Abstract
The ultramicroanalytic system (SUMA), created in the 1980s, is a complete system of reagents and instrumentation to perform ultramicroassays combining the sensitivity of the micro-enzyme-linked immunosorbent assay (ELISA) tests with the use of ultramicrovolumes. This technology permitted establishing large-scale newborn screening programs (NSPs) for metabolic and endocrine disorders in Cuba. This article summarizes the main results of the implementation during the 30 years of SUMA technology in NSP for 5 inherited metabolic diseases, using ultramicroassays developed at the Department of Newborn Screening at the Immunoassay Center. Since 1986, SUMA technology has been used in the Cuban NSP for congenital hypothyroidism, initially studying thyroid hormone in cord serum samples. In 2000, a decentralized program for the detection of hyperphenylalaninemias using heel dried blood samples was initiated. These successful experiences permitted including protocols for screening congenital adrenal hyperplasia, galactosemia, and biotinidase deficiency in 2005. A program for the newborn screening of CH using the thyroid-stimulating hormone Neonatal ultramicro-ELISA was fully implemented in 2010. Nowadays, the NSP is supported by a network of 175 SUMA laboratories. After 30 years, more than 3.8 million Cuban newborns have been screened, and 1002 affected children have been detected. Moreover, SUMA technology has been presented in Latin America for over 2 decades and has contributed to screen around 17 million newborns. These results prove that developing countries can develop appropriate diagnostic technologies for making health care accessible to all.
Introduction
From January 1959, health and human life became a constant concern of the Cuban reality. Sociopolitical changes in the island brought a different conception of health services, being conceived by the state as an integrated system, with emphasis on disease prevention and universal access to services. This conception has significantly impacted on the progresses of medicine in Cuba. 1-2
Among the efforts to advance this concept, the creation and opening of the Immunoassay Center (CIE) in 1987 are included, an organization in charge of developing and producing kits for diagnostic purposes where its leading products are the ultramicroanalytic system (SUMA) or SUMA technology, 3 part of BioCubaFarma business group, which gathers major centers for scientific research and production in the area of biotechnology and the pharmaceutical industry in our country.
In the late 1970s, a small group of young researchers from the National Center for Scientific Research (CNIC), with the aim of contributing to the reduction in infant mortality in Cuba and by the existing need in the country of counting on economical and reliable methods for diagnosis, began to develop a sustainable test for the quantification of serum α-fetoprotein in pregnant women. 4
This small group, led from the beginning by Dr Jose Luis Fernandez Yero, who became later CIE’s Founding-Director for 25 years, laid out a strategy to achieve a type of test that would be compatible with the conditions of a country with limited financial resources and underdeveloped infrastructure; it should also have had low operating costs and could be easily adapted to the Cuban health system. That is how the SUMA technology was born, an own health technology and especially “appropriate” to the conditions and realities of the country. 3-4 In the 1980s, the development of the SUMA technology created the basis for establishing large-scale newborn screening programs (NSPs) for metabolic and endocrine disorders in Cuba.
Newborn screening programs are essential, preventive public health programs for early identification of disorders in newborns that can affect the quality of their life and expectancy. Early detection, diagnosis, and treatment of certain genetic, metabolic, or infectious congenital disorders can lead to significant reductions in death, disease, and associated disabilities. 5,6
This article reviews the main results on the implementation of this technology in NSP for 5 inherited metabolic disorders: congenital hypothyroidism (CH), congenital adrenal hyperplasia (CAH), biotinidase deficiency (BIOT), phenylketonuria (PKU), and galactosemia (GAL), using ultramicroassays, developed for almost 3 decades in the Department of Newborn Screening at the CIE. Besides, the article approaches the main challenges that SUMA technology, applied to the newborn screening, will be facing in the next years.
Methodology
Ultramicroassays for Neonatal Screening Supported by SUMA Technology
The SUMA technology is a complete system of reagents and instrumentation to perform ultramicro-enzyme-linked immunosorbent assay (UMELISA) or ultramicrotest (UMTEST), which comprises fully computerized spectrofluorometers for automatic reading, validation, interpretation, and quantification of results, plate washers, manual and automatic punchers, and reagent kits designed to perform UMTESTs (10-30 μL volumes of samples and reagents). The technology is manufactured by the CIE, Havana, Cuba, and it combines the sensitivity of the micro-ELISA tests with the use of ultramicrovolumes. 7 Since the birth of the methodology, the development and use of dedicated software for each of the tests have been a complementary tool that gave independence and added value to the technology. 8
Currently, neonatal SUMA’s assays include a set of 8 kits for newborn diagnosis of inherited metabolic diseases that CIE provides for the Cuban National Health Care System. Six of them use dried blood spots (DBS) on filter paper and the other 2 use serum samples. At present, the latter kits (TSH UMELISA and T4 UMELISA) are used for the confirmation of CH.
This article focuses on the characteristics of assays using DBS samples collected between the 5th and 7th days of age, with analytical quality evaluated and certified, and comparable with other existing commercial methods. 9
Thyroid-stimulating hormone (TSH) NEONATAL UMELISA, an ultramicroassay developed by CIE to determine neonatal TSH levels in DBS, is a sandwich-type, heterogeneous enzyme immunoassay using as solid-phase UMELISA strips (10 µL of solution per well) coated with monoclonal antibodies anti-TSH beta chain. Dried blood spots are eluted with a solution that contains sheep polyclonal anti-TSH/alkaline phosphatase conjugate, and the eluate is placed in the coated reaction opaque polystyrene UMELISA plates, where an antibody/TSH/antibody–enzyme complex is formed. A posterior washing of the plates eliminates the unbound conjugate and other components of the samples. When the fluorogenic substrate (4-metilumbeliferil phosphate) is added to the wells, the enzyme of the conjugate hydrolyzes it, and subsequent fluorescence intensity will be in proportion to TSH concentrations in the samples. 10
The thyroxine (T4) Neonatal UMELISA is an additional competitive ultramicromethod for CH screening, designed for the determination of total T4 in DBS, where the natural antigen and the enzyme-labeled antigen compete for a limited number of binding sites on the antibody. This assay uses as a solid-phase UMELISA strips coated with specific rabbit polyclonal anti-T4 antibodies. The DBS are eluted with a solution that contains the T4/alkaline phosphatase conjugate, and the eluate is placed in the coated reaction opaque polystyrene UMELISA plates. When samples are incubated in the reaction wells, antibody–antigen or antibody–antigen–enzyme complex are formed. A posterior washing of the plates eliminates the unbound conjugate and other components of the samples. When the fluorogenic substrate is added to the wells, the conjugated enzyme hydrolyzes it so that the intensity of fluorescence will be inversely proportional to the total T4 concentrations present in the samples. 11
The UMTEST PKU is a modification of the traditional McCaman and Robin method to determine the concentration of phenylalanine (Phe) in DBS. This ultramicrofluorometric test, supported in the SUMA technology, requires less time and volume than the traditional method by McCaman et al. The test is based on the reaction of Phe present in the sample with ninhydrin, in optimal conditions of pH and temperature, forming a low-level fluorescent complex. With the addition of copper ions, this complex becomes fluorescent, increasing its intensity due to the previous addition of
Initially, these 3 assays used 5-mm DBS samples, standards, and controls. 12 With the increase in tests done in NSP, it was necessary to use smaller blood punches in order to make a more rational use of the samples, so these kits were modified into 3-mm DBS discs at the beginning of this century.
The 17-hydroxyprogesterone (17OHP) Neonatal UMELISA is a simple and rapid competitive UMELISA assay designed to determine 17OHP levels in DBS. The assay is based on competition between 17OHP in blood specimens and 17OHP-alkaline phosphatase conjugate for a limited number of binding sites on specific rabbit polyclonal anti-17OHP antibodies. Standards, control, and samples blood spots are eluted with 17OHP-alkaline phosphatase conjugate in a danazol solution, and the eluates are placed in the coated reaction opaque polystyrene UMELISA plates. When samples are incubated in the strip wells, an antibody/17OHP or antibody/17OHP-alkaline phosphatase conjugate complex is formed. A posterior washing of the plates eliminates the unbound components. When the fluorogenic substrate (4-metilumbeliferil phosphate) is added to the wells, the enzyme of the conjugate hydrolyzes it, and subsequent fluorescence intensity will be inversely proportional to the amount of 17OHP in the sample. The 17OHP Neonatal UMELISA described initially used 40 µL of eluting solution, 13 which it was subsequently modified to 70 µL in the year 2010.
The UMTEST GAL is a simple, rapid, quantitative UMTEST based on the fluorometric method introduced by Fujimura et al adapted to SUMA technology for the detection of total galactose (GAL) in DBS. A methanol/acetone/water solution is used for deproteination. Galactosemia and galactose-1-phosphate are determined by measuring the change in fluorescence of nicotinamide adenine dinucluotide (reduced form NADH+H) at 450 nm, treated with β-galactose dehydrogenase and alkaline phosphatase. Both reactions occur in optimal conditions of pH and temperature. The resulting fluorescence is directly proportional to the amount of GAL in DBS. 14 The ultramicroassay described initially used 70 µL of deproteination solution, which was subsequently modified to 30 µL in the year 2014.
In all quantitative assays described, automatic validation and interpretation of the results are done using a dedicated SUMA assay software. 8 The UMTEST BIOTINIDASE is a simple qualitative visual ultramicroassay based on the colorimetric method introduced by Heard et al for the detection of BIOT in dried blood samples spotted on filter paper using N-biotinyl-p-aminobenzoate (B-PAB) as a substrate. According to the controls and samples evaluation, the ultramicroassay could clearly discriminate between normal newborns and those ones with low or absent biotinidase enzymatic activity. Results are obtained by a simple visual inspection of the colorimetric reaction. Those samples that developed a purple color are considered to have a normal biotinidase activity, and therefore, they are normal. The samples that developed a clear purple color are considered to have low biotinidase activity, and it was suspected that they are partial deficient. Those that present light yellow color are considered to have little or none activity, and they are classified as profound deficient. The ultramicroassay described initially used 40 µL of B-PAB, 15 which was subsequently modified to 70 µL in the year 2007. Table 1 shows the SUMA assays and the cutoff values used for the newborn screening of the 5 disorders included in the Cuban NSP.
Ultramicroanalytic System (SUMA) Assays and Disorders Included in the Cuban Newborn Screening Program.a

Abbreviations: GAL, galactosemia; Phe, phenylalanine; PKU, phenylketonuria; TSH, thyroid-stimulating hormone; UMELISA, ultramicro-enzyme-linked immunosorbent assay; UMTEST, ultramicrotest.
aSource: Newborn screening programs (NSP) database network of SUMA laboratories, Immunoassay Center.
bSince 1986 to 2010 NSP for congenital hypothyroidism (CH) used the TSH UMELISA in cord serum samples.
Cuban NSP Strategy
Cuban NSP is managed by the Ministry of Public Health through its Program for Comprehensive Care for Women and Children, which coordinate the actions of the health institutions in charged for the diagnosis, confirmation, control of the cases, treatment, follow-up of patients, and attention to families. The algorithm of the Cuban NSP is shown in Figure 1.
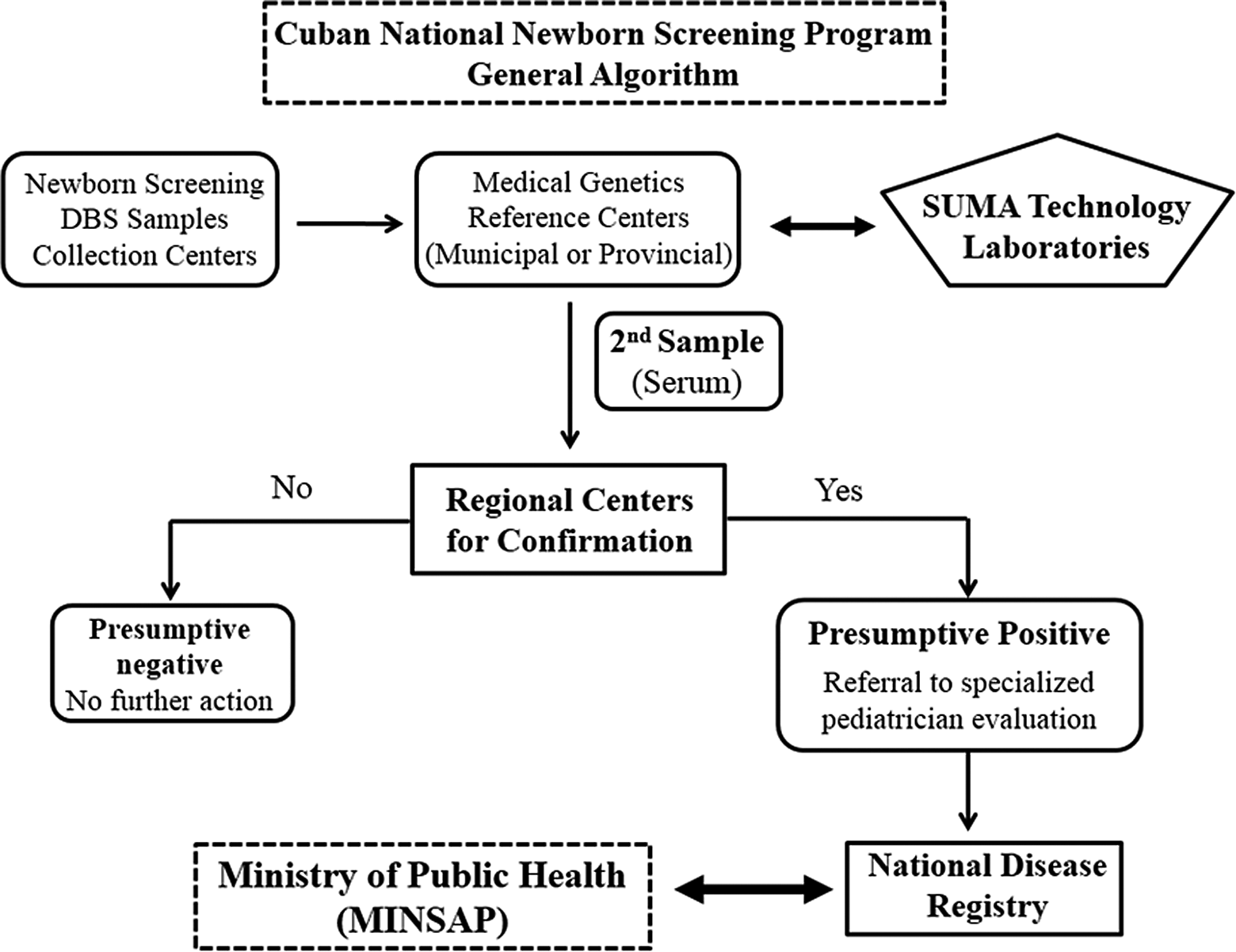
Cuban National Newborn Screening Program Algorithm.
Newborn screening tests were initially conducted by a national laboratory network made up by 32 laboratories. In the year 2010, all municipalities had at least a SUMA laboratory, which operated as a comprehensive active screening center for studying people according to their age, physical condition, and risk of contracting transmissible and nontransmissible diseases, in coordination with the primary health-care system, through polyclinics and family doctors. Thus, the number of laboratories covering the entire national territory performing neonatal screening tests has increased up to 175.
Technical and analytical assistance, replacement, and upgrading of equipment and reagents are supplied by Tecnosuma Internacional SA (TISA), which manages 5 regional technical assistance centers spread throughout the country.
The DBS samples are collected in the municipal collection centers preferably between the 5th and 7th days of life and sent to medical genetics reference centers, which are responsible for recording the data, and sent DBS samples to laboratories within 72 hours. Specimens are analyzed quickly after arrival in order to have the results ready in <24 hours. Thyroid-stimulating hormone, Phe, Gal, 17OHP, and BIOT activity are measured by the SUMA technology test for newborn screening previously described.
All newborns with a positive screening test are referred immediately for biochemical and clinical confirmation and follow-up at the regional pediatric centers. These institutions of the health system are nationally controlled by the National Institute of Endocrinology (CH and CAH) and the National Medical Genetics Center (GAL, PKU, and BIOT).
Confirmation is performed on serum samples that are sent to referral centers. Infants with negative serum results with respect to the cutoff value used for a given analyte are considered as simple false positives, and no other further investigations are made. Generally, patients with a positive confirmation serum result are referred to Provincial Pediatric Specialized Services for starting treatment. The DNA testing is only performed on PKU and CAH patients to study phenotype–genotype correlation. 16,17
In the specific case of the CAH program, due to the high false-positive rate (FPR), neonates with serum 17OHP levels ≥ cutoff value without clinical signs are considered as hyper-17OHP infants and are submitted to clinical and biochemical follow-up monitoring until the normalization of hormone values. Treatment with hydrocortisone is begun in all newborns with 17OHP levels ≥ cutoff value and clinical or biochemical signs of CAH. Patients with suggestive salt-wasting crises are given fludrocortisone doses.
Some quality indicators of the Cuban NSP were analyzed. The NSP database network of SUMA laboratories was reviewed from 1986 to December 2015 to determine the number of live births, screened infants, and confirmed cases. In addition, the number of positives (true and false positives) was recorded to calculate the positive predictive value (PPV) of the screening test and the incidence of the disorders. Further indicators such as coverage of the program, time of collection, percentage of unsatisfactory or invalid specimens, sample transit, and processing time in the laboratory and delivery time of results were evaluated in the period 2010 to 2015.
Results
Cuban NSP Results
In Cuba, the Cuban NSP using SUMA technology was initiated with an original development strategy, organizational model, and technological resources by a research and development-closed cycle through which scientific findings and technological developments are part of a combined effort to solve a specific health problem with feedback to improve it. The program was organized to be incorporated into the existing health-care system and make the most efficient use of limited resources. The evolution and the main results of the Cuban NSP are shown in Table 2.
Some Main Results of the Cuban Newborn Screening Program (1986-2015).a

aSource: Newborn screening program (NSP) database network of ultramicroanalytic system (SUMA) laboratories, Immunoassay Center.
The program for CH began in May 1986 and since 1988 has spread throughout the country. After Canada, Cuba was a leading pioneer nation of the Americas that provided a program with full coverage of the CH detection for newborn babies.
Until 2007, the study of TSH was exclusively done in cord serum using the TSH UMELISA, 18 when a pilot program for the implementation of the TSH Neonatal UMELISA was started.
In this case, SUMA technology has made possible to provide early detection of CH to all the Cuban children born since that date. Through December 31, 2015, a total of 3 847 656 children had been studied, of which 868 had that pathology at birth. Quality control in NSP requires precise assessment of target variables of development of the children, which should be identified and defined operationally throughout the entire program. The NSP for CH has made efforts to complement all the specialities. Nowadays, when the TSH value in DBS samples is over 15 mUI/L, the result is considered positive, and this leads to collection of a second sample for the determination of TSH and T4 in serum. Until this result is known, therapy with
Coordinators of the NSP have emphasized the value of studying the neurodevelopment in infants with CH, providing complementary data for the clinical management of the patients and continuous feedback to determine screening program efficacy and performance. The follow-up of the patients provides information that could lead to new means for optimal intervention in these and other affected children. 19,20
If they had not been detected by the screening provided with the SUMA test and provided with the medical treatment required by these cases, those children would had developed severe mental retardation due to the low levels of thyroid hormone production.
During 2000, the Ministry of Health has been reformulating the NSP for the gradual introduction of UMTEST PKU allowing the decentralization of the Cuban program for the detection of hyperphenylalaninemias using heel dried blood samples. Guthrie’s BIA was used in Cuba, since 1983 until 2000, 21 with an incidence of 1:50 000. 22 A pilot program for the newborn screening of hyperphenylalaninemias started in that year using the UMTEST PKU. 7
This ultramicrofluorometric test, supported in the SUMA technology, is a simple, specific, precise, accurate, and rapid assay that permits the processing of many samples at low cost and guarantees efficiency and reliable results. The UMTEST PKU requires less time and volume than the traditional method by McCaman et al. 23 With regard to the Guthrie’s BIA, previously used in the Cuban neonatal PKU screening program, it needs over 16 hours to perform the assay, while the total time required by the UMTEST PKU is about 2 hours. The diagnosis and treatment time are reduced, and the newborn screening quality is increased using the ultramicrofluorometric method. In addition, the UMTEST PKU gives the possibility of automatic validation and interpretation from data processing and permits the development of decentralized programs for the neonatal screening of hyperphenylalaninemias. The analytical performance characteristics of this method and its practical application in the Cuban PKU NSP have demonstrated its suitability for the neonatal screening of PKU. 7
These successful experiences permitted developing screening programs for CAH, Gal, and BIOT in 2005. Additionally, a decentralized NSP for CH in DBS using the TSH Neonatal UMELISA was fully implemented in 2010.
As shown in Table 2, after 30 years, more than 3.8 million Cuban newborns have been screened with this technology, and 1002 affected children have been detected. The current global incidence of inherited metabolic diseases identified by the Cuban NSP screening program was 2.02 per 10 000 live births (1:4890), with an annual rate of 25 cases/year. In the program, the age at serum sampling for the confirmatory testing was conducted between 9.0 to 20.0 days with a mean of 13.7 ± 9.9 days. The mean age for the start of the treatment was estimated at 21.8 ± 9.6 days.
The PPV and FPR are 2 aspects of the program that should be analyzed. The percentage of false positives among screened infants is maintained below 0.70% for the majority of the disorders, except for CAH screening. Table 2 shows that in general the PPVs in the programs are low, being higher only for CH screening. In some cases (Gal, PKU, and BIOT), this is related to the low incidence of these disorders, but in CH and CAH programs, there exists other factors that influence the PPV values.
In 2010, with the implementation of TSH UMELISA Neonatal in the Cuban NSP for CH, there was a marked decrease in the FPR. In the period 1986 to 2010, detecting TSH in cord serum samples, the FPR was 1.14%, whereas the use of DBS samples and the introduction of TSH UMELISA Neonatal diminished this rate to 0.21%. Thus, the PPV increased from 2.2 % to 5.8% in the last 6 years.
However, the magnitude of false-positive results generated in the CAH screening program has represented a great challenge for the public health system. In December 2015, the FPR for the CAH program was 1.54%. That’s why, in spite of the incidence of the disease, the PPV has been very low. A previous work had analyzed that a unique cutoff value of 55 nmol/L (calculated by 98th percentile) is used in the program, and more than 50% of false-positive results occurred in infants with a birth weight (BW) <2500 g or gestational age (GA) <37 weeks and was concluded that using an optimized cutoff level for BW or GA could lead to a reduction in the percentage of recalled babies and improve the PPV. 24 Taking into consideration these studies, 3 different cutoff values using BW and GA were applied in the Cuban NSP for CAH since November 2015.
Some Cuban NSP efficiency indicators in the period 2010 to 2015 are summarized in Table 3. In this study period, the program was able to achieve over 98% coverage. By the end of 2013, the coverage reached values higher than 99%, which have been maintained for the last 3 years. The analysis done to the rest of the indicators gathered in Table 3 shows that in spite of counting on a decentralized program, the percentage of unsatisfactory specimens has appropriately progressed in the last years, whereas an adequate and sustained work is observed collecting samples between the fifth and sixth day of life, processing time in the laboratory before 48 hours and informing the results in less than 72 hours.
Cuban Quality Newborn Screening Program Indicators (2010-2015).a
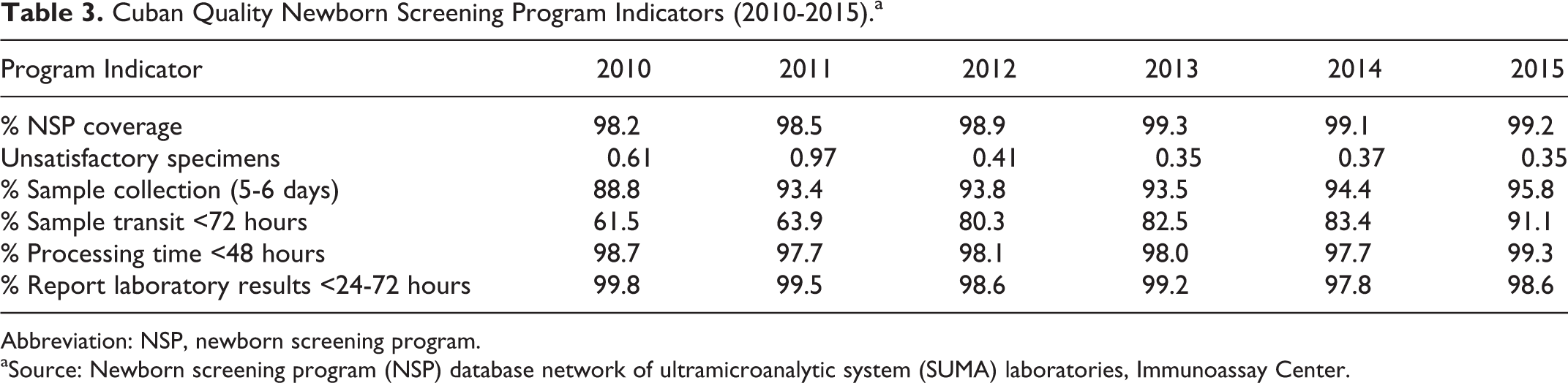
Abbreviation: NSP, newborn screening program.
aSource: Newborn screening program (NSP) database network of ultramicroanalytic system (SUMA) laboratories, Immunoassay Center.
Nevertheless, diminishing of sample transit to the laboratory is an important issue to work in because this is an essential element that would improve significantly the efficiency of the program, favoring the quality of the samples due to temperature excursions to which they may be subjected and increase the diagnostic opportunity of the program.
Finally, it should be mentioned that SUMA neonatal kits are regularly monitored for accuracy of results. Monthly, all laboratories in Cuba and most in Latin America participate in the Integral Scheme for Assurance and External Quality Assessment (EIAEEC) implemented since 1988 and provided by TISA. 25,26 Additionally, some SUMA laboratories are integrated in International monitoring External Quality Assurance Schemes, such as the US Center for Disease Control Newborn Screening Quality Assurance Program 27 and External Quality Assurance Program for NBS (PEEC-PN) from Argentina. 28 It is important to emphasize that the EIAEEC, as part of the SUMA technology, was the first EQAS for NBS, implemented in Latin America in 1988, and plays an important role because it includes the entire region. 28
Presence of SUMA Technology in Latin America and Other World Regions
From the consolidation of SUMA technology, and always with the main objective to guarantee national health programs, CIE has also dedicated to propose, design, and expand health programs in other countries, mainly in the Latin American region.
In Latin America, a sustained and significant growth in newborn screening activities has been palpable through the last 20 years, highlighted by the implementation of new national or regional NSP, the increases in the percentage of population covered, the expansion of the diseases included in the screening panels, and the increased involvement of the government and public health authorities. 6,29
In 1990, within this context, TISA is created who is in charge of marketing, extending health programs across the country, and makes them function stably, safely, and with the required quality.
The SUMA technology was installed in Brazil for the first time, 30 and then other countries such as Colombia, Mexico, Venezuela, Peru, Ecuador, Nicaragua, Bolivia, China, and Angola have followed. At present, more than 750 SUMA technology laboratories are installed worldwide.
Moreover, SUMA technology, which has been present in Latin America for 2 decades, has contributed to screening more than 17 million newborns, and more than 5000 affected patients have been detected during that period.
Challenges of the SUMA Technology
In Cuba, the implementation of SUMA technology has provided a model of a successful public health NSP for the past 30 years. However, this way has not been without its difficulties and challenges.
The SUMA technology and its application in the NSP have important challenges. Projects for the development of new tests and to improve existing ones together with assimilation and development of new technologies give continuity to the efforts for continuous improvement in neonatal screening in the country.
First, it is necessary to develop new affordable methods to complete the diseases included in the basic newborn screening panel and begin working on the development of methodologies to quickly count on with an expanded sustainable newborn screening panel in Cuba.
In this sense, in 2016, regional pilot screening projects are planning to test new assays for cystic fibrosis and maple urine disease supported in the SUMA technology. Successful results will permit proposing to the Ministry of Health to expand NSP of these disorders to the national territory.
Along with this, it should be considered to decrease the time of collection of the sample at 3 to 5 days of life, which would increase the diagnostic opportunity of the program and favor the future introduction of other diseases to the NSP, where time collection is crucial to ensure reliable results.
Second, although SUMA technology emerged as a semiautomatic technology mainly intended for laboratories of low and medium sample processing capacity, the global trend toward centralization of the newborn screening laboratories, increasing the amount of samples to be processed, it has made necessary the automation of this technology.
That is why a new automated analyzer for Neonatal Screening of SUMA technology SUMAutoLab has been developed. 31 The instrument presents a robotic arm for liquid handling with ceramic-coated stainless steel tips, a robotic arm to transport the plates to the various devices, programmable temperature incubators, shakers, plate washer, and fluorescence reader. The equipment is controlled by specific software that optimally schedules the different tasks and provides the necessary information both in preparation and during the run. The SUMAutoLab will allow the customers to increase efficiently the volume of sample processing in the laboratory, and it also results in more reliable and accurate diagnoses, drastically reducing the number of handling errors by operators. 31
Finally, the assimilation of new technologies when economically possible will favor the detection of new analytes, will increase the number of diseases, and will improve the protocols for newborn screening, confirmation, and follow-up. For example, it is known that PKU screening could be dramatically improved by adding a Phe/tyrosine (tyr) ratio, but unfortunately, the current technology does not allow us to apply this methodology. However, since 2010, an high-performance liquid chromatography method for simultaneously quantifying serum Phe and tyr is used to perform confirmatory and differential diagnosis of hyperphenylalaninemias, as well as biochemical monitoring of Cuban PKU patients. 32
Current economic conditions (mainly related to communications and transport) do not allow us to centralize the neonatal screening. In the future, with the introduction of a greater number of diseases to this program and the use of more expensive technologies (eg, tandem mass spectrometry), it will require to reevaluate the algorithm of the current NSP.
Final Considerations
Since the second half of the 20th century, the world is witnessing an unprecedented scientific–technological progress in all spheres of society. Impact of technologies in clinical practice and in the organization and delivery of services has been exponential, and today, it is possible to have more and better diagnostic and therapeutic prevention methods. 33
Contemporary world health situation demands the society to place in the foreground of analysis issues like equity, health promotion, and prevention, quality of care, ethical dilemmas, technological needs recognition, and the use of appropriate technologies as key factors for the development of adequate national health policies. 4,34 -36
Today, when referring to appropriate technologies, it should prevail the idea that their use must be determined by national technological capabilities and its correspondence with the social, economic, political, and cultural context of a particular country or region. 4,33 Thus, the solutions for massive and free application of preventive diagnostic systems and treatment of inherited metabolic disorders in the developing world could not come only from by simple technological assimilation because the economic implications would be unaffordable for those countries.
Therefore, in the case of Cuba, it was necessary to find own technological solutions adapted to its conditions of development and socioeconomic infrastructure, to thereby further progress in improving its public health system. 1 -3
The profound political, social, and economic changes that occurred in Cuba since 1959 have led to a significant improvement in the quality of life that shows health indicators of developed countries. In Cuba, health is seen as a strategic objective in the growth and development of society, where the state assumes full responsibility for the health care of the entire population. 1 -3
The SUMA technology is a genuine product of the Cuban technical–scientific revolution, developed in the early 80s of the last century as a response to a problem of our own health conditions of development: the need for making early diagnosis of diseases in large populations. 3,4
For almost 3 decades, the CIE has developed and commercialized SUMA technology, an appropriate methodology for diagnosis, to solve major health problems, introducing and maintaining quality indicators comparable with similar technologies in the field of clinical diagnostic.
Health programs supported by SUMA technology products include prenatal and neonatal screening, epidemiological surveillance, blood certification, and nontransmissible chronic diseases, giving a solution to the variety of diagnosis made in the laboratory. 3,4
Specifically, in the field of neonatal screening, through the development and implementation of SUMA technology, NSP for inherited metabolic diseases have been functioning in Cuba for more than 30 years and have expanded their coverage and now reach more than 99% of newborns.
In this manner, Cuban NSPs have been a triumph of public health system; through these programs, each year infants born with potentially devastating but treatable diseases are identified, treated, and preserved from an early death or mental retardation.
Conclusions
Genetics and immunology have experienced major scientific and technological progress in recent times, mainly in the industrialized world. The development of these sciences and the diagnosis of inherited metabolic diseases differ in Cuba in relation to other developing countries of the region and the world, given the advantages of our social and public health models.
This article has reviewed main elements related to the design and development of SUMA technology, an own and appropriate diagnostic procedure, which has largely allowed improving health indicators of the Cuban people over the last 30 years, being the main incentive for the continuous progress of this technology.
The creation of the CIE and the implementation of SUMA technology marked a significant moment in the scientific development of the Caribbean Island. In 2015, Cuba reached the lowest infant mortality rate in America, a figure that ratifies it as a leading country in this indicator, internationally regarded as a reflection of the health of the population and sociocultural development.
In these results, it has been important the expansion of NSP for 5 inherited metabolic disorders using SUMA technology, which places Cuba among the most developed in the region in the field of neonatal screening and demonstrate that developing countries can develop own and appropriate diagnostic technologies, that correctly applied can make health care accessible to all.
Footnotes
Acknowledgments
The authors would like to thank all employees who have served for 3 decades from Immunoassay Center. Also thank all the people and institutions from the Cuban public health system, who have been the most responsible for the results reported in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
