Abstract
The mucopolysaccharidosis (MPS) disorders are rare genetic diseases caused by deficiencies in lysosomal enzymes involved in the degradation of glycosaminoglycans, leading to pulmonary, cardiac and neurological dysfunctions, skeletal anomalies, impaired vision, and/or hearing and shortened life spans. Whereas in the past, few individuals with MPS reached adulthood, better diagnosis, multidisciplinary care, and new therapies have led to an increasing number of adult patients with MPS. Therefore, fertility and pregnancy questions in this patient population are becoming more important. Management of fertility issues and pregnancy in patients with MPS is challenging due to the lack of documented cases and a dearth in the literature on this topic. This review presents multidisciplinary expert opinions on managing fertility and pregnancy based on case studies and clinical experience presented at a meeting of MPS specialists held in Berlin, Germany, in April 2015. An overview of the existing literature on this subject is also included.
Introduction
The mucopolysaccharidosis (MPS) disorders comprise a group of rare lysosomal storage disorders caused by deficiencies in enzymes involved in the catabolism of glycosaminoglycans (GAGs; Appendix A). 1,2 Glycosaminoglycans are hydrophilic polymers of highly modified hexose saccharides found in part of the connective tissue matrix. Deficient catabolism leads to intra- and extracellular accumulation of GAGs in a variety of tissues and subsequent multi-organ dysfunctions. Features of MPS include short stature, skeletal, and joint abnormalities (hip dysplasia, knee/ankle valgus, kyphosis/scoliosis, atlanto-axial instability, chest deformities, joint stiffness/hypermobility), impaired respiratory function (upper/lower airway obstruction, restrictive lung disease), cardiac dysfunction (valve stenosis/insufficiency, left ventricular hypertrophy, abnormal diastolic function), neurological disease (spinal cord compression), hepatosplenomegaly, impaired vision, and hearing loss (Figure 1). 1 Patients with severe forms of MPS I (MPS IHS OMIM #607015 and MPS IH OMIM #607014), II (OMIM #309900) and VII (OMIM #253220), and all patients with MPS III (OMIM #252900, #252920, #252930, #252940) may show cognitive and neurological decline. 1

Adult patients with mucopolysaccharidosis (MPS) VI showing their characteristic appearance. Reproduced from Harmatz and Shediac 3 , with permission from Frontiers in Bioscience.
More patients with MPS are surviving to adulthood due to earlier diagnoses, multidisciplinary care, and the availability of specific therapies such as enzyme replacement therapy (ERT) and hematopoietic stem cell transplantation (HSCT). 4,5 Therefore, patients with MPS are presenting new challenges for adult care physicians. Specifically, adults with MPS are considering having children and as such, clinicians will likely encounter fertility and pregnancy questions in this patient population. To address this changing clinical picture, we present multidisciplinary expert opinions on managing fertility and pregnancy in MPS and provide an overview of the existing literature on this subject.
Methods
Expert opinions on managing fertility and pregnancy in MPS are based on presentations and discussions of case studies from an advisory board meeting with MPS specialists held on April 18, 2015 in Berlin, Germany, and on the existing relevant literature. The advisory board of international experts on fertility issues and pregnancies in patients with MPS included pediatric and adult geneticists, an adult metabolic specialist, a pediatric endocrinologist, an adult respiratory and critical care specialist, a cardiologist, a nurse practitioner, a pediatrician, a subspecialist in maternal and fetal medicine, an obstetric anesthesiologist, and a genetic counselor.
Published articles on this subject were identified by searching the following terms in PubMed: “(“mucopolysaccharidoses” [MeSH Terms]) and (pregnancy or mother or father or fertility or puberty [MeSH Terms]).” Relevant papers were selected from this search, and reference lists were searched for additional material on the subject not identified by the PubMed search.
Evidence from Literature
Pubertal Development and Fertility in MPS
Data on pubertal development in patients with MPS are limited. Children with MPS IH and MPS III may show precocious puberty (<9 years for boys and <8 years for girls). 6,7 In some cases, this may be due to having a HSCT rather than MPS. 6 Delayed puberty (14 years for boys, 13 years for girls) can occur in patients with MPS IVA (OMIM #253000) and MPS VI (OMIM #253200). 7,8
Our literature search revealed limited published material on the impact of MPS on fertility (1 human study and 4 animal studies). Glycosaminoglycans have been shown to accumulate in the gonads (testes, ovaries), placenta, and pituitary of animals and humans with MPS and may interfere with reproductive function. 9 -11 Glycosaminoglycan accumulation in the testes and ovaries, a decrease in sperm count, and changes in sperm morphology (loss of head and tail) have been reported for MPS I mice. 11,12 Poor fertility is documented in female MPS I and MPS II mice. 13 Mucopolysaccharidosis VII mice showed accumulation of GAGs in gonads and uteri but normal maturation of sperm, follicles, and corpora lutea. 14 Although male MPS VII mice are reproductively sterile as a result of reduced mobility and/or impaired cognitive function, females can mate but are unable to keep their young due to insufficient lactation. 14,15
Fertility studies in humans with MPS are limited. Yasuda et al showed reduced spermatogenesis in 1 patient with MPS IVA. 10 No other significant fertility issues in humans with MPS have been described. Some published case studies describe female or male patients with MPS having children, 16 -27 but it was not reported whether any of these patients had difficulties conceiving.
Allogenic HSCT has been shown to adversely impact fertility due to delayed consequences of chemotherapy and radiotherapy causing gonadal failure and hormone deficiency characterized by abnormally low blood testosterone and estradiol levels. 28 Irradiation of the uterus can cause poor embryo implantation or, in the case of pregnancy, intrauterine growth restriction, prematurity, and uterine rupture. 28 However, the conditioning regimen is often less aggressive in transplants for metabolic diseases. Two case reports of pregnancies in women with MPS I who had HSCT at 14 months and 4 years show that early transplantation does not necessarily preclude pregnancy in these patients. 18,19
Pregnancy and Delivery in MPS
A number of case studies in the literature address pregnancy and delivery in women with MPS, 16 -20,22,27,29 with many reporting high-risk pregnancies and deliveries.
A woman with severe MPS IVA developed polyhydramnios and dyspnea by the 21st week of gestation, leading to delivery of a premature child (1.18 kg, 38 cm) at 28 weeks. The child showed anemia, hyperbilirubinemia, apnea, and bronchopulmonary dysplasia but was discharged in good condition. 17 Severe myelopathy in the third trimester of pregnancy due to compression of the cervical spinal cord was reported for a 25-year-old woman with MPS VI. 22 An MPS IH patient, homozygous for the W402X mutation, who had a successful bone marrow transplantation at the age of 14 months became pregnant at the age of 21 years. Because of her poor clinical condition (severe gibbus abnormality, subluxation of the hips, and instability of the knees), the patient decided to terminate her pregnancy at 9 weeks due to concerns about her own health and her ability to care for the baby. 19 A female with MPS I who discontinued ERT when she became pregnant gave birth prematurely, through normal delivery, to a girl of 1.25 kg at 29 weeks gestation. 20 Her clinical condition deteriorated rapidly after discontinuation of ERT.
Other case reports have described successful pregnancies and deliveries in women with MPS. 16,18,22 For example, a woman with MPS IHS treated with allogenic bone marrow transplantation at the age of 4 years had 4 successful pregnancies without reproductive assistance between the ages of 18 and 26 years. 18 Delivery occurred between 37 and 39 weeks of gestation by cesarean section. A woman with MPS IS (OMIM #607016) receiving ERT during pregnancy delivered a healthy boy by cesarean section at 37 weeks. 16 Women with milder attenuated forms of MPS have been reported to have children before they were diagnosed. 21,26 Most MPS mothers deliver by cesarean section (with spinal or epidural anesthesia), mainly due to cephalopelvic disproportion secondary to maternal short stature. 16 -18,21,22 Clinical guidelines are lacking in this area as most case studies describe the results rather than the suggested management.
The literature regarding pregnancy in patients with MPS having cognitive impairment is very limited. Case reports of patients with MPS IH or MPS HIS and a woman with mild MPS II concerned patients with only mild cognitive delay. 18,19,25 One published case report described a woman with MPS IIIB who had a daughter. 26 Cognitive and behavioral retardation and deterioration led to domestic conflicts. However, she was diagnosed very late, at 57 years of age, after her daughter had left the parental home.
Complications during pregnancy or delivery can arise from exacerbation of preexisting clinical manifestations of MPS due to the physiological adaptations of the body to pregnancy or during general or regional anesthesia for cesarean section. 17,22 Clinical issues of MPS that can cause problems during pregnancy include cardiac and respiratory manifestations (eg, aortic or mitral valve insufficiency/stenosis, left ventricular hypertrophy, abnormal diastolic function, upper and lower airway obstruction, extrathoracic and lung restriction), short stature, kyphoscoliosis, hip dysplasia, spinal cord compression, and hepatosplenomegaly. 30 -33 Cardiac and respiratory manifestations can worsen with increased cardiac output and respiratory drive, diaphragmatic splinting due to the gravid uterus, reduced residual capacity, increased oxygen requirements, decreased venous outflow, vasodilatation, and airway edema that occur during pregnancy. 34,35 Diaphragmatic splinting from the enlarging uterus can occur in women with MPS due to short stature, kyphosis/scoliosis, or hepatosplenomegaly causing restrictive pulmonary/respiratory function from around 28 weeks onward. 34 Patients with a small abdomen are also at increased risk of reflux or aspiration due to increasing intra-abdominal pressure during pregnancy. 34 Weight gain and lordosis associated with pregnancy increase the risk of hyperlordosis, resulting in lower back pain and instability, particularly in the many patients with MPS having preexisting spine abnormalities. 36 Finally, in patients who receive steroids as pretreatment with ERT to avoid infusion-associated reactions, insufficiency of the hypothalamic–pituitary–adrenal axis should be considered. During normal pregnancy, there is an upregulation of the hypothalamic–pituitary–adrenal axis and failure to provide this activation poses a substantial risk to both mother and child. 37,38
Few studies in the literature have addressed anesthesia during delivery for women with MPS. 27,29 One case report described effective epidural anesthesia for cesarean section in a woman with MPS IH. 27 Recently, a woman with MPS IVA was reported to have inadequate pain management during labor with epidural anesthesia, probably due to a combination of lumbar canal stenosis, thecal sac compression, and kyphoscoliosis causing suboptimal spread of anesthetics in the epidural space. 29 Studies with nonpregnant patients with MPS, mostly children, suggest that general anesthesia can pose a high risk. 39 -42 Individuals may experience complications with bag mask ventilation, intubation, extubation, subglottic stenosis, and secretion clearance during anesthesia. 40 Cardiac problems, spinal cord compression, and atlantoaxial instability also increase anesthetic risk. 40,42 In addition, positioning of the patient during anesthesia can be difficult due to kyphosis. Airway management during general anesthesia is problematic in children with MPS. Therefore, consideration should be given to the preservation of spontaneous ventilation. Muscle relaxants should be used with great caution since loss of airway tone after paralysis may lead to total airway occlusion. Securing the airway with a fiber-optic technique, while the patient is awake, may be necessary. In case of severe airway difficulties during general anesthesia, emergency tracheostomy may be required. Tracheostomy, however, should be a last resort as it poses significant risk in patients with MPS for anatomical reasons and due to thickened skin and tissue from accumulated GAGs. 40
Evidence in the literature addressing the risks and/or advantages of ERT use during pregnancy and lactation is very limited. Castorina et al recently described a woman with MPS IS who continued ERT throughout pregnancy and while lactating for 3 months. 16 The child did not develop antidrug (laronidase) antibodies and had normal urinary GAG levels. No laronidase was detected in the breast milk.
Corridor of Uncertainty
The expert meeting in Berlin revealed several areas relating to fertility and pregnancy in patients with MPS that were underrepresented in the published literature. Very little is known about the social and psychological aspects of pregnancy and child rearing as it relates to individuals with MPS. The case reports and discussions in Berlin revealed that only a minority of adults with MPS choose to have children. The explanation for this is likely to be multifactorial and may include physical, mental, or financial hurdles associated with child rearing, difficulties in finding a partner possibly due to challenges in self-esteem, social connections, and social skill development, apprehension regarding difficult pregnancy and delivery, and concerns about the heritability of MPS. Patients who become parents often indicate that management of their disease is challenging and that they require extensive support from their family and partner in raising the children.
The expert advisory group revealed that most patients with MPS under their care were not well informed about the risks associated with pregnancy and delivery, or the genetic heritability component of MPS. In fact, few patients with MPS consider genetic testing of their partner prior to pregnancy to evaluate the risk of passing on MPS to their offspring.
There are a limited number of published papers focusing on fertility issues and management of complications during pregnancy in patients with MPS in clinical practice (1 case report on fertility and 8 on pregnancy). The case series presented in Berlin confirmed that women with MPS and partners of men with MPS in tertiary care are able to become pregnant without fertility assistance. In addition, most women delivered nearly full term, and all delivered healthy babies. However, these outcomes may not be representative of all MPS pregnancies as unsuccessful pregnancies in patients with no access to expert care may go unreported. Despite the successful outcomes of pregnancies in tertiary care, most women in the series experienced high-risk pregnancies and deliveries. Complications reported include lumbar pain, atrial tachycardia, problems related to disproportion in size between mother and child, the need for cesarean section, and umbilical hernia and deterioration of cardiac function after delivery. Basic care of the baby was difficult for some patients due to their short stature and other physical issues related to MPS.
Published evidence regarding the risks and advantages of ERT use during pregnancy and lactation in MPS mothers is limited to 1 case report. 16 However, several case reports of ERT use during pregnancy with good outcomes in a tertiary care setting were presented in Berlin. Enzyme replacement therapy use during lactation (for only 4 weeks) was reported for only one of the MPS cases. More evidence exists for other lysosomal storage diseases such as Gaucher disease, for which experts concluded that there is potentially more risk involved in stopping ERT than in continuing its use during pregnancy. 43,44 Regarding ERT use during lactation, the Berlin advisory group postulated that, if enzyme is indeed excreted in breast milk, it would likely be digested in the baby’s gastrointestinal tract and therefore not be absorbed in significant quantities.
There are currently no guidelines for managing fertility and pregnancy in MPS. Guidelines for anesthesia in patients with MPS have been published. 40 These should be consulted, even when general anesthesia is not planned for delivery, due to the potential need for emergency cesarean section.
Conclusions and Expert Opinions
The following MPS advisory panel opinions are based on the current literature and case reports presented in Berlin:
Expert Opinions on Evaluation of Puberty in Patients With MPS
Tanner staging should be assessed annually, starting at 7 years of age. Successful progression through puberty is required to establish fertility and pregnancy potential. If clinical signs of precocious or delayed puberty arise, hormone levels (luteinizing hormone [LH]/follicle-stimulating hormone [FSH], testosterone/estrogen) should be measured. Bone age in MPS can be challenging to determine due to difficulty comparing results to reference ranges based on an unaffected population. In patients with LH/FSH mediated-central precocious puberty (before the age of 8 years in girls and 9 years in boys), magnetic resonance imaging must be performed to determine the presence of a suprasellar or sellar mass. Patients with delayed puberty should be screened for thyroxine (T4), cortisol, and growth hormone to see whether there is pituitary dysfunction. If there are confirmed issues with puberty, hormone therapy can be discussed with the patient and his/her family (from age 12 years for girls and 13 years for boys).
Deviation from MPS-related growth curves should prompt evaluation by a pediatric endocrinologist. Growth hormone deficiency has sporadically been reported in patients with MPS 7,9,45 and may be accompanied by deficiencies in other pituitary hormones, including LH and FSH, which can impact growth, puberty, and fertility. If growth hormone is deficient, it is important to test for other pituitary hormone deficiencies that could result from the underlying lysosomal storage disease.
Expert Opinions on Management of Fertility Issues, Pregnancy, and Delivery in Patients With MPS
Counseling
All patients with MPS should be counseled to address questions about genetic heritability and expectations and needs with regard to fertility and pregnancy (Figure 2; Table 1). Appendix B provides information on the inheritance of MPS.
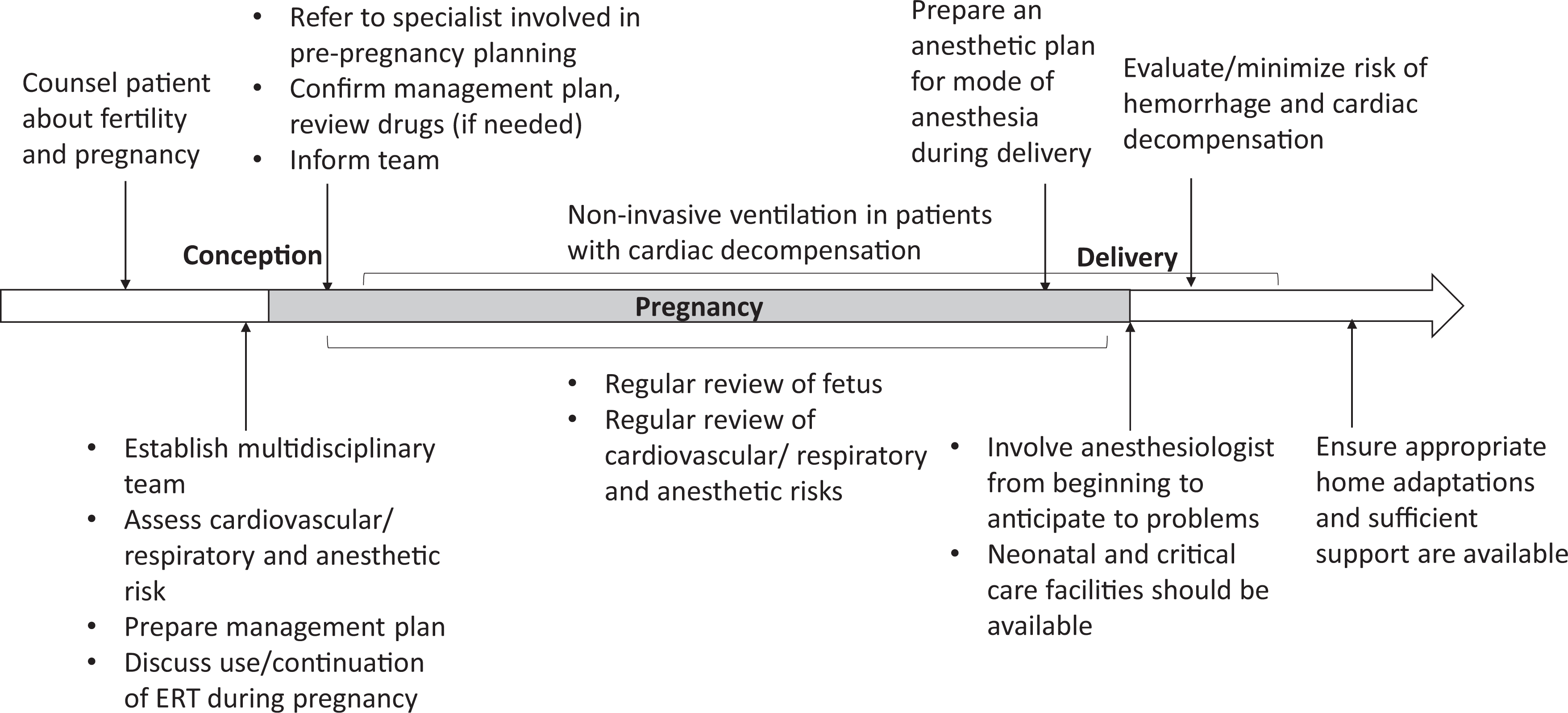
Planning of management and follow-up before and during pregnancy and during and after delivery. ERT indicates enzyme replacement therapy.
Counseling Recommendations for Fertility and Pregnancy.

Abbreviation: MPS, mucopolysaccharidosis.
Preconception counseling and fertility issues
In case of fertility issues in women with MPS, physical, psychological, and social maternal medical conditions should be assessed. Multidisciplinary preconceptual assessment and counseling (see below) is essential to fully elicit the potential physical impact of pregnancy. Mucopolysaccharidosis centers should have a link with the tertiary obstetric unit and contact an obstetrician to arrange multidisciplinary preconceptual counseling and investigation. If the maternal condition is such that there is a significant risk of severe morbidity- or mortality-related to pregnancy, discuss the option of surrogacy, adoption or to not have children. Once full multidisciplinary preconceptual assessment and counseling has taken place, and the maternal medical condition is optimized, the patient should be referred to a fertility specialist to evaluate intact endocrine/gonadal axis and uterine/fallopian tube/ovarian anatomy and function. If assisted reproduction is considered, the risk of multiple pregnancies should be minimized by avoiding multiple embryo implantation.
Pregnancy and delivery
Clinicians caring for adult patients embarking on pregnancy should be aware of how the medical aspects of MPS, including cardiac, pulmonary and skeletal manifestations, and anesthetic considerations, may impact pregnancy and delivery. Therefore, a detailed medical history is required, and clinicians need to be familiar with the specialized care involved in managing individuals with MPS disorders.
An individualized management approach is required when addressing the potential complications of pregnancy and delivery. Determining risk solely based on disease phenotype (eg, slowly vs rapidly progressing disease) should be avoided. Early planning of management and follow-up, before and during pregnancy, and during and after delivery, is the key to successful outcomes (Table 2).
Management Recommendations for Planning Pregnancy and Delivery.

Abbreviations: ERT, enzyme replacement therapy; MPS, mucopolysaccharidosis.
Patients may get practical advice and support from the MPS patient organization in their country.
Expert Opinions on the Use of ERT During Pregnancy or Lactation
When deciding whether to use ERT during pregnancy and lactation, physicians should closely follow product label recommendations. Possible risks and benefits to both mother and fetus should be considered. Regarding the initiation or continuation of ERT during pregnancy and lactation, a detailed assessment of the safety category listed for the ERT in the US and EU labels (applicable to all global labels) should be weighed against the risks of exacerbation of disease associated with cessation of ERT (Appendix C). 20
Sharing Experiences Among Health-Care Providers
Case reports of pregnancy and delivery in patients with MPS, as well as additional experience with ERT during pregnancy and lactation, should be published to facilitate experience with these high-risk and challenging cases. Detailed information should be recorded in a disease-specific pregnancy registry. Ongoing registries collecting data on pregnancy and lactation exist for MPS I (more info on www.MPSIregistry.com or call +1 (800) 745-4447), MPS IVA (Morquio A Registry Study; for more information, contact MARS@bmrn.com or call +1 (800) 983-4587), and MPS VI (more info on https://clinicaltrials.gov/ct2/show/NCT00214773). Although females with MPS II are very rare as it is an X-linked disorder, 1 if pregnancy occurs it may be reported to Shire (contact info on http://www.shiremedinfo.com). Pharmacovigilance needs to be contacted at each manufacturer if ERT is continued during pregnancy and/or lactation.
Footnotes
Appendix A
Overview of the Mucopolysaccharidosis (MPS) Disorders. 1,4
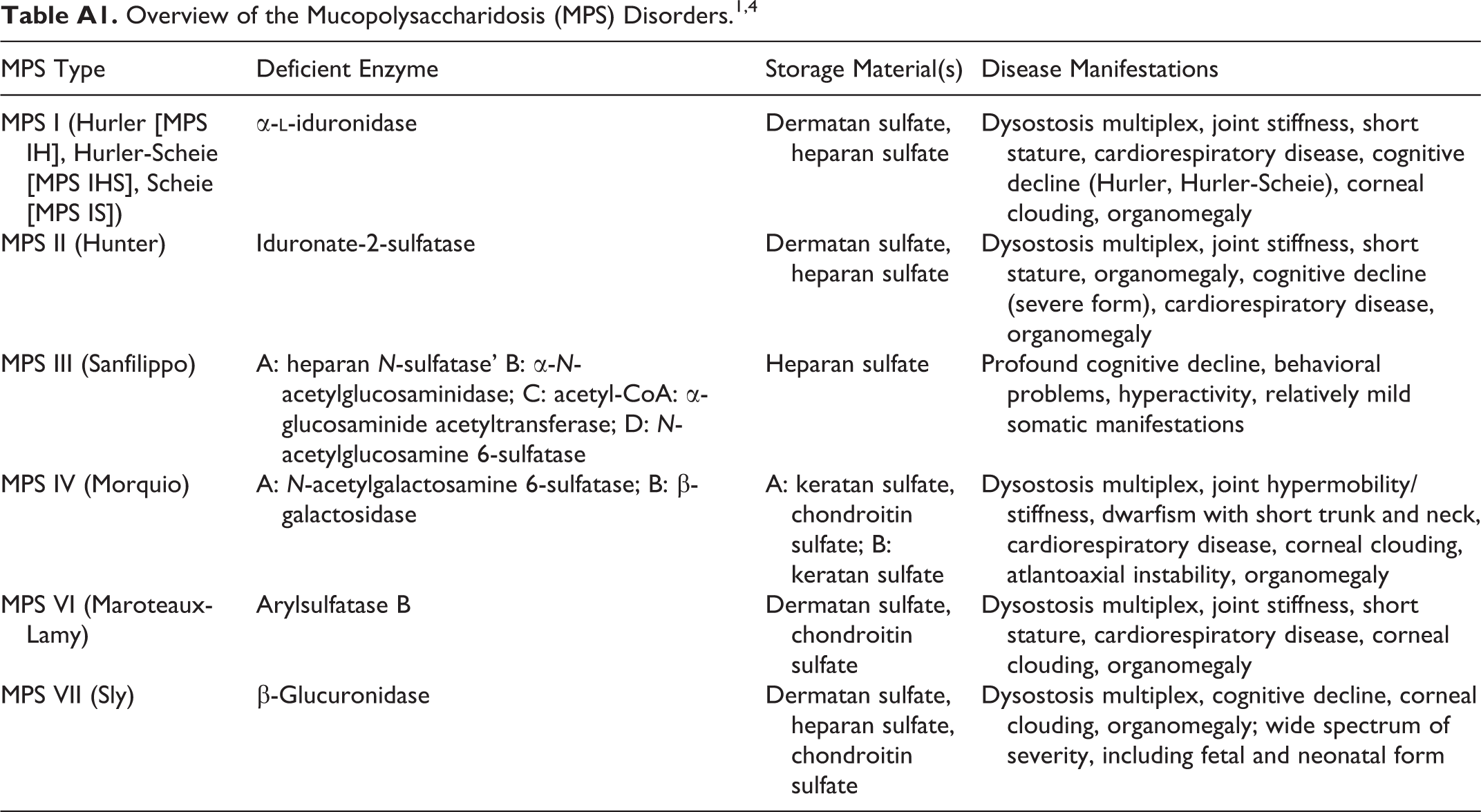
| MPS Type | Deficient Enzyme | Storage Material(s) | Disease Manifestations |
|---|---|---|---|
| MPS I (Hurler [MPS IH], Hurler-Scheie [MPS IHS], Scheie [MPS IS]) | α- |
Dermatan sulfate, heparan sulfate | Dysostosis multiplex, joint stiffness, short stature, cardiorespiratory disease, cognitive decline (Hurler, Hurler-Scheie), corneal clouding, organomegaly |
| MPS II (Hunter) | Iduronate-2-sulfatase | Dermatan sulfate, heparan sulfate | Dysostosis multiplex, joint stiffness, short stature, organomegaly, cognitive decline (severe form), cardiorespiratory disease, organomegaly |
| MPS III (Sanfilippo) | A: heparan N-sulfatase’ B: α-N-acetylglucosaminidase; C: acetyl-CoA: α-glucosaminide acetyltransferase; D: N-acetylglucosamine 6-sulfatase | Heparan sulfate | Profound cognitive decline, behavioral problems, hyperactivity, relatively mild somatic manifestations |
| MPS IV (Morquio) | A: N-acetylgalactosamine 6-sulfatase; B: β-galactosidase | A: keratan sulfate, chondroitin sulfate; B: keratan sulfate | Dysostosis multiplex, joint hypermobility/stiffness, dwarfism with short trunk and neck, cardiorespiratory disease, corneal clouding, atlantoaxial instability, organomegaly |
| MPS VI (Maroteaux-Lamy) | Arylsulfatase B | Dermatan sulfate, chondroitin sulfate | Dysostosis multiplex, joint stiffness, short stature, cardiorespiratory disease, corneal clouding, organomegaly |
| MPS VII (Sly) | β-Glucuronidase | Dermatan sulfate, heparan sulfate, chondroitin sulfate | Dysostosis multiplex, cognitive decline, corneal clouding, organomegaly; wide spectrum of severity, including fetal and neonatal form |
Appendix B
Appendix C
Overview of Safety Recommendations Listed for Enzyme Replacement Therapy Use During Pregnancy and Lactation in US and EU Labels.
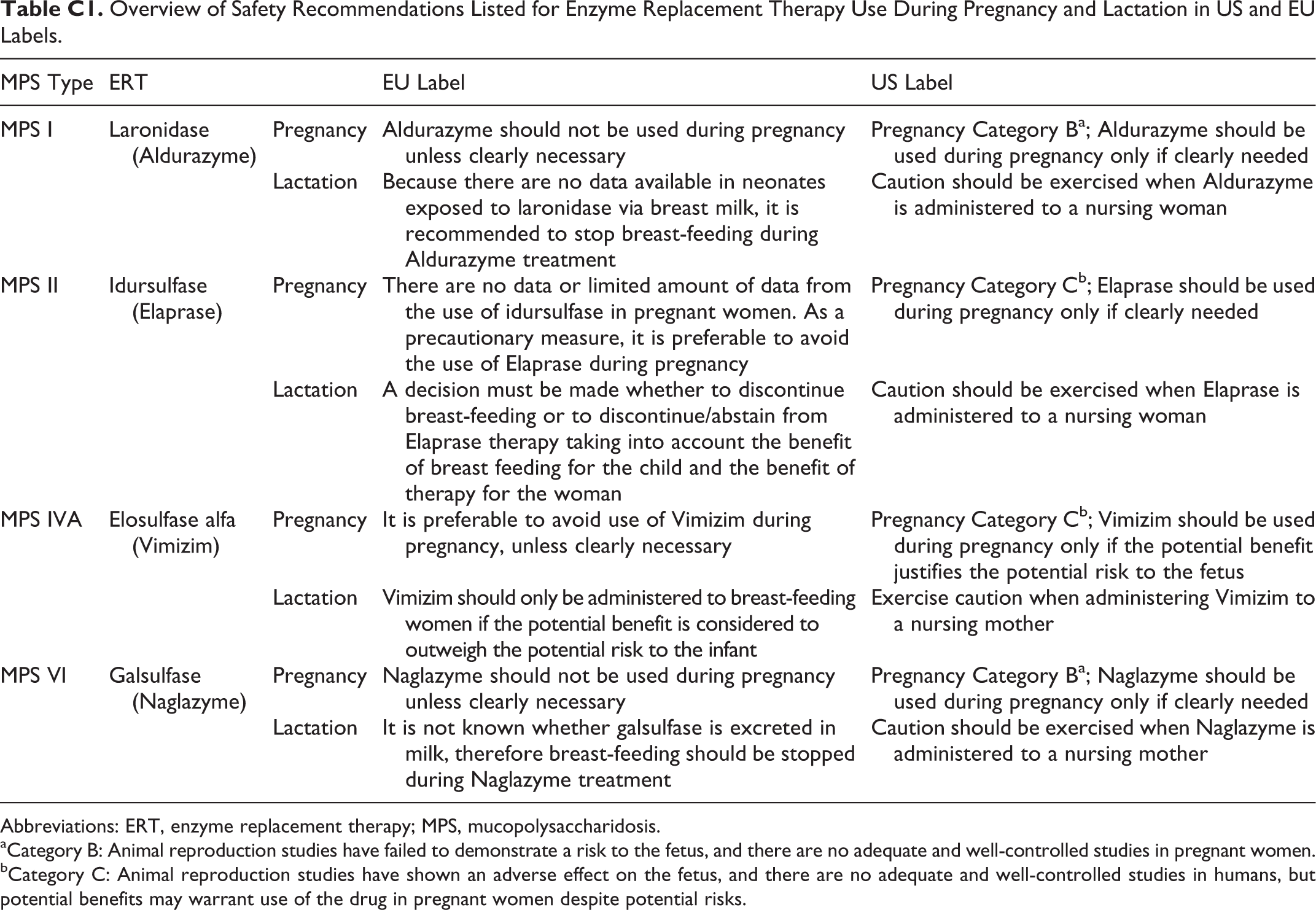
| MPS Type | ERT | EU Label | US Label | |
|---|---|---|---|---|
| MPS I | Laronidase (Aldurazyme) | Pregnancy | Aldurazyme should not be used during pregnancy unless clearly necessary | Pregnancy Category Ba; Aldurazyme should be used during pregnancy only if clearly needed |
| Lactation | Because there are no data available in neonates exposed to laronidase via breast milk, it is recommended to stop breast-feeding during Aldurazyme treatment | Caution should be exercised when Aldurazyme is administered to a nursing woman | ||
| MPS II | Idursulfase (Elaprase) | Pregnancy | There are no data or limited amount of data from the use of idursulfase in pregnant women. As a precautionary measure, it is preferable to avoid the use of Elaprase during pregnancy | Pregnancy Category Cb; Elaprase should be used during pregnancy only if clearly needed |
| Lactation | A decision must be made whether to discontinue breast-feeding or to discontinue/abstain from Elaprase therapy taking into account the benefit of breast feeding for the child and the benefit of therapy for the woman | Caution should be exercised when Elaprase is administered to a nursing woman | ||
| MPS IVA | Elosulfase alfa (Vimizim) | Pregnancy | It is preferable to avoid use of Vimizim during pregnancy, unless clearly necessary | Pregnancy Category Cb; Vimizim should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus |
| Lactation | Vimizim should only be administered to breast-feeding women if the potential benefit is considered to outweigh the potential risk to the infant | Exercise caution when administering Vimizim to a nursing mother | ||
| MPS VI | Galsulfase (Naglazyme) | Pregnancy | Naglazyme should not be used during pregnancy unless clearly necessary | Pregnancy Category Ba; Naglazyme should be used during pregnancy only if clearly needed |
| Lactation | It is not known whether galsulfase is excreted in milk, therefore breast-feeding should be stopped during Naglazyme treatment | Caution should be exercised when Naglazyme is administered to a nursing mother |
Abbreviations: ERT, enzyme replacement therapy; MPS, mucopolysaccharidosis.
aCategory B: Animal reproduction studies have failed to demonstrate a risk to the fetus, and there are no adequate and well-controlled studies in pregnant women.
bCategory C: Animal reproduction studies have shown an adverse effect on the fetus, and there are no adequate and well-controlled studies in humans, but potential benefits may warrant use of the drug in pregnant women despite potential risks.
Acknowledgments
The authors are grateful to Ismar Healthcare NV for their assistance in the writing of the manuscript, which was funded by BioMarin Pharmaceutical Inc. The current review was based on presentations and discussions from an expert meeting about fertility and pregnancy in MPS that was held on April 18, 2015, in Berlin, Germany. This meeting was coordinated and funded by BioMarin Pharmaceutical Inc. All authors participated in this meeting.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sue Graham and Stephen Sande are employees of BioMarin Pharmaceutical Inc, Zlatko Sisic is an employee of BioMarin Europe Ltd. All other authors received funding and travel support from BioMarin Pharmaceutical Inc to attend the advisory board meeting. In addition, Fiona Stewart reports personal fees and nonfinancial support from BioMarin and Shire and personal fees from Genzyme, outside the submitted work. Barbara Burton reports personal fees and clinical trial support from BioMarin, grants, personal fees and clinical trial support from Shire, personal fees and clinical trial support from Genzyme, personal fees from ReGenX Bio, and clinical trial support from Alexion, Ultragenyx and Cytonet, outside the submitted work. Nathalie Guffon reports grants for clinical trials from BioMarin, Shire, Genzyme, Sobi, and Merk Sereno, outside the submitted work. Susan L. Hale reports grants, personal fees and nonfinancial support from BioMarin, and grants from Genzyme and Shire outside the submitted work. Paul R. Harmatz reports consulting fees, grant support, and clinical trial support from BioMarin, consulting fees and clinical trial support from Shire, consulting fees and clinical trial support from Genzyme, consulting fees and clinical trial support from Alexion, Ultragenyx, and Armagen and consulting fees from PTC, Inventiva and Chiesi. John J. Mitchell has participated in BioMarin-sponsored clinical trials. He has received travel support and consulting fees from BioMarin. Jennifer Semotok has attended meetings and received travel support from Actellion and Genzyme. She did not receive any grants or personal fees or direct personal financial support from these companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The content of this manuscript was based on presentations and discussions during an expert meeting that was coordinated and funded by BioMarin Pharmaceutical Inc. Support in the process of manuscript development was also funded by BioMarin Pharmaceutical Inc.
