Abstract
Inborn errors of intermediary metabolism (IEiM) are complex diseases with high clinical heterogeneity, and some patients who have severe enzyme deficiencies or are subjected to stress (catabolism/infections) actually decompensate in the neonatal period. In this study, we performed metabolic tests on 2025 newborns in Mexico admitted to 35 neonatal intensive care units or emergency wards (NICUs/EWs) over a 6-year period, in whom a metabolic disorder was clinically suspected. Of these 2025 newborns with sickness, 11 had IEiM, revealing a prevalence of 1:184. Clinical characteristics and outcomes of the newborns with confirmed IEiM are shown. Of these 11 patients, 4 had isolated methylmalonic acidemia, 3 had maple syrup urine disease, 2 had urea cycle disorders, 1 had 3-hydroxy-3-methylglutaric acidemia, and 1 had isovaleric acidemia. During the first week of life (average 3 days), all of these newborns presented with impaired alertness, hypotonia, feeding difficulties, and vomiting along with metabolic acidosis and hyperammonemia. Of the 11 newborns with IEiM, 7 died, leading to a mortality rate of 64%. In conclusion, the differential diagnosis of newborns admitted to the NICU/EW must include IEiM, requiring systematic screening of this population.
Keywords
Introduction
Inborn errors of intermediary metabolism (IEiM) are a complex, heterogeneous group of genetic diseases. Most of them have severe neonatal onset and are a primary cause of death in newborns and infants. Unfortunately, newborns have a limited variety of responses to illness, and early signs and symptoms of IEiM are similar to the features of other, more common neonatal illnesses. 1 Specific and effective treatments are available for many IEiM, and early therapeutic intervention can prevent the worsening of disease. Even if therapy is unavailable, an accurate diagnosis is crucial for genetic counseling. 2
Inborn errors of intermediary metabolism represent a challenge to physicians. Prompt suspicion of these diseases in newborns and the knowledge of initial diagnostic approaches can aid in the selection of appropriate biochemical tests and measures of emergency management. 3 However, very early clinical onset of the severe neonatal forms of some of these disorders represents a logistics challenge to screening programs for newborns. Neonates with severe IEiM often become seriously ill during the first few days of life and must be admitted to the neonatal intensive care units or emergency wards (NICUs/EWs). 4 –6
Most children born in developing countries are not subject to mandatory neonatal screening for IEiM, making prompt and accurate diagnosis critical as soon as their clinical manifestations appear. 7 The Inborn Errors of Metabolism and Screening Laboratory of the National Institute of Pediatrics (INP) in Mexico City is a national reference center devoted to the diagnosis and management of aminoacidopathies, organic acidemias, and fatty acid oxidation disorders. 8 The objectives of the present study were to assess the prevalence of IEiM in newborns admitted to NICUs/EWs throughout Mexico, in whom a metabolic disorder was suspected and to describe their clinical characteristics and the outcomes of the first episode of metabolic decompensation.
Methods
Metabolic tests were performed in 2025 newborns (less than 30 days old at the time of the sample collection) in whom a metabolic disorder was clinically suspected and who were admitted to 35 NICUs/EWs, comprising 23 in the Mexico City metropolitan area and 12 in other Mexican states. The data collection period was from January 2007 to December 2012. Suspicion of a metabolic disorder was based on the clinical findings in each patient, for example, unexplained neurological signs and/or digestive symptoms and/or cardiac signs and/or positive family history or unexplained biochemical abnormalities, such as metabolic acidosis, hyperammonemia, or liver failure. Metabolic diagnostic tests included analysis of urinary organic acids, determination of plasma amino acid concentrations, and the quantification of amino acid and acylcarnitine concentrations in dried blood spot samples by tandem mass spectrometry (MS/MS), which was introduced in our laboratory since January 2010. The characteristic metabolic profile with the description of primary and secondary markers as well as the confirmatory tests was based on previously published data. 9,10 The markers were defined as follows, according to Millington and Stevens 11 : primary markers are those having potential diagnostic significance, while secondary ones are those biomarkers adding an element of risk for an underlying metabolic disorder, including certain biochemically related analyte ratios. The frequency of IEiM was calculated as the ratio of newborns diagnosed with IEiM to those suspected of having IEiM. The study was approved by the Institutional Review Board of INP, and the approval number is 017/2011.
Urinary Organic Acid Analysis
The concentrations of organic acids in urine were determined by gas chromatography–mass spectrometry (GC/MS) on an Agilent 6890N GC coupled to an MSD 5973 MS (5301 Stevens Creek Blvd, Santa Clara), as described by Sweetman. 12
Quantification of Plasma Amino Acids
Amino acid analysis was performed using reverse-phase high-performance liquid chromatography with fluorescence detection using an automated Waters system (34 Maple Street, Milford). 13
Quantification of Amino Acids, Acylcarnitines, and Succinylacetone
Dried blood spots were analyzed by electrospray ionization liquid chromatography-tandem MS (LC-MS/MS), using a Quattro Micro API tandem MS with commercial kits (NeoBase Non-derivatized MS/MS Kit; PerkinElmer 940 Winter Street Waltham, Massachusetts).
Results
During the study period, a total of 2025 samples from newborns admitted to NICU/EW with a suspicion of IEiM were analyzed. The proportion of premature versus full-term newborns was 33% versus 77%, respectively. From these samples, 11 newborns (8 male and 3 female) were diagnosed with 6 different IEiM. Four newborns were diagnosed with isolated methylmalonic acidemia (iMMA); 3 with maple syrup urine disease (MSUD); 2 with urea cycle disorders (UCDs), including one each with ornithine transcarbamylase deficiency (OTCD) and argininosuccinic acidemia; 1 with 3-hydroxy-3-methylglutaric acidemia; and 1 with isovaleric acidemia (IVA). Nonfatty acid oxidation disorder (FAO) was found in the studied patients. The prevalence of IEiM in newborns with sickness was 1:184; amino acid, acylcarnitine, and ratios determined by MS/MS as well as the confirmatory test findings are shown in Table 1. Consanguinity was found in 2 (18%) families. All the IEiM confirmed had an autosomal recessive inheritance, except 1 case with OTCD (X-linked inheritance). Four patients (36%) had a previously affected sibling who died due to similar clinical manifestations, with 1 patient diagnosed with IVA during postmortem. All 11 newborns were full term and were born after normal pregnancy and delivery. After birth, 10 were discharged home as healthy newborns, whereas 1 newborn with OTCD was kept in the hospital due to intrauterine growth delay and cephalohematoma.
Metabolic Screening and Confirmatory Tests Results of the Studied Patients.
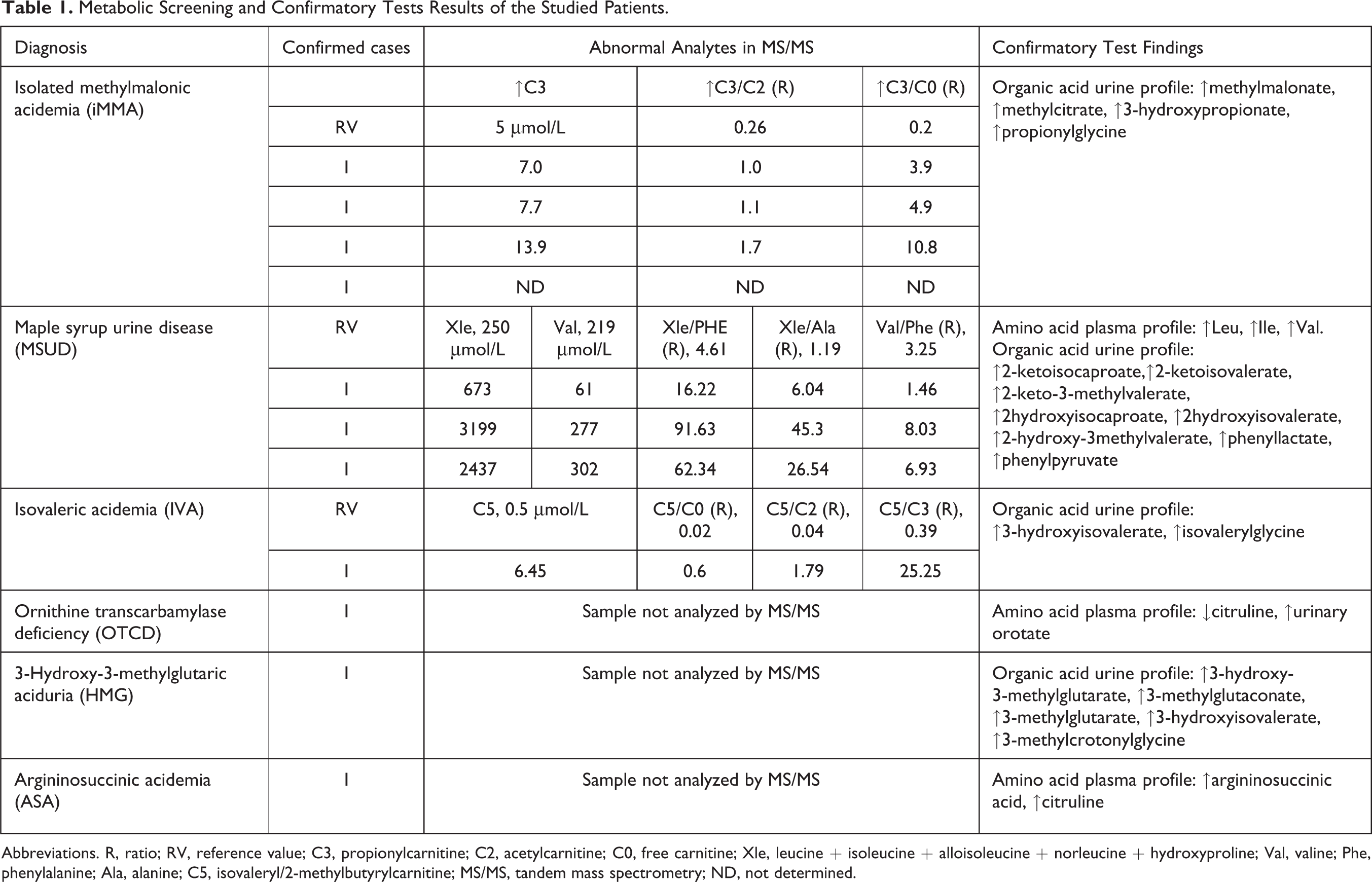
Abbreviations. R, ratio; RV, reference value; C3, propionylcarnitine; C2, acetylcarnitine; C0, free carnitine; Xle, leucine + isoleucine + alloisoleucine + norleucine + hydroxyproline; Val, valine; Phe, phenylalanine; Ala, alanine; C5, isovaleryl/2-methylbutyrylcarnitine; MS/MS, tandem mass spectrometry; ND, not determined.
The average age at the symptom onset was 3 days, and the mean gap between symptom onset and diagnosis was 14.5 days. Of the 11 patients, 6 died during the first hospitalization and 1 was diagnosed postmortem (Table 2).
Age at Clinical Onset, Diagnosis, Gap, and Status at Discharge From the Neonatal Intensive Care Unit and Emergency Wards (NICU/EWs) of 11 Mexican Newborns With IEiM.
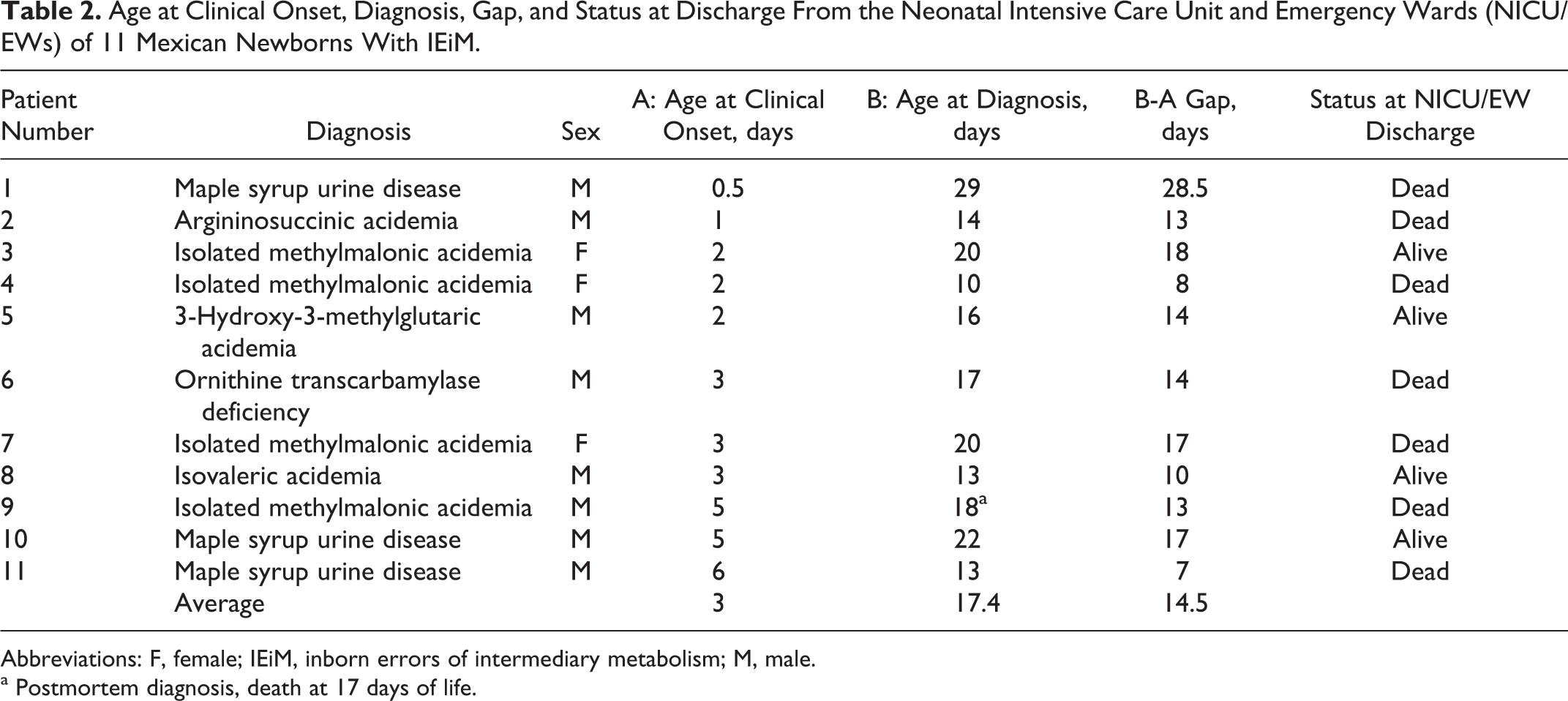
Abbreviations: F, female; IEiM, inborn errors of intermediary metabolism; M, male.
a Postmortem diagnosis, death at 17 days of life.
The main clinical features of this group of newborns are shown in Table 3. Neurological symptoms were the most frequent event in all newborns, followed by abnormalities in biochemical blood markers. Typical clinical manifestations included impaired alertness, stupor or coma, hypotonia, unexplained feeding difficulties and vomiting accompanied with metabolic acidosis and hyperammonemia, all occurring during the first week of life. Table 4 compares the IEiM detection and consanguinity rates in our population with similar studies performed on other populations.
Clinical Characteristics of Neonates Diagnosed With IEiM at Neonatal Intensive Care Units and Emergency Wards in Mexico.
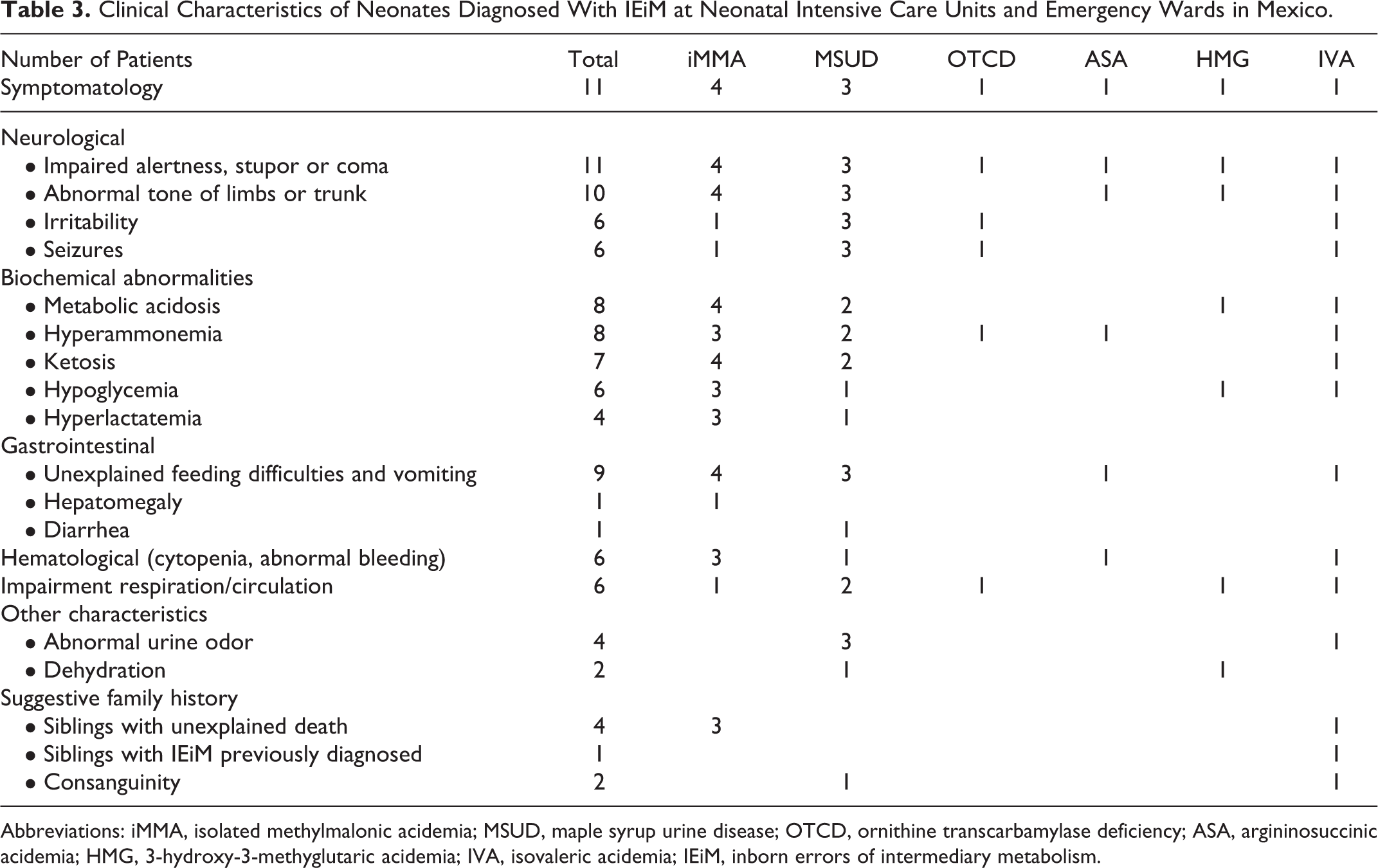
Abbreviations: iMMA, isolated methylmalonic acidemia; MSUD, maple syrup urine disease; OTCD, ornithine transcarbamylase deficiency; ASA, argininosuccinic acidemia; HMG, 3-hydroxy-3-methyglutaric acidemia; IVA, isovaleric acidemia; IEiM, inborn errors of intermediary metabolism.
Comparison of IEiM Detection (Total and by Individual Disease) and Consanguinity Rate in 3 Different Populations of Newborns With Sickness.
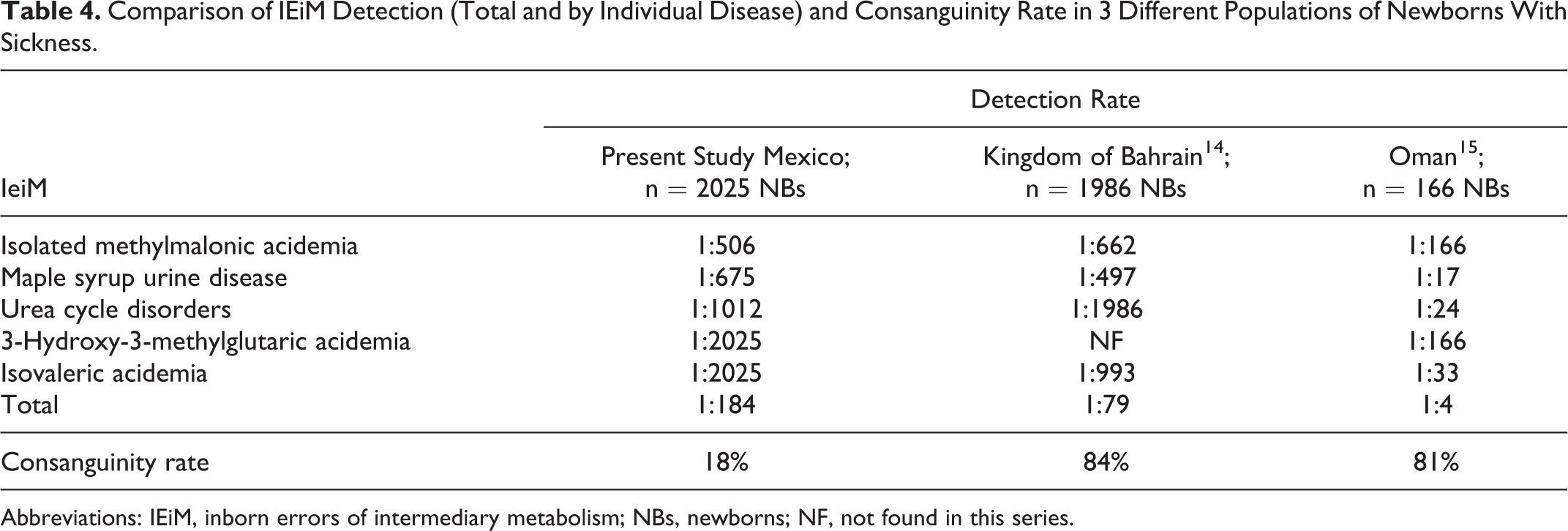
Abbreviations: IEiM, inborn errors of intermediary metabolism; NBs, newborns; NF, not found in this series.
Discussion
Although several studies have assessed the frequency of IEiM in children with sickness, 15 –18 only few others have analyzed its occurrence, specifically in critically ill newborns. 4,14 Our study population included newborns with critical illness admitted to NICU/EW and suspected of having a genetic metabolic disorder upon hospitalization. The prevalence of IEiM in this group was 1 in 184 newborns, less than the rates reported in Bahrain (1:79) and Oman (1:4). However, the consanguinity of these populations (84% and 81%, respectively) 14,15 was more frequent than in ours (18%).
Isolated methylmalonic acidemia was the most frequently detected genetic disorder in these patients, being reported in 1 in 506 newborns with sickness. It is known that iMMA is one of the most common IEiM detected worldwide, both in high-risk patients 8,14,17,19,20 and during screening of asymptomatic newborns. 21
It is noteworthy that not a single FAO was detected in the population studied; this could be due to some of the following reasons: (1) it is known that some patients among this group of disorders can present with sudden death syndrome and that they die before a diagnosis is made 22 ; (2) a false negative result was due to carnitine depletion in patients or because the sample was not collected at the moment of metabolic decompensation 23 ; and (3) a low prevalence of these disorders exists among Hispanic population as has been documented in some newborn screening programs. 21
Symptom onset was observed very early, that is, at an average of 3 days of life (range 0.5-6 days). All neonates with IEiM manifested the disease during the first week of life after an initial symptom-free period, but 1 neonate showed signs as early as 12 hours of life. The median gap between the onset of symptoms and a definitive diagnosis was 14.5 days (7-28.5 days); interestingly the patient who showed early symptoms had the longest delay in diagnosis (ie, 28.5 days). Initially this patient presented with irritability, dehydration, and fever and later developed abdominal distention, spasticity, seizures, and apnea requiring mechanical ventilation. He was initially thought to have neonatal sepsis, but due to adverse outcome he was suspected to have IEiM at the age of 20 days. A diagnosis of MSUD was confirmed at 29 days of life, and he died at the age of 2.3 months.
Of the 11 patients with IEiM, 7 (64%) died during their first hospitalization. Early onset of symptoms and high mortality rates in newborns with metabolic disorders, including organic acidemias and UCDs, have been documented in other population of newborns with sickness, ranging from 29% in France 4 to 36% in Thailand 24 and 50% in China. 6 Moreover, many infants with IEiM die during their first episode of metabolic decompensation before the disorder is diagnosed. 25 Fortunately, in some medical centers, the mortality rate showed a decrease during the first IEiM episode, due to rapid diagnosis and aggressive and timely treatment. 26,27
It is interesting to note that among the patients with autosomal recessive IEiM inheritance (10 of 11), there was a predominance of males (8 of 10). This has been reported in previous publications; Wasant et al studied 35 patients and found a male predominance of 2:1, and only 3 patients had an X-linked IEiM. 28 Similarly, Marin-Valencia et al found a male predominance of 1.4:1 among the 42 newborns studied (3 patients with an X-linked inheritance). 29 However, there are no further explanation that allows making any inference, and the number of patients studied to date is too small for any generalization.
As expected, the clinical manifestations of IEiM were nonspecific and generally multisystemic, affecting mainly the neurological, gastrointestinal, and hematological systems with impairments in respiration or circulation accompanied by biochemical abnormalities. The IEiM can result in acute or progressive intoxication, due to the accumulation of toxic compounds proximal to the metabolic block. However, these may manifest as nonspecific symptoms, attributable to other common causes, including sepsis, cardiac failure, or intraventricular hemorrhage. 1,30
We also found that 4 of the 11 patients had suggestive family histories, including siblings who died unexpectedly, siblings previously diagnosed with IEiM, or consanguineous parents (Table 3). Early onset, nonspecificity of symptoms, and high mortality rate of IEiM indicate that these diseases must be suspected and considered during the differential diagnosis of newborn with illness admitted to the NICU/EW, especially those with suggestive family history. High-risk metabolic screening must be part of the initial medical approach for these patients.
Newborn screening has changed the natural course of some IEiM, preventing sequelae and diminishing mortality rates. 31 However, the severe neonatal forms of these disorders pose a challenge to newborn screening programs, as their potentially devastating clinical symptoms may be present during the first few days of life, even before the Guthrie card is collected or abnormal results are reported. Many screening programs require specimens to be collected after 24 hours of age, with results generally reported when infants are between 6 and 15 days of age; by that time most infants with severe organic acidemias become seriously ill. 32 Fortunately, highly sensitive and accurate techniques such as MS/MS have shortened the turnaround time of diagnosis, allowing early collection of blood samples while providing immediate determination of amino acids, acylcarnitines, and their ratios, resulting in early diagnosis and prompt initiation of treatment. 33
It is important to emphasize that any child with an abnormal neonatal screening result suggestive of IEiM must be carefully evaluated and hospitalization must be considered, even in the absence of symptoms. 22 Rapid diagnostic confirmation and implementation of disease-specific treatment is essential for effective prevention. 33,34 The prevalence of IEiM presented in this study is useful for appropriate expansion of newborn screening in Mexico.
Conclusion
All newborns admitted to NICU/EW must be immediately and systematically screened for IEiM. Emergency treatment protocols should be initiated as soon as a metabolic disorder is suspected to reduce the mortality rates and sequelae associated with these diseases.
Footnotes
Acknowledgments
We would like to thank the patients and their families and physicians for their participation in this study. We also thank Aida Hernández and Ricardo Morales for their technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially funded by the Instituto Científico Pfizer de México, Fondo de Investigación 2011.
