Abstract
Background:
Graft selection in anterior cruciate ligament reconstruction (ACLR) may lead to long-term, graft-specific muscle adaptations, yet their effect on functional outcomes remains poorly understood. Magnetic resonance imaging (MRI)–based 3D modeling enables precise quantification of muscle volume changes, offering insight into the relationship between postoperative muscle preservation and patient-reported outcomes.
Purpose:
To radiologically evaluate thigh muscle volume changes and patient-reported outcomes at minimum 5-year follow-up among patients after primary anterior cruciate ligament reconstruction (ACLR) using allograft, bone–patellar tendon–bone autograft (BPTB), or hamstring autograft (gracilis and semitendinosus; HS) with MRI-based 3D modeling.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Patients aged 18 to 45 years (mean ± SD age, 26.9 ± 7.2 years) with a body mass index (BMI) <30 who underwent primary ACLR with either allograft, BPTB, or HS with minimum 5-year follow-up were identified via retrospective chart review. Exclusion criteria were any history of a knee flexor or extensor injury/surgery, revision ACLR procedures, any concomitant surgical procedures other than chondral debridement and/or meniscectomy/repair, or any subsequent lower extremity surgery since the index ACLR. Bilateral thigh 3.0-T MRI scans were obtained, and images were rendered using 3D modeling software. Volume measurements were performed for (1) the individual muscles, (2) each compartment, and (3) the entire thigh. Specific individual muscles analyzed included the quadriceps (rectus femoris, vastus lateralis, vastus medialis, vastus intermedius) and sartorius in the anterior compartment; the hamstrings (biceps femoris long and short heads, semitendinosus, semimembranosus) in the posterior compartment; and the gracilis and combined adductor muscle group in the medial compartment. Individual muscle volumes were calculated for each thigh and then normalized as a percentage of the total volume of their respective muscle compartments. These compartment-relative percentages in the operative thigh were subsequently compared with the corresponding percentages in the contralateral, nonoperative thigh. Multivariable linear regression linked ratios of injured to contralateral muscle preservation (total and compartmental) to International Knee Documentation Committee (IKDC) scores, adjusting for age, sex, BMI, and graft type. Paired Student t tests and chi-square tests were used for statistical analysis.
Results:
A total of 30 patients were included: 10 allograft, 10 BPTB, and 10 HS. The cohort consisted of 18 male patients (60%) and 12 female patients (40%), with a higher proportion of male patients in the BPTB group (9/10 male). Allograft patients (mean age, 34.6 years; range, 31-40 years) were significantly older at surgery than BPTB (mean age, 23.4 years; range, 17-33 years) and HS cohorts (mean age, 22.7 years; range, 16-28 years) (P < .001). The mean time interval from surgery to follow-up MRI was 6.7 ± 1.2 years. No significant differences in total thigh or individual muscle volumes were found between operative and nonoperative limbs with allograft. BPTB demonstrated a significant reduction in mean muscle volume percentage of the vastus intermedius in the operative thigh compared with the nonoperative thigh (13.98% vs 16.48%; P < .001). With HS, significant reductions were observed in mean muscle volume percentages of the gracilis (15.61% vs 27.35%; P < .001) and semitendinosus (7.49% vs 17.49%; P = .001) muscles in the operative thigh. Conversely, the semimembranosus exhibited a significant increase in mean muscle volume percentage in the operative thigh relative to the nonoperative thigh (42.50% vs 36.57%; P = .006). Across graft types, greater preservation of operative-limb muscle volume predicted better function: Total-thigh preservation correlated with higher IKDC scores (B = 61.8; P = .043), and anterior-compartment preservation showed the strongest association (B = 110.3; P = .004; model R2 = 0.48). No covariate (age, sex, BMI, graft type) independently influenced IKDC score.
Conclusion:
Five years after ACLR, thigh muscle adaptation depended on graft choice and carried meaningful functional consequences. Hamstring autografts were associated with lasting atrophy of the semitendinosus and gracilis with compensatory hypertrophy of the semimembranosus, whereas BPTB autografts were associated with selective loss of the vastus intermedius and no compensatory enlargement in the remaining quadriceps. Across all patients, larger differences in muscle volume between limbs, especially in the anterior compartment, were linked to lower IKDC scores.
Anterior cruciate ligament (ACL) injuries remain one of the most frequently encountered orthopaedic injuries among athletes, with incidence rates continuing to rise.6,21,28 Without treatment, ACL injuries can lead to significant functional impairments, tibiofemoral instability, and an increased risk of degenerative joint changes, especially in highly active individuals.4,17 Surgical reconstruction of the ACL has consistently demonstrated favorable results, including improved knee stability, reduced likelihood of subsequent knee surgeries, and fewer meniscal or cartilage injuries.1,4,24
Selecting the optimal graft is a key surgical decision, as different graft types have distinct biomechanical properties, donor-site morbidities, and long-term implications.8,13,23 Autografts, such as hamstring tendon (HS) and bone–patellar tendon–bone (BPTB), are frequently chosen for use in active individuals due to the relatively low failure rates of these grafts. 23 However, each autograft has tradeoffs. BPTB grafts can provide excellent long-term outcomes but may be associated with anterior knee pain and quadriceps dysfunction due to harvest site morbidity. 7 In contrast, HS grafts are considered less morbid but carry a risk of persistent hamstring atrophy, accompanied by compensatory hypertrophy in nearby posterior compartment muscles.3,19,25 Allografts circumvent donor-site morbidity altogether but may pose a risk of increased laxity and higher failure rates among younger, more demanding athletic populations.5,13,15,26
Although graft selection is often guided by graft strength, fixation properties, and retear risk, the long-term effects on muscle volume are less frequently considered, despite their potential functional implications. Muscle volume is a major determinant of torque generation across joints, 9 and reductions in thigh muscle volume after ACL reconstruction (ACLR) could directly contribute to postoperative strength deficits and impairments in knee function. 9 Although semitendinosus and gracilis adaptations have been well documented after HS autograft ACLR, relatively few studies have examined the volumetric effect of allografts or BPTB autografts, despite evidence suggesting that quadriceps dysfunction is a common consequence of BPTB harvest.3,18,25
To address these gaps, this study used 3D magnetic resonance imaging (MRI)–based volumetric analysis to evaluate long-term thigh muscle adaptations in patients who underwent ACLR using tibialis anterior allografts, BPTB autografts, or HS autografts. In addition to quantifying compartmental muscle volume relative to the contralateral limb, we assessed how muscle preservation ratios correlate with patient-reported outcomes. We proposed 2 primary hypotheses. First, in patients receiving BPTB or HS autografts, we expected persistent atrophy of donor-site muscles (eg, quadriceps or semitendinosus), accompanied by compensatory hypertrophy in adjacent muscles within the same compartment. Second, we hypothesized that greater volumetric asymmetry, reflecting lower muscle mass compared with the contralateral limb, would be associated with worse patient-reported outcome scores.
Methods
Patient Selection
After approval was granted by the institutional review board (IRB #00161193), patients who underwent ACLR with tibialis anterior allografts, ipsilateral BPTB autografts, or ipsilateral HS (gracilis and semitendinosus) autografts between December 2014 and December 2018 were identified at a single academic center for inclusion. Inclusion criteria were age between 18 and 45 years at the time of surgery and a body mass index (BMI) <30 at the time of surgery. Exclusion criteria were any history of a knee flexor or extensor injury/surgery, previous knee joint surgery, revision ACLR procedures, any concomitant surgical procedures other than chondral debridement and/or meniscectomy/repair, or any subsequent lower extremity surgery since the index ACLR.
A retrospective chart review was conducted to sequentially screen patients meeting the inclusion criteria. Those identified were contacted to confirm that they had not undergone any additional surgical procedures on either the ipsilateral or contralateral knee after their index ACLR. Relevant demographic and clinical data, including age, sex, and BMI, were extracted from the electronic medical record system (Epic; Epic Systems Corporation). Enrollment continued on a rolling sequential basis (moving from longest term follow-up to nearest term follow-up) until each graft group had recruited 10 patients who consented to and completed MRI scans. The final sample size was determined based on previously established methods using 3D MRI reconstruction in ACLR patients. 25 The 30 included procedures were performed by 4 fellowship-trained orthopaedic surgeons, with each surgeon performing a minimum of 6 surgeries within the cohort.
Digital Reconstruction of MRI and Segmentation Procedures
MRI scans were performed for each patient on both the injured and contralateral thighs, spanning from the lesser trochanter to the superior tibial tubercle. Imaging was conducted using a Siemens Magnetom Vida 3T scanner (Siemens Healthineers) with voxel dimensions of 1.5 × 1.5 × 3.0 mm. Digital Imaging and Communications in Medicine (DICOM) files were retrieved from the Picture Archiving and Communication System (IntelliSpace Radiology Enterprise 4.5; Philips North America Corporation) and analyzed using an open-source medical imaging platform (3D Slicer, Version 5.6.2; The Slicer Community, https://www.slicer.org).
T1-weighted images in axial, sagittal, and coronal planes were used for segmentation. Within 3D Slicer, the “Segment Editor” module was used to manually delineate anatomy in a systematic order starting with the bone, anterior compartment, posterior compartment, and medial thigh compartment. Surrounding structures such as skin, subcutaneous fat, and neurovascular elements were then segmented separately. The segmentation process began by outlining muscle compartment borders in the most superior axial plane and continued sequentially in a distal direction. Semiautomated tools within the software were used to improve accuracy and consistency.
To address potential differences in proximal visualization between limbs, a standardized adjustment was implemented. In cases where 1 limb had a longer scanned length, the 3D rendering was truncated to match the shorter limb, ensuring uniformity in volumetric comparisons. A reference line was drawn from the posterior aspect of the trochlear notch along the posterior femur to the most distal visible portion of each thigh. Using these delineated contours, 3D models of each thigh muscle compartment were reconstructed, and compartmental muscle volumes were calculated in accordance with previously published research using this method. 11 Volumetric data were extracted in cubic millimeters and centimeters using the statistical modules available in 3D Slicer (Figure 1).

3D magnetic resonance imaging rendering with corresponding axial segmentation.
Evaluation and Statistical Analysis
To ensure segmentation accuracy, interrater reliability was established before full cohort analysis. A subset of 10 patients was independently segmented by 2 orthopaedic residents (N.M. and R.M.R.) and the primary analyst (C.D.R.H.). An intraclass correlation coefficient (ICC) of 0.88 was calculated, demonstrating good consistency between the residents and the primary analyst. After this validation, the primary analyst completed the segmentation for the remaining subjects. All measurements and data entered were supervised by a fellowship-trained sports medicine surgeon.
Categorical variables, including sex distribution and operative laterality between graft groups, were compared using chi-square tests. The normality of muscle volume distributions within each graft group and individual muscle compartment was assessed using the Shapiro-Wilk test. For continuous variables, independent Student t tests were performed to compare mean values of age at ACLR, age at MRI acquisition, follow-up duration, and BMI across graft groups. A 2-tailed alpha level of .05 was used to determine statistical significance.
The volume of each thigh muscle as a percentage of the respective compartment was calculated and compared with the contralateral thigh using this formula:
To control for intersubject variability in body size and intrasubject variability regarding limb dominance, muscle volumes were normalized. Data were analyzed as a ratio of the specific compartment volume to the total thigh muscle volume. This internal normalization minimizes the confounding effect of absolute volume differences between dominant and nondominant limbs. Paired Student t tests were conducted to assess for statistically significant differences in muscle volume percentages between limbs, with significance set at P < .05. To minimize bias, the side of the injury was blinded during statistical analysis. Data were coded such that the analyst was unaware of which value corresponded to the operative or nonoperative limb during the computational processing.
To evaluate the relationship between postoperative thigh muscle preservation and patient-reported outcomes, 4 separate multivariable linear regression models were constructed. The dependent variable in each model was the International Knee Documentation Committee (IKDC) score. The independent variable of interest was the ratio of injured to contralateral muscle volume, analyzed for (1) total thigh volume and (2) each muscle compartment individually (anterior, medial, and posterior). Each model was adjusted for age at surgery, sex, BMI, and graft type (dummy-coded with allograft as the reference group). Multicollinearity was assessed using variance inflation factors, and model assumptions were verified using residual plots, histograms, and normal probability plots. The coefficient of determination (R2), regression coefficients (B), 95% CI, and P values were reported for each model and predictor. Based on an a priori calculation assuming a medium effect size (f2 = 0.15) at 80% power, it was determined that a larger sample would be required to fully detect effects of this magnitude in patient-reported outcomes. The primary variables of interest for the power analysis included patient-reported outcomes (IKDC and Single Assessment Numeric Evaluation [SANE] scores) as dependent variables and the ratios of injured to contralateral muscle volume preservation as the main independent predictors. Statistical significance was set at P < .05. All analyses were performed using SPSS Version 27 (IBM Corp).
Results
Participant Demographic Characteristics
A total of 30 patients (10 per graft group) met the inclusion/exclusion criteria and completed postoperative MRI (Table 1). Patients in the allograft cohort were significantly older at the time of ACLR (34.6 ± 5.2 years) compared with both the HS group (22.7 ± 4.3 years) and the BPTB group (23.4 ± 4.4 years) (P < .001). Similarly, at the time of MRI acquisition, the allograft group was significantly older (40.9 ± 4.9 years) than the HS (30.2 ± 4.3 years) and BPTB (29.7 ± 5.0 years) groups (P < .001). The HS group had a longer mean follow-up interval (7.5 ± 0.9 years) compared with both the allograft (6.3 ± 1.1 years) and BPTB (6.3 ± 1.3 years) groups (P = .033). The allograft cohort included 4 male patients (40%) and 6 female patients (60%), whereas the HS group had 5 male patients (50%) and 5 female patients (50%). The BPTB group had the highest proportion of male participants, with 9 male patients (90%) and only 1 female patient (10%) (P = .016). The mean BMI values were as follows: allograft, 23.4 ± 3.3 kg/m2; HS, 25.3 ± 3.9 kg/m2; and BPTB, 23.5 ± 2.6 kg/m2, without any significant differences between groups (P = .366). In the allograft group, 6 patients (60%) underwent surgery on the left knee and 4 (40%) on the right knee. In the HS group, 3 patients (30%) had left-sided ACLR, whereas 7 (70%) had right-sided procedures. The BPTB group had 5 patients (50%) with left knee ACLR and 5 (50%) with right knee ACLR (P = .171) (Table 1).
Demographic Characteristics of Study Participants a
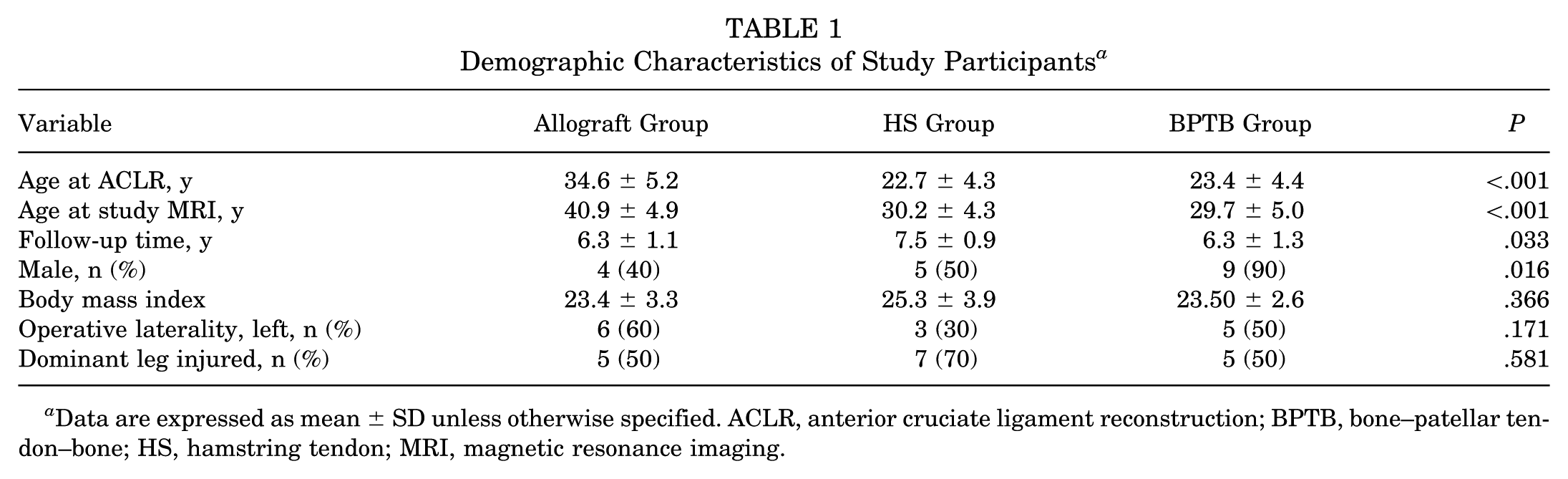
Data are expressed as mean ± SD unless otherwise specified. ACLR, anterior cruciate ligament reconstruction; BPTB, bone–patellar tendon–bone; HS, hamstring tendon; MRI, magnetic resonance imaging.
Muscle Volume Outcomes by Graft Type
Allograft Group
In the allograft group, no statistically significant differences were seen in overall or compartment-level muscle volumes between the operated (ipsilateral) and contralateral limbs. The mean anterior compartment volume measured 907.15 ± 69.3 cm3 on the ipsilateral side versus 940.06 ± 73.6 cm3 on the contralateral side (P = .088). Within this compartment, the rectus femoris measured 48.51 cm3 ipsilaterally and 49.02 cm3 contralaterally (P = .705), accounting for 5.36% and 5.21% of the compartment, respectively (P = .719). The vastus intermedius measured 191.17 cm3 ipsilaterally and 196.27 cm3 contralaterally (P = .232), representing 21.11% and 20.88% of the compartment (P = .654). The vastus lateralis measured 198.45 cm3 versus 202.06 cm3 (P = .417), with relative contributions of 21.89% and 21.51% (P = .317). The vastus medialis was 42.91 cm3 ipsilaterally and 41.90 cm3 contralaterally (P = .745), contributing 4.73% and 4.47% to the compartment (P = .651). The sartorius muscle measured 18.12 cm3 on the ipsilateral side and 18.78 cm3 on the contralateral side (P = .492), with relative contributions of 1.99% and 2.00% (P = .896).
The total posterior compartment volume was 516.04 ± 38.6 cm3 ipsilaterally and 532.18 ± 40.7 cm3 contralaterally (P = .314). Within this compartment, the long head of the biceps femoris measured 89.25 cm3 ipsilaterally and 91.40 cm3 contralaterally (P = .221), contributing 17.30% and 17.18% of the posterior compartment, respectively (P = .772). The short head of the biceps femoris measured 78.99 cm3 and 80.05 cm3 (P = .359), representing 15.32% and 15.06% of the compartment (P = .513). The semitendinosus measured 27.09 cm3 on the ipsilateral side and 26.89 cm3 on the contralateral side (P = .528), accounting for 5.25% and 5.06% of the compartment (P = .446). The semimembranosus muscle measured 19.80 cm3 ipsilaterally and 20.54 cm3 contralaterally (P = .265), with relative contributions of 3.83% and 3.86% (P = .947). The total medial compartment volume was 229.02 ± 18.4 cm3 on the ipsilateral side and 232.36 ± 16.9 cm3 on the contralateral side (P = .767). The adductor muscle group measured 181.95 cm3 ipsilaterally and 186.12 cm3 contralaterally (P = .505), representing 79.47% and 80.14% of the compartment (P = .661). The gracilis muscle measured 45.05 cm3 on the ipsilateral side and 45.74 cm3 on the contralateral side (P = .672), contributing 19.67% and 19.68% of the medial compartment (P = .978). Across all measured muscles and compartments, none of the differences between the ipsilateral (operated) and contralateral limbs were statistically significant (all P values >.05) (Table 2).
Allograft Muscle Volume Statistics

HS Autograft Group
In the HS autograft cohort, no significant differences were found in overall anterior or posterior compartment volumes between the operated and contralateral thighs. The anterior compartment measured 1127.15 ± 83.6 cm3 on the ipsilateral side and 1117.23 ± 81.2 cm3 on the contralateral side (P = .589). The posterior compartment volumes were similarly unaffected, measuring 537.63 ± 48.2 cm3 ipsilaterally and 539.69 ± 49.7 cm3 contralaterally (P = .945).
Among individual posterior compartment muscles, the biceps femoris long head volume was slightly higher on the ipsilateral side (162.54 ± 18.5 cm3) than the contralateral side (149.79 ± 17.2 cm3), although this difference was not statistically significant (P = .098). The biceps femoris short head also showed no significant volume difference (105.17 ± 11.8 vs 98.78 ± 10.5 cm3; P = .444). However, the semitendinosus volume was markedly lower in the operated limb (33.40 ± 4.2 cm3) compared with the contralateral limb (96.25 ± 8.1 cm3; P = .001), indicating substantial atrophy after tendon harvest. Similarly, the semimembranosus volume was slightly higher ipsilaterally (223.98 ± 21.3 cm3) compared with contralaterally (194.88 ± 19.5 cm3), although this difference did not reach statistical significance (P = .106).
Proportionally, the semitendinosus (7.49% vs 17.49%; P = .001) and gracilis (15.61% vs 27.35%; P < .001) contributed significantly less to their respective compartments on the operated limb, reflecting notable atrophy. Conversely, the semimembranosus demonstrated increased proportional volume in the ipsilateral thigh (42.50% vs 36.57%; P = .006), indicating possible compensatory hypertrophy. Similarly, the adductor group contributed a greater proportion to the medial compartment in the operated limb (84.39% vs 72.65%; P < .001), suggesting increased muscle recruitment in response to semitendinosus and gracilis deficits. These findings collectively indicate that semitendinosus and gracilis harvest resulted in significant atrophy of these muscles, with compensatory hypertrophy occurring in the semimembranosus and adductors (Table 3).
Hamstring Autograft Volume Statistics a

Boldface indicates statistical significance.
BPTB Autograft Group
In the BPTB autograft cohort, the anterior compartment volume was significantly lower on the operated thigh (1176.97 ± 64.7 cm3) compared with the contralateral thigh (1245.64 ± 59.3 cm3; P = .011). Among the quadriceps muscles, the vastus intermedius exhibited the most substantial reduction in volume, measuring 171.72 ± 18.0 cm3 on the ipsilateral side versus 212.08 ± 17.5 cm3 on the contralateral side (P = .003). Proportionally, the vastus intermedius contributed a smaller percentage to the anterior compartment on the operated limb (13.98%) compared with the contralateral limb (16.48%, P = .001), further emphasizing the localized atrophy in this muscle.
In contrast, no significant differences were observed in the remaining anterior compartment muscles, including the rectus femoris (59.12 ± 6.3 vs 60.71 ± 5.9 cm3; P = .495), vastus medialis (429.50 ± 41.2 vs 448.01 ± 39.7 cm3; P = .202), vastus lateralis (417.05 ± 38.5 vs 424.45 ± 37.3 cm3; P = .630), and sartorius (99.60 ± 9.8 vs 100.39 ± 9.6 cm3; P = .754). Additionally, the posterior compartment volumes (649.12 ± 52.7 vs 659.34 ± 51.1 cm3; P = .412) and medial compartment volumes (262.39 ± 23.9 vs 246.83 ± 22.6 cm3; P = .090) were not significantly different between limbs. These findings suggest that the effect of BPTB harvesting was primarily localized to the vastus intermedius, with minimal effects on other quadriceps muscles or adjacent compartments (Table 4).
BPTB Autograft Volume Statistics a

Boldface indicates statistical significance.
Patient-Reported Outcomes and Regression Analysis of Muscle Preservation
No significant differences in patient-reported outcomes were observed between graft types. The mean IKDC scores were comparable across groups, with patients in the HS group reporting the highest scores (85.6 ± 7.4), followed closely by the BPTB (83.8 ± 8.1) and allograft (83.6 ± 12.8) groups (P = .900). Similarly, mean SANE scores were consistent across cohorts, ranging from 8.0 to 8.2, with no statistically significant differences detected (P = .918) (Table 5). Multivariable regression analysis demonstrated that anterior thigh muscle preservation was the strongest and most consistent predictor of both SANE and IKDC scores. For SANE, greater anterior muscle preservation was significantly associated with higher scores (B = 11.8; 95% CI, 4.1 to 19.6; P = .004), and the overall model reached statistical significance (R2 = 0.417; F5,20 = 2.86; P = .042). Similarly, anterior preservation was also significantly associated with higher IKDC scores (B = 110.3; 95% CI, 38.9 to 181.7; P = .004), with a statistically significant model overall (R2 = 0.479; F6,19 = 2.92; P = .034). Total thigh preservation was associated with higher scores for both IKDC (B = 61.8; 95% CI, 2.1 to 121.6; P = .043) and SANE (B = 5.0; 95% CI, –1.5 to 11.6; P = .125), although only the IKDC association reached statistical significance and neither model was significant overall. Posterior compartment preservation was significantly associated with IKDC scores (B = 40.1; 95% CI, 0.2 to 80.0; P = .049) but not with SANE (B = 2.3; 95% CI, –2.3 to 6.8; P = .314), and both models failed to reach significance at the model level (R2 = 0.345 and 0.161, respectively). Medial compartment preservation was not significantly associated with either outcome (IKDC: B = −1.2; 95% CI, –31.3 to 28.8; P = .933; SANE: B = 0.15; 95% CI, –2.6 to 2.9; P = .910). Across all models, none of the covariates including sex, BMI, age at surgery, or graft type were independently associated with outcome scores (all P > .05) (Table 6).
Patient-Reported Outcomes (IKDC, SANE) of Patients in Each Graft Type a
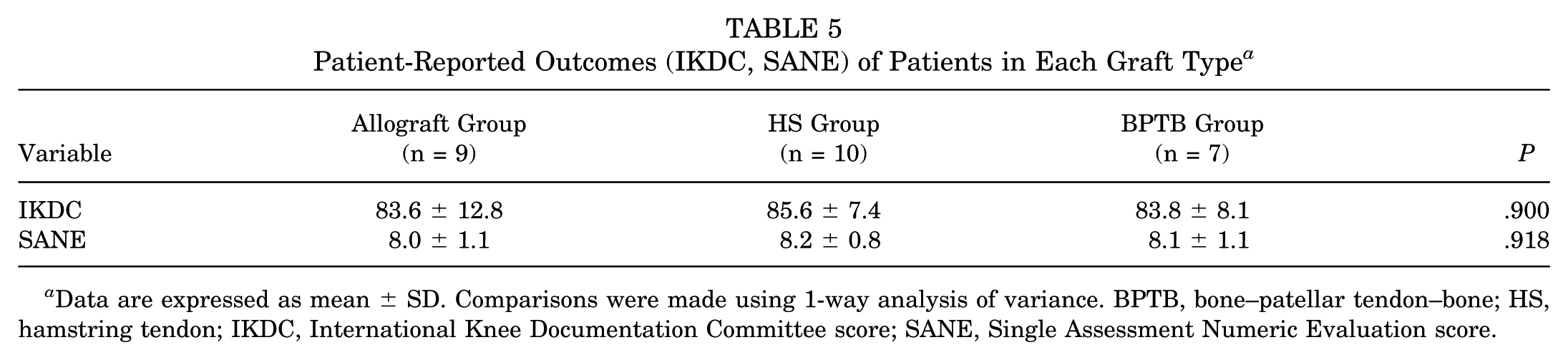
Data are expressed as mean ± SD. Comparisons were made using 1-way analysis of variance. BPTB, bone–patellar tendon–bone; HS, hamstring tendon; IKDC, International Knee Documentation Committee score; SANE, Single Assessment Numeric Evaluation score.
Multivariable Linear Regression Predicting IKDC and SANE From Ratios of Injured to Contralateral Muscle Preservation a

All regression models were adjusted for age at surgery, sex, and body mass index. I:C, ratio of injured to contralateral muscle volume; IKDC, International Knee Documentation Committee score; SANE, Single Assessment Numeric Evaluation score. Boldface indicates statistical significance at the alpha = .05 level.
Discussion
Over the 5-year follow-up, graft choice produced distinct and clinically meaningful patterns of thigh-muscle adaptation. Consistent with our first hypothesis, the hamstring autograft cohort showed marked, long-term atrophy of the semitendinosus and gracilis with a reciprocal hypertrophic response of the semimembranosus, suggesting redistribution of load within the posterior compartment. In the BPTB group, the vastus intermedius demonstrated significant volume loss; however, no compensatory enlargement was detected in the neighboring quadriceps heads, indicating that anterior-compartment recovery after BPTB harvest may rely more on strength rehabilitation than on intrinsic hypertrophy of the remaining muscle fibers. As hypothesized, the allograft cohort displayed no measurable side-to-side differences in total or compartmental muscle volume, reinforcing the advantage of avoiding donor-site morbidity when graft laxity risk is acceptable. Importantly, across all graft types, greater thigh-muscle asymmetry correlated with lower IKDC scores, underscoring the functional relevance of muscle preservation after ACLR.
Previous research examining muscle volume changes after hamstring autograft ACLR aligns with our findings of persistent semitendinosus and gracilis atrophy. Snow et al 25 reported a 40% to 50% reduction in gracilis and semitendinosus volumes at long-term follow-up after ACLR, with about an 8% increase in the biceps femoris long head. Konrath et al 14 similarly observed altered morphology and strength in the hamstring muscles up to 2 years postoperatively, noting that only about 35% of patients exhibited complete tendon regeneration in both semitendinosus and gracilis. Those with incomplete tendon regeneration experienced greater losses in donor muscle volume and length, which correlated significantly with reduced knee flexion strength. 14 Our findings reinforce these observations by demonstrating comparable semitendinosus and gracilis volume deficits but also reveal distinct patterns of compensatory hypertrophy in adjacent musculature. Although we observed an approximately 8% increase in the biceps femoris long head on the operated limb, this finding was not statistically significant, suggesting that hypertrophy of this muscle may not be predictable. Instead, our results suggest that the semimembranosus and adductor muscles undergo significant compensatory hypertrophy. These findings underscore the multifaceted interplay between muscle atrophy and hypertrophy in response to tendon harvesting, emphasizing the need for targeted rehabilitation strategies that optimize hamstring muscle recovery, mitigate imbalances, and address long-term functional deficits.
The present study further investigated patient-reported outcomes in individuals undergoing ACLR, with a focus on those with a minimum of 5-year follow-up. Although the sample size was limited, our findings revealed meaningful trends across graft types. Most notably, greater asymmetry in total thigh muscle volume between the operative and contralateral limbs was significantly associated with worse IKDC scores, suggesting that volumetric muscle preservation may play an important role in long-term knee function. Anterior compartment asymmetry demonstrated the strongest and most consistent association with both IKDC and SANE scores, underscoring the functional relevance of this region. Although greater anterior muscle asymmetry correlated with lower SANE scores, the corresponding model did not reach statistical significance, likely reflecting sample size limitations. These trends collectively support the hypothesis that postoperative muscle imbalance, particularly in the anterior compartment, may contribute to suboptimal patient-reported outcomes after ACLR and warrant further investigation in larger, prospective cohorts.
Quadriceps muscle atrophy in the postoperative period after ACLR is well documented.10,16,20 In our analysis, the BPTB cohort exhibited a compartment-specific deficit, characterized by a 19% absolute reduction in the vastus intermedius (P = .003) and a 17.6% overall decrease in anterior compartment volume (P = .011). These findings contrast with those of Noehren et al, 18 who reported more diffuse quadriceps atrophy, particularly affecting the vastus lateralis, along with histological disruptions suggestive of complex muscle adaptations. Similarly, Norte et al 19 observed substantial deficits exceeding 20% in the vastus lateralis and rectus femoris, while also noting relative preservation or compensatory hypertrophy in hip and adjacent thigh muscles after HS autograft ACLR. However, such widespread alterations were not observed in our study across any graft group, indicating that the pattern and severity of anterior thigh atrophy can vary significantly across studies. A key methodological difference between our study and that of Norte et al is the imaging field of view used for muscle assessment. Norte et al obtained MRI scans from the 12th thoracic vertebra distally to the ankle mortise, capturing a larger imaging field than was feasible in our study. Although Norte et al did not explicitly define the exact muscle cutoffs, it is likely that their imaging approach included more proximal regions of the rectus femoris, whereas our study's MRI field may have provided limited visualization of this muscle within the thigh. This discrepancy in imaging protocols may explain why rectus femoris differences were not detected in our study, despite previous findings suggesting its involvement in post-ACLR muscle adaptations. It is also possible that these discrepancies likely stem from differences in intraoperative techniques, graft harvesting, and postoperative rehabilitation protocols.20,22,27 Although literature on muscle volume loss remains limited, existing evidence consistently highlights significant quadriceps strength deficits after ACLR. 2 Because muscle volume is a key determinant of joint torque, it is likely that quadriceps atrophy contributes to functional impairments postoperatively. 9 Our findings suggest that the marked atrophy observed in the vastus intermedius and anterior compartment after BPTB ACLR may contribute to these deficits. Given the role of the vastus intermedius in knee extension strength, its disproportionate loss could have functional implications for postoperative recovery.
A recent exploratory MRI study by Ito et al 12 quantified lower extremity muscle volumes in Division I male football players and likewise highlighted persistent loss in the vastus intermedius after ACLR. That investigation provides valuable limb-to-limb data in a highly trained, sport-specific cohort, but its design differs from the present work in several key respects. First, Ito et al enrolled a homogeneous sample of collegiate football athletes (all male) and analyzed muscle size at a single follow-up visit that varied widely across individuals (mean 28 ± 19 months). In contrast, we prospectively selected equal numbers of patients who received hamstring autograft, BPTB autograft, or tibialis-anterior allograft and studied them at a uniform, long-term interval of 6 to 8 years. Our mixed-sex cohort therefore broadens generalizability and permits graft-specific contrasts that were not possible in the previous report. Second, the 2 studies approach muscle quantification differently. Ito et al normalized volumes to body size and converted each muscle to a Z-score, an approach that facilitates cross-muscle ranking but removes the absolute units, making it difficult to judge the clinical magnitude of these changes. We expressed every muscle as a percentage of its parent compartment, preserving volume units while reducing dominance bias, and we reported both compartment-wide and individual-muscle values in those same units. Although neither study corrects formally for multiple comparisons, our hypothesis testing was restricted a priori to graft-specific side-to-side differences within 3 thigh compartments, thereby limiting the total number of tests relative to the 38 paired contrasts performed by Ito et al. Furthermore, the earlier work was morphologic only; no strength, gait, or patient-reported outcome measures accompanied the MRI data. We incorporated validated patient-reported outcome measures (IKDC and SANE) and demonstrated that greater anterior-compartment preservation independently predicts higher IKDC scores at long-term follow-up. These methodological differences do not diminish the importance of the work by Ito et al; rather, the 2 studies complement each other, with one defining broad patterns of postoperative atrophy in elite athletes in an evaluation of 38 muscles of the thigh and lower leg and the other relating graft-specific morphology to patient experience in a more diverse population. Together, these studies advance understanding of long-term muscular adaptation after ACLR while highlighting the need for future, adequately powered investigations that integrate imaging, biomechanics, and outcome metrics within a single framework.
Limitations
This study has limitations inherent to its retrospective design. First, the absence of preoperative baseline imaging precludes assessing whether observed muscle volume deficits resulted from preexisting atrophy, neurogenic inhibition, or the surgical intervention itself. Second, the cohorts were not perfectly matched regarding age, sex, or athletic status, and the specific rationale for graft selection was based on subjective shared decision-making rather than a standardized protocol. To mitigate this heterogeneity and potential selection bias, we prioritized within-subject comparisons using the contralateral limb as an internal control. Third, we could not quantify specific rehabilitation dosing or adherence, as physical therapy was frequently performed at external facilities with variable documentation; consequently, variations in rehabilitation exposure remain a potential confounder. Fourth, although muscle volume serves as a proxy for functional capacity, this study did not directly correlate volumetric data with isokinetic strength or neuromuscular activation. Fifth, MRI-based analysis is subject to the resolution of image acquisition and segmentation technique, although our method aligns with validated approaches. Sixth, based on an a priori calculation assuming a medium effect size (f2 = 0.15; α = .05; 80% power), a sample of nearly 98 patients would be required to detect effects in patient-reported outcome measures of this magnitude. Seventh, quadriceps tendon autografts were excluded from this analysis as our institutional cohort for this graft type has not yet reached the minimum 5-year follow-up threshold.
Conclusion
Five years after ACLR, thigh muscle adaptation depended on graft choice and carried meaningful functional consequences. Hamstring autografts produced lasting atrophy of the semitendinosus and gracilis with compensatory hypertrophy of the semimembranosus, whereas BPTB autografts were associated with selective loss of the vastus intermedius and no compensatory enlargement in the remaining quadriceps. Across all patients, larger differences in muscle volume between limbs, especially in the anterior compartment, were linked to lower IKDC scores.
Footnotes
Final revision submitted February 2, 2026; accepted February 8, 2026.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research reported in this publication was supported by the LS Peery Foundation. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from The University of Utah Health (IRB Approval No. 00161193).

