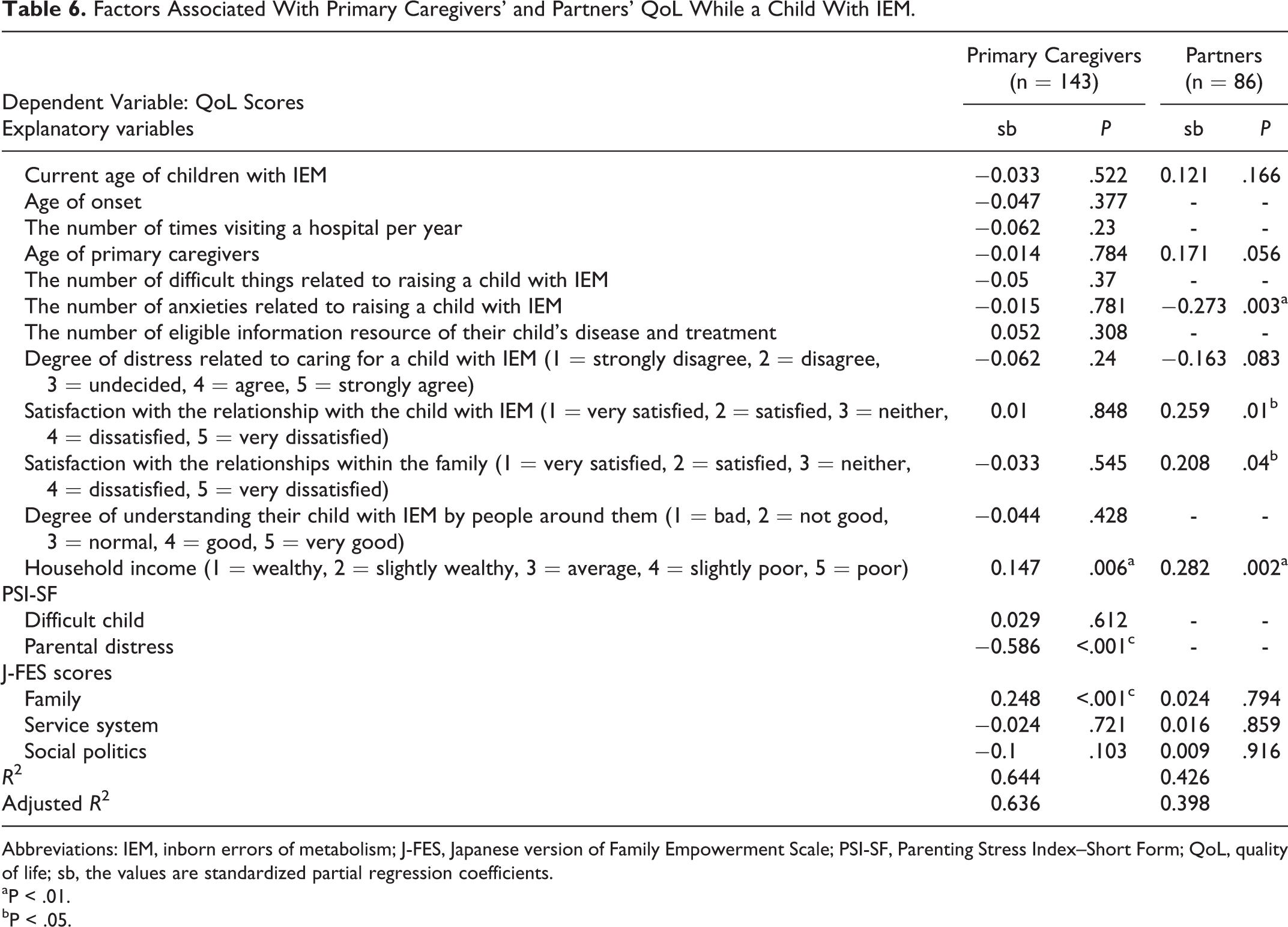
Abbreviations: IEM, inborn errors of metabolism; J-FES, Japanese version of Family Empowerment Scale; PSI-SF, Parenting Stress Index–Short Form; QoL, quality of life; sb, the values are standardized partial regression coefficients.
aP < .01.
bP < .05.
Partners of Primary Caregivers of Children With IEM
In total, 86 partners answered the questionnaires. A total of 88.4% of partners were fathers. Their characteristics are shown in Table 3, and Table 5 shows their scores on each scale. Supplementary Table 2 shows the details of predictive factors of QoL.
The mean age of partners was 43.3 ± 6.8 years (Table 3), and mean score of their QoL was 3.38 ± 0.48 points. In total, 93% of partners had some anxieties related to raising a child with IEM, and the distribution was similar to that of primary caregivers. The mean score of WHOQOL-26 was 3.38 ± 0.48 points (Table 5). Multiple regression analysis was performed with the total WHOQOL-26 score set as the dependent variable. The J-FES score provided by primary caregivers were assigned to their respective partners in this analysis because the J-FES score provided by the primary caregiver represents the empowerment represented by the entire family. Higher levels of household income (sb = 0.283, P = .002), fewer anxieties related to raising a child with IEM (sb = −0.273, P = .003), higher levels of satisfaction in the relationship with the child with IEM (sb = 0.259, P = .01), and higher levels of satisfaction with relationships within the family (sb = 0.208, P = .04) contributed to higher QoL of partners (adjusted R 2 = 0.398), as shown in Table 6.
Discussion
This was the first study to reveal current QoL and its associated factors in children with IEM, their siblings, primary caregivers, and their partners in Japan. Medical professionals and government employees, especially those involved in policy making for pediatric chronic illness, can now better understand QoL of these individuals and refer to these results when devising effective support to improve their QoL. The response rate was quite small because the questionnaires were mailed, and no incentive payments for families to cooperate with this study.
Children With IEM
Previous studies reported that QoL of children with IEM was lower than those with other pediatric chronic diseases. 24,25 Our results suggest that QoL tends to be lower in Japanese children with IEM, compared to the results of healthy Japanese children (73.2 points). 26 Additionally, in accordance with the results of previous studies in other countries, 5,24 QoL of children with IEM in Japan was affected by their condition. According to a previous study in France, the physical dimension of QoL was more affected by IEM than the psychological dimension. 24 Similar results can be seen in our study: the mean scores on the physical subscale of the KINDL tended to be lower than those of the emotional subscale, compared to the siblings’ mean score and the mean scores of healthy children (physical: 82.4 points, emotional: 88.0 points). 26 There are some possible explanations. One is respondent bias, as only children with IEM who were under stable conditions and not staying in hospital were enrolled in this study. Second is a considerable experience of physical symptoms: for example, it is well known that patients with citrin deficiency experience daily fatigue, even under stable conditions, which affected their QoL. 2,27 Third is that they had sufficient emotional support resources: the results by our questions. Interestingly, enough emotional support resources contribute to all dimensions of KINDL (QoL), and the number of siblings was negatively correlated with the self-esteem dimension of QoL of children with IEM. Generally, Japanese children’s self-esteem is potentially low, 28 and pediatric diseases affect their self-esteem and QoL. 29,30 Our results imply that the traits of their child–parent relationship and child–sibling relationship in the family with IEM and their effects on their QoL.
Siblings of Children With IEM
Although studies on siblings of children with chronic conditions, such as pediatric cancer and disabilities, exist, 31,32 no previous research has focused on the QoL of siblings of children with IEM. In these previous studies, it was reported that siblings of children with chronic conditions have anxiety for their future and problems with family relationships. However, siblings’ QoL was in the same range as that of healthy children 26 and tended to be slightly better than the QoL scores of children with IEM. Age was negatively correlated with the friends’ dimension of QoL of siblings, possibly because the relationships between children become more complicated when growing up. 33 As a next step, the study of siblings of children with IEM by qualitative approach might be needed to understand their anxiety for their future and problems with family relationships.
Primary Caregivers of Children With IEM
The QoL scores in primary caregivers were similar to those of healthy Japanese women (3.32 ± 0.41 points) and mothers raising children with food allergy (3.4 ± 0.4 points). 15,22 A previous study of parents raising children with IEM in Germany revealed predictors of parents’ QoL. Older age in parents was positively associated with QoL, specifically in the emotional dimension and self-development, 34 but the age of primary caregivers was not an associated factor in this study. It is difficult to conclude whether age affects the QoL due to the biased distribution of the primary caregivers’ ages; about 60% of the primary caregivers in this study were in their 40s. The researchers also reported the predictors of QoL to be family stress and social support. 34 In the current study, parental distress was the most influential factor of QoL of primary caregivers. For primary caregivers of children with IEM, the responsibility of managing the child’s health must be included in the parental role, and this might result in considerable distress for them. Referring to the results of our literature review, 6 we consider that the small number of clinical doctors specializing in IEM and the lack of information about raising a child with IEM are also related to parental distress. It is also known that parents feel guilty about passing on a genetic disease, 3 and this contributes to their distress. Additionally, more than half of the children with IEM were diagnosed as having amino acid disorder; therefore, many parents could be responsible for dietary therapies. In fact, this is supported by the fact that over 40% of the primary caregivers responded that they had difficulty with diet therapy (Supplementary Table 1). In the future, we should investigate the parental distress of primary caregivers of children with IEM and diet therapy, in order to help to think the concrete way to support their childrearing.
Family empowerment was the second most influential factor on primary caregivers’ QoL. This was the world’s first study to explore family empowerment in families raising a child with IEM. The family subscale represents their power and confidence in raising their child within the family. 19 We clarified that improving their family empowerment contributes to improving primary caregivers’ QoL, while raising a child with IEM. Therefore, we considered that supporting families as a whole is a significant perspective not only individual family members.
Partners of Primary Caregivers of Children With IEM
Compared to the national data on healthy Japanese men (3.24 ± 0.43 points), 15 the QoL of partners raising their child with IEM was higher in this study. The most positive influential factor of partners’ QoL was household income. As previously mentioned, all children with IEM receive medical aid for hospital admission and visits and therapeutic special milk in Japan. 35 However, this medical aid does not cover the cost of any low-protein diet food, and such foods are always priced higher than common foods. Low-protein diet food is necessary for their appropriate diet therapy. 36 We could not conclude the causal correlation between their household income and the burden of high-priced low protein food in this study. It might be needed to investigate the annual cost of low-protein food and reconsider the application of medical aid for those families. Anxiety negatively related to raising their child with IEM was the secondary associated factor of their QoL. One previous study reported that it was important for parents raising a child with special needs to express and share their anxieties. 37 A representative of a parental support group of patients with IEM suggested that the primary reasons for joining the group was having considerable anxiety and loneliness due to the rarity of the disease. 38 Medical professionals should introduce such families to peer groups such as parental support group for children with IEM and provide the environment where they can easily share their anxieties at the daily outpatient setting.
Conclusion
Our study was the first cross-sectional questionnaire survey, revealing QoL and associated factors in children with IEM, their primary caregivers, and other family members in Japan. We revealed that there are different associated factors of QoL or each participant group, and we believe that each group should be supported inclusively: from the perspective of individuality and the family as a whole. By effectively utilizing the registration system (JaSMIn), important outcomes for the development of new treatments and improving QoL will be reported in the future too. The next challenge would be the development of interventions for improving QoL for these individuals in an outpatient setting.
Supplementary Material
Supplementary Material
Supplemental material, Book1 for Quality of Life and Associated Factors in Japanese Children With Inborn Errors of Metabolism and Their Families by Keiko Yamaguchi, Rie Wakimizu and Mitsuru Kubota in Journal of Inborn Errors of Metabolism and Screening
Footnotes
Acknowledgments
The authors would like to express our greatest appreciation to Editage for their courteous English proofreading.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a research scholarship from the Pfizer Health Research Foundation, 2015 to 2016. The study was partially supported by the Practical Research Project for Rare/Intractable Diseases from the Japan Agency for Medical Research and Development (AMED).
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
